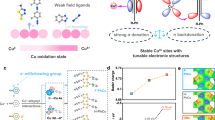
Abstract
Copper (Cu)-based single-atom catalysts (SACs) enable electrocatalytic CO2 reduction into methane (CH4) fuel for thermal power plant decarbonization, yet conventional Cu SACs face industrial deployment barriers like instability and sluggish kinetics caused by d − p orbital coupling. Herein, we develop a Cu–Ti1O3 catalyst with localized Cu single-atom sites by oxygen vacancy (Ov)-involved orbital engineering, achieving industrial-level CH4 production. Theoretical and in-situ studies reveal the intensification of the d − d coupling at Cu sites triggered by [Cu−Ov − Ti] motifs, which enhances d-π* polar interactions upon *CO2 and accelerates C − O bond cleavage in *OCH3 intermediate. As a result, Cu–Ti1O3 achieves a competitive performance, i.e., the highest Faradaic efficiency of 76% and a peak partial current density of 670 mA cm−2 toward CH4 (corresponding turnover frequency = 24,930 h−1), ~3.5-fold promotion over conventional Cu SACs. Furthermore, it demonstrates high durability (>1,230 hours) at an industrial-level current density, exceeding the longevity of conventional Cu SACs by over 20 times. Our findings highlight the prospect of d-orbital engineering in enabling industrial-level electrocatalytic methanation, offering promising implications for decarbonizing traditional power plants.
Similar content being viewed by others
Introduction
Carbon emissions from traditional thermal power plants significantly hinder carbon neutrality efforts, e.g., overseas coal-fired power plants currently emit 0.53 gigatons of CO2 per year1. To address this, abundant decarbonization technologies are being proposed, among which the electrocatalytic CO2 reduction reaction (CO2RR) emerges as a critical pathway for CO2 valorization and sustainable fuel production2,3. Specifically, electro-catalytic methanation (i.e., CO2-to-CH4 conversion) using flue gas provides a promising decarbonization strategy by coupling CO2 fixation with high-energy CH4 (55.5 MJ kg−1) production for thermal power plants4,5. Regarding catalyst candidates, copper (Cu)-based catalysts facilitate pathways for deeply reduced products, including CH4 (>2e⁻ transfer)6,7, but face challenges of sluggish kinetics (| Jtotal| <500 mA cm−2) and poor selectivity (<50%).
From a mechanistic perspective8,9, spatially coupled Cu–Cu sites promote C–C coupling over the CH4 pathway (e.g., 0.53 eV higher barrier for CH4 on bulk Cu10). Therefore, enhancing CH4 selectivity requires spatially isolated Cu single-atom catalysts (SACs). Current synthesis strategies for Cu SACs rely on embedding Cu into non-metallic matrices like carbon, C3N4, and so on (Supplementary Fig. 1a)6. For instance, N-coordinated Cu SACs have been confirmed to own a favored C1 selectivity (e.g., CO11, HCOOH12, and CH413,14). Nevertheless, the modern hard-soft acid-base (HSAB) theory15,16 illustrates that the inherently delocalized p-orbitals17 may cause a d-electronic delocalization via d–p orbital coupling, thus weakening hard-basic intermediate adsorption like *OCH3 and facilitating hydrogen evolution side reaction18 at Cu sites (Details in Supplementary Note 1)15. Moreover, Cu−X (X=C, N, O) coordinates are thermodynamically unstable19, causing severe reconstruction such as agglomeration of Cu atoms. This may facilitate C−C coupling20 and cause performance degradation under industrial-level current density (> 500 mA cm−2)21.
To these issues, constructing localized Cu single-atom sites (i.e., harder acid15) by introducing matrices with localized d/f valence orbitals may be essential22, and proposed solutions include constructing Cu1M single-atomic alloys (M = f/d-block elements) as well as Cu1/MOx SACs (Supplementary Fig. 1b, c). Although Cu1M systems feature localized d-orbitals, free-atom-like Cu sites (i.e., resembling d−p coupled molecular metal complexes) will cause an unstable condition23,24, thus favoring a tight *CO binding and predominant CO selectivity25. For Cu1/MOx SACs, the intrinsic charge polarization of M−O bonds26 and spin-orbit interaction triggered insurmountable energy barriers for direct d−d/f coupling27,28, finally causing dominative [Cu−O−M] motifs that facilitate indirect d−p−d/f coupling during catalyst synthesis29,30. In this indirect d−p−d/f coupling structure, the continuous electron-polaron accumulation at d-orbitals during CO2RR may destroy the Cu−O coordinates31, significantly threatening the robustness (Fig. 1a). For instance, Zhou et al. synthesized a Cu-Ce-Ox catalyst (i.e., d−p−f coupling) with isolated Cu sites32, achieving an efficient CO2-to-CH4 conversion (FE of CH4 (FECH₄) ≈ 65%), but durability remains poor (<10 h). Unlike typical electron-polaron trapping at metallic sites, electron in anatase titanium oxide (TiO2) prefers a free-carrier state, and can only be trapped at oxygen vacancy (Ov)31,33, providing a vital opportunity for Ov-induced metal-metal bond formation (strong d−d coupling, Fig. 1b), thereby constructing localized Cu single-atom sites (l-Cu sites) and stabilizing the active sites34,35. Inspired by these, employing Ov-rich TiO2 as matrices may be a promising strategy to realize strong d−d coupling and facilitate selective and stable methanation.
a Conventional Cu1/MOx SACs display a delocalized d−p−d orbital coupling involving lattice oxygen, causing two major challenges: sluggish kinetics and instability. b Electron-polaron accumulation at Ov sites induces strong d−d orbital coupling in a valent-variable system like Cu1/TiOx. c Synthesis of Cu–Ti1O3 via lattice O removal in Cu-embedded TiO2, obtaining a localized [Cu−Ov − Ti] motif with strong d−d orbital coupling. d Cu–Ti1O3 achieves enhanced d-π* polar interactions on *CO2 and accelerated C−O bond cleavage of *OCH3, thereby leading to highly efficient and stable CH4 production.
Herein, we have developed an oxygen-vacancy-involved orbital engineering method to construct [Cu−Ov−Ti] motifs through O removal in Cu-embedded TiO2 SAC (Fig. 1c). As-synthesized Cu-based SAC (marked as Cu–Ti1O3) achieves an efficient CO2-to-CH4 conversion in flow cells, including an excellent FECH4 of 76% at −0.88 V (potentials converting into the reversible hydrogen electrode (RHE), see “Methods”), and a record-high turnover frequency of CH4 to date (24,390 h−1 at −1.0 V). Furthermore, Cu–Ti1O3 also exhibits exceptional stability of >1230 h at an industrial-level current density (600 mA cm−2) in a zero-gap electrolyzer, over 12 times longer than a conventional Cu1/TiO2 catalyst with major [Cu−O−Ti] motifs (marked as Cu−O4(Ti)). Synergistic experimental/theoretical analyses reveal that Oᵥ-intensified d−d orbital coupling between Cu and Ti atoms enhances electronic localization of Cuδ+ sites (i.e., harder acidity than pristine Cuδ+ sites in Cu-O4(Ti)), strengthening d-π* polar interactions with adsorbed *CO2 (Fig. 1d) and stabilizing the Cu−O binding with hard-basic *OCH3. Simultaneously, in-situ time-field electrochemical electron magnetic resonance (E-EMR) reveals a new mechanism involving pseudo-lattice oxygen formation through *OCH3 insertion into Ov. This reversible pseudo-lattice oxygen formation route supplements a localized ultra-stable adsorption for *OCH3 surpassing the typical physico/chemical adsorption at conventional Cu single-atom sites (c-Cu sites) with majorly Cu−O coordination (i.e., bond dissociation enthalpy (BDE) of Cu−O = 263.1 vs. 41.9 kJ mol−1). Therefore, an accelerated C−O bond cleavage (~16.8% lower BDE) and rapid methanation generation on Cu–Ti1O3 are achieved compared with Cu-O4(Ti) (Supplementary Fig. 2). Overall, this work establishes an orbital engineering method for constructing localized Cu single-atom sites and provides a universal strategy to design durable SACs toward industrial-level CO2-to-fuel conversion.
Results
Synthesis and characterization
Density functional theory (DFT) calculations were first employed to evaluate the influence of d−d orbital coupling at Cu single-atom sites. The local density of states (LDOS) for Cu atom at [Cu−Ov−Ti] motifs exhibits intensified peaks relative to the conventional [Cu−O−Ti] structures, confirming localized electronic states of l-Cu sites (Fig. 2a, Supplementary Fig. 3). Integrated projected density of states (IPDOS) analysis identifies the dx2-y2 orbital as the frontier orbital of the Cu sites (Supplementary Fig. 4). The higher electron density within the dx2-y2 orbital (e.g., IPDOS = 0.980 for up-spin states, Fig. 2b and Supplementary Table 1) of l-Cu site likely facilitates strong d-π* polar-interactions on adsorbed *CO2, corroborating accelerated CO2 activation36,37, and leading to an enhanced CO2-to-CH4 conversion trend as reported by Huang’s group37. Furthermore, crystal orbital Hamilton population (COHP) analyses reveal a significantly larger negative integrated COHP (−ICOHP) value for the Cu−Ti bond on l-Cu sites (2.422 a.u.) versus c-Cu sites (0.002 a.u., Fig. 2c), indicating robust d−d coupling induced by oxygen vacancies. BDE assessments of *OCH3 confirm tighter Cu–O bonding post orbital engineering (BDECu–O = 263.1 kJ mol−1 on l-Cu sites, Fig. 2d) compared with unstable adsorption on pristine c-Cu sites (BDECu–O = 41.9 kJ mol−1, Supplementary Table 2). Meanwhile, a 16.8% reduction in BDEC−O at l-Cu sites suggests potential acceleration of C−O bond cleavage during CO2RR (inset slab model in Fig. 2d). Hence, d−d orbital coupling at l-Cu sites confers a kinetically enhanced methanation pathway.
a LDOS of Cu atom at l-Cu and c-Cu sites. b IPDOS of Cu−O−Ti and l-Cu sites around the Fermi level. c COHP analysis for d−d orbital coupling at c-Cu and l-Cu sites. d BDE evaluations illustrate that strong d−d coupling promotes C−O bond cleavage of *OCH3 on l-Cu sites compared with c-Cu sites. Source data for Fig. 2 are provided as a Source Data file.
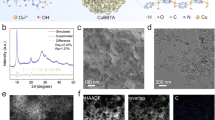
Guided by computational insights, we designed Cu–Ti1O3 SAC with partial Cu–Ti bonding toward CO2RR. In detail, Cu single-atom precursor was first fabricated through a hydrothermal method (Details in “Methods”), where Cu substitutes Ti in [TiO6] octahedra and forms Cu–O–Ti motifs38. Given that lattice oxygen removal facilitates adjacent metallic bonding39,40, Cu single-atom precursor was then calcined in 10% H2/Ar for 3 h (surface Cu: 2.67 wt%, Supplementary Table 3) to synthesize Cu–Ti1O3. As a control group, Cu–O4(Ti) catalyst was prepared by an air-calcination of Cu single-atom precursors. X-ray diffraction (XRD) patterns confirmed the major anatase phase (PDF#71-1167, Supplementary Fig. 5a) in Cu–Ti1O3, while Cu-related peaks were absent, indicating the lattice incorporation of Cu atoms. Compared to pristine TiO2, the (101) diffraction peak of Cu–Ti1O3 shifted to a smaller angle, which might result from the lattice expansion38. Meanwhile, Cu–Ti1O3 exhibited a higher 2θ angle for the (101) plane than Cu–O4(Ti), suggesting the lattice shrinkage caused by Ov accumulation (Supplementary Fig. 5b)41.
The morphologies and atomic structures of Cu–Ti1O3 and Cu–O4(Ti) were then examined by transmission electron microscopy (TEM) and aberration-corrected high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM). The TEM images illustrated a retained TiO2 particle size of ~20 nm after Cu embedding (Supplementary Fig. 6). High-resolution transmission electron microscopy (HRTEM) of the Cu–Ti1O3 revealed an ~0.194 nm interplanar spacing for the (200) plane (d200, Supplementary Fig. 6), indicating a lattice expansion compared with TiO2 (d200 = 0.189 nm). The energy-dispersive spectroscopy (EDS) mapping confirmed the uniform distribution of Cu, O, and Ti across the nanoparticles (Supplementary Fig. 7). In addition, HAADF-STEM revealed the dumbbell-shaped periodic arrays consisting of adjacent Ti atom pairs along the [100] direction in Cu–O4(Ti) and Cu–Ti1O3 (Fig. 3a, b). Bright spots that refer to Cu atoms were exclusively located at the original Ti sites, indicating the on-site substitution and formation of Cu single-atom sites in Cu–Ti1O3 and Cu–O4(Ti). Moreover, in the [100]-directed images (Fig. 3c), the distance between one bright (Cu) and one dark spot (Ti) in Cu–Ti1O3 was about 2.83 Å, shorter than that in Cu–O4(Ti) (2.97 Å), suggesting an atom approaching and a possible direct Cu−Ti bond formation triggered by Ov.
Aberration-corrected HAADF-STEM images of a Cu–Ti1O3 and b Cu–O4(Ti) along the [100] direction. c Left panel: Front and top views of Cu–Ti1O3, where the purple, blue, and red spheres represent the Cu, Ti, and O atoms, respectively. Right panel: the measured distance between the adjacent Cu and Ti atoms on Cu–Ti1O3 and Cu–O4(Ti). d Normalized XANES spectra at Cu K-edge. e Fourier-transformed k2-weighted EXAFS spectra at Cu K-edge for Cu foil, Cu2O, CuO, Cu–Ti1O3 and Cu–O4(Ti) samples. Source data for Fig. 3c–e are provided as a Source Data file.
Interfacial electronic states were then assessed by X-ray photoelectron spectroscopy (XPS). The high-resolution spectra of O 1s showed a higher Ov peak at 531.2 eV42 for Cu–Ti1O3 than Cu–O4(Ti) (ca. 18.5% vs. 10.8%, Supplementary Fig. 8). As for Cu 2p spectra (Supplementary Fig. 9a), Cu–O4(Ti) catalyst showed two main deconvoluted peaks at around 951.7 eV and 932.0 eV, respectively, attributed to Cu0/Cu+ species15. These two peaks exhibited a ~0.40 eV red-shift in Cu–Ti1O3, demonstrating an intensified electron density at interfacial Cu sites43. Cu LMM Auger spectra unveiled that both catalysts exhibited two fitted peaks at 917.7 and 916.7 eV (Supplementary Fig. 10), ascribed to the Cu2+ and Cu+ species, respectively44. In detail, Cu–Ti1O3 possessed a higher ratio of Cu+ with 86.1% compared with that of Cu–O4(Ti) with 67.9%, signifying the low valence state of surface Cu. In contrast, Ti 2p3/2 and 2p1/2 peaks showed a slight blue-shift (~0.13 eV) post orbital engineering, suggesting an electron outflow at interfacial Ti sites (Supplementary Fig. 9b)45. Taken together, a strong d−d coupling may appear in Cu–Ti1O3, which modulates the local electron density distribution and results in l-Cu sites46. Moreover, the structures of these two catalysts were also studied by Raman spectroscopy (Supplementary Fig. 11). The bands at 637, 517, 395, 200, and 146 cm−1 were observed for all of Cu SACs and TiO2, which could be ascribed to the vibrational mode of Eg(3), A1g & B1g(2), B1g(1), Eg(2), and Eg(1), respectively45. The Eg(1) band in Cu–Ti1O3 showed the highest wavenumber among all samples, indicating the highest Ov concentration45.
To further investigate the electronic states and coordination environment at Cu sites, X-ray absorption spectroscopy (XAS) was conducted at the Cu K-edge. X-ray absorption near-edge fine structure exhibited that the adsorption edges and corresponding first derivative peaks of two Cu SACs lay between those of Cu2O and CuO references (Fig. 3d and Supplementary Fig. 12), suggesting the existence of major Cuδ+ species in Cu SACs. The peak position of Cu–Ti1O3 catalyst (8982.3 eV) was lower than Cu–O4(Ti) catalyst (8982.7 eV, Supplementary Fig. 12), further indicating localized electronic states of l-Cu sites when Ov accumulation, consistent with the results of XPS. As for extended X-ray absorption fine structure (EXAFS) spectra, the Fourier-transformed EXAFS exhibited an intense peak at R ≈ 1.92 Å for Cu–O4(Ti) (Fig. 3e and Supplementary Fig. 13; Supplementary Table 4), which may be ascribed to Cu−O bonds15. While the wavelet-transformation analyses revealed the absence of Cu−Cu bonds in Cu–O4(Ti), confirming the formation of Cu−O−Ti motifs where an average Cu−Ti interatomic distance of 2.97 Å was observed (Supplementary Fig. 14)16. While in Cu–Ti1O3, a peak at R = 2.83 Å appeared, and wavelet-transformed EXAFS revealed a high-energy region at ~6.53 Å−1 (Supplementary Fig. 14), which together confirms the formation of Cu−Ti bonds46. The generation of Cu−Ti bonds, originating from Ov accumulation, implies a strong d−d orbital coupling in Cu–Ti1O3.
Electrochemical measurements
The catalytic performance of CO2RR was then tested in a flow cell with 1 M KOH electrolyte (Details in “Methods”). The linear sweep voltammetry curves with the CO2 flow revealed a higher reaction current for Cu–Ti1O3 than Cu–O4(Ti) (Supplementary Fig. 15). in situ electrochemical impedance spectroscopies (EIS) at –0.88 V were executed, which showed that Cu–Ti1O3 obtained lower charge-transfer resistance compared with Cu–O4(Ti) (Supplementary Fig. 16 and Table 5). Corresponding advanced distributed relaxation times analyses exhibited two characteristic peaks at –0.88 V over two catalysts, i.e., P2 and P3, which refer to the charge-transfer process of CO2RR and HER, respectively47. The interfacial charge transfer impedances of CO2RR on Cu–Ti1O3 were calculated as 48.62 Ω, lower than that over Cu–O4(Ti) (123.7 Ω), while the trend was opposite toward HER (Supplementary Table 6), indicating a more favorable CO2RR pathway on Cu–Ti1O3.
In electrolysis experiments, Cu–Ti1O3 achieved a peak FECH₄ of 76% at −0.88 V, about 2.2-fold higher than Cu–O4(Ti) (Fig. 4a and Supplementary Fig. 17). The FE ratios of CH4 to 2e−-product (CO + HCOOH) were calculated as 14.2 on Cu–Ti1O3, much larger than that of Cu–O4(Ti) (Fig. 4b and Supplementary Fig. 18), indicating a favorable multi-electron (>2e−) pathway on Cu–Ti1O3. Furthermore, partial current density of CH4 (JCH₄) on the Cu–Ti1O3 reached a maximal value of 670 mA cm−2 at −1.00 V (i.e., turnover frequency of CH4 (TOFCH₄) = 24,930 h−1, Fig. 4c), surpassing Cu–O4(Ti) (JCH₄ = 205 mA cm−2, TOFCH₄ = 7100 h−1). The corresponding JCH₄ normalized by electrochemically active surface area (ECSA) was also calculated to eliminate the influence of the reaction surface, and Cu–Ti1O3 delivered a 2.5 times greater current density than Cu–O4(Ti) (Supplementary Fig. 19). Furthermore, the energy efficiency (EE) of Cu–Ti1O3 attained a peak value of 38.7% at −0.88 V, exceeding that on Cu–O4(Ti) (19.2%, Fig. 4d). By tuning the CO2 flow rate to 50 sccm, the Cu–Ti1O3 achieved the largest FECH₄ of 79% at around −0.88 V, while the peak FECH₄ of the Cu–O4(Ti) was 38% (Supplementary Fig. 20). The pristine TiO2 showed that H2 was dominant product (FE > 95%) with no detection of carbonous product, confirming that Cu sites on the support facilitate the CO2RR (Supplementary Fig. 21). For mechanistic insight, Ov-free Cu1/MgO(Al) without other d-block elements was then synthesized and tested (Details in Supplementary Note 2, XRD/XPS/TEM/EDS in Supplementary Figs. 22 and 23), exhibiting a FECH₄ of less than 30% (Supplementary Fig. 24), further suggesting the essential role of Ov-intensified d−d orbital coupling. We then study the influence of impurities such as sulfur dioxide (SO2) and oxygen (O2) on the CO2RR performance of Cu–Ti1O3 at −0.88 V. Under pure CO2 flow, the Cu–Ti1O3 realized a high FECH₄ of ~76%. However, upon introducing 1% SO2 (~104 ppm) at t = 0.5 h, the total FE, along with FECH₄, decreased markedly (Supplementary Fig. 25a), which can be attributed to the preferential reduction of SO248. After 1 h of electrolysis (including a feed of 1% SO2 for 30 min), the FECH₄ recovered to >66%, suggesting that the influence of SO2 on the catalyst structure is largely reversible. A similar reversible trend was observed when feeding 10% O2 (~105 ppm, Supplementary Fig. 25b).
a Current densities (top) and FEs (bottom) of CO2RR product and H2 on Cu–Ti1O3 catalyst at different applied potentials in 1 M KOH (with 90% iR-correction). b FEs and corresponding ratios of CH4: (HCOOH + CO) on Cu–Ti1O3 and Cu–O4(Ti) at around −0.88 V. c TOFs of CH4 on Cu–Ti1O3 and Cu–O4(Ti) catalysts at different applied potentials (with 90% iR-correction). d Energy efficiency (EE) of CH4 on Cu–Ti1O3 and Cu–O4(Ti) catalysts (with 90% iR-correction). Cell resistance and pH of 1 M KOH electrolyte were measured as 0.98 ± 0.06 Ω and 14.00 ± 0.02, respectively, with an electrode surface area of 1 cm2. e Stability tests in a 5-cm2 zero-gap electrolyzer with 0.1 M KHCO3. f Comparison of this work with state-of-the-art CO2RR electrocatalysts, including JCH₄, CH4 selectivity, EECH₄, stability and TOFCH₄. Data are presented as mean ± s.d. (n = 3), with the error bars representing the s.d. Source data for Fig. 4 are provided as a Source Data file.
Both two catalysts were further integrated into a 5-cm2 zero-gap electrolyzer to evaluate the potential of our strategies for practical applications towards the CH4 formation (Supplementary Fig. 26). The Cu–Ti1O3 exhibited a higher FECH₄ of 75% than that of Cu–O4(Ti) (37%) at a constant current density of –0.6 A cm–2 (Supplementary Fig. 27). Catalyst stability was then evaluated at –0.6 A cm−2. Cu–Ti1O3 maintained continuous operation for >1230 h at an industrial current density of –0.6 A cm−2, exhibiting a cell voltage of −4.05 ± 0.16 V and an average FECH₄ of ~71% (Fig. 4e). Post-test XRD, HRTEM, aberration-corrected HAADF-STEM, and XPS confirmed structural integrity via persistent d−d coupling (Supplementary Figs. 28–32). In contrast, Cu–O4(Ti) showed rapid degradation: FECH₄ dropped to 23% within 64 h under identical conditions, with observed Cu nanoparticle formation that suggests Cu−O bond cleavage and metallic atomic aggregation. In addition, the Cu content of Cu–Ti1O3 after reaction was almost unchanged, yielding less Cu dissolution (5 ppb vs. 65 ppb for Cu–O4(Ti), Supplementary Tables 7 and 8), implying screened Cu corrosion. From the perspective of reaction activity and stability, Cu–Ti1O3 surpassed most of the advanced Cu SACs in CH4 production metrics (Fig. 4f and Supplementary Table 9). Moreover, techno-economic assessments based on a power plant model (Details in Supplementary Note 3 and Supplementary Fig. 33), utilizing established literature parameters21,49 and the durability data presented herein (Supplementary Table 10), demonstrate that a profitable pathway is feasible for this CO2-to-CH4 conversion system under a given carbon price (Supplementary Fig. 34).
Mechanism investigations
To gain insight into the role of orbital engineering in modulating the reaction mechanism, a series of in situ electrochemical spectroscopies was employed. In situ time-field E-EMR tracked Ov dynamics and intermediates during CO2RR. For Cu–Ti1O3, the time-field scans suggested that a signal related to Ov (centered at g-tensor of 1.997) maintained well after 10-min CO2 injection, excluding the influence of CO2 adsorption on the Ov concentrations (Fig. 5a and Supplementary Fig. 35). However, Ov gradually faded out under applied potential, and recovered when the potential was revoked (Fig. 5a). On the contrary, Cu–O4(Ti) catalyst exhibited a maintained Ov intensity throughout the catalytic process (Supplementary Fig. 36). Further quantitative spin counting (Fig. 5b) showed an initial Ov concentration of 1.08 × 1017 spins in Cu–Ti1O3, approximately 3 times higher than that in Cu–O4(Ti) (3.52 × 1016 spins). Under applied potential, Ov concentration of Cu–Ti1O3 dropped to 3.78 × 1016 spins, while recovering to 1.08 × 1017 spins at open-circuit potential (OCP), suggesting an intermediate-mediated (e.g., *OCH3) reversible Ov occupancy process. We then used heavier deuterium (D) from D2O solvent as the source of H to investigate the Ov concentration variation during CO2RR. Under applied potential, Ov concentration decreased to 5.81 × 1016 spins, higher than that using H2O as solvent under the same condition (Supplementary Fig. 37). As hydrogenation activity is weakened in D2O50, lower *OCD3 was produced during CO2RR, hence causing a lower occupancy rate of Ov. To reveal these short-lived intermediates, the spin traps were spiked under flow employing 5,5-dimethyl-1-pyrroline N-oxide (DMPO)51 (Supplementary Fig. 38). A typical six-finger peak (g = 2.0055, AN = 1.35 mT, AHβ = 0.74 mT) appeared (Fig. 5c), exhibiting a negative correlation with Ov, which demonstrated the replacement of Ov by alkoxy (i.e., *OCH3). Subsequently, this signal disappeared during the power outage, and signals associated with Ov gradually reverted to their initial state. The Ov concentration of Cu–Ti1O3 was also probed during water electrolysis, which retained well, indicating that H2O and *OH adsorption have only a minor influence on Ov stability (Supplementary Fig. 39). Signal reversibility during potential interruption confirmed Ov’s dynamic participation in CO2RR on Cu–Ti1O3, revealing a new *OCH3 insertion mechanism (Supplementary Fig. 40). Notably, as methoxyl radicals (*OCH3) are typically poorly detectable in bulk water solution, this result is more indicative of an association with the electrochemical interface. By regulating the calcination condition, the Ov concentration could be controlled rationally (Supplementary Fig. 41). EMR spectra showed that the Ov concentration of catalyst increased to higher values with 1.58 × 1017 spins when calcined in 10% H2/Ar for 4 h, which could further improve the FECH₄ to 78%. However, the TOFCH₄ per Ov of Cu–Ti1O3 showed the maximum value with 1.62 × 10−13 h−1 spin−1 (Supplementary Fig. 41e), indicating the most efficient CH4 formation on l-Cu sites.
a In-situ time-field E-EMR spectra in CO2-saturated 0.1 M KHCO3 at −0.88 V over Cu–Ti1O3 (with 90% iR-correction), referring to in-situ Ov evaluation. b Quantitative spin counts of signals related to Ov versus time over Cu–Ti1O3 and Cu–O4(Ti). c In-situ time-field E-EMR spectra with spin-trap agent (i.e., DMPO) in CO2-saturated 0.1 M KHCO3 at −0.88 V over Cu–Ti1O3 (with 90% iR-correction), corresponding to the in-situ concentration assessments for both Ov and *OCH3 intermediate during CO2RR. d In-situ Raman spectra of Cu–Ti1O3 and e Cu–O4(Ti) during a 10-min CO2RR in CO2-saturated 0.1 M KHCO3 at −0.88 V (with 90% iR-correction). Cell resistance and pH of electrolyte were measured as 0.98 ± 0.06 Ω and 6.83 ± 0.03, respectively, with an electrode surface area of 1 cm2. Source data for Fig. 5 are provided as a Source Data file.
The quasi-operando XPS also revealed the dynamic switch of Ov during CO2RR from a semi-quantitative perspective. For Cu–Ti1O3, Ov content decreased to 15.9% after 10-min electrolysis (intermediate-retained state), while recovering upon degassing (Supplementary Fig. 42a). This reversible change indicates pseudo-lattice oxygen formation by adsorbed intermediates. Concurrent Ti 2p binding energy upshift correlated with Ov depletion (Supplementary Fig. 42b), further evidencing intermediate-induced electronic perturbation. In stark contrast, Cu–O4(Ti) showed negligible Ov/Ti 2p changes (Supplementary Fig. 42c, d), confirming Cu−O−Ti motifs’ minimal reactivity impact. Complementary in-situ Raman (Supplementary Fig. 43) also revealed a reversible downshift of the Eg(1) band (147→143 cm−1) in Cu–Ti1O3 during reaction (Fig. 5d), while remaining invariant in Cu–O4(Ti) (Fig. 5e), consistent with the *OCH3 insertion mechanism mentioned above. These results elucidate the unique mechanistic pathway governing CH4 formation on Cu–Ti1O3, mediated through d−d orbital coupling at l-Cu sites.
CO2 temperature-programmed desorption (CO2-TPD) was then carried out to assess the CO2 adsorption capacity of the Cu SACs (Supplementary Fig. 44). Cu–Ti1O3 displayed a large chemisorption peak at a higher temperature (455 °C), compared with the weak chemisorption peak of Cu–O4(Ti) at 430 °C, manifesting the intensive CO2 adsorption ability52. In situ attenuated total reflection surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS, Supplementary Fig. 45) was then employed to probe intermediates. On Cu–Ti1O3, the 1355 cm−1 peak (i.e., *COOH)12 at −0.1 V disappeared distinctly at −0.5 V, indicating rapid conversion to *CO (Fig. 6a). Both catalysts exhibited linear-bonded *CO (*COL) at ~2075 cm−1 (Fig. 6a, b)53,54, with Cu–Ti1O3 showing stronger intensity over wider potentials, reflecting higher *CO coverages. COL signal on Cu–O4(Ti) red-shifted with increasing applied potential, ascribing to the Stark effect55. In contrast, COL peak on Cu–Ti1O3 intensified and blue-shifted with the potential increasing to –1.1 V. This phenomenon is ascribed to dipole–dipole coupling resulting from the increased *CO coverage at localized Cuδ+ sites and adjacent Ti atom55,56. The in-situ ATR-SEIRAS spectra of pristine Au film, which served as the SEIRAS-active template, were also measured during CO2RR (Supplementary Fig. 46). A peak observed at ~2100 cm−1 was assigned to adsorbed *CO57, confirming that the detected *COL peak on the Cu–Ti1O3 surface indeed stems from Cu–*CO interaction. The in-situ Raman spectra showed a band at around 358 cm−1 (Supplementary Fig. 47), which was attributed to the frustrated rotation of adsorbed *CO on Cu58. A distinct *CO signal at low cathodic potential further highlights the higher *CO coverage on Cu–Ti1O3 surface. Moreover, C–H bending modes of *OCH3 appeared53, yet notably red-shifted on Cu–Ti1O3 (1380 cm−1) versus Cu–O4(Ti) (1398 cm−1). This 18 cm−1 shift signifies reduced C–H vibrational frequency, consistent with stabilized *OCH3 insertion at Ov mentioned above35,59. Meanwhile, earlier emergence and intensified *OCH3 signal on Cu–Ti1O3 further evidence facilitated *CO hydrogenation toward CH4.
In-situ ATR-SEIRAS spectra on a Cu–Ti1O3 and b Cu–O4(Ti) in a potential window from OCP to −1.1 V in CO2-saturated 0.1 M KHCO3 (with 90% iR-correction). Cell resistance and pH of electrolyte were measured as 0.98 ± 0.06 Ω and 6.83 ± 0.03, respectively, with an electrode surface area of 1 cm2. c The charge density difference analyses of *CO2 on Cu–Ti1O3 and Cu–O4(Ti). Green isosurfaces indicate negative-charged regions, and the yellow isosurfaces refer to positive-charged regions when CO2 is adsorbed on catalysts. d Free energy diagram of CO2-to-CH4 conversion on Cu–Ti1O3 and Cu–O4(Ti) at 298.15 K. Source data for Fig. 6a, b, d are provided as a Source Data file.
DFT calculations were then carried out to assess the effect of orbital engineering on catalytic performance (Model details in Supplementary Data 1). The CO2 adsorption features were first evaluated (Supplementary Table 11), and detailed charge density difference (CDD) analyses revealed an enhanced electron donation from Cu sites to *CO2 on Cu–Ti1O3 (Fig. 6c). The highest CDD value on Cu–Ti1O3 was calculated as −1.8× 10−3 e Bohr−3 (63.6% larger than that on Cu–O4(Ti)), showing a negative charge cloud (blue color in Fig. 6c) surrounded *CO2, indicating a stronger d-π* polar-interaction. Moreover, the free energy diagrams illustrate how the CO2RR pathways proceed on Cu single-atom sites (Fig. 6d). The overall free energy change (ΔG) for methanation (i.e., *CO2→* + H2O(l)+CH4(g)) on both Cu SACs shows energy downhill, indicating a thermodynamically favorable methanation. With the Brønsted−Evans−Polanyi relation, we further assessed the reaction kinetics in CO2-to-CH4 conversion. For *CO2→*COOH conversion, Cu–Ti1O3 displayed a 0.52 eV smaller energy rise relative to Cu–O4(Ti), suggesting a promoted CO2 activation resulting from stronger d-π* polar-interactions36,37, which has been mentioned in Fig. 2b. For the vital *OCH3 intermediate, ΔG for *OCH2→*OCH3 step was only 0.01 eV on Cu–Ti1O3, ~100% reduction from that on Cu–O4(Ti), demonstrating the accelerated kinetics induced by the new *OCH3 insertion mechanism at l-Cu sites. Meanwhile, ΔG for hydrogenation of *OCH3 was 1.57 eV and 0.97 eV on Cu–O4(Ti) and Cu–Ti1O3, respectively (Fig. 6d), revealing faster *OCH3-to-*CH4 converting kinetics at l-Cu sites. Taken together, the DFT calculations indicate a more favorable CO2-to-CH4 conversion on Cu–Ti1O3, which is attributed to the localization of Cu electronic states and subsequently enhanced d-π* polar-interaction and Cu–O bonding.
Discussion
In summary, we employed oxygen-vacancy-involved orbital engineering to construct localized Cu single-atom sites on Ov-rich TiO2 toward efficient methanation. Oxygen vacancies intensify d−d orbital coupling between Cu and Ti, heightening the electronic localization at l-Cu sites. This coupling strengthens the d-π* polar-interaction on *CO2, and helps switch the O in *OCH3 to pseudo-lattice oxygen. These synergistic effects promote the CO2 activation and targeted cleavage of the C−O bond in *OCH3, thus driving efficient CH4 production. In electrocatalytic experiments, Cu–Ti1O3 catalyst achieves a FECH₄ of 76%, a peak TOFCH₄ of 24,390 h−1, and record-long durability of >1230 h at industrial-level current densities. This work highlights the prospect of d-orbital engineering in enabling industrial-level electrocatalytic methanation on Cu SACs, and provides a promising catalyst design strategy toward sustainable CH4 production for decarbonizing traditional thermal power plants.
Methods
Chemicals and materials
Potassium hydroxide (KOH, ≥85.0%), Sodium hydroxide (NaOH, ≥85.0%), acetone (99.7%), ethanol (99.7%), glycerol (99.7%), Copper chloride dihydrate (CuCl2·2H2O, ≥99.0%), Titanium oxysulfate dihydrate (TiOSO4·2H2O, ≥99.0%), Magnesium nitrate hexahydrate (Mg(NO3)2·2H2O, ≥99.0%) and Aluminum nitrate nonahydrate (Al(NO3)3·9H2O, ≥99.0%) were purchased from the Sinopharm Chemical Reagent Company. Ni foam was purchased from the Fangdou Technology Co., LTD (99% metal basis) with 0.3 mm thickness and 0.1 mm pore size. The Nafion solution (Dupont, D-520 dispersion, 5% w/w in water and 1-propanol) and triethylene glycol were purchased from Alfa Aesar. Gas diffusion layer (SGL Carbon, 28BC) was purchased from Fuel Cell Store. The hydroxide exchange membrane (Fumasep FAB-PK-130, 3 × 2 cm) for the flow cell was purchased from Fumatech, which was activated by soaking in 1 M KOH for 24 h for further test. The hydroxide exchange membrane (Grade 60, 3 × 3 cm) for the zero-gap electrolyzer was purchased from Dioxide Materials, which was activated by soaking in 1 M KOH for 24 h before the test. CO2 gas (99.99%), Ar gas (99.99%), N2 gas (99.99%), and all the standard gases were purchased from Shanghai Tomoe Gas Company.
Fabrication of Cu–Ti1O3
In a typical procedure, 2.2 g TiOSO4·2H2O, 0.15 g CuCl2·2H2O, 12 mL deionized water, 6 mL glycerol, and 20 mL ethanol were mixed and stirred for 1 h. The mixed solution was then moved into a 100 mL autoclave and kept at 200 °C for 8 h, followed by natural cooling to room temperature. Finally, the precipitate was washed thoroughly with alcohol and dried at 60 °C in a vacuum oven overnight. The sample was then calcined at 300 °C in 10% H2/Ar for 3 h to obtain the Cu–Ti1O3 catalyst.
Fabrication of Cu–O4(Ti)
Cu–O4(Ti) were prepared in a similar way as Cu–Ti1O3 nanoparticles, except that the final sample was calcined at 300 °C in the air for 3 h.
Preparation of electrodes
For the CO2RR in a flow cell, a well-dispersed catalyst slurry containing 18 mg of catalysts, 3 mL of acetone, and 120 μL of Nafion (Dupont, D-520 dispersion, 5% w/w in water and 1-propanol) ionomer solution was slowly dropped onto a gas diffusion layer (SGL Carbon, 28BC), attaining a catalyst loading of ~0.3 mg cm−2 measured by 0.01 mg analytical balance.
Materials characterizations
XRD was tested on a Bruker SMART APEX (II)-CCD. XPS data were characterized by a Thermo Scientific K-Alpha XPS. Scanning electron microscopy measurements were carried out by a Hitachi S4800 SEM. HRTEM measurements were performed by a JEM 2100 F (JEOL, Japan). 1H-nuclear magnetic resonance (1H-NMR) measurements were performed on a Bruker AVANCE AV III HD 500. Moreover, Athena and Artemis software with a standard IFEFFIT package were used to process XAS data.
Electrocatalytic CO2 reduction measurements
For CO2RR in flow-cells, the electrochemical tests were conducted using a 1-cm2 cell with a Gamry (Model: Reference 3000) electrochemical workstation at room temperature (25 ± 1 °C). The flow-cell configuration consists of a gas diffusion layer loaded with catalysts as the working electrode, a Ni foam as the anode, an Ag/AgCl electrode as the reference, and an anion exchange membrane to separate the cathode and anode chambers (Supplementary Fig. 48). The chamber volume of the cathode and anode is 0.5 cm3. All applied potentials were converted to the RHE scale according to the Nernst equation:
The electrode potential was iR-corrected using the following equation:
where Esample was the corrected potential at the cathode, Eapplied was the applied potential, and Itotal was the total current. Rcell was the cell resistance of 0.98 ± 0.06 Ω, which was measured via potentiostatic EIS.
1 M KOH solution (pH = 14.00 ± 0.02) was utilized as the electrolyte that was prepared before electrochemical tests to ensure freshness. The reaction was performed with a steady stream of CO2 (30 mL min−1) measured by a soap bubble flowmeter. The gas product was collected and analyzed by a gas chromatograph (Shanghai Ramiin GC 2060) while the liquid products were quantified by 1H-NMR spectroscopy (Bruker AVANCE AV III HD 500) using a method previously reported60.
The gas product FE was calculated by the following equation:
where N was the number of charge transfers (H2: 2; CH4: 6; C2H4: 8); Sx was the peak area of the gas product and S was the peak area of corresponding standard gas; C was the concentration of standard gas; v was the flow rate of CO2; I was the real-time current during CORR; F was the Faraday constant (96485 C mol–1).
The liquid product FE was calculated by the following equation:
where N was the number of charge transfers (Acetate: 4; EtOH: 8; n-PrOH: 12); V was the electrolyte volume (20 mL); C was the concentration of products; Q was the reaction charge.
The ECSA was calculated by assuming a monolayer of Pb adatoms over a Cu surface and 2e− Pb oxidation with a conversion factor of 310 μC cm−2 61. The EIS was performed at an open-circuit potential state in the frequency range from 100 kHz to 100 MHz. The Pb underpotential deposition was conducted in an Ar-saturated 0.1 M HClO4 aqueous solution with 10 mM Pb(ClO4)2 by using cyclic voltammetry. This is the deposition of an adlayer of a foreign metal (i.e., Pb2+) on a substrate (i.e., surface Cu site) at potentials higher than the thermodynamic reduction potential of the electrodeposited metal. The foreign metal tends to deposit on the Cu rather than TiO2 due to higher electrical conductivity, leading to accurate deposition on the Cu surface62. Before the experiment, the potential was first set at −0.20 V vs. RHE for 150 s, then swept from −0.0840 to 0.161 V vs. RHE with a sweep rate of 10 mV s−1.
Stability tests in a zero-gap electrolyzer
The zero-gap electrolyzer (Shanghai Keqi, active area 5 cm2, Supplementary Fig. 26) was utilized, which was established with a well-established membrane electrode assembly containing a cathode electrode, an anion membrane (Dioxide Materials, Grade 60), and an anode electrode (Ti mesh loaded with 2 mg cm−2 IrOx). 0.05 M HCl-humidified CO2 was supplied with a flow rate of 30 sccm through the flow channels in the cathode flow field to reduce (bi)carbonate salt accumulation according to previous work63, and 0.1 M KHCO3 (pH = 6.83 ± 0.03) was fed into the anode flow channels with a flow rate of 15 sccm. The stability tests were performed at a constant current of −3 A. During operation, the anolyte was replaced regularly, and the precipitated salt was further removed by separating the device to inhibit channel block and active site shield.
Calculation for EE
For flow-cell systems, the \({{{\rm{EE}}}}_{{{{\rm{CH}}}}_{4}}\) was calculated based on the assumption that the overpotential of oxygen evolution is zero. The methane \({{{\rm{EE}}}}_{{{{\rm{CH}}}}_{4}}\) can be calculated as follows:
where Eapplied is the potential applied in the experiment, \({{{\rm{FE}}}}_{{{{\rm{CH}}}}_{4}}\) is the measured FE of CH4 in percentage, and \({E}_{{{{\rm{CH}}}}_{4}}\) = 0.17 V vs. RHE for CO2RR64.
For the zero-gap electrolyzer, \({{{\rm{EE}}}}_{{{{\rm{CH}}}}_{4}}\) is calculated as follows:
where Efull-cell applied is the full-cell voltage applied in the zero-gap electrolyzer.
Calculation for TOF
The TOF of CH4 is calculated according to the following equation:
Where n is the number of electrons transferred to CH4 with 8, F is the Faradaic constant with 96485 C mol−1, mcat. is the catalyst mass in the electrode, \({{{\rm{\alpha }}}}\) is the mass ratio of surface Cu active sites in the catalyst measured by XPS, and MCu is the atomic mass of Cu.
In-situ electrochemical electron magnetic resonance
The in-situ E-EMR was carried out on a self-made in-situ cell according to our previous report with the employment of a magnetic resonance spectrometer (CIQTEK, EPR200, China)65. The 0.17 M of DMPO (Dojindo Laboratories, Japan) was selected as the trap agent without additional purification. In the Schlenk line, the just-prepared electrode was cleaned in argon and then pumped with CO2. The electrochemical analyzer (CHI852D, CH Instrument, USA) applied multi-step potentials and recorded current response with sequential 10 min of 0, −0.88, and 0 V vs. RHE. The collaboration between magnetic resonance and electro-analysis was ensured through a serial communication port. The obtained raw data were processed and analyzed based on Easyspin 6.0.666,67.
In-situ electrochemical infrared spectroscopy experiments
The in-situ attenuated total reflection surface enhanced infrared absorption (ATR-SEIRAS) measurements were conducted on a home-made spectro-electrochemical cell (Supplementary Fig. 31). A ~60-nm-thick SEIRA-active Au film was pre-deposited on the reflection plane of a semicircular cylinder Si prism. Then, the catalyst was drop-cast on the above film. The operando ATR-SEIRAS measurements were performed in an optical system embedded in the spectroscopic chamber. To reach a high signal/noise ratio, the spectrum was obtained by averaging 256 interferograms. The incident angle of the IR beam was set to 55°. The spectra were shown as absorbance defined as A = ‒logI1/I2, where I1 and I2 represent the irradiation intensity of incident and reflective beams, respectively.
For nano Au film preparation, a hemicylindrical Si prism was polished and then cleaned with the RCA method: soaking it in a 1:1:5 (v/v) solution of NH3, H2O2, and H2O, and afterward in a 1:1:5 (v/v) solution of HCl, H2O2, and H2O at about 80 °C, followed by rinsing with water. Then the total reflecting plane was immersed in 40% NH4F solution for 90 s to terminate the Si surface. Deposition of Au was performed at 60 °C by dropping a mixture of a plating solution and 2% HF (2:1 v/v) onto the Si surface, and the deposition time was 60 s. The composition of the plating solution was 0.015 M NaAuCl4·2H2O + 0.15 M Na2SO3 + 0.05 M Na2S2O3·5H2O + 0.05 M NH4Cl. After deposition, the prism was rinsed with deionized water to finish the deposition.
For catalyst deposition, a well-dispersed catalyst slurry containing 18 mg of catalysts, 3 mL of acetone, and 120 μL of Nafion ionomer solution was slowly dropped onto Au film, attaining a catalyst loading of ~0.3 mg cm−2 that can cover the Au film.
In-situ Raman experiments
A Raman spectrometer (HORIBA Jobin Yvon Inc., Model: HR Evolution) coupled with an optical microscope was used for in-situ Raman spectroscopy. A near-infrared laser (λ = 532 nm) was used as the excitation source. The types of objective and grating were 50XL and 500 nm, respectively. A homemade spectro-electrochemical cell equipped with a Pt wire counter electrode and an Ag/AgCl reference electrode was used, with the working electrode prepared by coating the catalyst onto glassy carbon. Each potential was maintained for at least 10 min before collecting the spectra.
Quasi-in-situ XPS measurement
All samples were prepared in the glovebox with an oxygen concentration of less than 4 ppm. The samples were prepared through 10-min CO2RR in Ar-saturated 1 M KOH at around −0.88 V, and were then packaged with liquid nitrogen protection to stabilize the surface reaction intermediate for studying its influence on Ov. For the exploration of the Ov structure after reaction over catalysts, the samples were prepared in the same reaction conditions and were then dried under vacuum at 120 °C for 24 h to remove the surface species.
Theoretical computational
DFT calculations were performed with the Vienna ab initio simulation package (VASP), and the detailed calculation methods and parameters are presented in Supplementary Note 4 and Supplementary Data 1. Generally, all structures were relaxed by a conjugate gradient method until the residual force component was <0.04 eV Å−1, and the convergence criterion of total energy in the self-consistent field method was set to 10−6 eV. The original structure of anatase was employed for calculations with the parameters that a = b = 3.7825 Å, c = 9.6150 Å, and α = b = γ = 90.0000°. With the relaxed cell structure, the (100) plane was utilized for further assessments according to XRD and TEM results. DFT + U calculations on the density of states were performed for both Cu−Ti1O3 and Cu−O4(Ti), and Lobster was employed for COHP analyses68.
Furthermore, (100) surface was also employed for CO2RR evaluations, using a slab model consisting of three atomic layers and 2 × 1-unit cells for the lateral dimensions, where the thickness of the vacuum was set at 12 Å to ensure that there was no superficial interaction between different layers. The total size for the established slab model was 7.6134 × 9.5168 × 21.3227 Å3, and α = β = γ = 90.0000°. The Brillouin zone of the supercell was sampled by a 3 × 3 × 1 uniform k-point mesh. All the energy calculations were based on a two-step process involving the structure relaxation in vacuum (i.e., electron energy, EDFT) and a subsequent single-point calculation using an implicit solvation model.
The bond dissociation enthalpies of Cu–O and O–C in Cu–*O–CH3 were calculated based on a similar method reported previously15:
where \({H}_{\ast {{{\rm{OCH}}}}_{3}}\) and H*O are the enthalpies of the catalyst with adsorption of the corresponding molecules, \({H}_{\ast {{{\rm{O+CH}}}}_{3}}\) and \({H}_{\ast {{{\rm{O+CH}}}}_{3}}\) are the enthalpies of the corresponding bond-breaking models.
The Gibbs free energy change of formation (at T K) for each elementary step was calculated as:
where ∆Esol was the solvation correction, \({\Delta G}_{{{\rm{cor}}}}\left(T\right)={\Delta E}_{{{\rm{ZPE}}}}-T\Delta S+\Delta U(T)\) was obtained by processing frequency calculation results, and ∆G298.15K was used to construct the free energy profiles. The computational hydrogen electrode model was also employed69, in which the chemical potential of (H+ + e–) is equal to that of half a H2 molecule with a pH correction of kBTln([H+]).
Data availability
The data that support the findings of this study are available in the paper and its Supplementary Information. Source data are provided as a Source Data file. Source data are provided with this paper.
References
Guo, P. et al. Carbon dioxide emissions from global overseas coal-fired power plants. Nat. Clim. Chang. 14, 1151–1157 (2024).
O’Brien, C. P. et al. CO2 electrolyzers. Chem. Rev. 124, 3648–3693 (2024).
Rakhsha, A. et al. Tandem gold/copper catalysis and morphological tuning via wrinkling to boost CO2 electroreduction into C2+ products. Nano Energy 133, 110457 (2025).
Stanley, J. S. et al. Sorbent-mediated electrocatalytic reduction of dilute CO2 to methane. J. Am. Chem. Soc. 147, 16099–16106 (2025).
Sun, Z. et al. Integrated CO2 capture and methanation from the intermediate-temperature flue gas on dual functional hybrids of AMS/CaMgO||NixCoy. Sep. Purif. Technol. 307, 122680 (2023).
Woldu, A. R. et al. Experimental and theoretical insights into single atoms, dual atoms, and sub-nanocluster catalysts for electrochemical CO2 reduction (CO2RR) to high-value products. Adv. Mater. 36, 2414169 (2024).
Xu, Y. et al. Optimizing the selectivity of CH4 electrosynthesis from CO2 over cuprates through Cu─O bond length descriptor. Angew. Chem. Int. Ed. 64, e202503745 (2025).
Yang, Q. et al. Ethylene electrosynthesis at low voltages enabled by dopant-induced modulation of the rate-determining step. Nat. Synth. 4, 1396–1407 (2025).
Wu, M. et al. Emerging atomically precise metal nanoclusters and ultrasmall nanoparticles for efficient electrochemical energy catalysis: synthesis strategies and surface/interface engineering. Electrochem. Energy Rev. 7, 10 (2024).
Santatiwongchai, J., Faungnawakij, K. & Hirunsit, P. Comprehensive mechanism of CO2 electroreduction toward ethylene and ethanol: the solvent effect from explicit water–Cu(100) interface models. ACS Catal. 11, 9688–9701 (2021).
Pérez-Rodríguez, S. et al. Single-atom Cu-N-C catalysts for the electro-reduction of CO2 to CO assessed by rotating ring-disc electrode. J. Energy Chem. 88, 169–182 (2024).
Dong, J. et al. Continuous electroproduction of formate via CO2 reduction on local symmetry-broken single-atom catalysts. Nat. Commun. 14, 6849 (2023).
Shen, W. et al. Self-healing Cu single-atom catalyst for high-performance electrocatalytic CO2 methanation. Nat. Commun. 16, 7943 (2025).
Cai, R. et al. The proximal protonation source in Cu-NHx-C single-atom catalysts selectively boosts CO2 to methane electroreduction. Angew. Chem. Int. Ed. 64, e202424098 (2025).
Kong, S. et al. Delocalization state-induced selective bond breaking for efficient methanol electrosynthesis from CO2. Nat. Catal. 6, 6–15 (2023).
Liu, Z. et al. Switching CO2 electroreduction toward ethanol by delocalization state-tuned bond cleavage. J. Am. Chem. Soc. 146, 14260–14266 (2024).
He, S. et al. The p-orbital delocalization of main-group metals to boost CO2 electroreduction. Angew. Chem. Int. Ed. 57, 16114–16119 (2018).
Sun, T. et al. Ferromagnetic single-atom spin catalyst for boosting water splitting. Nat. Nanotechnol. 18, 763–771 (2023).
Yang, Y. et al. Ligand-tuning copper in coordination polymers for efficient electrochemical C-C coupling. Nat. Commun. 15, 6316 (2024).
Lu, T. et al. Local coordination environment-driven structural dynamics of single-atom copper and the CO2 electroreduction pathway. J. Am. Chem. Soc. 147, 26425–26436 (2025).
Lv, X., Liu, Z., Yang, C., Ji, Y. & Zheng, G. Tuning structures and microenvironments of Cu-based catalysts for sustainable CO2 and CO electroreduction. Acc. Mater. Res. 4, 264–274 (2023).
Wang, M. et al. f-p-d coupling-induced bonding covalency boosts C–C coupling in electrocatalytic CO2 reduction over Er–O–Cu sites. J. Energy Chem. 108, 239–245 (2025).
Réocreux, R. & Stamatakis, M. One decade of computational studies on single-atom alloys: Is in silico design within reach? Acc. Chem. Res. 55, 87–97 (2022).
Greiner, M. T. et al. Free-atom-like d states in single-atom alloy catalysts. Nat. Chem. 10, 1008–1015 (2018).
Xu, M. et al. Kinetically matched C-N coupling toward efficient urea electrosynthesis enabled on copper single-atom alloy. Nat. Commun. 14, 6994 (2023).
Yang, C.-L. et al. Sulfur-anchoring synthesis of platinum intermetallic nanoparticle catalysts for fuel cells. Science 374, 459–464 (2021).
Li, M. et al. Reinforcing Co–O covalency via Ce(4f)–O(2p)–Co(3d) gradient orbital coupling for high-efficiency oxygen evolution. Adv. Mater. 35, 2302462 (2023).
Zhu, J. et al. Frustrated Lewis pair mediated f-p-d orbital coupling: Achieving selective seawater oxidation and breaking *OH and *OOH scaling relationship. Angew. Chem. Int. Ed. 64, e202414721 (2025).
Zheng, B. et al. Rare-earth doping in nanostructured inorganic materials. Chem. Rev. 122, 5519–5603 (2022).
Wang, X. et al. Orbital-level band gap engineering of RuO2 for enhanced acidic water oxidation. Nat. Commun. 16, 4845 (2025).
Franchini, C., Reticcioli, M., Setvin, M. & Diebold, U. Polarons in materials. Nat. Rev. Mater. 6, 560–586 (2021).
Zhou, X. et al. Stabilizing Cu2+ ions by solid solutions to promote CO2 electroreduction to methane. J. Am. Chem. Soc. 144, 2079–2084 (2022).
Setvin, M. et al. Direct view at excess electrons in TiO2 rutile and anatase. Phys. Rev. Lett. 113, 086402 (2014).
Hao, Y.-N., Chen, T., Zhang, X., Zhou, H. & Ma, Y. Ti-Ti sigma bond at oxygen vacancy inducing the deep defect level in anatase TiO2 (101) surface. J. Chem. Phys. 150, 224702 (2019).
Wang, R. et al. Compressive-strained rutile TiO2 enables O2 mono-hydrogenation for singlet oxygen electrosynthesis. Nat. Synth. 4, 754–764 (2025).
Li, R. et al. Strong dipole-promoted N-O bond hydrogenolysis enables ampere-level electrosynthesis of methylamine. Nat. Chem. 17, 1152–1160 (2025).
Wu, Q.-J. et al. Atomically precise copper nanoclusters for highly efficient electroreduction of CO2 towards hydrocarbons via breaking the coordination symmetry of Cu site. Angew. Chem. Int. Ed. 62, e202306822 (2023).
Hiramatsu, W. et al. Surface oxygen vacancies on copper-doped titanium dioxide for photocatalytic nitrate-to-ammonia reduction. J. Am. Chem. Soc. 147, 1968–1979 (2025).
Kang, S. H. et al. Chemically stable low-dimensional electrides in transition metal-rich monochalcogenides: theoretical and experimental explorations. J. Am. Chem. Soc. 144, 4496–4506 (2022).
Li, Y. et al. Recent progress on titanium sesquioxide: Fabrication, properties, and applications. Adv. Funct. Mater. 32, 2203491 (2022).
He, Y. et al. In situ fabrication of atomically adjacent dual-vacancy sites for nearly 100% selective CH4 production. Proc. Natl. Acad. Sci. USA 121, e2322107121 (2024).
Wei, Z.-W. et al. Reversed charge transfer and enhanced hydrogen spillover in platinum nanoclusters anchored on titanium oxide with rich oxygen vacancies boost hydrogen evolution reaction. Angew. Chem. Int. Ed. 60, 16622–16627 (2021).
Greczynski, G., Haasch, R. T., Hellgren, N., Lewin, E. & Hultman, L. X-ray photoelectron spectroscopy of thin films. Nat. Rev. Methods Primers. 3, 40 (2023).
Pauly, N., Tougaard, S. & Yubero, F. LMM Auger primary excitation spectra of copper. Surf. Sci. 630, 294–299 (2014).
Wu, T. et al. Identifying the origin of Ti3+ activity toward enhanced electrocatalytic N2 reduction over TiO2 nanoparticles modulated by mixed-valent copper. Adv. Mater. 32, 2000299 (2020).
Shen, Y. et al. Room-temperature photosynthesis of propane from CO2 with Cu single atoms on vacancy-rich TiO2. Nat. Commun. 14, 1117 (2023).
Zhang, W. et al. Electrolyte design using “Porous Water” for high-purity carbon monoxide electrosynthesis from dilute carbon dioxide. Angew. Chem. Int. Ed. 64, e202424104 (2025).
Luc, W. et al. SO2-induced selectivity change in CO2 electroreduction. J. Am. Chem. Soc. 141, 9902–9909 (2019).
Zhou, B. et al. Electrosynthesis of CO from an electrically pH-shifted DAC post-capture liquid using a catalyst: support amide linkage. Joule 9, 101883 (2025).
Li, J. et al. Copper-catalysed electrochemical CO2 methanation via the alloying of single cobalt atoms. Angew. Chem. Int. Ed. 64, e202417008 (2025).
Cai, J. et al. Accurate removal of trace 17β-estradiol and estrogenic activity in blended systems under a photoelectrocatalytic circulating flow. Environ. Sci. Technol. 55, 12585–12595 (2021).
Yao, F. et al. Ordered copper triangular atomic sites for industrial-grade electromethanation of CO2 via self-regulated adsorption of reactants. Angew. Chem. Int. Ed. 64, e202511459 (2025).
Zhao, J. et al. Modulation of *CHxO adsorption to facilitate electrocatalytic reduction of CO2 to CH4 over Cu-based catalysts. J. Am. Chem. Soc. 145, 6622–6627 (2023).
Ji, Y. et al. Selective CO-to-acetate electroreduction via intermediate adsorption tuning on ordered Cu–Pd sites. Nat. Catal. 5, 251–258 (2022).
Malkani, A. S. et al. Understanding the electric and non-electric field components of the cation effect on the electrochemical CO reduction reaction. Sci. Adv. 6, eabd2569 (2020).
Green, I. X., Tang, W., Neurock, M. & Yates, J. T. Jr. Spectroscopic observation of dual catalytic sites during oxidation of CO on a Au/TiO2 catalyst. Science 333, 736–739 (2011).
Guan, Y. et al. Exposure of Au atom on Au(111) in metal nanoclusters for pH-universal electrocatalysis. Angew. Chem. Int. Ed. 64, e202508459 (2025).
Zhang, L. et al. Oxophilicity-controlled CO2 electroreduction to C2+ alcohols over Lewis acid metal-doped Cuδ+ catalysts. J. Am. Chem. Soc. 145, 21945–21954 (2023).
Delmo, E. P. et al. In situ infrared spectroscopic evidence of enhanced electrochemical CO2 reduction and C-C coupling on oxide-derived copper. J. Am. Chem. Soc. 146, 1935–1945 (2024).
Liu, Z. et al. Surface energy tuning on Cu/NC catalysts for CO electroreduction. ACS Catal. 12, 12555–12562 (2022).
Gao, J. et al. Solar reduction of carbon dioxide on copper-tin electrocatalysts with energy conversion efficiency near 20%. Nat. Commun. 13, 5898 (2022).
Sebastián-Pascual, P. & Escudero-Escribano, M. Surface characterization of copper electrocatalysts by lead underpotential deposition. J. Electroanal. Chem. 896, 115446 (2021).
Hao, S. et al. Acid-humidified CO2 gas input for stable electrochemical CO2 reduction reaction. Science 388, eadr3834 (2025).
Xu, Z. et al. Electroreduction of CO2 to methane with triazole molecular catalysts. Nat. Energy 9, 1937–1406 (2024).
Cai, J., Niu, T., Shi, P. & Zhao, G. Boron-doped diamond for hydroxyl radical and sulfate radical anion electrogeneration, transformation, and voltage-free sustainable oxidation. Small 15, 1970261 (2019).
Stoll, S. CW-EPR Spectral Simulations: Solid State. In Electron Paramagnetic Resonance Investigations of Biological Systems by Using Spin Labels, Spin Probes, and Intrinsic Metal Ions Part A, 121–142 (Academic Press, 2015).
Stoll, S. & Schweiger, A. EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J. Magn. Reson. 178, 42–55 (2006).
Maintz, S., Deringer, V. L., Tchougréeff, A. L. & Dronskowski, R. LOBSTER: a tool to extract chemical bonding from plane-wave based DFT. J. Comput. Chem. 37, 1030–1035 (2016).
Nørskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Acknowledgements
We thank the following funding agencies for supporting this work: National Natural Science Foundation of China (No. 22409031, 22306135, 22509185, 22222901, 22175022), Natural Science Foundation of Zhejiang Province (No. LY24B030009, LQN26B030010), the Science and Technology Commission of Shanghai Municipality (No. 23DZ1200800), the National key research and development program of China (No. 2022YFC2904900), the ARC Centre of Excellence for Green Electrochemical Transformation of Carbon Dioxide (CE230100017) funded by the Australian Government, the China Postdoctoral Science Foundation (2025M781024). The XAS research was undertaken on the MEX-1 beamline (23877) at the Australian Synchrotron, part of ANSTO.
Author information
Authors and Affiliations
Contributions
X.Lv, F.Huang, and G.Zheng proposed and supervised the project. Z.Liu, J.Cai, and X.Lv designed the experiments. Z.Liu, S.Dong, C.Qin, Z.Lv, J.Wang, and J.Gong performed material synthesis, electrochemical experiments, structural characterizations, and corresponding data processing. X.Lv performed DFT calculations. J.Cai performed the in-situ time-field E-EMR experiments. Y.Yang collected the XAS data, and Z.Liu analyzed the data. G.Zheng, X.Lv, F.Huang, F.Li, and J.Cai acquired funding support for this project. Z.Liu and X.Lv wrote the manuscript; Q.Han participated in the key discussion and revision of the manuscript; Y.Yang, J.Cai, and F.Li contributed to manuscript editing. All the authors discussed the results and provided significant assistance during the manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Shu Ping Lau and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, Z., Cai, J., Dong, S. et al. Engineering d-orbital of copper single-atom sites toward industrial-level electrocatalytic methanation. Nat Commun 17, 2723 (2026). https://doi.org/10.1038/s41467-026-69260-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69260-z