Abstract
In nasopharyngeal carcinoma, cisplatin is known to be associated with poor treatment compliance and notable side effects. More effective and safer platinum drugs are needed for the treatment of patients with nasopharyngeal carcinoma. In 2021, our multicenter, randomized, phase 3 trial reported that lobaplatin and fluorouracil induction chemotherapy plus concurrent chemoradiotherapy resulted in non-inferior survival and fewer toxic effects than did cisplatin-based therapy in nasopharyngeal carcinoma. Data from the 10-year survival analysis are updated here. With a median follow-up of 10.6 years in the intention-to-treat population, 10-year progression-free survival is 70.7% in the lobaplatin-based therapy group vs. 71.9% in the cisplatin-based therapy group (HR 1.02, 95% CI 0.72–1.43; log-rank p = 0.885). The difference between the groups is 1.2% (95% CI −6.7–9.1, pnon-inferiority = 0.015), which is lower than the prespecified non-inferiority margin of 10%. The results are similar when we analyze patients in the per-protocol population. In the univariable and multivariable analyses, stage is an independent prognostic factor for progression-free survival (p = 0.001). The subgroup analyses suggest that the non-inferiority of lobaplatin-based therapy did not differ among specific populations. The incidence of late toxic effects is similar between the therapy groups, except for grades 1–2 peripheral neuropathy (p = 0.033), grades 1–2 deafness/otitis (p = 0.021), and grades 1–2/3 nephrotoxicity (p = 0.005; p = 0.021), the incidence of which is greater in the cisplatin-based therapy group than in the lobaplatin-based therapy group. Our findings suggest that lobaplatin and fluorouracil induction chemotherapy plus lobaplatin-based concurrent chemoradiotherapy is an alternative doublet treatment strategy to cisplatin-based concurrent chemoradiotherapy in patients with locoregionally advanced nasopharyngeal carcinoma.
Similar content being viewed by others
Introduction
Approximately 129,000 new cases of nasopharyngeal carcinoma were diagnosed globally in 2018, accounting for 0.7% of all cancers diagnosed1,2,3. Nevertheless, its global geographical distribution is extremely unbalanced; >70% of new cases are in Southern China, southeast Asia, and northern Africa1,2,4. Despite recent improvements in the survival of patients with locoregionally advanced nasopharyngeal carcinoma, notable side effects and therapy compliance are still not satisfactory in patients with this disease4,5.
Cisplatin-based induction chemotherapy plus concurrent chemoradiotherapy is generally recommended as the standard first-line therapy for locoregionally advanced nasopharyngeal carcinoma. Nevertheless, cisplatin-based therapy is known to be related to poor toxicity. Thus, effective and safer platinum drugs for treating locoregionally advanced nasopharyngeal carcinoma are needed. Lobaplatin, a third-generation platinum-based drug, leads to the formation of DNA adducts and causes cell apoptosis6,7. Our phase 2 trial regarding lobaplatin-based induction chemotherapy plus concurrent chemoradiotherapy for stage III–IVB nasopharyngeal carcinoma reported encouraging antitumor activity and safer treatment-related toxicity8. In 2021, our multicenter, randomized, phase 3 trial (No. ChiCTR-TRC-13003285) compared the efficacy and safety of lobaplatin and induction chemotherapy plus concurrent chemoradiotherapy with cisplatin and fluorouracil induction chemotherapy plus concurrent chemoradiotherapy in locoregionally advanced nasopharyngeal carcinoma, with non-inferior 5-year progression-free survival and fewer treatment-related toxicities4. A durable long-term survival benefit is imperative.
In this work, we sought to determine the 10-year survival and late toxicity of lobaplatin and fluorouracil induction chemotherapy plus lobaplatin-based concurrent chemoradiotherapy compared with those of cisplatin and fluorouracil induction chemotherapy plus cisplatin-based concurrent chemoradiotherapy in patients with locoregionally advanced nasopharyngeal carcinoma. The associated late toxic effects of these therapies were also reported.
Results
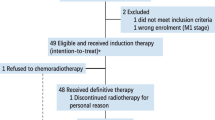
Between June 7, 2013, and June 16, 2015, 515 patients were screened for eligibility, 502 of whom were randomly assigned to receive lobaplatin-based therapy (n = 252) or cisplatin-based therapy (n = 250) (Fig. 1). Measurements of plasma Epstein-Barr virus DNA (EBV-DNA) were available for 478 patients (237 patients [94%] in the lobaplatin-based therapy group and 241 patients [96.4%] in the cisplatin-based therapy group). Baseline characteristics were balanced between the two therapy groups (Table 1).
aOne patient without adequate hematological function, two patients without adequate renal function, one patient without adequate hepatic function, and four patients aged >60 years were excluded. bOther drugs included concurrently administered carboplatin and cetuximab. GP gemcitabine and cisplatin.
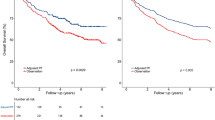
The data cutoff date for this updated analysis was April 30, 2025. The median follow-up was 10.6 years (IQR 10.5–10.7) for the intention-to-treat population. In the intention-to-treat population, 10-year progression-free survival rate was 70.7% (95% CI 65.1–76.8) in the lobaplatin-based therapy group vs. 71.9% (95% CI 66.4–77.8) in the cisplatin-based therapy group (HR 1.02, 95% CI 0.72–1.43, log-rank p = 0.885). The difference between the therapy groups was 1.2% (95% CI −6.7–9.1%, pnon-inferiority = 0.015), which was lower than the prespecified non-inferiority margin of 10% (Fig. 2A; Supplementary Table 1). Sixty-nine of 252 patients (27%) in the lobaplatin-based therapy group and 68 of 250 patients (27.2%) in the cisplatin-based therapy group experienced a progression-free survival event in the intention-to-treat population. The proportional hazards assumption was met (Supplementary Table 2). There were no differences between the therapy groups regarding the secondary endpoints of overall survival, locoregional recurrence-free survival, and distant metastasis-free survival in the intention-to-treat population (Fig. 2B–D; Supplementary Table 1). In the per-protocol population, 10-year progression-free survival rate was 70.1% (95% CI 64.3–76.3) in the lobaplatin-based therapy group vs. 72.5% (95% CI 66.9–78.5) in the cisplatin-based therapy group (HR 1.08, 95% CI 0.77–1.52, log-rank p = 0.664) (Supplementary Fig. 1A; Supplementary Table 1). The difference between the therapy groups was 2.4% (95% CI −5.6–9.5, pnon-inferiority = 0.033), which was lower than the prespecified non-inferiority margin of 10% (Supplementary Fig. 1B–D; Supplementary Table 1). Sixty-eight of 252 patients (27%) in the lobaplatin-based therapy group and 66 of 250 patients (26.4%) in the cisplatin-based therapy group experienced a progression-free survival event in the per-protocol population. The proportional hazards assumption was met (Supplementary Table 2). Differences were not observed between the therapy groups in the per-protocol analysis for the secondary endpoints (Supplementary Fig. 1B–1D, Supplementary Table 1). Summaries and time points of patients lost to follow-up after treatment are presented in Supplementary Table 3–4.
A Progression-free Survival, B Overall Survival, C Locoregional Recurrence-free Survival, D Distant Metastasis-free Survival.
In the univariable and multivariable analyses, stage was an independent prognostic factor for progression-free survival, locoregional recurrence-free survival, and distant metastasis-free survival in the intention-to-treat population and the per-protocol population, marginal significance for overall survival in the intention-to-treat population (Table 2, Supplementary Table 5). Results were similar when we excluded patients lost to follow-up in the sensitivity analyses (Supplementary Table 6).
At the last follow-up on April 30, 2025, total of 71 (14.1%) patients of local recurrence (35 of 252 patients [13.9%] in the lobaplatin-based therapy group vs. 36 of 250 patients [14.4%] in the cisplatin-based therapy group) and 85 (16.9%) patients of distant metastasis (42 of 252 patients [16.7%] in the lobaplatin-based therapy group vs. 43 of 250 patients [17.2%] in the cisplatin-based therapy group) were observed (Supplementary Table 7). Fourteen new cases of local recurrence (2.8%) were observed, including 5 of 252 cases (2.1%) in the lobaplatin-based therapy group vs. 9 of 250 patients (3.6%) in the cisplatin-based therapy group). In addition, 12 new cases of distant metastasis (2.4%) were reported, comprising 8 of 252 cases (3.1%) in the lobaplatin-based therapy group vs. 4 of 250 cases (1.6%) in the cisplatin-based therapy group. Furthermore, 70 of 502 cases (16.9%) died, with 35 of 252 cases (14%) in the lobaplatin-based therapy group vs. 35 of 250 cases (14%) in the cisplatin-based therapy group (Supplementary Table 8).
We performed subgroup analyses for progression-free survival, overall survival, locoregional recurrence-free survival, and distant metastasis-free survival in patients stratified by the following covariates: sex, age, Karnofsky scale, stage, EBV-DNA, and hospital. No interactions between these covariates and therapy were observed, irrespective of the intention-to-treat population or the per-protocol population, suggesting that the non-inferiority of lobaplatin-based therapy did not differ among specific populations. The details of the analyses are shown in Fig. 3 and Supplementary Fig. 2.
A Progression-free Survival, B Overall Survival, C Locoregional Recurrence-free Survival, D Distant Metastasis-free Survival. An unstratified Cox proportional hazards model was used to calculate HRs and 95% CIs, and to execute the interaction test. HR hazard ratio. EBV-DNA Epstein-Barr virus DNA. 1 = Sun Yat-sen University Cancer Center. 2 = Other hospitals.
During the entire therapy and follow-up course, the proportions of the most common late adverse events (i.e., those occurring in >10% of patients) were similar between the lobaplatin-based therapy group and the cisplatin-based therapy group (dry mouth: 189 patients [75%] vs. 184 patients [73.5%]; deafness/ otitis: 104 patients [41.3%] vs. 138 patients [55.5%]; neck tissue damage: 90 patients [35.7%] vs. 97 patients [38.9%]). After multivariable analyses, the incidence of grades 1–2 peripheral neuropathy (28 patients [11.2%] vs. 15 patients [6%], p = 0.033), grades 1–2 deafness/otitis (116 patients [46%] vs. 88 patients [34.9%], p = 0.021), and grades 1–2 nephrotoxicity (22 patients [8.8%] vs. 6 patients [2.4%], p = 0.005) was higher in the cisplatin-based therapy group than in the lobaplatin-based therapy group. In addition, the incidence of grade 3 nephrotoxicity (10 patients [4%] vs. 1 patient [0.4%], p = 0.021) was also higher in the cisplatin-based therapy group compared with the lobaplatin-based therapy group (Table 3). No grade 4–5 late adverse events were recorded. Details of the acute adverse events in the two therapy groups were reported in our previous study4. Temporal trends in late toxicities are presented in Supplementary Fig. 3. Descriptions of patients with missing toxicity data in each therapy group are showed in Supplementary Table 9.
Details regarding radiotherapy compliance, relative dose intensity, and dose reductions are provided in Supplementary Table 10.
Discussion
Clinical trials frequently include multiple endpoints that mature at different times. The data from updates provide an opportunity to disseminate additional results of the trial. In this phase 3 trial, the 10-year survival analysis provided decisive evidence of the efficacy of lobaplatin and fluorouracil induction chemotherapy plus lobaplatin-based concurrent chemoradiotherapy in locoregionally advanced nasopharyngeal carcinoma. Compared with the cisplatin-based therapy group, the lobaplatin-based therapy group demonstrated in non-inferior 10-year progression-free survival.
Reducing the toxicity associated with treatment should be a constant endeavor in our field. The significant survival and adverse effects associated with chemotherapy and radiotherapy for nasopharyngeal carcinoma render this effort even more crucial. The rationale for substituting cisplatin with next-generation platinum salts during the induction and concurrent phases may imply at least similar activity, with the benefits of reduced toxicity, improved quality of life, and increased convenience9. Several studies support these promising alternative regimens in locoregionally advanced nasopharyngeal carcinoma. A retrospective study performed by Liang et al. revealed that the 5-year progression-free survival rates (68% vs. 79% vs. 70%), 5-year local relapse-free survival rates (86% vs. 88% vs. 84%), 5-year distant metastasis-free survival rates (74% vs. 86% vs. 84%), and 5-year overall survival rates (77% vs. 88% vs. 77%) of cisplatin-based therapy, carboplatin-based therapy, and lobaplatin-based therapy were similar in locoregionally advanced nasopharyngeal carcinoma. Notably, patients in the cisplatin-based therapy group were more susceptible than those in the lobaplatin-based therapy group to vomiting (any grade, 96% vs. 64%), leucopenia ( ≥ grade 3, 89% vs. 75%), nephrotoxicity (any grade, 18% vs. 2%), and dermatitis ( ≥ grade 2, 61% vs. 10%). More late toxicities (e.g., grade 1–2 otitis, grade 1 deafness, and anygrade superficial soft tissue fibrosis) were also observed in the cisplatin-based therapy group10. In a meta-analysis of 1907 participants, significant differences in 5-year progression-free survival (p = 0.64), 5-year locoregional relapse-free survival (p = 0.26), 5-year distant metastasis-free survival (p = 0.96), and 5-year overall survival (p = 0.87) between cisplatin-based chemotherapy and other platinum-based chemotherapies (e.g., carboplatin, nedaplatin, and lobaplatin) were not observed. Furthermore, toxicity, including nausea (p = 0.01), vomiting (p < 0.0001), and weight loss (p = 0.0001), was lower in the other platinum-based therapy groups11. The results of a phase 3 trial revealed that the 5-year survival rate in the nedaplatin-based therapy group was not inferior to that in the cisplatin-based therapy group (progression-free survival rate, 79.8% vs. 81.4%, pnon-inferiority = 0.002; locoregional relapse-free survival rate, 92.6% vs. 89.6%, p = 0.17; distant metastasis-free survival rate, 85.9% vs. 90.4%, p = 0.17; overall survival rate, 88.8% vs. 89.4%, p = 0.63), with a lower incidence of late grade 3–4 auditory toxic effects (7.7% vs. 21%, p = 0.04)12.
We aimed to address this issue by providing high-quality phase 3 data that demonstrated promising results in the exploration of lobaplatin-based therapy for locoregionally advanced nasopharyngeal carcinoma. Our final survival analysis served as a confirmatory phase 3 trial to further validate the clinical strategy of lobaplatin-based therapy for patients with locoregionally advanced nasopharyngeal carcinoma.
Based on evidence available by 2012, platinum combined with fluorouracil was considered the standard induction regimen for nasopharyngeal carcinoma13. Accordingly, we designed the chemotherapy schedules on the basis of these considerations. In a phase 3 trial, Zhang et al. found that the addition of induction therapy with gemcitabine plus cisplatin to chemoradiotherapy improved recurrence-free survival among patients with high-risk locoregionally advanced nasopharyngeal cancer14. A 2019 phase 3 trial showed that the addition of cisplatin, fluorouracil, and docetaxel induction chemotherapy to concurrent chemoradiotherapy significantly improved failure-free survival in locoregionally advanced nasopharyngeal carcinoma with long-term follow-up15. Further validation is required to determine whether the lobaplatin plus gemcitabine and lobaplatin, fluorouracil, and docetaxel regimens can be applied to clinical practice with non-inferior survival and fewer toxic effects.
Chemotherapy combined with radiotherapy is crucial for treating locoregionally advanced nasopharyngeal carcinoma. At present, the National Comprehensive Cancer Network guidelines recommend induction chemotherapy followed by concurrent chemoradiation as level 2A evidence for stage II–IVA nasopharyngeal carcinoma16. Chemotherapy and radiotherapy inevitably induce acute and late toxicities, and the latter can emerge months or even years after treatment completion2. These issues highlight the importance of close follow-up. The investigation of late toxicities is important for long-term patient well-being. While radiotherapy kills cancer cells, it can also affect surrounding normal tissues. If the radiation field is near the ear or includes ear structures, the incidence of ear damage increases. Radiation-induced oxidative stress can destroy inner ear cells and promote cell damage, inflammation and other post-irradiation effects17. This is common in radiotherapy for nasopharyngeal carcinoma. Ear damage can be categorized into the following types, which may occur alone or simultaneously: external ear damage; middle ear damage, such as secretory otitis; and inner ear damage, such as sensorineural hearing loss and tinnitus18,19. The addition of different chemotherapy regimens to radiotherapy may influence the incidence of late toxicity2. In this final analysis, compared with the cisplatin-based regimen, the lobaplatin-based regimen resulted in a lower incidence of deafness or otitis in patients with locoregionally advanced nasopharyngeal carcinoma. Our data are consistent with previous findings regarding the association between platinum-based chemoradiotherapy and hearing impairment. A previous study suggested that patients with locoregionally advanced nasopharyngeal carcinoma who received cisplatin-based therapy had a high incidence of all otological complications (hearing impairment [46%], tinnitus [48%], and otorrhea [68%])20. In a study by Liang et al., the incidence of hearing impairment was greater in patients undergoing concurrent cisplatin-based therapy than in those undergoing concurrent lobaplatin-based therapy10. Cisplatin induces apoptosis by inhibiting the activity of antioxidant enzymes21. Some scholars believe that cisplatin has a toxic effect on the stria vascularis, resulting in a reduction in the endocochlear potential related to striatal edema22. In 2022, Dillard et al. conducted a meta-analysis of 66 studies on hearing loss related to platinum-based treatment; their findings revealed that ototoxicity was less prevalent in patients treated with carboplatin/nedaplatin/lobaplatin than in those treated with cisplatin23. Cell apoptosis and significantly increased antioxidant gene expression were observed in response to only cisplatin. Cisplatin significantly damages sensory hair cells. Other platinum agents are less ototoxic than cisplatin because they likely have different ototoxic mechanisms than cisplatin does24. Thus, compared with other platinum agents, radiotherapy-induced ototoxicity and cisplatin may exhibit a synergistic effect on cochlear damage. Further rigorous studies are needed to explore this issue.
This trial has several limitations. First, as all patients were sourced from endemic areas in China where non-keratinizing nasopharyngeal carcinoma comprises more than 95% of cases, the generalizability of these findings to non-Asian participants in non-endemic regions needs to be investigated further. Second, the long-term toxicity data presented here may not, to some extent, clearly support the premise of a more favorable toxicity profile. Data completeness over the 10-year follow-up was inevitably limited and may have led to an underestimation of some late adverse events; competing late adverse events may dilute differences between treatment groups. These considerations highlight the need for further studies with more comprehensive follow-up to clarify the potential benefits of lobaplatin-based therapy. Third, quality of life-related data obtained from questionnaires (e.g., the Functional Assessment of Cancer Therapy—Head and Neck) were not collected25,26, which could have placed data on late toxic effects in context and helped assess differences in quality of life for patients receiving lobaplatin-based therapy compared with patients receiving cisplatin-based therapy.
Taken together, the findings of our 10-year analysis suggest a non-inferior alternative antitumor efficacy regimen involving the use of lobaplatin in induction chemotherapy plus concurrent chemoradiotherapy while minimizing acute and late toxic effects by eliminating cisplatin, thereby improving the net benefit for patients with nasopharyngeal carcinoma.
Methods
Study design and participants
This is a multicenter, open-label, non-inferiority, randomized, controlled, phase 3 trial. The Institutional Ethics Review Board of Sun Yat-sen University Cancer Center approved the trial protocol (No. B2012-003-01). The trial was performed in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice guidelines as defined by the International Conference on Harmonisation. Written informed consent was obtained from all patients before enrollment. Patients could withdraw consent at any time after enrollment and could discontinue the trial if severe coexisting conditions occurred.
The study design was previously reported4. We enrolled patients from hospitals in China with previously untreated, non-distant-metastatic, non-keratinizing, stage III–IVB nasopharyngeal carcinoma (7th edition of the International Union Against Cancer Staging System). To be eligible for the study, the following inclusion criteria had to be met: age 18–60 years; a Karnofsky performance status score of at least 70; loss of no more than 10% of body weight; adequate hematological parameters (absolute neutrophil count ≥ 2000/μL, platelet count ≥ 100,000/μL, and hemoglobin concentration ≥ 90 g/L); adequate renal function (creatinine clearance ≥ 80 mL/min); and adequate hepatic function (serum bilirubin, alanine aminotransferase, and aspartate aminotransferase concentrations ≤ 2.0 times the upper limit of normal). The main exclusion criteria were previous malignancies; the presence of recurrent or distant metastasis; previous radiotherapy, chemotherapy, immunotherapy, or surgery; severe coexisting illness; and pregnancy or lactation. The full list of eligibility and ineligibility criteria is provided in the Protocol (Supplementary Note 2).
Randomization and masking
Patients were randomly assigned (1:1) to one of four blocks to receive lobaplatin and fluorouracil induction chemotherapy plus lobaplatin-based concurrent chemoradiotherapy (lobaplatin-based therapy group) or cisplatin and fluorouracil induction chemotherapy plus cisplatin-based concurrent chemoradiotherapy (cisplatin-based therapy group). The identity of each group was known only to the statistician. The clinical trial center of Sun Yat-sen University Cancer Center conducted the centralized randomization procedure using a computer-generated random number code. The investigators and patients were aware of the therapy group assignment (open-label), whereas the Central Imaging Review Committee was masked to the treatment assignment. Several quality control measures were implemented to ensure scientific integrity (Protocol [Supplementary Note 2]).
The computer-generated random number
Randomization was conducted at the Clinical Trial Center of Sun Yat-sen University Cancer Center. The computer-generated random number sequence was generated using a block design with a block size of four27,28,29. In our trial, the stage and hospital could significantly influence the treatment. If these characteristics were unevenly distributed between therapy groups, the evaluation of the results could be affected. Stratified randomization is an important trial design that divides participants into several levels on the basis of certain important characteristics and then randomly assigns them within each level. Stratified analysis is the analysis of survival data using this method, considering the effect of different stratification factors on survival, which can be used to accurately evaluate treatment effects30. Blocks of four have been used in several trials of nasopharyngeal carcinoma5,14,31,32.
Procedures
Pretreatment evaluation included a complete medical history; physical examination; hematologic and biochemical analyses; nasal endoscopy or rhinosinusal endoscopy; contrast-enhanced MRI or CT of the nasopharynx and neck (CT was indicated only in patients with contraindications to MRI); chest imaging (X-ray or CT); and liver imaging (external ultrasonography or CT), a bone scan or 18F-FDG PET-CT was mandatory for distant metastasis staging. All patients were referred for a dental examination before therapy. Pretreatment evaluations were performed within 2 weeks before randomization.
Patients in the lobaplatin-based therapy group received two cycles of induction lobaplatin (30 mg/m², 2 h intravenously, on days 1 and 22) and fluorouracil (800 mg/m², continuous 120 h infusion, on days 1–5 and 22–26), administered once every 3 weeks. For patients assigned to the cisplatin-based therapy group, induction chemotherapy was given at 3 week intervals as follows: cisplatin at a dose of 100 mg/m2, 4 h intravenously on days 1 and 22, and fluorouracil at a dose of 800 mg/m² as a continuous 120 h infusion on days 1–5 and 22–26. Subsequently, both groups received concurrent chemoradiotherapy with either lobaplatin (30 mg/m², 2 h intravenously) or cisplatin (100 mg/m², 4 h intravenously) every 3 weeks on days 43 and 64. Two cycles were given concurrently with intensity-modulated radiotherapy (IMRT), according to previously published guidelines4. The radiotherapy plans were centrally administered and reviewed in accordance with the criteria recommended in international guidelines33. Dose modifications of chemotherapy due to adverse events were permitted as specified in the Protocol (Supplementary Note 2).
Acute toxic effects observed during treatment were graded in accordance with the Common Toxicity Criteria for Adverse Events (version 3.0). Radiotherapy toxicity was assessed using the late Radiation Morbidity Scoring Scheme of the Radiation Therapy Oncology Group (Supplementary Data 1)34. Patients were removed from the trial if they experienced disease progression or severe comorbidities or if they refused to receive therapy at any time during the trial. After the primary treatment was completed, the patients were followed up every 3 months for the first 3 years, every 6 months for 4–5 years, and annually thereafter. The nasopharynx was assessed by nasal endoscopy or rhinosinusal endoscopy approximately 4 weeks after the completion of radiotherapy. During routine follow-up, hematological and biochemical analyses, nasal endoscopy or rhinosinusal endoscopy, enhanced MRI or CT of the nasopharynx and neck, chest radiography, and abdominal scan were performed, or when tumor recurrence or metastasis was clinically suspected. Tumor recurrence or metastasis was confirmed on the basis of the results of biopsy or fineneedle aspiration. For lesions that were not accessible, the clinical diagnosis was also accepted on the basis of the presence of at least two classic radiological features on CT, MRI, abdominal ultrasonography, or ¹⁸F-FDG PET-CT (Protocol).
Outcomes
The primary endpoint was progression-free survival, which was defined as the time from the date of randomization to the date of locoregional recurrence, distant metastasis, or death from any cause, whichever occurred first. The following secondary endpoints were also analyzed: overall survival (interval between randomization and the date of death), locoregional recurrence-free survival (interval between randomization and the date of locoregional recurrence or death from any cause), distant metastasis-free survival (interval between randomization and the date of distant metastasis or death from any cause), toxicity profile, and treatment compliance.
Statistical analysis
The present trial adopted a non-inferiority margin of 10%4,35. This margin was selected to ensure preservation of an appropriate proportion of the treatment effect of the standard regimen36,37,38,39,40,41,42. Non-inferiority would be concluded if the upper limit of the 95% confidence interval (CI) for the difference in progression-free survival between the lobaplatin-based group and the cisplatin-based group did not exceed 10%. Based on these assumptions, a minimum of 246 patients per group (492 in total, allowing for a 10% dropout or loss to follow-up) was required to provide 80% power with a one-sided type I error rate of 2.5%4.
Efficacy analyses were performed in patients who received at least one cycle of concurrent chemoradiotherapy. The safety analysis population comprised patients who received at least one cycle of induction chemotherapy4.
Survival was estimated via the Kaplan-Meier method, with Greenwood’s formula used to calculate 95% CIs and the log-rank test used to establish significance. Time-to-event data were censored for patients who were lost to follow-up and patients with no events observed at the date of the last follow-up. For patients who had no observed events, the follow-up times and statuses were recorded according to the final follow-up visit (April 30, 2025). We generated hazard ratios with the Cox proportional-hazards model with the assumption of proportional hazards and assessed them on the basis of Schoenfeld residuals. The Bonferroni method was applied to control the type I error in multiple toxicity comparisons.
SPSS (IBM, version 26.0) and SAS (version 9.4) were used for statistical analyses. The statistical test for the primary endpoint was one-sided, and p < 0.025 was considered significant. All statistical tests for the other endpoints were two-sided, and p < 0.05 was considered significant. The database is preserved on the Research Data Deposit public platform (No. RDDA2021002107). The trial is registered with the Chinese Clinical Trial Registry (No. ChiCTR-TRC-13003285).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All requests for data will be reviewed by the clinical site of Sun Yat-sen University Cancer Center, and the study sponsor, to verify if the request is subject to any intellectual property or confidentiality obligations. A proposal with detailed description of study objectives and statistical analysis plan will be needed for evaluation of the request. Additional materials might also be required during the process of evaluation. Data are available to request 12 months after the publication of this article. Requests for access to the De-identified participant data from this study can be submitted via e-mail to lvxing@sysucc.org.cn with detailed proposal for approval. Please allow one month for response to requests. A signed data access agreement with the sponsor is required before accessing the shared data. The study protocol is available as Supplementary Note 2in the Supplementary Information file. The remaining data are available within the Article, Supplementary Information or Source Data file. Source Data are provided with this paper. Source data are provided with this paper.
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Chen, Y. P. et al. Nasopharyngeal carcinoma. Lancet 394, 64–80 (2019).
Ferlay, J., Ervik, M. & Lam, F. Global Cancer Observatory: cancer today. Lyon, France: International Agency for Research on Cancer 2018. https://gco.iarc.fr/today (IARC, 2021).
Lv, X. et al. Induction chemotherapy with lobaplatin and fluorouracil versus cisplatin and fluorouracil followed by chemoradiotherapy in patients with stage III-IVB nasopharyngeal carcinoma: an open-label, non-inferiority, randomised, controlled, phase 3 trial. Lancet Oncol. 22, 716–726 (2021).
Sun, Y. et al. Induction chemotherapy plus concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: a phase 3, multicentre, randomised controlled trial. Lancet Oncol. 17, 1509–1520 (2016).
Welink, J., Boven, E., Vermorken, J. B., Gall, H. E. & van der Vijgh, W. J. F. Pharmacokinetics and pharmacodynamics of lobaplatin (D-19466) in patients with advanced solid tumors, including patients with impaired renal or liver function. Clin. Cancer Res. 5, 2349–2358 (1999).
McKeage, M. J. Lobaplatin: a new antitumour platinum drug. Expert Opin. Investig. Drugs 10, 119–128 (2001).
Ke, L. R. et al. Safety and efficacy of lobaplatin combined with 5-fluorouracil as first-line induction chemotherapy followed by lobaplatin-radiotherapy in locally advanced nasopharyngeal carcinoma: preliminary results of a prospective phase II trial. BMC Cancer 17, 134 (2017).
Cavalieri, S. & Licitra, L. Next generation platinum salt in nasopharygeal carcinoma. Lancet Oncol. 22, 577–578 (2021).
Liang, X., Liu, Q., Yao, W. & Yang, S. Efficacy and toxicity of three concurrent chemoradiotherapy regimens in treating nasopharyngeal carcinoma: comparison among cisplatin, nedaplatin, and lobaplatin. Medicine 101, e31187 (2022).
Li, Z. et al. Comparing the efficacy and safety of cisplatin and other platinum-based chemotherapies in locally advanced nasopharyngeal carcinoma: a systematic review and meta-analysis. BMC Cancer 22, 616 (2022).
Tang, Q. N. et al. Effect of concurrent chemoradiotherapy with nedaplatin vs cisplatin on the long-term outcomes of survival and toxic effects among patients with stage II to IVB nasopharyngeal carcinoma: a 5-year follow-up secondary analysis of a randomized clinical trial. JAMA Netw. Open. 4, e2138470 (2021).
Chan, A. T. et al. Nasopharyngeal cancer: EHNS-ESMO-ESTRO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 23, vii83–vii85 (2012).
Zhang, Y. et al. Gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma. N. Engl. J. Med. 381, 1124–1135 (2019).
Li, W. F. et al. Concurrent chemoradiotherapy with/without induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: long-term results of phase 3 randomized controlled trial. Int J. Cancer 145, 295–305 (2019).
NCCN Clinical Practice Guidelines in Oncology: Head and Neck Cancers, version 2. (NCCN, 2025).
Lin, D. et al. Comparative analysis of hearing loss induced by cisplatin versus carboplatin in locally advanced nasopharyngeal carcinoma: early to mid-term findings. Clin. Otolaryngol. 50, 690–697 (2025).
Mujica-Mota, M. A., Lehnert, S., Devic, S., Gasbarrino, K. & Daniel, S. J. Mechanisms of radiation-induced sensorineural hearing loss and radioprotection. Hear Res. 312, 60–68 (2014).
Ishak, E. M., Gallitto, M., Golub, J. S., Sisti, M. B. & Wang, T. J. C. Radiation-induced sensorineural hearing loss and potential management. Pr. Radiat. Oncol. 14, 212–215 (2024).
Yeh, S. A., Tang, Y., Lui, C. C., Huang, Y. J. & Huang, E. Y. Treatment outcomes and late complications of 849 patients with nasopharyngeal carcinoma treated with radiotherapy alone. Int J. Radiat. Oncol. Biol. Phys. 62, 672–679 (2005).
Rybak, L. P., Mukherjea, D. & Ramkumar, V. Mechanisms of cisplatin-induced ototoxicity and prevention. Semin Hear. 40, 197–204 (2019).
Rybak, L. P., Whitworth, C. A., Mukherjea, D. & Ramkumar, V. Mechanisms of cisplatin-induced ototoxicity and prevention. Hear Res. 226, 157–167 (2007).
Dillard, L. K. et al. Global burden of ototoxic hearing loss associated with platinum-based cancer treatment: a systematic review and meta-analysis. Cancer Epidemiol. 79, 102203 (2022).
Li, K. H., Zhao, Y. Y., Cheng, H. L., Yang, J. J. & Chien, C. Y. Ototoxicity among cisplatin, carboplatin, and oxaliplatin in zebrafish model. Environ. Toxicol. 39, 4058–4065 (2024).
Cella, D. F. et al. The functional assessment of cancer therapy scale: development and validation of the general measure. J. Clin. Oncol. 41, 5335–5344 (2023).
List, M. A. et al. The performance status scale for head and neck cancer patients and the functional assessment of cancer therapy-head and neck scale. a study of utility and validity. Cancer 77, 2294–2301 (1996).
Kernan, W. N., Viscoli, C. M., Makuch, R. W., Brass, L. M. & Horwitz, R. I. Stratified randomization for clinical trials. J. Clin. Epidemiol. 52, 19–26 (1999).
Efird, J. Blocked randomization with randomly selected block sizes. Int J. Environ. Res. Public Health 8, 15–20 (2011).
Bruce, C. L. et al. Choosing and evaluating randomisation methods in clinical trials: a qualitative study. Trials 25, 199 (2024).
Guidelines for random allocation in drug clinical trials of China. 5 https://www.bjdth.com.cn/Sites/Uploaded/File/2024/07/026385550472401280739347963.pdf (2022).
Chen, Y. P. et al. Metronomic capecitabine as adjuvant therapy in locoregionally advanced nasopharyngeal carcinoma: a multicentre, open-label, parallel-group, randomised, controlled, phase 3 trial. Lancet 398, 303–313 (2021).
Yang, Y. et al. Camrelizumab versus placebo in combination with gemcitabine and cisplatin as first-line treatment for recurrent or metastatic nasopharyngeal carcinoma (CAPTAIN-1st): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 22, 1162–1174 (2021).
Lee, A. W. et al. International guideline on dose prioritization and acceptance criteria in radiation therapy planning for nasopharyngeal carcinoma. Int J. Radiat. Oncol. Biol. Phys. 105, 567–580 (2019).
Cox, J. D., Stetz, J. & Pajak, T. F. Toxicity Criteria Of The Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J. Radiat. Oncol. Biol. Phys. 31, 1341–1346 (1995).
Tang, L. Q. et al. Concurrent chemoradiotherapy with nedaplatin versus cisplatin in stage II-IVB nasopharyngeal carcinoma: an open-label, non-inferiority, randomised phase 3 trial. Lancet Oncol. 19, 461–473 (2018).
Fleming, T. R., Odem-Davis, K., Rothmann, M. D. & Li Shen, Y. Some essential considerations in the design and conduct of non-inferiority trials. Clin. Trials 8, 432–439 (2011).
Odem-Davis, K. & Fleming, T. R. Adjusting for unknown bias in non-inferiority clinical trials. Stat Biopharm Res. 5, 795910 (2013).
Kay, R. Statistical principles for clinical trials. J. Int Med Res. 26, 57–65 (1998).
Piaggio, G. et al. Reporting of noninferiority and equivalence randomized trials: extension of the CONSORT 2010 statement. JAMA 308, 2594–2604 (2012).
D’Agostino, R. B. Non-inferiority clinical trials: concepts and issues. Stat. Med. 22, 169–186 (2003).
EMA. Guideline on the choice of the non-inferiority margin (EMA, 2005).
FDA. Guidance for industry: non-inferiority clinical trials (FDA, 2016).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 81872375, No. 82172863, No. 82402382), Natural Science Foundation of Guangdong Province (No. 2021A1515010118), Guangdong Medical Science and Technology Research Fund Project (No. A2024037), and Guangdong Basic and Applied Basic Research Foundation (No. 2025A1515010914). We are grateful to Prof. Qing Liu (Department of Medical Statistics and Epidemiology, Sun Yat-sen University Cancer Center) for assistance with the statistical analysis. We thank the patients and their families who participated in this trial. The paper was edited by SPRINGER NATURE editing service (No. 8MB6W1KS).
Author information
Authors and Affiliations
Contributions
X.L. and X.G. were the principal investigators and participated in trial design, study management, data and toxicity review, review of the report, and final approval. Xu.C., J.Y.Z., and H.Y.H. contributed to the trial design, writing of the protocol, recruitment and treatment of the patients, data and trial management, data analysis and interpretation, writing, and final approval of the report. Xu.C., X.L., and X.G. verified the data in the study. L.G., K.J.C., H.Y.M., F.H., Y.M.L., X.M.L., Z.H.L., Y.X.H., and Y.R.B. were involved in trial design, recruitment and treatment of the patient data and trial management, and review of the report. Y.Y.H., W.Z.Q., C.N.Q., and S.X.W. participated in the recruitment and treatment of the patients, data and trial management, and report preparation. Xu.C., Z.C.L., C.X.L., and Xi.C. were responsible for statistical analyses and interpretation, as well as toxicity and data review. L.R.K., W.Z.Q., Y.D., Y.X., Z.Y.L., J.J.M., H.L., and W.X.X. contributed to patient accrual, writing of the report, and review of the completed report. Xu.C., L.G., X.L., and X.G. were involved in trial management and toxicity review. All authors reviewed and approved the final draft. All authors fully accessed to all the data in the study, and the corresponding author had final responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Competing interests
The author declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Melvin Lee Kiang Chua, who co-reviewed with Jessie Yip (ECR); Xianming Tan, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cao, X., Zhou, JY., Huang, HY. et al. Final survival analysis of induction chemotherapy with lobaplatin and fluorouracil versus cisplatin and fluorouracil followed by concurrent chemoradiotherapy in nasopharyngeal carcinoma: a multicenter, randomized, phase 3 trial. Nat Commun 17, 2604 (2026). https://doi.org/10.1038/s41467-026-69315-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69315-1