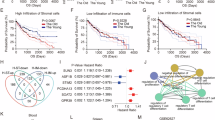
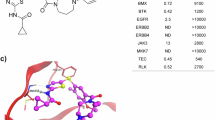
Abstract
Although cancer immunotherapy has recently revolutionized treatment, the low response rate to existing immune checkpoint blockade (ICB) underscores the need for new druggable targets. Here, we find that SOAT1 is selectively expressed in cancer stem cell (CSC) and pharmacological inhibition with STK results in robust anti-tumor effects across various preclinical mouse models, including colon, liver, lung, breast, and melanoma cancer, with low toxicity. Mechanistically, treatment with STK (or gene knockdown of Soat1) induces the release of 20(S)-Hydroxycholesterol (20SOHC) from the tumor cells, and downstream activation of the trans-cellular 20SOHC (tumor)- GPR132 pathway in regulatory T cell (Treg), ultimately resulting in the suppression of Treg functions and enhanced dendritic cells and cytotoxic CD8+ T cell responses. Importantly, STK treatment synergizes with anti-PD-1 or anti-CTLA-4 ICB therapy. Thus, our findings identify SOAT1 as a CSC metabolism checkpoint that facilitates immune evasion and SOAT1 inhibition as a promising strategy for advanced cancer immunotherapy.
Similar content being viewed by others
Data availability
The single-cell RNA-sequence raw data are deposited at Gene Expression Omnibus (GEO) at GSE286015 (scRNA-seq for Hepatocellular carcinoma, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE286015) and GSE286092 (scRNA-seq for Lung carcinoma, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE286092). The Bulk RNA-sequence raw data are deposited at Gene Expression Omnibus (GEO) at GSE309517. All data are included in the Supplementary Information or available from the authors, as are unique reagents used in this Article. Source data are provided with this paper.
References
Zebley, C. C., Zehn, D., Gottschalk, S. & Chi, H. T cell dysfunction and therapeutic intervention in cancer. Nat. Immunol. 25, 1344–1354 (2024).
Mellman, I., Chen, D. S., Powles, T. & Turley, S. J. The cancer-immunity cycle: Indication, genotype, and immunotype. Immunity 56, 2188–2205 (2023).
Pittet, M. J., Di Pilato, M., Garris, C. & Mempel, T. R. Dendritic cells as shepherds of T cell immunity in cancer. Immunity 56, 2218–2230 (2023).
Chi, H., Pepper, M. & Thomas, P. G. Principles and therapeutic applications of adaptive immunity. Cell 187, 2052–2078 (2024).
Albelda, S. M. CAR T cell therapy for patients with solid tumours: key lessons to learn and unlearn. Nat. Rev. Clin. Oncol. 21, 47–66 (2024).
Agudo, J. & Miao, Y. Stemness in solid malignancies: coping with immune attack. Nat. Rev. Cancer 25, 27–40 (2025).
Tay, C., Tanaka, A. & Sakaguchi, S. Tumor-infiltrating regulatory T cells as targets of cancer immunotherapy. Cancer Cell 41, 450–465 (2023).
Song, Y., Liu, J., Zhao, K., Gao, L. & Zhao, J. Cholesterol-induced toxicity: An integrated view of the role of cholesterol in multiple diseases. Cell Metab. 33, 1911–1925 (2021).
Singh, S. R. et al. The lipolysis pathway sustains normal and transformed stem cells in adult Drosophila. Nature 538, 109–113 (2016).
Aggarwal, P. et al. Disruption of the lipolysis pathway results in stem cell death through a sterile immunity-like pathway in adult Drosophila. Cell Rep. 39, 110958 (2022).
Wang, Y. et al. Novel Arf1 inhibitors drive cancer stem cell aging and potentiate anti-tumor immunity. Adv. Sci. (Weinh.) 11, e2404442 (2024).
Qian, H. et al. Structural basis for catalysis and substrate specificity of human ACAT1. Nature 581, 333–338 (2020).
Barker, N. et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457, 608–611 (2009).
Shachaf, C. M. et al. MYC inactivation uncovers pluripotent differentiation and tumour dormancy in hepatocellular cancer. Nature 431, 1112–1117 (2004).
Wang, J. L. et al. Functional screening and rational design of compounds targeting GPR132 to treat diabetes. Nat. Metab. 5, 1726–1746 (2023).
Yan, C. et al. Exhaustion-associated cholesterol deficiency dampens the cytotoxic arm of antitumor immunity. Cancer Cell 41, 1276–1293.e1211 (2023).
Lim, S. A. et al. Lipid signalling enforces functional specialization of T(reg) cells in tumours. Nature 591, 306–311 (2021).
Lu, Z. et al. ATF3 and CH25H regulate effector trogocytosis and anti-tumor activities of endogenous and immunotherapeutic cytotoxic T lymphocytes. Cell Metab. 34, 1342–1358.e1347 (2022).
Ma, X. et al. Cholesterol Induces CD8(+) T Cell Exhaustion in the Tumor Microenvironment. Cell Metab. 30, 143–156.e145 (2019).
Swamy, M. et al. A cholesterol-based allostery model of T cell receptor phosphorylation. Immunity 44, 1091–1101 (2016).
Yang, W. et al. Potentiating the antitumour response of CD8(+) T cells by modulating cholesterol metabolism. Nature 531, 651–655 (2016).
Jiang, Y. et al. Proteomics identifies new therapeutic targets of early-stage hepatocellular carcinoma. Nature 567, 257–261 (2019).
Yue, S. et al. Cholesteryl ester accumulation induced by PTEN loss and PI3K/AKT activation underlies human prostate cancer aggressiveness. Cell Metab. 19, 393–406 (2014).
Huang, B., Song, B. L. & Xu, C. Cholesterol metabolism in cancer: mechanisms and therapeutic opportunities. Nat. Metab. 2, 132–141 (2020).
Wang, G. et al. Arf1-mediated lipid metabolism sustains cancer cells and its ablation induces anti-tumor immune responses in mice. Nat. Commun. 11, 220 (2020).
Pai, M. Y. et al. Drug affinity responsive target stability (DARTS) for small-molecule target identification. Methods Mol. Biol. 1263, 287–298 (2015).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587.e3529 (2021).
Gao, R. et al. Delineating copy number and clonal substructure in human tumors from single-cell transcriptomes. Nat. Biotechnol. 39, 599–608 (2021).
Jin, S. et al. Inference and analysis of cell-cell communication using CellChat. Nat. Commun. 12, 1088 (2021).
Qiu, X. et al. Reversed graph embedding resolves complex single-cell trajectories. Nat. Methods 14, 979–982 (2017).
Borcherding, N., Bormann, N. L. & Kraus, G. scRepertoire: an R-based toolkit for single-cell immune receptor analysis. F1000Res 9, 47 (2020).
Schubert, M. et al. Perturbation-response genes reveal signaling footprints in cancer gene expression. Nat. Commun. 9, 20 (2018).
Ma, H., Fang, W., Li, Q., Wang, Y. & Hou, S. X. Arf1 ablation in colorectal cancer cells activates a super signal complex in DC to enhance anti-tumor immunity. Adv. Sci. (Weinh.) 10, e2305089 (2023).
Borah, K. et al. A quantitative LC-MS/MS method for analysis of mitochondrial-specific oxysterol metabolism. Redox Biol. 36, 101595 (2020).
Long, T., Sun, Y., Hassan, A., Qi, X. & Li, X. Structure of nevanimibe-bound tetrameric human ACAT1. Nature 581, 339–343 (2020).
Acknowledgments
The State Key Laboratory of Genetic Engineering (Fudan University, Shanghai, China) and the Animal Facility of the Institute of Development Biology (Fudan University, Shanghai, China) are appreciated for their technological assistance. This work was supported by the National Key R&D Program of China (Grant No. 2024YFA1306000 to S.X.H and 2023YFA1800202 to S.X.H) and the National Natural Science Foundation of China (Grant No. 8245010 to S.X.H).
Author information
Authors and Affiliations
Contributions
Yahui Ding: Writing original draft, Visualization, Investigation, Formal analysis, Data curation, Conceptualization, Software, Resources. Wanqi Fang: Writing original draft, Investigation, Formal analysis, Data curation, Conceptualization, Resources. Ruiqing Xiang: Investigation, Formal analysis. Haitao Liu: Investigation, Data curation. Menglin Huang: Formal analysis, Data curation. Yingran Shen: Formal analysis, Data curation. Ying Chen: Formal analysis, Data curation. Guohao Wang: Formal analysis, Data curation. Zhaocai Zhou: Supervision, Project administration. Yun Ling: Supervision, Project administration. Ling Sun: Supervision, Project administration, Funding acquisition, Conceptualization. Yuetong Wang: Writing review&editing, Validation, Project administration, Methodology, Funding acquisition, Conceptualization. Steven X. Hou: Writing-review & editing, Supervision, Project administration, Funding acquisition, Conceptualization. All authors have reviewed and approved the submission. All data were generated in-house, and no paper mill was used. All authors agree to be accountable for all aspects of work, ensuring integrity and accuracy.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests or personal relationships that could have appeared to influence the work reported in this paper.
Peer review
Peer review information
Nature Communications thanks Sergei Gryaznov and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ding, Y., Fang, W., Xiang, R. et al. Inhibition of the cancer stem cell immune checkpoint SOAT1 suppresses regulatory T cell functions through a trans-cellular 20(S)-Hydroxycholesterol-GPR132 pathway in mice. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69305-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69305-3