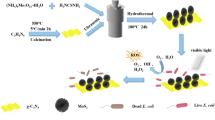
Abstract
Solar disinfection (SODIS), as a point-of-use (POU) water disinfection strategy for controlling waterborne microorganisms, serves millions of residents daily in over 50 low- to middle-income countries lacking basic drinking water services. However, SODIS is time consuming (6-48 h of sunlight exposure) due to its strong dependence on UV photons, which account for only ~4% of the solar energy. Thus, it is desirable to capture additional energy from visible-light photons (~50% of the solar energy) to accelerate the slow kinetics. Here, we use phosphorene nanoflakes (PNs) and BiOI nanosheets (BS) as model materials to construct a heterojunction photocatalyst, illustrating that simultaneously modulating the interfacial interaction and band alignment between the heterojunction components can achieve a dual optimization of the kinetic and thermodynamic constraints in photo-induced charge carriers, effectively enhancing the utilization of visible-spectrum energy for microbial inactivation. Notably, a subminute photocatalytic water disinfection performance is demonstrated by the PNs/BS heterojunction, completely inactivating 7 log of E. coli within 45 s under real sunlight. This results in a first-order disinfection rate ~221 times greater than that of commercial P25 TiO2. This work provides insights into the design of potent antimicrobial photocatalysts for POU water disinfection applications.
Similar content being viewed by others
Introduction
Even today, over 800 million individuals, primarily living in low- to middle-income countries, continue to endure the absence of basic drinking water services, which typically include centralized water treatment and distribution systems1,2. In these regions, up to 80% of diseases can be traced to waterborne microorganisms, with diarrheal diseases alone accounting for over 480,000 deaths annually3,4. Unfortunately, this adverse situation shows a limited prospect of resolution in the foreseeable long duration, as the deployment of basic drinking water services is impeded by exorbitant capital expenses required and the lack of pre-existing infrastructures (such as electrical grids) in these areas4. Consequently, for now, the residents in these regions are compelled to rely on point-of-use (POU) disinfection technologies at the household or community levels to mitigate the threat of waterborne pathogens, despite their limited effectiveness. Among these, solar disinfection (SODIS) is widely adopted, providing daily services to millions of people in over 50 countries5. This is due to its inherent simplicity and minimal infrastructure requirement, as it only necessitates exposing microbe-contaminated water in transparent containers to sunlight, thereby enabling microbial inactivation. However, SODIS generally demonstrates a relatively low disinfection efficacy, requiring a time-consuming exposure period of 6–48 h under natural sunlight5. This results from the heavy dependence of SODIS on UV photons, which constitute only ~4% of the solar energy and experience rapid attenuation as they propagate through water6.
To accelerate the slow kinetics, one approach is leveraging advanced forms of SODIS, such as photocatalysis, to capture additional energy from visible-light photons (~50% of the solar energy) for water disinfection7. Generally, a photocatalyst with an appropriate bandgap can harness visible light to generate electron-hole pairs, which initiate redox reactions, leading to the formation of reactive oxygen species (ROS). Theoretically, the redox potentials relevant to ROS formation are −0.33 V (O2/•O2−) and 0.28 V (O2/H2O2) for electron-driven oxygen reduction, and 1.90 V (H2O/•OH) for hole-driven water oxidation, all referenced to NHE at pH 7 (see refs. 7,8). These ROS can subsequently disrupt the essential macromolecules of pathogens, affecting multiple levels such as membrane integrity and metabolism, antioxidant enzyme activity, and genetic material, thereby accelerating their photoinactivation8,9,10. To gain photocatalytic antimicrobial materials with strong visible-light-harvesting abilities, a universal strategy involves the construction of heterojunctions by integrating semiconductor components with complementary bandgaps11,12,13,14,15,16,17. However, effectively utilizing absorbed visible light for microbial inactivation requires minimizing energy losses both during charge carrier transfer across the heterojunction interfaces and during the subsequent ROS generation by these carriers, a limitation that current heterojunctions remain unable to overcome. Specifically, for most existing heterojunctions: (i) the restricted interfacial interaction between components leads to a slow charge carrier transfer process at the interface regions, causing energy loss from a kinetic perspective; (ii) the limited redox capabilities of charge carriers prevent the sufficient generation of ROS, resulting in energy loss from a thermodynamic perspective. Consequently, the disinfection rates of heterojunctions reported to date remain unsatisfactory, necessitating light exposure of tens to hundreds of minutes to achieve a 5-to-7-log inactivation of bacteria3,18.
In this study, we use visible-light-responsive phosphorene nanoflakes (PNs) and BiOI nanosheets (BS) as illustrative materials to show that optimizing the compatibility between the macroscopic geometric architecture and microscopic band structure of heterojunction components can effectively address both the kinetic and thermodynamic constraints of photo-induced charge carriers (see Supplementary Information for additional information on the properties of these two components). On one hand, both PNs and BS possess a 2D nanostructure, enabling them to form intimate “face to face” contacts with robust interfacial interactions for fast charge carrier transfer (2D/2D heterojunctions provide larger interfacial contact areas than other counterparts, e.g., 0D/2D or 1D/2D)19. On the other hand, a well-defined S-scheme band alignment results in the accumulation of electrons and holes with enhanced redox abilities for elevated ROS generation. Benefiting from this dual-level optimization, the energy losses of visible-light energy are effectively suppressed from both kinetic and thermodynamic perspectives. With an optimal content of 0.8 wt% PNs, the resulting PNs/BS heterojunction exhibits a subminute photocatalytic water disinfection ability, achieving a complete inactivation of 7 log of E. coli within 45 s under real-sunlight illumination. Furthermore, integrating the photocatalyst into a continuous-flow fixed-bed reactor enables sustainable water purification for up to 24 h, highlighting its potential for practical POU water disinfection applications.
Results
Design, preparation, and characterization of PNs/BS heterojunction
Among the two illustrative heterojunction components selected in this study, special focus should be placed on the design and synthesis of PNs. As distinct from most semiconductor materials (including BiOI), phosphorene demonstrates a unique layer-dependent direct bandgap accompanied by tunable redox capabilities of its charge carriers20. Thus, we initiate our study by employing density functional theory (DFT) calculations to investigate the bandgap energy (Eg) of PNs to instruct the reasonable control over their layer thickness, thereby optimizing their performance as a component in the heterojunction. Our investigation reveals that the Eg values associated with PNs, for layers numbering one, two, three, and four, are 1.70, 1.48, 0.93, and 0.66 eV, respectively (Supplementary Fig. 1). This indicates that the control over the layer number of PNs is more necessary compared to BS. Given that, while ensuring the full utilization of the entire visible-light spectrum, a reduced number of layers in PNs can lead to enhanced redox capabilities of charge carriers. Moreover, the synthesis of ultrathin PNs is expected to enhance their contact with BS, which is beneficial for strengthening the interfacial interaction between the two components.
Nevertheless, the most commonly used liquid exfoliation method for PNs production from bulk black phosphorus (BP) proves inadequate in fulfilling this requirement, as multilayers are typically obtained (e.g., 5–8 phosphorene atomic layers using N-methyl-2-pyrrolidone (NMP), and even thicker with other solvents)21,22,23,24,25,26. This is due to the surface energy of phosphorene (~58.6 mJ/m2) is mismatched with that of the widely employed exfoliation solvents, such as NMP, acetone, ethanol, isopropanol (IPA), and dimethylformamide (DMF), hindering the enthalpy minimization of the mixed system27,28. However, considering water’s significantly higher surface energy of ~102 mJ/m2 and its miscibility with the aforementioned organic solvents, an organic-water mixture with an optimal surface energy for effective PNs production is achievable. Thus, we investigate the exfoliation efficiencies of 21 distinct organic-water mixtures for PNs production employing the Lambert-Beer law as a fundamental analysis principle (Supplementary Fig. 2). As anticipated, we ascertain a diverse range of mixtures exhibiting exceptional exfoliation performances, enabling the highly efficient generation of PNs. According to the experimental results, the optimal solvents are identified as an ethanol/water system (Vethanol/Vwater = 50%) and a NMP/water system (VNMP/Vwater = 80%). Given the potential challenge encountered in removing NMP with a high boiling point, the ethanol/water system is selected for PNs production (see Supplementary Information for details of analysis).
Through the liquid exfoliation of bulk BP in the determined ethanol/water system, the outcome yields PNs characterized by a superior crystalline state, as validated by selected-area electron diffraction and Raman assessments (Supplementary Fig. 3). As depicted in Supplementary Fig. 4, the vertical extent data extracted along the marked lines reveal a thickness of ~1.7–2.1 nm, which corresponds to 2–3 atomic layers, confirming the successful production of ultrathin PNs29. PNs/BS materials are then prepared by a wet chemistry method, in which PNs are mixed with BS precursors (KI and Bi(NO)3·5H2O), enabling the in situ growth of BS on PNs. According to the weight ratios of PNs to BS (x wt%), the samples are denoted as xPNs/BS. As shown in Supplementary Fig. 5, the X-ray diffraction (XRD) pattern of 0.0PNs/BS clearly indicates its alignment with the tetragonal phase of BiOI (JCPDS No. 10-0445). A comparison of the XRD patterns between 0.0PNs/BS and 0.8PNs/BS underscores that the incorporation of PNs has a negligible impact on the crystalline structure of BS. Moreover, it is noteworthy that the XRD peaks associated with BP are conspicuously absent within the pattern of 0.8PNs/BS. This absence is attributed to the fact that PNs are loaded at a relatively low concentration and exhibit a remarkable degree of dispersion. The transmission electron microscope (TEM) image of PNs reveals a thin 2D flake-like structure with well-defined edges and sleek external surfaces, whereas the TEM image of 0.0PNs/BS displays a 2D sheet-like architecture marked by irregular edges and rough external surfaces (Fig. 1a, b). These 2D morphological features are further corroborated by scanning electron microscope (SEM) observations (Supplementary Figs. 6 and 7). The high-resolution TEM (HRTEM) images of PNs and 0.0PNs/BS unveil their high crystalline quality (Supplementary Fig. 8). The image in Fig. 1c illustrates a TEM view of 0.8PNs/BS, revealing the stacking of BS onto the surface of ultrathin PNs, establishing a “face-to-face” contact between the two 2D materials. The HRTEM image of 0.8PNs/BS demonstrates lattice fringes that are consistent with the (002) facet of BP and the (110) facet of BiOI (Fig. 1d)30,31. The energy dispersive X-ray spectroscopy (EDS) and X-ray photoelectron spectroscopy (XPS) survey results of 0.8PNs/BS concur with the findings of TEM and HRTEM by confirming the elemental composition of Bi, O, I, and P in the material (Supplementary Fig. 9). The high-angle annular dark field-scanning transmission electron microscope (HAADF-STEM) image along with the corresponding elemental mapping images of 0.8PNs/BS provide additional evidence of the consistent hybridization of PNs and BS in a uniform manner (Supplementary Fig. 10). Therefore, all the aforementioned results strongly validate the successful preparation of a heterojunction of PNs/BS.
TEM images of a PNs, b BS, and c 0.8PNs/BS. d HRTEM image of 0.8PNs/BS. TEM and HRTEM images represent one of three independent experiments showing consistent results.
Strong interfacial interaction for improved charge carrier transfer
The observed “face to face” contact between the two 2D components, PNs and BS, within the constructed heterojunction, is anticipated to create a strong interfacial interaction between the layers. To confirm this, synchrotron-based X-ray absorption near-edge structure (XANES) analysis is employed. The Bi L3-edge absorption edge position of 0.8PNs/BS shows a negative shift compared to that of 0.0PNs/BS, suggesting the occurrence of electron transfer from PNs to BS within the heterojunction (Fig. 2a). To further validate the electron transfer between the two materials, DFT calculations are performed. Figure 2b shows the differential charge density of the composite system, with blue and yellow regions representing charge depletion and accumulation, respectively. The result indicates that the surface of BS attracts electrons from neighboring PNs. Such electron transfer behavior is often associated with the establishment of an intimate electronic interaction between distinct heterojunction moieties. To rationalize this observation, the work function (Φ) values of the two components are calculated, resulting in 4.61 eV for phosphorene and 5.89 eV for BiOI (Fig. 2c, d). The disparity in Φ, in theory, can promote a directional electron transfer between PNs and BS, thereby establishing an internal electric field that induces interlayer electronic coupling. More importantly, such a strong interfacial electronic interaction is expected to facilitate a fast charge transfer process between the two materials.
a XANES Bi-L3 edges of 0.0PNs/BS and 0.8PNs/BS. b Side-view differential charge density map of PNs/BS, with yellow and blue regions representing the areas of net electron accumulation and depletion, respectively. Average potential profiles along Z-axis direction for c phosphorene and d BiOI. Transient absorption spectra of e 0.0PNs/BS and f 0.8PNs/BS recorded at specified delay times. Corresponding kinetic decay curves of g 0.0PNs/BS and h 0.8PNs/BS.
Building on this expectation, femtosecond transient absorption spectroscopy is carried out. As illustrated in Fig. 2e, f, excitation at 350 nm induces distinct positive peaks for both 0.0PNs/BS and 0.8PNs/BS in the timescale of 0.5–200 ps, indicating the dominance of excited-state absorption during this interval. In contrast, the time range of 500 ps–3 ns is characterized by prominent negative peaks, demonstrating that the gradual relaxation of excited-state population back to ground state over time. Interestingly, for 0.8PNs/BS, in addition to a ground-state bleaching peak at ~526 nm, a stronger negative feature is evident on the longer wavelength side. This can be attributed to the stimulated emission resulting from the electron-hole recombination between PNs and BS. Figure 2g illustrates the normalized recovery kinetics of 0.0PNs/BS at 520 nm (fitted using a two-exponential function), reflecting the interband diffusion and the trapping by shallow trap states of photo-induced electrons. The integration of PNs with BS gives rise to an additional ultrafast pathway (~2.1 ps), which can be attributed to the transfer of photo-induced electrons between the two components (Fig. 2h). This ultrafast transfer process is anticipated to mitigate the recombination of electrons and holes occurring within each individual component, effectively preventing photon energy loss from a kinetic perspective.
To validate this, the charge carrier separation efficiencies of 0.0PNs/BS and 0.8PNs/BS are systematically investigated using a range of characterization techniques. First, transient photo-current (TPC) response measurement is conducted. As depicted in Supplementary Fig. 12a, the TPC response of 0.8PNs/BS, which is significantly higher than that of 0.0PNs/BS, suggests an improved efficiency in the separation of electron-hole pairs upon the incorporation of PNs with BS. Additionally, the results from electrochemical impedance spectroscopy (EIS) measurement demonstrate a smaller semicircle radius in the Nyquist plot of 0.8PNs/BS compared with that of 0.0PNs/BS, implying a reduced charge transfer resistance for 0.8PNs/BS, thereby promoting more efficient charge carrier separation (Supplementary Fig. 12b). Furthermore, the steady-state photoluminescence (PL) results of the samples reveal a reduced PL intensity for 0.8PNs/BS in contrast to 0.0PNs/BS, suggesting a suppressed charge carrier recombination in 0.8PNs/BS (Supplementary Fig. 12c). Moreover, the transient-state PL results shown in Supplementary Fig. 12d exhibit an increase in average PL lifetime, from 19.8 ns for 0.0PNs/BS to 23.9 ns for 0.8PNs/BS. This further supports the higher electron-hole separation efficiency of 0.8PNs/BS, in agreement with the TPC, EIS, and steady-state PL results. Hence, all the above-mentioned experimental findings collectively validate that the energy loss during charge transfer process is effectively mitigated.
Boosted redox capacity for elevated ROS production
The separated photo-induced charge carriers can subsequently participate in catalytic redox reactions for ROS generation. The redox capacities of these charge carriers are determined by the band alignment type of the heterojunction, which, in turn, governs the energy efficiency of the ensuing ROS generation process. Considering this, band-structure characterizations are performed for the two heterojunction components. First, the Eg of BS is derived from the plot of (αhν)n versus hν shown in Supplementary Fig. 13a, where α represents absorption coefficient, h stands for Planck constant, and ν denotes light frequency32. In this equation, n is set to 1/2 due to the indirect bandgap nature of BS. Consequently, the determined Eg value of BS stands at 1.68 eV. Subsequent to this, XPS valence-band spectroscopy analysis is conducted for BS. As illustrated in Supplementary Fig. 13b, the valence-band value of BS is observed to be 1.36 V versus NHE. This leads to the calculation of the conduction-band value of BS, which is found to be −0.32 V versus NHE (Supplementary Table 1). Furthermore, the Eg of PNs is ascertained through UV–visible (UV–vis) absorption spectroscopy analysis. As shown in Supplementary Fig. 13c, the identified absorption edge of PNs at 731 nm corresponds to an Eg value of 1.70 eV. We then execute a Mott-Schottky test, revealing the n-type semiconductor characteristic of PNs (Supplementary Fig. 13d). Thus, the flat-band potential of PNs, extracted from the Mott-Schottky result, can offer a rough estimation for the conduction-band value. Consequently, the conduction-band value of PNs is established at −0.44 V versus NHE, and the valence-band value is then calculated to be 1.26 V versus NHE (Supplementary Table 1).
Combining the band structures of PNs and BS with the above experimental and DFT findings on the electron transfer direction between them, a typical S-scheme band alignment is expected at the PNs/BS interface (Fig. 3a–c). In this band alignment, PNs, characterized by a more negative conduction-band position, act as the reduction-type component, while BS, with a more positive valence-band position, function as the oxidation-type component. In the absence of light irradiation, electrons spontaneously transfer from PNs to BS, establishing an internal electric field across the heterojunction directed from PNs to BS. Under light irradiation, this built-in field facilitates the recombination of photo-excited electrons in the conduction band of BS with photo-induced holes in the valence band of PNs. Consequently, energetically favorable charge carriers are preserved in the conduction band of PNs and the valence band of BS. Such an S-scheme band alignment is anticipated to augment the ROS-production abilities of charge carriers, effectively mitigating photon energy loss from a thermodynamic perspective.
a–c Schematic illustration showing the formation of the S-scheme band alignment at the PNs/BS interface (EC, EV, and EF denote the conduction band, valence band, and Fermi level of the heterojunction components; Φ1 and Φ2 represent the Φ values of BS and PNs; the “+” and “−” symbols represent electron depletion and accumulation regions; the green and purple spheres denote photo-generated electrons and holes). Steady-state concentrations of d •O2−, e 1O2, and f •OH, and g accumulated concentrations of H2O2 over different PNs/BS heterojunctions. The error bars (mean ± standard deviation) are obtained based on three independent photocatalytic experiments.
To confirm this, the visible-light-induced ROS generation of the samples is evaluated using specific reactions involving probe molecules. Specifically, nitroblue tetrazolium (NBT) can react with •O2−, leading to a reduction in the absorption intensity at 259 nm. As depicted in Fig. 3d, 0.8PNs/BS demonstrates the highest NBT consumption among the tested samples, corresponding to a •O2− steady-state concentration of 4.90 × 10−9 M in its reaction system, significantly surpassing the concentration observed in 0.0PNs/BS system (1.25 × 10−9 M). Furthermore, furfuryl alcohol (FFA), a widely acknowledged probe for 1O2 generation, is monitored for its decay to quantitatively assess the 1O2-generation activities of the samples (Fig. 3e). The 1O2 steady-state concentration in 0.8PNs/BS system is determined to be 2.02 × 10−13 M, exceeding the corresponding value in 0.0PNs/BS system (1.38 × 10−13 M). Moreover, p-chlorobenzoic acid (pCBA) is used as a reactive agent to evaluate the •OH-generation abilities of the materials. As shown in Fig. 3f, 0.8PNs/BS exhibits the highest •OH-production capacity, as evidenced by its more rapid pCBA decomposition compared to the other tested samples. The corresponding •OH steady-state concentration is determined to be 5.20 × 10−14 M, surpassing the value observed in 0.0PNs/BS system (2.17 × 10−14 M). The results of electron spin resonance (ESR) spectroscopy further support these findings, offering additional evidence of the enhanced generation activities of •O2−, 1O2, and •OH over 0.8PNs/BS relative to 0.0PNs/BS (Supplementary Fig. 14). We then examine the H2O2-production capacities of the samples using iodometric analysis (Fig. 3g). After 60 min of reaction, the accumulated H2O2 concentrations in 0.0PNs/BS and 0.8PNs/BS systems are quantified to be 3.3 and 6.6 μM, respectively, indicating the superior H2O2-production activity of 0.8PNs/BS. Collectively, these experimental findings unequivocally confirm the effective mitigation of energy loss during ROS generation process.
Visible-light photocatalytic water disinfection performance
An assessment of the photocatalytic disinfection activities of the samples is then carried out. Gram-negative bacterium E. coli, with an initial concentration of 7 log, is used as a process indicator in all experiments unless otherwise indicated. Visible light with an optical power density of ~100 mW cm−2 is generated using a solar simulator. As depicted in Supplementary Fig. 15, PNs demonstrate a minimal photocatalytic disinfection effect against E. coli, with bacterial concentration decreasing by less than 1 log after 60 min of illumination. Nonetheless, their incorporation markedly improves the photocatalytic antibacterial performance of BS across varying loading amounts (Fig. 4a). Notably, 0.8PNs/BS shows the highest photocatalytic water disinfection performance, achieving a complete 7-log inactivation of E. coli (> 99.99999% disinfection efficiency) in only 5 min of visible-light irradiation. In contrast, control experiments conducted under solely visible-light exposure or in the presence of 0.8PNs/BS under a dark condition result in disinfection efficiencies below 0.25 log after 120 min, demonstrating that bacterial inactivation is attributed to visible-light photocatalysis (0.8PNs/BS can harvest the entire visible-light spectrum, as evidenced by UV–vis diffuse reflectance spectroscopy) (Supplementary Fig. 16). The contributions of various ROS during the disinfection process of E. coli are investigated through a scavenger study. The experimental results illustrated in Supplementary Fig. 17 indicate the participation of four distinct ROS for bacterial inactivation, with contributions of •O2− > 1O2 > H2O2 > •OH. These ROS contribute to E. coli inactivation by disrupting cell membrane permeability, inhibiting energy metabolism, and damaging intracellular components (Supplementary Fig. 18). The mechanism of photocatalytic water disinfection by 0.8PNs/BS is shown in Supplementary Fig. 19. The broad-spectrum photocatalytic antibacterial activity of 0.8PNs/BS is then demonstrated by its effective disinfection of other waterborne bacteria, including E. faecalis, S. aureus, and P. aeruginosa (Supplementary Fig. 20). Notably, the approach proposed in this study, which strategically optimizes the compatibility between the macroscopic geometric architecture and microscopic band structure of heterojunction components, exhibits broad applicability, as evidenced by the significantly enhanced photocatalytic antibacterial activities observed upon the integration of PNs with BiOBr or BiOCl nanosheets (Supplementary Figs. 21 and 22).
a Visible-light photocatalytic water disinfection activities of PNs/BS materials. b Visible-light photocatalytic water disinfection performance comparison of 0.8PNs/BS with the literature values of other photocatalysts. The plot gives the log inactivation efficiency of E. coli with respect to illumination time: (1) 0.8PNs/BS (this work), (2) (Al2O3@v-MoS2)/Cu (ref. 36), (3) SGO-ZnO-Ag (ref. 37), (4) Cu-MoS2 (ref. 7), (5) F-g-C3N4-30-EP (ref. 38), (6) CoFe2O4/HTCC (ref. 13), (7) g-C3N4-TiO2 (ref. 14), (8) BiVO4 nanotube (ref. 39). c Water disinfection performance comparison between 0.8PNs/BS and P25 TiO2 under real-sunlight illumination. d Visible-light photocatalytic water disinfection activity of a continuous-flow fixed-bed reactor, constructed based on our previously reported method (ref. 6). The error bars (mean ± standard deviation) are obtained based on three independent photocatalytic experiments.
The photocatalytic antibacterial performance of 0.8PNs/BS, with a 7-log inactivation of E. coli within 5 min under visible-light illumination, is considered one of the most remarkable based on a thorough review of the existing literature, particularly in terms of the extent of the log reduction in bacterial concentration and the rapidity of disinfection process (Fig. 4b and Supplementary Table 2). Compared with those that achieve a complete inactivation of E. coli within 30 min, such as (Al2O3@v-MoS2)/Cu, Cu-MoS2, and F-g-C3N4-30-EP, the bacterial concentration reduction achieved by 0.8PNs/BS surpasses them by 1-2 log. As for disinfection systems containing an initial bacterial concentration of 7 log, the reported antimicrobial materials thus far necessitate a minimum of 60 min to achieve the total inactivation of E. coli, whereas these materials, such as SGO-ZnO-Ag, are accompanied by a certain degree of biological intolerance. To the best of our knowledge, no previously reported photocatalysts have demonstrated such an extraordinary disinfection efficacy when confronted with an initial E. coli concentration of 7 log.
Moreover, the photocatalytic water disinfection efficacy of 0.8PNs/BS is compared with that of the widely-studied P25 TiO2 (20 nm). As shown in Fig. 4c, 0.8PNs/BS outperforms P25 TiO2 under real-sunlight irradiation (10:00 AM, local time, September 6, 2023, Northeast Normal University, 43.83°N, 125.43°E), achieving a complete inactivation of 7 log of E. coli in only 45 s. This corresponds to a first-order disinfection rate (k) of 9.30 min−1 (R2 = 0.97), which exceeds that of P25 TiO2 (k = 0.042 min−1, R2 = 0.95) by approximately 221-fold. More importantly, 0.8PNs/BS demonstrates its applicability in a continuous-flow fixed-bed reactor, enabling water purification for up to 24 h, underscoring its promise for practical POU water disinfection applications (Fig. 4d).
In conclusion, by utilizing PNs and BS as model materials, we demonstrate that optimizing the compatibility between the macroscopic geometric architecture and microscopic band structure of heterojunction components can effectively overcome both the kinetic and thermodynamic limitations of photo-induced charge carriers. Experimental findings, supported by DFT calculations, reveal a strong interfacial interaction between the two components, enabling an ultrafast electron transfer process (~2.1 ps). Furthermore, the established S-scheme band alignment promotes the efficient accumulation of electrons and holes with enhanced redox capabilities. Consequently, the energy from visible spectrum is effectively utilized for bacterial inactivation. Under real sunlight, the synthesized 0.8PNs/BS exhibits a subminute photocatalytic water disinfection ability, achieving a complete inactivation of 7-log E. coli within 45 s. This delivers E. coli inactivation ~221 times faster than commercial P25 TiO2. Additionally, incorporating 0.8PNs/BS into a continuous-flow fixed-bed reactor allows for sustainable water purification for up to 24 h, underscoring its suitability for practical POU water disinfection.
Methods
Chemicals
Iodine and tin were supplied by Guangfu Technology Development Co., Ltd. (Tianjin, China). Red phosphorus was obtained from Hengxing Chemical Reagent Manufacturing Co., Ltd. (Tianjin, China). KI, KBr, KCl, and potassium hydrogen phthalate were purchased from Kermel Chemical Reagent Co., Ltd. (Tianjin, China). Bi(NO)3·5H2O was purchased from Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). NMP was purchased from Damao Chemical Reagent (Tianjin, China). Ethanol, acetone, IPA, DMF, and FFA were purchased from Fuyu Fine Chemical Co., Ltd. (Tianjin, China). NBT, pCBA, L-histidine, and P25 TiO2 were sourced from Rhawn Technology Development Co., Ltd. (Shanghai, China). Ethylenediamine tetraacetic acid (EDTA) disodium salt was purchased from Beijing Chemical Works (Beijing, China). 4-hydroxy-2,2,6,6-tetramethylpiperidinyloxy (TEMPOL) was supplied by Acros Organics (New Jersey, USA). All chemicals employed in this study were of analytical grade.
Synthesis of materials
Synthesis of bulk BP: A bulk BP crystal was synthesized using a well-established method12. In a standard experimental setup, commercial red phosphorus (2 g) was dispersed in 15 mL of H2O and hydrothermally treated at 200 °C for 12 h to remove surface oxide layers, and then dried in an oven9. A vacuum-sealed quartz tube was loaded with 30 mg of iodine, 80 mg of tin, and 500 mg of red phosphorus. This quartz tube was subsequently positioned within a muffle furnace and subjected to a controlled temperature program. Specifically, the temperature was elevated to 600 °C utilizing a controlled heating ramp rate of 3 °C/min, and subsequently maintained at this temperature for 120 min. The subsequent phase involved a controlled cooling process, gradually lowering the temperature to 490 °C with a cooling rate of 0.4 °C/min, and holding it at this temperature for 120 min. Then, the temperature was progressively reduced to 120 °C with a cooling rate of 0.9 °C/min. Finally, the quartz tube was allowed to cool naturally to room temperature, culminating in the formation of bulk BP crystal.
Solvent survey-informed PNs synthesis: In a standard procedure, 10 mg of BP powders were dispersed in 20 mL of the corresponding solvents, which included NMP, acetone, ethanol, IPA, and DMF, as well as their respective aqueous solutions. The resulting mixtures were subjected to an ultrasonic exfoliation process for a duration of 90 min, utilizing a water bath sonicator. Subsequently, the dispersions were subjected to a controlled centrifugation process at 45×g for a duration of 30 min. Following centrifugation, the supernatant samples containing exfoliated materials were collected for further analysis, using a UV–vis spectrophotometer (Hitachi U-2900). To ensure optimal accuracy and comparability, the absorbance of the samples was measured at 665 nm, since all selected solvents demonstrated near-complete transparency at this wavelength. Based on the experimental results of solvent survey, an ethanol/water system (Vethanol/Vwater = 50%) was selected as the exfoliating medium for PNs production. PNs synthesis involved a sequential process of tip sonication followed by ice-bath sonication of bulk BP. Initially, 40 mg of bulk BP was ground and subsequently introduced into the ethanol/water exfoliating medium (40 mL). The dispersion was then subjected to N2 bubbling for 5 min, followed by a tip sonication treatment operating at an ice-cooled environment for 4 h. The sonicator was operated in 1 s on/2.5 s off cycles. Subsequently, the obtained dispersion underwent a further ice-bath sonication for 2 h (the purpose of ice bath is to maintain a constant temperature). Following this, the dispersion was centrifuged at 2862×g for 10 min to eliminate any non-exfoliated bulk BP, leading to the formation of PNs dispersion (0.128 mg mL−1).
Synthesis of BS: In a standard procedure, both 35 mL of KI (11.4 mM) and 35 mL of Bi(NO3)3·5H2O (11.4 mM) were prepared as aqueous solutions and then mixed. The resulting mixture was stirred for 1 h at room temperature. Following this, the mixture was heated at 80 °C for 4 h in a programmable temperature chamber. The resulting materials were collected through suction filtration and subsequently dried in an oven at 60 °C, yielding BS.
Synthesis of BiOBr and BiOCl nanosheets: BiOBr nanosheets were synthesized using a method analogous to the synthesis of BS, except that KI aqueous solution was replaced by 35 mL of KBr (11.4 mM) aqueous solution. BiOCl nanosheets were synthesized using a method analogous to the synthesis of BS, except that KI aqueous solution was replaced by 35 mL of KCl (11.4 mM) aqueous solution.
Synthesis of PNs/BS: In a standard procedure, 35 mL of KI (11.4 mM) aqueous solution was combined with varying amounts of PNs dispersion, which was then blended with 35 mL of Bi(NO3)3·5H2O (11.4 mM) aqueous solution. The resulting mixtures underwent stirring at room temperature for 1 h. Following this, the mixtures were subjected to heating at 80 °C for 4 h within a programmable temperature chamber. The resultant materials were collected via suction filtration and subsequently dried at 60 °C in an oven, resulting in the formation of PNs/BS samples. The weight ratios of PNs to BS were set at 0.0, 0.1, 0.5, 0.8, and 1.0 wt%, leading to the corresponding PNs/BS samples designated as 0.0PNs/BS, 0.1PNs/BS, 0.5PNs/BS, 0.8PNs/BS, and 1.0PNs/BS, respectively.
Synthesis of PNs/BiOBr and PNs/BiOCl: PNs/BiOBr samples with varying PNs loading levels were synthesized using a method analogous to the synthesis of PNs/BS, with the sole exception of replacing KI aqueous solution by 35 mL of KBr (11.4 mM) aqueous solution. PNs/BiOCl samples were synthesized using a method analogous to the synthesis of PNs/BS, with the sole exception of replacing KI aqueous solution by 35 mL of KCl (11.4 mM) aqueous solution.
Characterization of materials
Raman data were collected with a Horiba LabRAM HR Evolution spectrometer. Atomic force microscope (AFM) images were acquired utilizing a Busker Dimension ICON instrument. XRD data were recorded using a Bruker D8 Advance diffractometer. TEM and HAADF-STEM images were acquired using a JEOL 2100 F instrument. SEM images were observed with a Hitachi SU8600 instrument. XPS measurements were performed using a Thermo Fisher Scientific Escalab 250Xi spectrometer. XANES analysis was carried out using a Super Photon Ring-8 GeV synchrotron facility. Femtosecond transient absorption spectroscopy measurements were conducted with an Ultrafast System LLC Helios pump-probe system. Steady-state and transient-state PL spectra were recorded on an Edinburgh Instruments FSL980 spectrophotometer. UV–vis diffuse reflectance spectra were recorded using a Shimadzu UV 3600 spectrometer.
Computational method
DFT calculations were performed using a PWmat code, with exchange-correlation interaction modeled through generalized gradient approximation (GGA) with Perdew–Burke–Ernzerhof (PBE) functional. Computational parameters included an energy cutoff of 70 Ryd and a Monkhorst-Pack k-point mesh of 4 × 4 × 1. Geometry optimization was conducted with convergence thresholds set to 1.0 × 10−5 eV for energy and 0.01 eV/Å for force. Surface models were constructed with a 30 Å vacuum layer to prevent the interaction between periodic replicas. To account for Van der Waals interaction, the DFT-D2 method of Grimme was employed. Φ was determined as the difference between vacuum level and Ef.
Electrochemical and photoelectrochemical measurement
TPC, EIS, and Mott-Schottky analyses were carried out employing an electrochemical workstation (EC 550, Wuhan Gaoss Union Technology Co., Ltd.) in conjunction with a conventional three-electrode cell configuration. In this configuration, a Pt plate served as counter electrode, while a Ag/AgCl (saturated KCl) electrode was utilized as reference electrode. The electrolyte solution used was a 0.5 M Na2SO4 aqueous solution. The preparation of working electrode involved the following steps. Initially, an indium-tin oxide (ITO) glass was cleaned by ultrasonication using ethanol and IPA for 10 min each, and subsequently dried at 60 °C in an oven. Then, 1 mg of material was dispersed in an ethanol solution (Vethanol/Vwater = 50%), and 5 μL of Nafion was introduced. This mixture underwent ultrasonication for 5 min to yield a homogeneous suspension. A 10 μL aliquot of this suspension was deposited onto the ITO glass substrate. Afterward, the ITO glass was heated at 80°C for 30 min to ensure optimal adhesion. Following cooling to room temperature, the resulting product was catalyst-ITO working electrode. EIS and Mott-Schottky measurements were performed under dark conditions, while TPC measurements were conducted under illumination provided by a 300 W Xe lamp (PLS-SXE300, Beijing Perfect Light Technology Co., Ltd.). All measurements were carried out at room temperature.
ROS measurement
In a typical procedure, a material concentration of 1 mg/mL was employed, with a total reaction volume of 30 mL. A 300 W Xe lamp (PLS-SXE300, Beijing Perfect Light Technology Co., Ltd.), coupled with a 400 nm UV-blocking filter, served as the light source (optical power density of ~100 mW cm−2). To determine the steady-state concentrations of •O2−, 1O2, and •OH, we analyzed the decay kinetics of specific probe molecules9. NBT was employed to detect •O2−, FFA was used as an indicator for 1O2, and pCBA acted as a reactive agent for •OH. The rate constants for the reactions between •O2− and NBT, 1O2 and FFA, as well as •OH and pCBA were determined to be 5.9 × 104, 1.2 × 108, and 5.2 × 109 M−1 s−1, respectively. Notably, to accurately ascertain the steady-state concentration of 1O2, 1 mM of IPA was introduced to quench •OH, taking into account the second-order reaction rate constant of 1.5 × 1010 M−1 s−1 between •OH and FFA33.
The quantification of accumulated H2O2 was achieved through an iodometric methodology34. Under an acidic condition, H2O2 could precipitate I− to form I3−, yielding a distinctive absorption peak at 350 nm. A typical experimental process involved a mixture of 1 mL of KI solution (0.4 M), 1 mL of potassium hydrogen phthalate solution (0.1 M), and 3 mL of reaction solution. This mixture was shielded from light and allowed to incubate for 30 min. Subsequently, the resulting solution was analyzed using a Hitachi U-2900 UV–vis spectrophotometer, with a focus on measuring the absorbance at 350 nm. All ROS measurement experiments were performed in triplicate to enhance the statistical rigor of the findings.
A Bruker A300 spectrometer was carried out for ESR measurement. To achieve this, 10 mg of photocatalyst was uniformly dispersed in water or methanol through ultrasonication. Subsequently, 200 µL of the resulting mixed solution was combined with 100 µL of trapping agent solution and loaded into a capillary tube for ESR testing. For the ESR analyses of •O2− and •OH, 5,5-dimethyl-1-pyrroline-N-oxide (DMPO) was employed as a trapping agent. Meanwhile, the ESR assessment of 1O2 utilized 2,2,6,6-tetramethylpiperidine (TEMP) as a trapping agent. Notably, methanol-dispersed photocatalyst was used for •O2− ESR testing, while water-dispersed photocatalyst was employed for •OH and 1O2 ESR evaluations.
Disinfection performance
E. coli K-12 was chosen as a process indicator to assess the photocatalytic water disinfection activities of the synthesized materials. Bacterial cells were cultured in Nutrient Broth at 37°C for 16 h to reach a logarithmic growth phase. Subsequently, they were collected via centrifugation at 2862×g for 10 min, washed with deionized (DI) water, and re-suspended in fresh DI water to achieve a concentration of ~107 cfu/mL. For all disinfection experiments, a material dosage of 1 mg/mL was applied, along with a bacterial suspension volume of 30 mL. Disinfection was conducted using a 300 W Xe lamp (PLS-SXE300, Beijing Perfect Light Technology Co., Ltd.) equipped with a 400 nm UV-blocking filter. Its optical power density was maintained at ~100 mW cm2, corresponding to the solar irradiance intensity at the Earth’s surface. A water cooling system was used to maintain the bacterial suspension at a constant temperature of ~25 °C. At predetermined intervals, the E. coli concentration of the suspension was assessed using the plate counting method. All bacterial inactivation experiments were performed in triplicate to enhance the statistical rigor of the findings.
Scavenger study
To determine the contributions of •O2−, 1O2, •OH, and H2O2 to E. coli inactivation, four scavengers were used, including TEMPOL (2 mM, targeting •O2−), L-histidine (0.5 mM, targeting 1O2), IPA (1 mM, targeting •OH), and Fe-EDTA (0.5 mM, targeting H2O2).
Data availability
Source data are provided with this paper and are available in Figshare repository with the identifier https://doi.org/10.6084/m9.figshare.3064586035. All the raw data relevant to the study are available from the corresponding author upon request.
References
Ryberg, E. C., Chu, C. & Kim, J. H. Edible dye-enhanced solar disinfection with safety indication. Environ. Sci. Technol. 52, 13361–13369 (2018).
Jeon, I., Ryberg, E. C., Alvarez, P. J. J. & Kim, J. H. Technology assessment of solar disinfection for drinking water treatment. Nat. Sustain. 5, 801–808 (2022).
Wang, W. et al. Carbon nitride based photocatalysts for solar photocatalytic disinfection, can we go further? Chem. Eng. J. 404, 126540 (2021).
Chu, C., Ryberg, E. C., Loeb, S. K., Suh, M. J. & Kim, J. H. Water disinfection in rural areas demands unconventional solar technologies. Acc. Chem. Res. 52, 1187–1195 (2019).
McGuigan, K. G. et al. Solar water disinfection (SODIS): a review from bench-top to roof-top. J. Hazard. Mater. 235–236, 29–46 (2012).
Yang, H. et al. Visible-light-induced rapid elimination of antibiotic resistance contaminations using graphitic carbon nitride tailored with carrier confinement domains. Adv. Mater. 37, 2410221 (2024).
Liu, C. et al. Rapid water disinfection using vertically aligned MoS2 nanofilms and visible light. Nat. Nanotechnol. 11, 1098–1104 (2016).
Xia, P. et al. Designing a 0D/2D S-scheme heterojunction over polymeric carbon nitride for visible-light photocatalytic inactivation of bacteria. Angew. Chem. Int. Ed. Engl. 59, 5218–5225 (2020).
Xia, D. et al. Red phosphorus: an Earth-abundant elemental photocatalyst for “green” bacterial inactivation under visible light. Environ. Sci. Technol. 49, 6264–6273 (2015).
Zhou, Y. et al. Bacterial inactivation by dichloride radical in water: molecular-level reaction kinetics and disinfection mechanism. Water Res. 288, 124558 (2026).
Yang, H. et al. Magnetic carbon nanotubes/red phosphorus/graphitic carbon nitride heterojunction for highly efficient visible-light photocatalytic water disinfection. Chem. Eng. J. 466, 143309 (2023).
He, D. et al. Black phosphorus/graphitic carbon nitride: a metal-free photocatalyst for “green” photocatalytic bacterial inactivation under visible light. Chem. Eng. J. 384, 123258 (2020).
Wang, T. et al. Enhanced visible-light-driven photocatalytic bacterial inactivation by ultrathin carbon-coated magnetic cobalt ferrite nanoparticles. Environ. Sci. Technol. 52, 4774–4784 (2018).
Li, G. et al. Enhanced visible-light-driven photocatalytic inactivation of Escherichia coli using g-C3N4/TiO2 hybrid photocatalyst synthesized using a hydrothermal-calcination approach. Water Res. 86, 17–24 (2015).
Liang, J., Liu, F., Deng, J., Li, M. & Tong, M. Efficient bacterial inactivation with Z-scheme AgI/Bi2MoO6 under visible light irradiation. Water Res. 123, 632–641 (2017).
Xia, D. et al. Enhanced photocatalytic inactivation of Escherichia coli by a novel Z-scheme g-C3N4/m-Bi2O4 hybrid photocatalyst under visible light: the role of reactive oxygen species. Appl. Catal. B-Environ. 214, 23–33 (2017).
Wang, W. et al. Photocatalytic hydrogen evolution and bacterial inactivation utilizing sonochemical-synthesized g-C3N4/red phosphorus hybrid nanosheets as a wide-spectral-responsive photocatalyst: the role of type I band alignment. Appl. Catal. B-Environ. 238, 126–135 (2018).
You, J., Guo, Y., Guo, R. & Liu, X. A review of visible light-active photocatalysts for water disinfection: features and prospects. Chem. Eng. J. 373, 624–641 (2019).
Low, J., Cao, S., Yu, J. & Wageh, S. Two-dimensional layered composite photocatalysts. Chem. Commun. 50, 10768–10777 (2014).
Liu, H., Du, Y., Deng, Y. & Ye, P. D. Semiconducting black phosphorus: synthesis, transport properties and electronic applications. Chem. Soc. Rev. 44, 2732–2743 (2015).
Ran, J., Zhu, B. & Qiao, S. Z. Phosphorene co-catalyst advancing highly efficient visible-light photocatalytic hydrogen production. Angew. Chem. Int. Ed. Engl. 56, 10373–10377 (2017).
Ran, J. et al. Metal-free 2D/2D phosphorene/g-C3N4 Van der Waals heterojunction for highly enhanced visible-light photocatalytic H2 production. Adv. Mater. 30, 1800128 (2018).
Zhu, M., Zhai, C., Fujitsuka, M. & Majima, T. Noble metal-free near-infrared-driven photocatalyst for hydrogen production based on 2D hybrid of black Phosphorus/WS2. Appl. Catal. B-Environ. 221, 645–651 (2018).
Yasaei, P. et al. High-quality black phosphorus atomic layers by liquid-phase exfoliation. Adv. Mater. 27, 1887–1892 (2015).
Kang, J. et al. Solvent exfoliation of electronic-grade, two-dimensional black phosphorus. ACS Nano 9, 3596–3604 (2015).
Dhanabalan, S. C. et al. Emerging trends in phosphorene fabrication towards next generation devices. Adv. Sci. 4, 1600305 (2017).
Hernandez, Y. et al. High-yield production of graphene by liquid-phase exfoliation of graphite. Nat. Nanotechnol. 3, 563–568 (2008).
Coleman, J. N. et al. Two-dimensional nanosheets produced by liquid exfoliation of layered materials. Science 331, 568–571 (2011).
Hanlon, D. et al. Liquid exfoliation of solvent-stabilized few-layer black phosphorus for applications beyond electronics. Nat. Commun. 6, 8563 (2015).
Lei, W. et al. Hybrid 0D-2D black phosphorus quantum dots-graphitic carbon nitride nanosheets for efficient hydrogen evolution. Nano Energy 50, 552–561 (2018).
Sun, L. et al. Enhanced visible-light photocatalytic activity of BiOI/BiOCl heterojunctions: key role of crystal facet combination. Acs. Catal. 5, 3540–3551 (2015).
Liu, J. et al. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science 347, 970–974 (2015).
Mostafa, S. & Rosario-Ortiz, F. L. Singlet oxygen formation from wastewater organic matter. Environ. Sci. Technol. 47, 8179–8186 (2013).
Wei, Z. et al. Efficient visible-light-driven selective oxygen reduction to hydrogen peroxide by oxygen-enriched graphitic carbon nitride polymers. Energ. Environ. Sci. 11, 2581–2589 (2018).
He, D. et al. Source data for 2D/2D phosphorene/BiOI S-scheme heterojunction for subminute photocatalytic water disinfection under real sunlight. Figshare https://doi.org/10.6084/m9.figshare.30645860 (2025).
Wu, T. et al. Solar-driven efficient heterogeneous subminute water disinfection nanosystem assembled with fingerprint MoS2. Nat. Water 1, 462–470 (2023).
Gao, P., Ng, K. & Sun, D. D. Sulfonated graphene oxide-ZnO-Ag photocatalyst for fast photodegradation and disinfection under visible light. J. Hazard. Mater. 262, 826–835 (2013).
Teng, Z. et al. Edge-functionalized g-C3N4 nanosheets as a highly efficient metal-free photocatalyst for safe drinking water. Chem 5, 664–680 (2019).
Wang, W. et al. Visible-light-driven photocatalytic inactivation of E. coli K-12 by bismuth vanadate nanotubes: bactericidal performance and mechanism. Environ. Sci. Technol. 46, 4599–4606 (2012).
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (42130705 (J.Q.), 42377048 (J.Q.), 42407082 (D.H.), 42422702 (Y.Z.), and 22176030 (Y.Z.)), China Postdoctoral Science Foundation (2022M720700 (D.H.)), and Fundamental Research Funds for the Central Universities (2412023QD027 (D.H.)).
Author information
Authors and Affiliations
Contributions
D.H. and K.Z. conceived and designed the experiments. C.L. and H.Y. conducted the DFT calculations. J.D. contributed to data analysis. D.H. wrote the manuscript. Y.Z. and J.Q. revised the manuscript and supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yongfa Zhu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
He, D., Zhang, K., Liu, C. et al. 2D/2D phosphorene/BiOI S-scheme heterojunction for subminute photocatalytic water disinfection under real sunlight. Nat Commun 17, 2267 (2026). https://doi.org/10.1038/s41467-026-69101-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69101-z