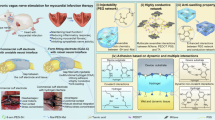
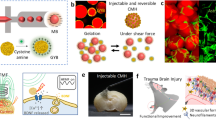
Abstract
Deep brain stimulation (DBS) is effective for treating neurological and psychiatric disorders. However, its tethered configuration, invasiveness, and limited tissue compatibility motivate wireless, minimally invasive alternatives. Here, we develop an in situ-gelled injectable conductive hydrogel (ICH), enabling wireless neuromodulation via electric-field localization under volume conduction. The ICH forms in vivo through bio-catalyzed polymerization and electrostatic self-assembly, yielding a stable, highly conductive, tissue-soft, and biocompatible network. Under high-frequency capacitive coupling, impedance difference between the ICH and surrounding brain tissue induces interfacial polarization and charge accumulation, locally concentrating the electric field to activate nearby neurons. This mechanism is supported by enhanced calcium signaling, increased c-Fos expression, and electrophysiological evidence of balanced basal ganglia-cortical activity. In a Parkinson’s disease rat model, ICH-mediated stimulation improved locomotor behavior, preserved dopaminergic neurons, and restored functional connectivity and structural integrity as revealed by fMRI. This injectable hydrogel bioelectronics provides a platform for minimally invasive, wireless neuromodulation therapies.
Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the article and its supplementary files. Any additional requests for information can be directed to, and will be fulfilled by the corresponding authors. Source data are provided with this paper.
Code availability
The custom pipeline used for fMRI data preprocessing and functional connectivity analysis in this study integrates established software packages and does not involve core analytical algorithms. The code is therefore available from the corresponding author upon reasonable request.
References
Fisher, R. S. & Velasco, A. L. Electrical brain stimulation for epilepsy. Nat. Rev. Neurol. 10, 261–270 (2014).
Kringelbach, M. L., Jenkinson, N., Owen, S. L. & Aziz, T. Z. Translational principles of deep brain stimulation. Nat. Rev. Neurosci. 8, 623–635 (2007).
Cagnan, H., Denison, T., McIntyre, C. & Brown, P. Emerging technologies for improved deep brain stimulation. Nat. Biotechnol. 37, 1024–1033 (2019).
Zhang, T. et al. Piezoelectric ultrasound energy–harvesting device for deep brain stimulation and analgesia applications. Sci. Adv. 8, eabk0159 (2022).
Singer, A. et al. Magnetoelectric materials for miniature, wireless neural stimulation at therapeutic frequencies. Neuron 107, 631–643 e635 (2020).
Wang, Q. et al. Lead-free dual-frequency ultrasound implants for wireless, biphasic deep brain stimulation. Nat. Commun. 15, 4017 (2024).
Lozano, A. M. et al. Deep brain stimulation: current challenges and future directions. Nat. Rev. Neurol. 15, 148–160 (2019).
Krauss, J. K. et al. Technology of deep brain stimulation: current status and future directions. Nat. Rev. Neurol. 17, 75–87 (2021).
Hescham, S. A. et al. Magnetothermal nanoparticle technology alleviates parkinsonian-like symptoms in mice. Nat. Commun. 12, 5569 (2021).
Zhang, Y. et al. Transcranial nongenetic neuromodulation via bioinspired vesicle-enabled precise NIR-II optical stimulation. Adv. Mater. 35, e2208601 (2023).
Kozielski, K. L. et al. Nonresonant powering of injectable nanoelectrodes enables wireless deep brain stimulation in freely moving mice. Sci. Adv. 7, eabc4189 (2021).
Munshi, R. et al. Magnetothermal genetic deep brain stimulation of motor behaviors in awake, freely moving mice. eLife 6, e27069 (2017).
Chen, R., Romero, G., Christiansen, M. G., Mohr, A. & Anikeeva, P. Wireless magnetothermal deep brain stimulation. Science 347, 1477–1480 (2015).
Wu, X. et al. Tether-free photothermal deep-brain stimulation in freely behaving mice via wide-field illumination in the near-infrared-II window. Nat. Biomed. Eng. 6, 754–770 (2022).
Kim, Y. J. et al. Magnetoelectric nanodiscs enable wireless transgene-free neuromodulation. Nat. Nanotechnol. 20, 121–131 (2025).
Kim, T. et al. Deep brain stimulation by blood–brain-barrier-crossing piezoelectric nanoparticles generating current and nitric oxide under focused ultrasound. Nat. Biomed. Eng. 7, 149–163 (2023).
Jin, S. et al. Instant noninvasive near-infrared deep brain stimulation using optoelectronic nanoparticles without genetic modification. Sci. Adv. 11, eadt4771 (2025).
Benfenati, F. & Lanzani, G. Clinical translation of nanoparticles for neural stimulation. Nat. Rev. Mater. 6, 1–4 (2020).
Deng, J. et al. Electrical bioadhesive interface for bioelectronics. Nat. Mater. 20, 229–236 (2021).
Liu, Y. et al. Soft and elastic hydrogel-based microelectronics for localized low-voltage neuromodulation. Nat. Biomed. Eng. 3, 58–68 (2019).
Chong, J. et al. Highly conductive tissue-like hydrogel interface through template-directed assembly. Nat. Commun. 14, 2206 (2023).
Liu, Y. et al. Morphing electronics enable neuromodulation in growing tissue. Nat. Biotechnol. 38, 1031–1036 (2020).
Inoue, A., Yuk, H., Lu, B. & Zhao, X. Strong adhesion of wet conducting polymers on diverse substrates. Sci. Adv. 6, eaay5394 (2020).
Zhang, J. et al. Engineering electrodes with robust conducting hydrogel coating for neural recording and modulation. Adv. Mater. 35, e2209324 (2023).
Cheng, S., Zhu, R. & Xu, X. Hydrogels for next generation neural interfaces. Commun. Mater. 5, 99 (2024).
Li, J. et al. PEDOT:PSS-based bioelectronics for brain monitoring and modulation. Microsyst. Nanoeng. 11, 87 (2025).
Li, W., Li, Y., Song, Z., Wang, Y.-X. & Hu, W. PEDOT-based stretchable optoelectronic materials and devices for bioelectronic interfaces. Chem. Soc. Rev. 53, 10575–10603 (2024).
Strakosas, X. et al. Metabolite-induced in vivo fabrication of substrate-free organic bioelectronics. Science 379, 795–802 (2023).
Wang, Y. et al. Dopant-enabled supramolecular approach for controlled synthesis of nanostructured conductive polymer hydrogels. Nano Lett. 15, 7736–7741 (2015).
Wang, Z., Chen, Z., Ma, Z. & Han, H. Label-free mode based on ferrocene/PEDOT:PSS–PPy for molecularly imprinted electrochemically ultrasensitive detection of amino acids. Anal. Chem. 96, 14298–14305 (2024).
Setka, M. et al. Raman and XPS studies of ammonia sensitive polypyrrole nanorods and nanoparticles. Sci. Rep. 9, 8465 (2019).
Boehler, C., Carli, S., Fadiga, L., Stieglitz, T. & Asplund, M. Tutorial: guidelines for standardized performance tests for electrodes intended for neural interfaces and bioelectronics. Nat. Protoc. 15, 3557–3578 (2020).
Kohn, P., Schroter, K. & Thurn-Albrecht, T. Interfacial polarization and field-induced orientation in nanostructured soft-ion conductors. Phys. Rev. Lett. 102, 216101 (2009).
Erfani, R., Marefat, F., Sodagar, A. M. & Mohseni, P. Modeling and characterization of capacitive elements with tissue as dielectric material for wireless powering of neural implants. IEEE Trans. Neural Syst. Rehab. Eng. 26, 1093–1099 (2018).
Huang, L. & Hu, A. P. Defining the mutual coupling of capacitive power transfer for wireless power transfer. Electron. Lett. 51, 1806–1807 (2015).
Theodoridis, M. P. Effective capacitive power transfer. IEEE Trans. Power Electron. 27, 4906–4913 (2012).
Wang, Z. et al. SAR and temperature: simulations and comparison to regulatory limits for MRI. J. Magn. Reson. Imaging 26, 437–441 (2007).
Xicoy, H., Wieringa, B. & Martens, G. J. The SH-SY5Y cell line in Parkinson’s disease research: a systematic review. Mol. Neurodegener. 12, 10 (2017).
Limousin, P. & Foltynie, T. Long-term outcomes of deep brain stimulation in Parkinson disease. Nat. Rev. Neurol. 15, 234–242 (2019).
Florence, G., Sameshima, K., Fonoff, E. T. & Hamani, C. Deep brain stimulation: more complex than the inhibition of cells and excitation of fibers. Neuroscientist 22, 332–345 (2015).
Herrington, T. M., Cheng, J. J. & Eskandar, E. N. Mechanisms of deep brain stimulation. J. Neurophysiol. 115, 19–38 (2015).
Prasad, A. et al. Comprehensive characterization and failure modes of tungsten microwire arrays in chronic neural implants. J. Neural Eng. 9, 056015 (2012).
Yuk, H., Lu, B. & Zhao, X. Hydrogel bioelectronics. Chem. Soc. Rev. 48, 1642–1667 (2019).
Shi, L. H. et al. High-frequency stimulation of the subthalamic nucleus reverses limb-use asymmetry in rats with unilateral 6-hydroxydopamine lesions. Brain Res. 1013, 98–106 (2004).
Chen, J. et al. Non-Faradaic optoelectrodes for safe electrical neuromodulation. Nat. Commun. 15, 405 (2024).
Palasz, E. et al. BDNF as a promising therapeutic agent in Parkinson’s disease. Int. J. Mol. Sci. 21, 1170 (2020).
Wu, N., Sun, X., Zhou, C., Yan, J. & Cheng, C. Neuroblasts migration under control of reactive astrocyte-derived BDNF: a promising therapy in late neurogenesis after traumatic brain injury. Stem Cell Res. Ther. 14, 2 (2023).
Burciu, R. G. et al. Functional MRI of disease progression in Parkinson disease and atypical Parkinsonian syndromes. Neurology 87, 709–717 (2016).
Tessitore, A., Cirillo, M. & De Micco, R. Functional connectivity signatures of Parkinson’s disease. J. Parkinsons Dis. 9, 637–652 (2019).
Zhao, S. et al. Full activation pattern mapping by simultaneous deep brain stimulation and fMRI with graphene fiber electrodes. Nat. Commun. 11, 1788 (2020).
Driscoll, N. et al. MXene-infused bioelectronic interfaces for multiscale electrophysiology and stimulation. Sci. Transl. Med. 13, eabf8629 (2021).
Boutet, A. et al. Predicting optimal deep brain stimulation parameters for Parkinson’s disease using functional MRI and machine learning. Nat. Commun. 12, 3043 (2021).
Subramanian, L. et al. Real-time functional magnetic resonance imaging neurofeedback for treatment of Parkinson’s disease. J. Neurosci. 31, 16309–16317 (2011).
Filippi, M., Sarasso, E. & Agosta, F. Resting-state functional MRI in Parkinsonian syndromes. Mov. Disord. Clin. Pract. 6, 104–117 (2019).
Pelled, G., Bergman, H., Ben-Hur, T. & Goelman, G. Manganese-enhanced MRI in a rat model of Parkinson’s disease. J. Magn. Reson. Imaging 26, 863–870 (2007).
Nyatega, C. O., Qiang, L., Adamu, M. J. & Kawuwa, H. B. Gray matter, white matter and cerebrospinal fluid abnormalities in Parkinson’s disease: a voxel-based morphometry study. Front. Psychiatry 13, 1027907 (2022).
Yang, K. et al. White matter changes in Parkinson’s disease. NPJ Parkinsons Dis. 9, 150 (2023).
Zhai, H. et al. Voxel-based morphometry of grey matter structures in Parkinson’s disease with wearing-off. Brain Imaging Behav. 17, 725–737 (2023).
Gillies, G. E., Murray, H. E., Dexter, D. & McArthur, S. Sex dimorphisms in the neuroprotective effects of estrogen in an animal model of Parkinson’s disease. Pharmacol. Biochem. Behav. 78, 513–522 (2004).
Gillies, G. E., Pienaar, I. S., Vohra, S. & Qamhawi, Z. Sex differences in Parkinson’s disease. Front. Neuroendocrinol. 35, 370–384 (2014).
Acknowledgements
This work was supported by the funds from the National Natural Science Foundation of China under grant No. 32471387 (awarded to Zhiqiang Luo) and No. 325B2052 (awarded to R.S.), and by the Ministry of Science and Technology of China under grant No. 2023YFF0714204 (awarded to J.W.). We would like to thank ZMT ZurichMedTech AG for providing Sim4Life software.
Author information
Authors and Affiliations
Contributions
Zhiqiang Luo, J.W., and C.Y. supervised the project. M.Y., W.L., P.C., Zhuang Liu, and R.S. designed the ICH and experiments. M.Y., W.L., P.C., N.Y., and Zhikun Li conducted fabrication and testing of materials. M.Y., W.L., Q.W., Bingqing Xue, and Dingke Zhang conducted the in vitro experiments. M.Y., R.S., C.G., and J.S conducted the in vivo rat experiments. M.Y. and Zhuang Liu conducted MRI experiments. M.Y., W.L., P.C., Zhuang Liu, R.S., C.Y., J.W., and Zhiqiang Luo prepared the manuscript. M.Y., W.L., Zhuang Liu, R.S., and Baochun Xu processed the data and drew the figures. C.M., C.G., Donghui Zhang, and J.F. polished the manuscript. All authors discussed and agreed with the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Hossein Montazerian, Daniel Simon, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, M., Liu, W., Chen, P. et al. Injectable hydrogel bioelectrostimulator for wireless deep brain neuromodulation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69226-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69226-1