Abstract
Recent studies on the electrocatalytic oxygen transfer from water to organic compounds have gained significant attention due to their sustainability and selectivity. However, the direct coactivation of inert hydrocarbons and water typically requires high oxidation potentials, leading to oxygen evolution reactions and low Faradaic efficiencies. Herein, a Ni-activated tungsten-oxygen covalency anode is designed for the efficient oxygen transfer from water to benzylic C(sp3)–H bonds via a Ni-regulated interfacial water structure between the anode and electrolyte. Both experimental and theoretical results reveal the critical role of W–O covalency sites with Ni-heteroatoms for boosting efficient oxygen transfer via breaking the dense interfacial hydrogen bond network and inhibiting the undesired oxygen evolution reactions, facilitating the coactivation of oxygen species and C(sp3)–H bonds. Thus, a Faradaic efficiency of > 56% in a water-involved system has been achieved. This work provides important insight into designing electrocatalytic systems for inert C–H oxidation.
Similar content being viewed by others
Introduction
The production of functional oxygenated compounds via the activation of hydrocarbons using various oxidants accounts for approximately one-quarter of all industrial catalytic processes1,2. Moreover, over 30% of cradle-to-gate greenhouse gas emissions in chemical production can be attributed to the intensive energy input and uncontrolled overoxidation associated with traditional thermochemical methods3,4. Recent works on the electrocatalytic oxidation of hydroxyl/carbonyl groups5,6,7, hydroxyl/carbonyl-α C–H bonds8,9, various types of biomasses10,11,12,13,14 and abundant hydrocarbons15,16,17 have demonstrated the significant potential of using water molecules as a sustainable oxygen source18,19 to produce functional organic compounds. However, the direct coactivation of even inert hydrocarbons and H2O typically requires relatively high oxidation potentials19,20, often leading to undesirable competitive oxygen evolution reactions (OER) and overoxidation, resulting in the formation of acids and esters16,17, among other products. Though several works have put forward strategies such as electron density regulation21, oxophilicity tuning22,23,24, halogen mediation25,26, and electrolyte engineering27,28, et al. for facilitating the oxidation of olefins while depressing the OER process and overoxidation, the activation of even inert C(sp3)–H bonds still suffers from unsatisfactory Faradaic efficiencies15,16,17 (FE ~ 31%) especially in additive-free catalytic systems due to ultra-high oxidation potentials of inert C(sp3)–H bonds as compared with that of OER. It is still highly alluring to put forward efficient strategies for activating C(sp3)–H bonds for effective K/A oil production while inhibiting the OER process in a water-involved electrocatalytic system.
Herein, we show that Ni-activated tungsten-oxygen covalency (Ni-WOC) can efficiently boost the oxygen transfer from water to inert benzylic C(sp3)–H bonds via loosening the originally dense interfacial water structure and promoting the C(sp3)–H bond activation on the in situ formed tungsten-oxygen covalency, effectively producing ketones and alcohols without overoxidation products (e.g., acids and esters). A reconstruction method is suggested to prepare highly dispersed and amorphous WOC sites with better catalytic performance than crystalline WO3. Enabled by favorable lattice matching that ensures the reconstruction durability, the synergy between the Ni dopants and reconstructed WOC sites can largely disrupt the interfacial hydrogen bond network and simultaneously boost the co-activation of water molecules and C–H bonds. In situ attenuated total reflection surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS), ab initio molecular dynamics (AIMD), and density functional theory (DFT) studies reveal the critical role of Ni dopants in weakening the hydrogen bond network over the adjacent Ni-WOC species for faster oxygen transfer from water to substrates, simultaneously depressing the unwanted OER process via regulating the reaction path. Concurrently, the efficient oxidation of C(sp3)–H bonds is achieved by the coactivation of C(sp3)–H bonds and adsorbed oxygen on certain W sites of Ni-WOC. The combination of theoretical and experimental results demonstrates the boosted oxygen transfer process from water to C(sp3)–H bonds on W–O covalency via the rationally introduced Ni heteroatoms, providing a Faradaic efficiency up to > 56% (Table S2).
Results
Oxygen transfer from water to benzylic C(sp3)–H bonds
To introduce uniformly dispersed Ni dopants, we employed a carbothermal method to synthesize the Ni-doped tungsten carbide29 (Ni/W2C) pre-catalyst by thermal pyrolysis of the mixture of ammonium metatungstate, nickel nitrate hexahydrate, and dicyandiamide, in N2 atmosphere at 1000 °C (for experimental details, please see Methods section). The Ni contents of Ni/W2C samples can be precisely tuned from 0 via 0.8 and 1.6 to 3.1 wt.% by varying the amounts of nickel nitrate hexahydrate in the precursor (Table S1). As the Ni contents increased from 0 wt.% to 3.1 wt.%, the as-formed tungsten carbide samples all exhibited a similar structure of crystalline nanoclusters supported on layered graphene with mean sizes of 3.4-3.7 nm (Fig. 1a and S1). Accordingly, the characteristic X-ray diffraction (XRD) peaks (Fig. S2) of the (0 0 2) facet of α-phase W2C (JCPDS# 35-776)29,30,31 gradually shifted from 38.1° to 38.6°, indicating more Ni dopants were introduced to the crystal lattice of W2C. Indeed, the uniformly dispersed Ni dopants in the crystal lattice of W2C nanoclusters have been directly revealed by the aberration-corrected high-angle-annular-dark-field scanning transmission electron microscopy (AC-HAADF-STEM) (Fig. 1a) with distinguishable differences in contrast of W and Ni atoms32 in atomic resolution AC-HAADF-STEM images (Fig. 1b, c). The homogeneous distribution of Ni dopants in W2C clusters was further confirmed by corresponding energy-dispersive X-ray spectroscopy (EDS) mapping and line scan results (Figs. 1a and S3–4). X-ray photo-electron spectroscopy (XPS) analysis results (Fig. 1d and S5) further confirmed the presence of W and Ni atoms mainly in the form of metallic state, aligning well with the typical electronic configuration of transition metal carbides29,33,34.
a The AC-HAADF-STEM image of Ni1.6/W2C pre-catalysts and corresponding STEM-EDS elemental mapping results of W Lα line and Ni Kα line. b The atomic resolution AC-HAADF-STEM image of Ni1.6/W2C pre-catalysts (the blue circles indicate Ni dopants). c The corresponding intensity profiles of line Ⅰ and line Ⅱ in Fig. 1b. d The XPS spectra of W 4 f and Ni 2p of Ni1.6/W2C pre-catalysts.
The obtained Ni/W2C samples were investigated as potential anode pre-catalysts for the oxygenation of benzylic C(sp3)–H bonds on ethylbenzene via oxygen transfer from water, which is utilized as a model reaction (Fig. 2a) in a homemade undivided three-electrode system (for experimental details, please see Methods section). In this system, no oxygenated products were generated without an applied bias (Fig. S6), regardless of whether Ni/W2C was used as a pre-catalyst or not. At 1.8 V vs. Ag/AgCl, the system without pre-catalysts (Fig. S6) only exhibited very low yield ( < 3%) and FE ( < 17%). However, all Ni/W2C pre-catalysts with different Ni contents (Fig. S6) exhibited higher yields (from 7.8% to 10%) and FEs (from 42.3% to 45.4%) at a fixed condition within 2 h. More importantly, with optimized Ni content, the Ni1.6/W2C pre-catalyst can successfully achieve the continuous oxygenation of benzylic C(sp3)–H bonds with a yield up to 90% within 48 h and a regio-selectivity of > 99% to 1-Phenylethanol and acetophenone (Fig. 2b and S7), indicating an efficient electrocatalytic oxygen transfer process. The Ni1.6/W2C pre-catalyst with optimized Ni content was employed for further studies.
a Schematic illustration of oxygen transfer from water to benzylic C(sp3)–H bonds. b Time-dependent yields of the electrocatalytic oxygen transfer process over 48 h at the anode. The inset picture shows the yield change from 0 to 0.8 h. Reaction conditions: 1.8 V vs. Ag/AgCl, with a catalyst loading of 1 mg/cm2. c The catalyst loading-dependent yields of oxygenated products and corresponding Faradaic efficiencies at 2.0 V vs. Ag/AgCl. d The potential-dependent yields of oxygenated products and corresponding Faradaic efficiencies with a catalyst loading of 1.5 mg/cm2. e GC spectra of the electrolyte after oxidation using H218O as the oxygen source at different reaction times. Anisole (at the retention time of 4.6 min) was used as the internal standard for quantification. f, g The mass spectrum of 1-phenylethanol and acetophenone generated at the anode using H218O as the oxygen source in a 4-hour reaction.
It should be noted that the conversion and FE for oxygenated products are closely tied to the catalyst loading (Fig. 2c), highlighting the essential role of anode catalysts in facilitating the electrocatalytic oxygen transfer process. Moreover, this oxygen transfer process can be efficiently achieved over a broad range of working potentials (Fig. 2d), from 1.6 V to 2.2 V (vs. Ag/AgCl), with satisfactory FE ranging from 47% to 56% (Fig. 2d). And the FE of 56.5% under optimized conditions (Fig. 2c, d and S8) is among the highest ones (Table S2) in currently all reported additive-free inert C(sp3)–H oxidation system using water as oxygen source, indicating more favorable oxygenation process compared with OER pathway with a FE > 50%.
The isotope labeling experiments (Fig. 2e) using H218O further confirmed an efficient oxygen transfer process from water to C(sp3)–H bonds, with only 18O-labeled products detected (Fig. 2f, g and S9). All these results reveal an electrocatalytic oxygen transfer process from water to C(sp3)–H bonds, as depicted in Fig. 2a.
In situ formation of Ni-activated W–O covalency sites
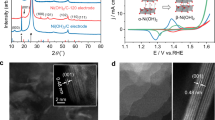
Interestingly, the progressive activation behavior (Fig. 2b inset) of the pre-catalyst was observed at the earlier stage of the oxygenation reaction of ethylbenzene, manifested by a notable enhancement in catalytic activity. We thus turned our interest to the identification of real active sites. AC-HAADF-STEM images and the corresponding EDS mapping results of the used catalysts (Fig. 3a-b) show highly dispersed and amorphous structures of W and Ni species (Fig. 3b), speaking for a reconstruction process of the W2C components to generate the real catalytic centers. The atomic-resolved AC-HAADF-STEM image (Fig. 3c) of the reconstructed sample directly demonstrates the homogeneous distribution of Ni atoms over amorphous clusters, which shows the replacement of W to Ni atoms with obvious contrast difference (Fig. 3d) and the comparable Ni/W–W distance without obvious loss of Ni sites. The fast degradation of metal carbide nanocrystals into an amorphous state within around 10 min was further revealed by the ex situ X-ray diffraction (XRD) analysis (Fig. S10).
a The AC-HAADF-STEM image of the used Ni1.6/W2C pre-catalysts. b The AC-HAADF-STEM image and corresponding STEM-EDS mapping results of the used Ni1.6/W2C pre-catalysts. c The atomic-resolved AC-HAADF-STEM image of the used Ni1.6/W2C pre-catalysts. d The corresponding intensity profiles of line Ⅰ and line Ⅱ in Fig. 3c. e Schematic illustration of the in situ formation of highly active Ni-WOC sites. f–h Fitted curves of time-dependent ex situ XPS spectra of O 1 s, Ni 2p, and W 4 f obtained under 1.8 V versus Ag/AgCl, respectively. i Yields of ethylbenzene oxygenation using different types of catalysts (reaction conditions: 1.8 V versus Ag/AgCl, with a catalyst loading of 1 mg/cm2).
The ex situ XPS studies further suggested a migration process of oxygen species (Fig. 3f) to generate WOC or Ni-WOC with W6+ (36.1 eV and 38.3 eV) or Ni2+ (856.5 eV) centers29,31,35,36,37,38 from original metallic W0 and Ni0 states (Fig. 3g, h and Table S3), respectively. Typical peaks of adsorbed hydroxyl groups36,39,40,41 centered at 532.5 eV (Fig. 3f) gradually decreased with prolonged reaction times, suggesting an oxygen transformation/transfer process from pre-adsorbed hydroxyl species to lattice oxygen for the formation of Ni-WOC, with lattice oxygen signal35,36,40,42 (530.8 eV) becoming dominant after 10 min. Specifically, at the initial stage of the reaction (1 min), a significant accumulation of hydroxyl groups at W sites was observed (Fig. 3f), serving as the primary oxygen source for the formation of lattice oxygen in the WOC structure. From 1 min to 30 min, the ratio of lattice oxygen increased from 21 % to 41% (Fig. 3f), while the peak area of hydroxyl groups remained relatively constant (Fig. 3f). This indicates an ongoing oxygen species migration process where hydroxyl groups transform into lattice oxygen, converting carbide into oxide, and the consumed hydroxyl groups can be readily replenished from the electrolyte, ensuring a continuous and abundant oxygen supply throughout the reaction period. Indeed, the potential-dependent in situ Raman spectra and the ex situ Raman spectra of the reconstructed samples confirmed the formation of tungsten-oxygen species (Fig. S11) according to the ever-weakened W–C signal43 from 690 cm-1 to 810 cm-1 and the accumulation of W=O signal40,43,44,45 at around 940 cm-1, as well as the W–O–W signal45 at around 330 cm-1. All these results, that the co-retention of Ni and W sites in amorphous clusters and the significant emergence of lattice oxygen, indicate the oxygen species-migration process as depicted in Fig. 3e for the in situ formation of Ni-WOC as the possible real active sites for the oxygen transfer process.
The in situ generated highly dispersed Ni-WOC sites have provided fully exposed active sites with the essential synergy between WOC sites and uniformly dispersed Ni heteroatoms, which is of key importance for boosting the oxygen transfer process. Specifically, the yield (3.5 %) and FE (24.3%) toward oxygenated products on crystalline WO3 (c-WO3, Fig. 3i and S12) anode is slightly lower than those of pristine WOC (4.0 % yield, 33.9% FE) or Ni/NC (3.7 % yield, 27.3% FE) anodes, indicating a slightly promoted oxygen transfer reaction on highly dispersed WOC or Ni sites. More importantly, the Ni-WOC anode provided a yield of 10% and an FE of 45.4% under a fixed condition (Fig. 3i and S12), which is 2.5 times the yield of the WOC anode and 2.1 times the yield of the mechanical mixture of WOC and Ni/NC. The decreased electrochemical surface area (ECSA) of Ni-WOC catalysts from 43.63 mF·cm-2 before reconfiguration to 25.01 mF·cm-2 after reconfiguration (Fig. S13) rules out the influence of ECSA on the enhanced catalytic efficiency. The well-maintained layered graphene-supported structures after reconfiguration (Fig. S14) further validate the unique role of Ni dopants, rather than any morphological changes in support, in enhancing the final catalytic performance. All these results reveal the essential importance of the in situ formation process for highly dispersed Ni-WOC sites with obvious synergy between Ni heteroatoms and WOC sites for the enhanced oxygen transfer process, which cannot be solely attributed to the existence of tungsten oxides or the changes in surface area.
Boosted water diffusion over Ni-activated W–O covalency
The involvement of oxygen transfer in the formation of Ni-WOC also plays a crucial role in the subsequent oxygenation reaction, in which the arrangement of water molecules (the oxygen sources) at the interface between the Ni-WOC surface and electrolyte dominates the initial oxygen diffusion46,47 efficiencies. The AIMD simulation results (Fig. 4a-c, S15, and Supplementary Data 1-4) theoretically indicated the weakened hydrogen bond (H-bond) networks (Fig. 4a, b) over the surface Ni sites of the Ni-WOC model, manifested by the decreased number of H-bonds from 4.8 on the WOC model to 2.9 on the Ni-WOC model in the area with a vertical distance (z) of 3.36 Å from the surface Ni atoms. This is to say that there is a void in the water layer over Ni sites as reflected by the absence of interfacial water (Fig. 3c) in the domain with a radius (r) of 2.16 Å to Ni sites.
a The equilibrium configuration of interfacial water structure and H-bond network over the Ni-WOC model revealed by AIMD simulation. b The calculated number of hydrogen bonds over the dopant site of WOC and Ni-WOC models, respectively. c The calculated radial distribution functions (RDFs) between water and surface dopant sites of WOC and Ni-WOC models, respectively. g(r) indicates the RDFs, and r indicates the distances between water molecules and dopant sites. d Time-resolved ATR-SEIRAS spectra of water structure collected at 2.0 V versus Ag/AgCl on WOC and Ni-WOC-based anode, respectively. e, f Potential-dependent DRT spectra of WOC and Ni-WOC-based anode, respectively. g Schematic illustration of oxygen diffusion from bulk liquid to Ni-WOC surface through the broken H-bond network caused by Ni heteroatoms.
The loosened water network may benefit the penetration of water molecules through the Helmholtz layer to the Ni-WOC surface48,49, as experimentally confirmed by ATR-SEIRAS analysis (Fig. 4d). At a constant working potential of 2.0 V vs. Ag/AgCl, the non-hydrogen-bonded water signal28,47,50,51 (at 3580 cm–1) predominates in the interfacial water structure over the Ni-WOC-based anode throughout the reaction period (Fig. 4d). In contrast, on the WOC-based anode, 2-coordinated hydrogen-bonded and 4-coordinated hydrogen-bonded water signals28,47,50,51 (at around 3220 cm–1 and 3400 cm–1) dominate during the reaction (Fig. 4d). The increased ratio of non-hydrogen-bonded water molecules over the Ni-WOC-based anode indicates an efficient diffusion process. It is noteworthy that, with a fixed and low concentration (0.05 M) of ClO4– in all the experiments, the ATR-SEIRAS results (Fig. S16) reveal negligible ClO4– signals52,53 in the region around 1100 cm–1 on both WOC and Ni-WOC catalyst surfaces throughout the reaction period. To further assess the universality of Ni heteroatoms in regulating the interfacial water structure, we conducted in situ ATR-SEIRAS experiments in electrolytes containing different anions, including SO42– and HPO42– (Fig. S17). The population of weakly hydrogen-bonded water molecules on the Ni-WOC-based electrode surface increased significantly by factors of 1.63, 1.43 and 1.56 in LiClO4, K2HPO4 and K2SO4 electrolytes, respectively, as compared to those on the WOC-based electrode (Fig. S18). These results further confirm the dominant role of Ni dopants in regulating the interfacial water structure, rather than the influence of anions. More importantly, potential-dependent distribution of relaxation times (DRT) spectra (Fig. 4e-f and S19) directly demonstrated the depressed diffusion resistance of oxygen species54,55 from 392.8 Ω (at 0.8 V vs. Ag/AgCl) over the WOC-based anode (Table S4) to 336.1 Ω over the Ni-WOC-based anode. All these results unveil the critical role of Ni heteroatoms in breaking the originally dense interfacial H-bond network over the Ni-WOC surface and ensuring a fast oxygen diffusion process from electrolyte to adsorption sites on Ni-WOC, as depicted in Fig. 4g.
Mechanism of oxygen transfer over Ni-activated W–O covalency
We then investigated the role of Ni in modulating the active sites for pre-adsorption and activation of water molecules transferred from the electrolyte to the Ni-WOC surface. Density functional theory (DFT) calculation results (Figs. S20–22, Fig. 5a, b, and Supplementary Data 5-6) reveal an upward shift in the d-band center of metal sites (Fig. S20) from –1.959 eV (WOC) to –1.902 eV (Ni-WOC) upon Ni doping. The Bader charge (Fig. S21a–c) and differential charge density analysis results (Fig. S21d) further demonstrate the role of the Ni heteroatom as an electron acceptor to attract 0.22 e– from the adjacent WOC site (1 in Fig. 5a). Accordingly, the W (1 in Fig. 5a) atom adjacent to the Ni atom of the Ni-WOC model has the strongest coupling interaction with the pre-adsorbed water molecule with an adsorption energy of –0.921 eV (Fig. 5b and Fig. S22). The RDF between the claimed W1 atom and water molecules (Fig. S23) also confirms the hydrophilic nature of this site with a W–O distance of 2.25 Å, consistent with the calculated adsorption energy (Fig. 5b). It should be noted that the Ni site is not a favorable absorption site for water molecules with an adsorption energy of 0.085 eV, matching well with the trend of water affinity revealed by the AIMD simulation results (Fig. 4a–c).
a Schematic illustration of absorbed water at site 1. b The adsorption energy of water at Ni and surrounding W sites of the Ni-WOC model. c, d The mass spectra of products obtained by using D2O on Ni-WOC-based anodes and using H2O on 18O-labeled Ni-WOC-based anodes, respectively. e–g Potential-dependent in situ ATR-SEIRAS spectra of oxygen transfer process from 0.2 V to 2.4 V (vs. Ag/AgCl) on WOC-based and Ni-WOC-based anodes, respectively. h–j Potentiostatic in situ ATR-SEIRAS spectra of oxygen transfer process before and after the addition of ethylbenzene at 2.0 V (vs. Ag/AgCl) on Ni-WOC-based anodes. k The Gibbs free energy profile and schematic illustration of OER pathways on WOC and Ni-WOC model, and oxygen transfer pathway on Ni-WOC model.
We further investigated the possible active oxygen species that might be derived from water for the subsequent oxygenation of C(sp3)–H bonds. The isotope reaction using D2O as the oxygen source (Fig. 5c and S24a-b) did not provide deuterated products even with a reaction time of 4 h, excluding the direct participation of hydroxide species (*OH) or oxyhydroxide species (*OOH) during the oxygen transfer process. Another control reaction using 18O-labeled Ni-WOC as the anode electrocatalyst did not give detectable amounts of 18O-labeled products (Fig. 5d and S24c, d), speaking for the absorbed oxygen (*O) from water activation rather than the lattice oxygen (Olat) as the majority of the oxygen species for the oxygen transfer process over the Ni-WOC anode. This result that *O plays as an active species for inert bonds activation is consistent with previously reported cases18,56.
After determining the adsorption site and the forms of possible active oxygen species, we turned our focus to the following coactivation of oxygen species and C(sp3)–H bonds on the Ni-WOC anode. The gradually enhanced ATR-SEIRAS signals (Fig. 5e) of hydrated tungstate57 (centered at 1746 cm–1), distinct from regular water bending modes (Fig. S25), observed on Ni-WOC anode at ever-enlarged working potentials higher than 1.0 V (vs. Ag/AgCl) demonstrated a more favorable diffusion process of oxygen species to Ni-WOC anode surface as compared to bare WOC anode. With a similar trend of potential induced enhancement on the pre-adsorption of ethylbenzene58,59 (Fig. 5f), the Ni-WOC anode directly triggered the transfer of oxygen to ethylbenzene via the formation of W–O–C bridge according to a more pronounced ATR-SEIRAS signal at around 1240 cm–1 of ethyl α-C bonded to metal through oxygen60 (Fig. 5g) on the Ni-WOC anode. The potential-dependent blue shift of the W–O–C signal from 1240 cm–1 at 1.0 V (vs. Ag/AgCl) to 1246 cm–1 at 2.4 V (vs. Ag/AgCl) further indicates an intensive electrocatalyzed oxygen transfer process, which contrasts sharply with rather weak W–O–C and unactivated –CH2– (1214 cm–1) signals58,59 on the WOC anode. To further validate the proposed oxygen transfer process, we conducted potentiostatic in situ ATR-SEIRAS experiments at 2.0 V versus Ag/AgCl both before and after the addition of ethylbenzene (Fig. 5h–j) to track the emergence of key intermediates. The ever-existing signals of hydrated tungstate57 at 1746 cm–1 both before and after the addition of ethylbenzene (Fig. 5h) indicate the strong affinity of W sites for water, facilitating subsequent water activation. Additionally, an emergent band corresponding to the C–C stretching mode of ethylbenzene58,59 near 1040 cm–1 after the addition of ethylbenzene (Fig. 5j) further confirms the efficient adsorption of ethylbenzene on Ni-WOC. Following the facile adsorption of water and ethylbenzene, the emerged signals at the region near 1246 cm–1 after adding ethylbenzene (Fig. 5i) have indicated a C–H activation process through the formation of W–O–C structure60. Collectively, the Ni heteroatoms boost facile water adsorption (Fig. 5e, h) on neighboring W sites and thus promote C–H activation by forming W–O–C intermediate (Fig. 5g and Fig. 5j) on the Ni-WOC-based electrode. These results indicate that the oxygen transfer process is induced by the coactivation of water and C–H bonds boosted by Ni heteroatoms, while the formation of the W–O–C structure plays a key intermediate role in the oxygen transfer process.
Theoretically, we further investigated the possible role of Ni-WOC in regulating reaction pathways from OER to oxygenation of C(sp3)–H bonds. As revealed by DFT calculations (Fig. 5k, S26–27, and Table S5, 6), the energy barrier of each step for the formation of *O as active oxygen species on the Ni-WOC model has decreased dramatically. The DRT spectra also indicate improved reaction kinetics, corresponding to a timescale of 10–3 s at the cathode upon introducing Ni dopants (Fig. S28 and Table S7). The kinetic impedance is reduced to 8.0 Ω for the Ni-WOC catalyst, compared to 14.0 Ω for the WOC catalyst (Table S7). The integration of theoretical and experimental results highlights the crucial role of Ni dopants in efficiently activating water molecules for generating active oxygen species from an intrinsic kinetics perspective. However, as the DFT calculation suggested, the ΔG value for the hydroxylation step from *O to *OOH as the rate-determining step (RDS) for OER has increased from 3.02 eV on the WOC model to 3.33 eV on the Ni-WOC model. Experimentally, the suppression of OER by Ni doping is directly evidenced by the decrease in FE for O2 from 55.2% on the WOC-based electrode to 34.1% on the Ni-WOC-based electrode (Fig. S29). More importantly, the ΔG value for the adsorption of ethylbenzene with the formation of *O on the Ni-WOC model is as low as 2.83 eV, making it more favorable for the oxygenation of C(sp3)–H bonds rather than the formation of *OOH from *O in OER. The displacement of OER by ethylbenzene oxidation is further experimentally manifested by linear sweep voltammetry curves (Fig. S30a), where both the appearance of a distinct oxidation peak at 1.72 V (vs. Ag/AgCl) and a simultaneous decrease in the OER current after adding ethylbenzene into the original water-based electrolyte were observed. Additionally, the isotope kinetic effect (KIE) was also tested to reveal the changes of the rate-determining step (Fig. S30b–d). The calculated KIE value (\({j}_{{\mbox{H}}}/{j}_{{\mbox{D}}}\), \({j}_{{\mbox{H}}}\): the current density measured in H2O-based electrolyte, and \({j}_{{\mbox{D}}}\): the current density measured in D2O-based electrolyte) at 1.72 V (vs. Ag/AgCl) decreases from 1.40 (OER alone) to 1.22 (with ethylbenzene), indicating that the isotopic effect of water oxidation becomes less dominant when C–H activation and oxidation are involved. All these results have clearly elucidated the essential role of Ni heteroatoms in boosting the coactivation of water and C(sp3)–H bonds while suppressing the undesired OER process (from *O to *OOH) from an intrinsic kinetics perspective, well explaining the high FE values for producing oxygenated compounds as supported by both theoretical and experimental evidence.
Performance of the electrocatalytic oxygen transfer reaction
Taken together, the Ni heteroatoms regulate interfacial water structure for fast oxygen diffusion from electrolyte to Ni-WOC surface, simultaneously boosting the coactivation of oxygen species and C(sp3)–H bonds on W–O covalency sites with inhibited OER process. In this way, the Ni-WOC-based anode has provided satisfactory FEs ranging from 43.2% to 64.8% in a general scope of benzylic C(sp3)–H bonds (Fig. 6a), including secondary (Ethylbenzene, Propylbenzene) and tertiary (iso-Propylbenzene, Diphenylmethane) C(sp3)–H bonds with electron-withdrawing (3-Ethyl-Fluorobenzenen) or donating (o-Ethyltoluene, p-Ethyltoluene) groups. It should be noted that the current substrate scope is still limited to benzylic C(sp3)–H bonds, and the near-unity regioselectivity is governed by the inherent reactivity of the benzylic site itself, requiring further research to expand this promising oxygen transfer system.
a Yields and Faradaic efficiencies for oxygenated products of different kinds of benzylic C(sp3)–H bonds over Ni-WOC-based anode. b The durability test for the Ni-WOC-based anode was performed with each cycle of 2 h. c Oxygenation performance of the electrocatalytic systems for benzylic oxidation over Ni-WOC-based anode. Reaction conditions: 2.0 V versus Ag/AgCl, with a catalyst loading of 1.5 mg/cm2.
Additionally, Ni-WOC-based anode has provided durable capability for oxygen transfer from water to C(sp3)–H bonds with high FEs (from 55.2% to 58.4%) for oxygenated products (Fig. 6b) without obvious decay in catalytic performance for more than 6 circles of reuse. More importantly, after a long-term electrolysis, the Ni-WOC structure remains durable with abundant amorphous features revealed by TEM (Fig. S31a, b) and XRD (Fig. S32). XPS analysis indicates a raised Ni2+/Ni0 ratio from 0.15 to 10.11 and W6+/W0 ratio from 0.37 to 21.22 (Fig. S33a, b and Table S3), along with the significant emergence of lattice oxygen (Fig. S33c). The EDS mapping (Fig. S31e, f) confirms the homogeneous distribution of Ni atoms over the whole support of the spent catalyst. Quantitative EDS spectra (Fig. S31d and Table S8) and the inductively coupled plasma atomic emission spectroscopy (ICP-AES) (Table S8) analysis results further demonstrate the consistent Ni/W atomic ratios before (0.095 via ICP-AES) and after (0.086 via ICP-AES and 0.107 via EDS) long-term uses, speaking for the co-retention of Ni-W complex during the long-term electrolysis.
Besides the excellent capability and durability, the Ni-WOC-based electrocatalytic devices utilizing water as an oxygen source for additive-free oxygenation of inert C(sp3)–H bonds are competitive when compared with the state-of-the-art electrocatalytic systems reported in the literature15,17,61,62,63 (Fig. 6c and Table S2). Taking ethylbenzene as an example, the Ni-WOC-based anode has provided a satisfactory FE up to 58.4% (Fig. 6c and Table S2). And a continuous oxygen transfer process for a high yield up to 90% has been achieved on Ni-WOC-based anode, which is among the competitive ones in the currently reported cases (Fig. 6c and Table S2). Accordingly, the Ni-WOC-based anode has exhibited an energy efficiency (ηE) of 10.9 mol/kW·h (Fig. 6c and Table S2). Additionally, the additive-free feature of the Ni-WOC anode has significantly reduced the costs associated with material and product purification (Fig. 6c and Table S2).
Discussion
In summary, we have demonstrated the essential role of Ni-activated W–O covalency sites in boosting the oxygen transfer process from water to benzylic C(sp3)–H bonds to produce K/A oil. Ni heteroatoms modulate the electronic deficiency of neighboring W sites, resulting in an enhanced oxygen transfer process which improves both mass transfer and intrinsic kinetics. Specifically, Ni heteroatoms disrupt the water structure at the anode/electrolyte interface, breaking the dense hydrogen bond network and facilitating the rapid diffusion of water molecules to the W–O covalency sites of Ni-WOC. Additionally, the coactivation of oxygen species and C(sp3)–H bonds is enhanced at the W–O covalent sites adjacent to Ni heteroatoms, further promoting the effective oxygenation process. More importantly, the undesired OER process is inhibited by Ni heteroatoms via a regulated reaction pathway, thereby providing satisfactory FEs for effective C(sp3)–H oxygenation reactions using water as an oxygen source in a general scope of substrates. This work not only highlights the importance of catalyst-tuned interfacial water structure for the oxidation of organic compounds but also provides important insights into designing electrocatalytic systems for inert C–H oxidation using water as the oxygen source.
Methods
Chemicals and materials
Ethylbenzene (C8H10, 99.0%), Water-18O (H218O, 98.0%), Deuterium Oxide (D2O, 99%), Propylbenzene (C9H12, 99%), Sulfolane (C4H8O2S, 99.0%), p-Ethyltoluene (C9H12, 98%), and o-Ethyltoluene (C9H12, 98%) were purchased from Adamas. iso-Propylbenzene (C9H12, 99%) and 1-Ethyl-3-Fluorobenzenen (C8H9F, 98%) were purchased from Macklin. Ammonium Metatungstate ((NH4)6H2W12O40·xH2O, 99.5%), Nickel (II) Nitrate Hexahydrate (Ni(NO3)2·6H2O, 99%), Urea (CH4N2O, 99.5%),1, 4-Benzenedicarboxylic Acid (C8H6O4, 99%), Lithium Perchlorate (LiClO4, 99.95%), Triethylene Diamine (C6H12N2, 98%), and N, N-Dimethylformamide (C3H7NO, 99.5%) were purchased from Aladdin. Anisole (C7H8O, 99.0%) and Diphenylmethane (C13H12, 99.0%) were purchased from TCI. Potassium Hydroxide (KOH, 95%) and Ethanol (C2H5OH, 99.8%) were purchased from Greagent. Dicyandiamide (C2H4N4, 99.5%) was purchased from Acros Organics. Nafion solution (5%) was purchased from DuPont. Nitrogen (N2, 99.999%) and Argon (Ar, 99.999%) were purchased from Air Liquide. Carbon Cloth (W0S1002) was purchased from CeTech. All chemicals and materials were used as received without further purification.
Synthesis of Ni/W2C and W2C pre-catalysts
Synthesis of Ni/W2C
Typically, 0.50 g ammonium metatungstate and 10.00 g dicyandiamide were mixed with different contents of nickel (II) nitrate hexahydrate (23.60, 47.20, 94.40 mg) in 50 mL deionized water. The mixture was heated at 80 °C under continuous stirring to remove the water. The obtained homogeneous solid mixture with a light green color was ground into powder and transferred into a cylindrical crucible with a lid. The crucible with the precursor was heated at 1000 °C for 2 h with a heating rate of 2.5 °C min−1 under a highly pure nitrogen gas atmosphere. The final product was collected after naturally cooling down and denoted as Ni0.8/W2C (with 23.60 mg nickel nitrate hexahydrate), Ni/W2C (with 47.20 mg nickel nitrate hexahydrate), and Ni3.1/W2C (with 94.40 mg nickel nitrate hexahydrate), respectively.
Synthesis of W2C
The synthesis of W2C follows the same procedure as that of Ni/W2C, with the exception that nickel nitrate hexahydrate is omitted from the precursor.
Synthesis of c-WO3 catalysts
The crystalline WO3 was synthesized by heating the monoclinic WO3·H2O, which was obtained by the Freedman method. Typically, 10 mL of 1 M Na2WO4·2H2O was added to 90 mL of 3 M HCl. The as-formed yellow mixture was stirred overnight at 300 rpm at room temperature. The sample obtained was collected via vacuum filtration and washed with deionized water until the pH of the rinsed solution reached approximately 6. The obtained yellow powder was dried at 60 °C overnight to get the monoclinic WO3·H2O. The monoclinic WO3 was obtained by heating WO3·H2O at 350 °C for 5 h in air with a heating rate of 5 °C min−1. The final product collected after naturally cooling down was denoted as c-WO3. The XRD patterns of the synthesized WO3·H2O and WO3 samples have demonstrated their crystalline structure, as shown in Fig. S2c, d.
Synthesis of Ni/NC catalysts
Briefly, the urea was heated to 550 °C for 4 h in air with the heating rate of 2.3 °C/min to form the polymeric carbon nitride (PCN). 6.19 g PCN powder was mixed with a homogeneous mixture of 48.7 mg nickel (II) nitrate hexahydrate, 0.91 g 1,4-benzenedicarboxylic acid and 0.64 g triethylene diamine in N, N-dimethylformamide (DMF) solution. The resulting solid powder after removing solvent was heated to 900 °C for 1 h with a heating rate of 1.5 °C/min under an N2-protected atmosphere. The obtained black solid samples were denoted as Ni/NC.
Preparation of working electrodes
The catalyst ink was prepared by mixing 10 mg corresponding catalyst and a solution containing 300 μL Nafion solution (5%), 700 μL deionized water, and 1400 μL Ethanol. The obtained mixture was sonicated for 2 h to get a homogeneously dispersed ink.
The working electrode was prepared by homogeneously dipping specific volumes of the ink onto a 2×2 cm2 carbon cloth, followed by drying at 120 °C for 1 h. The as-obtained electrode was used for further electrocatalytic tests.
Electrocatalytic oxygen transfer reaction tests
All the electrocatalytic experiments were performed in a homemade undivided cell with a three-electrode system (Fig. S33) on a potentiostat (CHI 660E, Shanghai CH Instruments Company). Working electrodes (the anode) were composed of catalysts supported on a carbon cloth. A Titanium (Ti) mesh with a size of 2 × 2 cm2 and an Ag/AgCl electrode was employed as the cathode and reference electrode, respectively. For the oxygen transfer reaction, the potentiostatic test was conducted in an Ar-saturated electrolyte composed of water/sulfolane solution containing 25 mM ethylbenzene and 50 mM LiClO4. The sulfolane was used as a bipolar solvent to improve the homogeneous dispersion of the ethylbenzene phase in the electrolyte. The influence of the volume ratio of sulfolane was shown in Fig. S8.
The electrocatalytic continuous oxygen transfer reaction was conducted continuously for 48 h without intervals. The electrocatalytic stability tests of electrodes for oxygen transfer reaction were evaluated via six repeated cycles at the same working potential, with each cycle of 2 h.
The generated anode products were analyzed by GC-MS (Shimadzu QP2010SE equipped with SH-Rxi-5Sil MS column) to determine the components of products and calculate the yield and Faradaic efficiency.
The Faradaic efficiency (FE) was calculated as follows:
where Ni (mole) is the number of moles for the specific product (alcohols and ketones); n is the number of electrons exchanged for product formation, which is 2 for alcohols and 4 for ketones; F is the Faradaic constant of 96485 C mol−1; Q is the measured passed charge.
The energy efficiency (ηE) was calculated as follows:
where N (mole) is the number of moles for the conversed substrates; W (kW·h) is the consumed electronic work during the electrocatalytic process.
Electrochemically active surface area measurement
The electrochemically active surface area (ECSA) of the catalysts, both before and after the reconstruction, was measured in 0.1 M KOH electrolyte using the cyclic voltammetry method. Working electrodes (the anode) were composed of catalysts supported on a 2 × 2 cm2 carbon cloth. A Titanium (Ti) mesh with a size of 2 × 2 cm2 and an Ag/AgCl electrode was employed as the cathode and reference electrode, respectively. For the fresh catalysts, the cyclic voltammetry was performed by scanning from 0.816 V (vs. Ag/AgCl) to 0.916 V (vs. Ag/AgCl) at scanning rates of 2, 4, 6, 8, 10, and 15 mV·s-1. The ECSA measurement for reconstructed samples follows the same procedure, with the exception that the working electrode was changed to a reconstructed one. The ECSA was calculated as follows:
where Cdl is the double layer capacitance measured from cyclic voltammetry method; Cs is the reference capacitance.
In situ Raman spectroscopy studies
The in situ Raman measurements were conducted on a Renishaw inVia Qontor Raman microscope using a custom-designed in situ electrochemical cell (Fig. S34a). Specifically, the measurements were performed using a 532 nm semiconductor laser at 2.5 mW power and a 50× microscope objective, with frequencies calibrated against silicon wafers beforehand. The experiments utilized an undivided three-electrode cell equipped with an embedded quartz window. The electrode configuration consisted of a platinum wire counter electrode, an Ag/AgCl reference electrode, and a working electrode made from carbon cloth loaded with catalyst at 1 mg cm−2. Water-sulfolane mixture (volume ratio of 3:1) with 25 mM ethylbenzene and 50 mM LiClO4 was used as electrolyte. A potentiostat (Vertex.C, Ivium Technologies B.V.) was used to control the potential during the Raman test. Each spectrum in the in situ potentiodynamic Raman test under different applied bias ranging from 0.4 V to 1.8 V (vs. Ag/AgCl) was collected for approximately 10 min every 0.2 V.
In situ infrared absorption spectroscopy studies
The attenuated total reflection surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) measurements were run on a Nicolet iS50 FTIR spectrometer equipped with an HgCdTe (MCT) detector using a custom-designed in situ electrochemical cell (Fig. S34b). All measurements are conducted at the Kretschmann configuration, incorporating an Au film-coated Si window with catalyst ink dropped on it. The Au film functions both as a current collector and a SEIRA-active substrate, enabling in situ analysis of the anode/electrolyte interfaces close to the catalyst with high sensitivity. Water-sulfolane mixture (volume ratio of 3:1) with 25 mM ethylbenzene and 50 mM LiClO4 was used as electrolyte, with an Ag/AgCl electrode and a Pt mesh as the reference electrode and cathode, respectively. The IR spectra were collected with p-polarized IR radiation at the spectral resolution of 8 cm−1. The incidence angle is ca. 60°. All spectra were shown in the absorbance unit as −log(I/I0), where I and I0 represent the intensities of the reflected radiation for the sample and reference single-beam spectra, respectively.
In situ potentiostatic ATR-SEIRAS spectra for the surface water structure were collected continuously at working conditions (2.0 V vs. Ag/AgCl) every 2 min. The whole experimental period is 30 min.
In situ potentiodynamic ATR-SEIRAS spectra for the oxygen transfer process at a scan rate of 5 mV·s−1 were continuously collected every 200 mV at the cell voltage from 0.2 to 2.4 V (vs. Ag/AgCl).
In situ potentiostatic ATR-SEIRAS spectra for the oxygen transfer process at a constant working potential of 2.0 V vs. Ag/AgCl were continuously collected every 1 min. The ethylbenzene was added to the in situ electrochemical cell between 9 and 10 min.
Each single-beam spectrum was averaged from 32 scans, corresponding to a period of ca. 14 s. The concentration of LiClO4 (50 mM) was fixed throughout all experiments to rule out the potential influence of anions on water structure.
Distribution of relaxation times analysis
Potentiostatic electrochemical impedance spectra (EIS) were collected in the range from 0.1 Hz to 1 MHz every 0.2 V at the bias ranging from 0.4 to 1.6 V (vs. Ag/AgCl) (see Fig. S19). To determine the oxygen species diffusion process over the Ni-WOC anode, 1 M KOH was used as the electrolyte. The distribution of relaxation times64,65,66 (DRT) is used to analyze the EIS data.
DRT impedance, ZDRT(f), at a frequency f, can be expressed as
Where R∞, τ, γ(lnτ) and f are the ohmic resistance, relaxation time, distribution function of relaxation time, and frequency, respectively. Moreover, polarization resistance, Rpol, is computed by:
Density functional theory calculations
Density functional theory (DFT) calculations were conducted utilizing the Vienna Ab Initio Simulation Package (VASP)67,68. The generalized gradient approximation (GGA) with the Perdew-Burke-Ernzerh (PBE) of exchange-correlation functional69 was employed. The DFT-D3 method70,71 was employed to incorporate van der Waals interactions. A plane-wave expansion with a kinetic energy cutoff of 450 eV was employed. The convergence criteria for energy and force were set to 10−5 eV and 0.02 eV Å−1, respectively. K-points were set to 3 × 3 × 1 for geometry optimization and energy calculation. A vacuum layer with a thickness of 15 Å along the z-direction was applied to avoid unreasonable interactions among periodical images along the z-direction.
The amorphous W–O covalency model was constructed by using ab initio molecular dynamics. Briefly, the originally crystalline Ni-doped WO3 model was treated using ab initio molecular dynamics by VASP with all layers fully relaxed. The system was maintained at 600 K in an NVT ensemble framework to obtain the amorphous structure. The total duration of the AIMD simulation reached 5 ps.
The Gibbs free energy change for each step of the oxidation reaction was calculated as follows:
where ΔE, ΔZPE, and ΔS are the changes in the reaction energy, zero-point energy, and entropy, respectively. Each step and the corresponding ∆G values of the OER and oxygenation pathway are listed in Tables S5 and6.
Ab initio molecular dynamics simulation
The ab initio molecular dynamics (AIMD) simulation for the structure of interfacial water over the Ni-WOC and WOC models was carried out using the open source CP2K/Quickstep package72 based on a hybrid Gaussian plane wave (GPW) scheme. The orbitals are described by an atom-centered Gaussian-type basis set. An auxiliary plane wave basis set with a cutoff energy of 400 Ry is used to re-expand the electron density in the reciprocal space. Perdew-Burke-Ernzerhof (PBE) functional69 with DFT-D3 dispersion correction70,71 was used. The core electrons were represented by analytic Goedecker-Teter-Hutter (GTH) pseudopotentials73,74. For valence electrons, the Gaussian basis sets were double-ζ basis functions with one set of polarization functions (DZVP)75.
The initial model of catalyst/water interfaces was constructed using the previously obtained amorphous W–O covalency model for DFT calculations. The simulation cell was filled with 116 water molecules and a vacuum layer of approximately 20 Å in z-direction. The entire simulation cell has a size of 15.44×15.44×46.00 Å3 as shown in Fig. S15.
All AIMD simulations were performed in the NVT ensemble, with Nosé–Hoover chain thermostat76 to keep a constant temperature of 298.15 K. A time step of 0.5 fs was adopted for the Velocity-Verlet algorithm77. The structural relaxation process lasted for 2 ps to obtain equilibrium configurations, followed by a simulation of 10 ps for the production process used for the final analysis. The simulation was conducted in periodic boundary conditions.
The hydrogen bond analysis follows a typical criterion of a 30° angle cutoff and a 3.5 Å bond length cutoff. To ensure the accuracy of the simulation, all presented results are statistical averages calculated over a 10 ps simulation, corresponding to 20,000 steps. This extensive sampling effectively filters out any anomalous simulation artifacts. Additionally, simulations of both the Ni-WOC and WOC models were conducted under identical conditions, providing clear evidence of the disruption of hydrogen bond networks near the doped Ni sites.
Structural characterization
Aberration-corrected high-angle annular dark field scanning transmission (AC-HAADF-STEM) images were recorded on a Hitachi HF5000 scanning/transmission electron microscope at an acceleration voltage of 200 kV with a probe spherical aberration corrector. The corresponding Energy Dispersive X-ray Spectra (EDS) were collected by an INCA X-Max 80 TEM EDS spectrometer. Transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM) and HAADF-STEM images were recorded by a Talos F200X G2 microscope with an acceleration voltage of 200 kV. Powder X-ray diffraction (XRD) patterns were recorded on a Rigaku Miniflex 600 C X-ray diffractometer with Cu Kα radiation (λ = 1.5418 Å) and operated at a scan rate of 5° min−1. X-ray photoelectron spectroscopy (XPS) experiments were performed using a Kratos Axis Ultra DLD spectrometer with monochromated Al Kα radiation. The inductively coupled plasma atomic emission spectroscopy (ICP-AES) measurement was conducted on an iCAP6300 spectrometer for metal element analysis. The Raman spectroscopy was conducted on a Renishaw inVia Qontor Raman microscope with the laser wavelength of 532 nm.
Data availability
All the relevant data are included in this paper and its Supplementary Information. The data that support the findings of this study are available from the corresponding authors upon request. Source data are provided with this paper.
References
Haber, J. in Handbook of Heterogeneous Catalysis. 3359–3384 (John Wiley & Sons, Ltd, 2008).
Zheng, R., Liu, Z., Wang, Y., Xie, Z. & He, M. The future of green energy and chemicals: Rational design of catalysis routes. Joule 6, 1148–1159 (2022).
Leow, W. R. et al. Electrified hydrocarbon-to-oxygenates coupled to hydrogen evolution for efficient greenhouse gas mitigation. Nat. Commun. 14, 1954 (2023).
Cullen, L., Meng, F., Lupton, R. & Cullen, J. M. Reducing uncertainties in greenhouse gas emissions from chemical production. Nat. Chem. Eng. 1, 311–322 (2024).
Qin, Y. et al. Adjacent-ligand tuning of atomically precise Cu−Pd sites enables efficient methanol electrooxidation with a CO-free pathway. Angew. Chem. Int. Ed. 64, e202420817 (2025).
Shi, R. et al. Electrochemical oxidation of concentrated benzyl alcohol to high-purity benzaldehyde via superwetting organic-solid-water interfaces. Sci. Adv. 10, eadn0947 (2024).
Tian, B. et al. Parameterization and quantification of two key operando physio-chemical descriptors for water-assisted electro-catalytic organic oxidation. Nat. Commun. 15, 10145 (2024).
Li, Y. et al. Accelerated selective electrooxidation of ethylene glycol and inhibition of C–C dissociation facilitated by surficial oxidation on hollowed PtAg nanostructures via in situ dynamic evolution. JACS Au 5, 714–726 (2025).
Shi, K., Si, D., Teng, X., Chen, L. & Shi, J. Pd/NiMoO4/NF electrocatalysts for the efficient and ultra-stable synthesis and electrolyte-assisted extraction of glycolate. Nat. Commun. 15, 2899 (2024).
Zhang, N. et al. Electrochemical oxidation of 5-hydroxymethylfurfural on nickel nitride/carbon nanosheets: reaction pathway determined by in situ sum frequency generation vibrational spectroscopy. Angew. Chem. Int. Ed. 58, 15895–15903 (2019).
Gu, K. et al. Defect-rich high-entropy oxide nanosheets for efficient 5-hydroxymethylfurfural electrooxidation. Angew. Chem. Int. Ed. 60, 20253–20258 (2021).
Pei, A. et al. Enhanced electrocatalytic biomass oxidation at low voltage by Ni2+-O-Pd interfaces. Nat. Commun. 15, 5899 (2024).
Zhang, P. et al. Paired electrocatalytic oxygenation and hydrogenation of organic substrates with water as the oxygen and hydrogen source. Angew. Chem. Int. Ed. 58, 9155–9159 (2019).
Chi, H. et al. Electrosynthesis of ethylene glycol from biomass glycerol. Nat. Commun. 16, 979 (2025).
Sun, Y. et al. Highly selective electrocatalytic oxidation of benzyl C–H using water as safe and sustainable oxygen source. Green Chem 22, 7543–7551 (2020).
Li, Z. et al. Electrocatalytic ethylbenzene valorization using a polyoxometalate@covalent triazine framework with water as the oxygen source. Chem. Commun. 57, 7430–7433 (2021).
Seo, B. et al. Electrochemical oxidation of toluene with controlled selectivity: The effect of carbon anode. Appl. Surf. Sci. 534, 147517 (2020).
Liu, C., Chen, F., Zhao, B.-H., Wu, Y. & Zhang, B. Electrochemical hydrogenation and oxidation of organic species involving water. Nat. Rev. Chem. 8, 277–293 (2024).
Leng, B.-L., Lin, X., Chen, J.-S. & Li, X.-H. Electrocatalytic water-to-oxygenates conversion: redox-mediated versus direct oxygen transfer. Chem. Commun. 60, 7523–7534 (2024).
Kawamata, Y. et al. Scalable, electrochemical oxidation of unactivated C–H bonds. J. Am. Chem. Soc. 139, 7448–7451 (2017).
Lin, X. et al. Direct oxygen transfer from H2O to cyclooctene over electron-rich RuO2 nanocrystals for epoxidation and hydrogen evolution. Angew. Chem. Int. Ed. 61, e202207108 (2022).
Dorchies, F. et al. Controlling the hydrophilicity of the electrochemical interface to modulate the oxygen-atom transfer in electrocatalytic epoxidation reactions. J. Am. Chem. Soc. 144, 22734–22746 (2022).
Chung, M., Jin, K., Zeng, J. S., Ton, T. N. & Manthiram, K. Tuning single-atom dopants on manganese oxide for selective electrocatalytic cyclooctene epoxidation. J. Am. Chem. Soc. 144, 17416–17422 (2022).
Chung, M. et al. Direct propylene epoxidation via water activation over Pd-Pt electrocatalysts. Science 383, 49–55 (2024).
Leow, W. R. et al. Chloride-mediated selective electrosynthesis of ethylene and propylene oxides at high current density. Science 368, 1228–1233 (2020).
Gao, Y. et al. Membrane-free electrosynthesis of epichlorohydrins mediated by bromine radicals over nanotips. J. Am. Chem. Soc. 146, 714–722 (2024).
Yao, J. et al. Interfacial hydrogen-bond network regulation tuned water dissociation enables selective chlorination of alkenes. J. Am. Chem. Soc. 147, 8024–8031 (2025).
Yan, M., Yang, R., Liu, C., Gao, Y. & Zhang, B. In situ probing the anion-widened anodic electric double layer for enhanced faradaic efficiency of chlorine-involved reactions. J. Am. Chem. Soc. 147, 6698–6706 (2025).
Lin, X. et al. Electrochemical activation of C–H by electron-deficient W2C nanocrystals for simultaneous alkoxylation and hydrogen evolution. Nat. Commun. 12, 3882 (2021).
Pol, S. V., Pol, V. G. & Gedanken, A. Synthesis of WC nanotubes. Adv. Mater. 18, 2023–2027 (2006).
Yang, B. et al. Optimization of hydrogen adsorption on W2C by late transition metal doping for efficient hydrogen evolution catalysis. Mater. Today Nano 23, 100350 (2023).
Yan, J. et al. Single atom tungsten doped ultrathin α-Ni(OH)2 for enhanced electrocatalytic water oxidation. Nat. Commun. 10, 2149 (2019).
Goto, Y. et al. Formation of Ni3C nanocrystals by thermolysis of nickel acetylacetonate in oleylamine: characterization using hard X-ray photoelectron spectroscopy. Chem. Mater. 20, 4156–4160 (2008).
Bayer, B. C. et al. In situ observations of phase transitions in metastable nickel (carbide)/carbon nanocomposites. J. Phys. Chem. C 120, 22571–22584 (2016).
Cui, Y. et al. Tungsten oxide/carbide surface heterojunction catalyst with high hydrogen evolution activity. ACS Energy Lett 5, 3560–3568 (2020).
Li, H. et al. Operando unveiling of hydrogen spillover mechanisms on tungsten oxide surfaces. J. Am. Chem. Soc. 147, 6472–6479 (2025).
Xu, K. et al. Catalytically efficient Ni-NiOx-Y2O3 interface for medium temperature water-gas shift reaction. Nat. Commun. 13, 2443 (2022).
Mansour, A. N. Characterization of NiO by XPS. Surf. Sci. Spectra 3, 231–238 (1994).
Duan, M. et al. Synergizing inter and intraband transitions in defective tungsten oxide for efficient photocatalytic alcohol dehydration to alkenes. JACS Au 2, 1160–1168 (2022).
Ahmadi, M. et al. WO3 nano-ribbons: their phase transformation from tungstite (WO3·H2O) to tungsten oxide (WO3). J. Mater. Sci. 49, 5899–5909 (2014).
Liu, F., Chen, X., Xia, Q., Tian, L. & Chen, X. Ultrathin tungsten oxide nanowires: oleylamine assisted nonhydrolytic growth, oxygen vacancies and good photocatalytic properties. RSC Adv. 5, 77423–77428 (2015).
Dupin, J.-C., Gonbeau, D., Vinatier, P. & Levasseur, A. Systematic XPS studies of metal oxides, hydroxides and peroxides. Phys. Chem. Chem. Phys. 2, 1319–1324 (2000).
Chen, Z. et al. Eutectoid-structured WC/W2C heterostructures: a new platform for long-term alkaline hydrogen evolution reaction at low overpotentials. Nano Energy 68, 104335 (2020).
Gabrusenoks, J., Veispals, A., Von Czarnowski, A. & Meiwes-Broer, K.-H. Infrared and Raman spectroscopy of WO3 and CdWO4. Electrochimica Acta 46, 2229–2231 (2001).
Daniel, M. F., Desbat, B. & Lassegues, C. Infrared and Raman study of WO3 tungsten trioxides and WO3, xH2O tungsten trioxide tydrates. J. Solid State Chem. 67, 235–247 (1987).
Gomez, A., Piskulich, Z. A., Thompson, W. H. & Laage, D. Water diffusion proceeds via a hydrogen-bond jump exchange mechanism. J. Phys. Chem. Lett. 13, 4660–4666 (2022).
Sun, Q. et al. Understanding hydrogen electrocatalysis by probing the hydrogen-bond network of water at the electrified Pt–solution interface. Nat. Energy 8, 859–869 (2023).
Yang, C., Fontaine, O., Tarascon, J.-M. & Grimaud, A. Chemical recognition of active oxygen species on the surface of oxygen evolution reaction electrocatalysts. Angew. Chem. Int. Ed. 56, 8652–8656 (2017).
Wang, H. et al. Breaking hydrogen bond network enhances oxygenic photosynthesis of photosystem II. Fundam. Res. S2667325825001190 https://doi.org/10.1016/j.fmre.2025.03.011 (2025).
Li, P. et al. Hydrogen bond network connectivity in the electric double layer dominates the kinetic pH effect in hydrogen electrocatalysis on Pt. Nat. Catal. 5, 900–911 (2022).
Chen, X. et al. Tuning the hydrogen bond network inside the Helmholtz plane for industrial hydrogen evolution. Adv. Funct. Mater. 35, 2503701 (2025).
Klassen, B., Aroca, R. & Nazri, G. A. Lithium perchlorate: Ab initio study of the structural and spectral changes associated with ion pairing. J. Phys. Chem. 100, 9334–9338 (1996).
Xuan, X., Wang, J., Tang, J., Qu, G. & Lu, J. A vibrational spectroscopic study of ion solvation in lithium perchlorate/propylene carbonate electrolyte. Phys. Chem. Liq. 39, 327–342 (2001).
Li, Y. et al. Application of distribution of relaxation times method in polymer electrolyte membrane water electrolyzer. Chem. Eng. J. 451, 138327 (2023).
Giesbrecht, P. K. & Freund, M. S. Investigation of water oxidation at IrO2 electrodes in Nafion-based membrane electrode assemblies using impedance spectroscopy and distribution of relaxation times analysis. J. Phys. Chem. C 126, 17844–17861 (2022).
Ran, P. et al. Disordered Ru–O6 octahedrons for efficient and selective electro-oxidation of sulfide to sulfoxide via boosted surface oxygen kinetics. J. Am. Chem. Soc. 147, 26254–26266 (2025).
Zaki, M. I., Mekhemer, G. A. H., Fouad, N. E. & Rabee, A. I. M. Structure–acidity correlation of supported tungsten(VI)-oxo-species: FT-IR and TPD studies of adsorbed pyridine and catalytic decomposition of 2-propanol. Appl. Surf. Sci. 308, 380–387 (2014).
Fishman, A. I., Klimovitskii, A. E., Skvortsov, A. I. & Remizov, A. B. The vibrational spectra and conformations of ethylbenzene. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 60, 843–853 (2004).
Wang, J., Qiu, X., Wang, Y., Zhang, S. & Zhang, B. Vibrational spectra and quantum calculations of ethylbenzene. Chin. J. Chem. Phys. 25, 526–532 (2012).
Tope, B. B. et al. Catalytic mechanism of the dehydrogenation of ethylbenzene over Fe–Co/Mg(Al)O derived from hydrotalcites. Appl. Catal. Gen. 407, 118–126 (2011).
Kato, S., Iwase, K., Harada, T., Nakanishi, S. & Kamiya, K. Aqueous electrochemical partial oxidation of gaseous ethylbenzene by a ru-modified covalent triazine framework. ACS Appl. Mater. Interfaces 12, 29376–29382 (2020).
Wilsey, M. K. et al. Selective toluene electrooxidation to benzyl alcohol. J. Am. Chem. Soc. 147, 36117–36135 (2025).
Xiong, P. et al. Site-selective electrooxidation of methylarenes to aromatic acetals. Nat. Commun. 11, 2706 (2020).
Ciucci, F. & Chen, C. Analysis of electrochemical impedance spectroscopy data using the distribution of relaxation times: a Bayesian and hierarchical Bayesian approach. Electrochimica Acta 167, 439–454 (2015).
Wan, T. H., Saccoccio, M., Chen, C. & Ciucci, F. Influence of the discretization methods on the distribution of relaxation times deconvolution: implementing radial basis functions with DRTtools. Electrochimica Acta 184, 483–499 (2015).
Chen, J., Quattrocchi, E., Ciucci, F. & Chen, Y. Charging processes in lithium-oxygen batteries unraveled through the lens of the distribution of relaxation times. Chem 9, 2267–2281 (2023).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
McGrath, M. J. et al. Toward a Monte Carlo program for simulating vapor–liquid phase equilibria from first principles. Comput. Phys. Commun. 169, 289–294 (2005).
Goedecker, S., Teter, M. & Hutter, J. Separable dual-space Gaussian pseudopotentials. Phys. Rev. B 54, 1703–1710 (1996).
Hartwigsen, C., Goedecker, S. & Hutter, J. Relativistic separable dual-space Gaussian pseudopotentials from H to Rn. Phys. Rev. B 58, 3641–3662 (1998).
VandeVondele, J. & Hutter, J. Gaussian basis sets for accurate calculations on molecular systems in gas and condensed phases. J. Chem. Phys. 127, 114105 (2007).
Evans, D. J. & Holian, B. L. The Nose–Hoover thermostat. J. Chem. Phys. 83, 4069–4074 (1985).
Verlet, L. Computer ‘Experiments’ on classical fluids. I. Thermodynamical properties of Lennard-Jones molecules. Phys. Rev. 159, 98–103 (1967).
Acknowledgements
This work was supported by Shanghai Science and Technology Committee (23XD1421800 for X.-H. L.), Shanghai Shuguang Program (21SG12 for X.-H. L.), State Key Laboratory of Synergistic Chem-Bio Synthesis (sklscbs202551 for X.-H. L.), and Shanghai Municipal Science and Technology Major Project (J.-S. C.).
Author information
Authors and Affiliations
Contributions
X.-H. L., B.-L. L., and X. L. designed the experiments. B.-L. L. planned and performed catalyst synthesis, conducted performance tests and analyzed data. B.-L. L. finished the theoretical calculation. D.-H. Y. contributed to the substrate scope section. Q.-Y. L. and S.-N. Z. helped to analyze the theoretical calculation. X.-H. L. and B.-L. L. cowrote the original manuscript. X.-H. L. and J.-S. C. oversaw all the research phases. All of the authors discussed the results and commented on the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Tiancheng Mu and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Leng, BL., Lin, X., Dong, HY. et al. Regulating interfacial water for oxygen transfer to benzylic C(sp3)–H bonds via Ni-activated tungsten-oxygen covalency. Nat Commun 17, 2355 (2026). https://doi.org/10.1038/s41467-026-69054-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69054-3