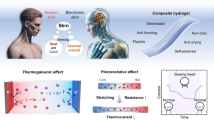
Abstract
Bridging biological and artificial systems, intelligent interfaces drive the demand for flexible electronics that emulate the skin’s multifunctionality. However, achieving such multifunctionality in a compact, self-sustained form remains challenging, as multimodal sensors often rely on rigid materials, discrete components, and external power sources. Herein, this study presents a single-component poly(vinyl alcohol) hydrogel e-skin integrating thermogalvanic, piezoionic, and diffusion mechanisms for self-powered sensing of skin temperature, arterial pulsation, and sweat secretion, simultaneously. The hydrogel features high stretchability, low modulus, and a prismatic architecture synergizing ionic polarization. Moreover, a temporal machine learning model with local attention is developed to decouple multimodal signals. Of practical importance, an active multimodal signal generator wristband is developed as a multifunctional human-machine interface for physiological detection, robotic control, and haptic feedback reproduction. Hence, this hydrogel e-skin represents an efficient material platform for intelligent interactions, showing broad potential for real-time health monitoring, robotic control, and virtual reality.
Similar content being viewed by others
Introduction
As human-machine integration technologies continue to develop, wearable electronic interfaces featuring multimodal sensing and closed-loop interactive capabilities have become a key enabler for intelligent devices1,2. To this end, flexible sensors are essential for real-time health monitoring of physiological parameters, such as body temperature, physical movements, and biochemical markers, which serve as the basis for embodied intelligence and human-machine interaction3,4. To date, single-signal monitoring wearable sensors have seen substantial development, with demonstrated applications across various physiological parameters5,6,7. To accelerate next-generation intelligent systems, current research has been increasingly focusing on integrated multifunctional platforms8,9. One feasible design strategy involves using thin, flexible substrates to accommodate multiple sensor modules that are based on different working mechanisms10,11. These modules, which may differ in response times and signal processing pathways, are arranged through stacked or parallel configurations into a representative architecture that enables coordinated multimodal sensing12,13. Despite the use of compact components, integrating different sensors and supporting circuitry often results in structurally complex, bulky devices that rely on external power sources14,15. As a result, such configurations not only add redundancy and complicate fabrication, but also compromise sensitivity and wearability, limiting their practical effectiveness in real-world scenarios16,17,18.
Unlike artificial electronic systems that rely on multiple discrete modules and complex circuitry, human skin exemplifies a seamlessly integrated, multimodal sensory system19,20,21,22. It features specialized dendritic endings of afferent nerve fibers that simultaneously perceive a diverse range of stimuli—temperature, pressure, and biochemical markers—with rapid response and high spatial resolution23. Inspired by natural skin, engineering electronic skins (e-skins) has attracted growing attention, driving the development of single-component multimodal sensors that simultaneously respond to multiple external stimuli24,25,26. These developments can simplify fabrication, lower energy consumption, and enable lightweight, miniaturized devices27,28. Recent advances in multimodal sensors and e-skins have demonstrated simultaneous perception of temperature, pressure, strain, and humidity by incorporating ionic conductors, conductive polymers, or hybrid nanocomposites (Supplementary Table 1)29. However, most existing single-component multimodal sensors suffer from asynchronous signal acquisition and redundant outputs, causing significant coupling errors and reduced sensing accuracy30. They also remain limited in system-level integration, often lacking capabilities of remote operation and independent energy supply31,32. In addition, only a few prototypes have achieved closed-loop perception and feedback, which is essential for practical human-machine interaction. Therefore, a major challenge lies not only in achieving skin-like multifunctionality but also in coordinating and integrating different mechanisms within a single material system33,34. Synergistic integration of these components is crucial for developing skin-like wearable devices that can simultaneously respond to multiple stimuli, enable closed-loop feedback, and operate with self-sustained power.
In this work, we present a hydrogel e-skin with self-powered multimodal sensing and closed-loop interactive capabilities. A high-strain (>800%), low-modulus (<300 kPa) poly(vinyl alcohol) (PVA) hydrogel is developed to achieve soft and conformal contact with the skin, which is critical for capturing subtle physiological signals. Multimodal sensing of body temperature, arterial pulsation, and sweat secretion is integrated within the PVA hydrogel, enabling real-time, non-invasive health monitoring. Each sensing function is modeled after a receptor mechanism in human skin: (i) thermal sensing via the thermogalvanic effect mimics the ionic current behavior of free nerve endings; (ii) piezoionic-enabled pulse monitoring reflects the mechanosensitive response of Merkel cells; and (iii) diffusion-based salt ion concentration sensing draws inspiration from the stretch-sensitive signaling of Ruffini endings (Fig. 1a). Notably, the thermogalvanic, piezoionic, and diffusion effects support both sensing and self-powering, enabling the hydrogel sensing unit to actively generate stimuli-responsive electrical signals. To integrate these mechanisms more effectively, the PVA hydrogel is engineered into a novel prismatic architecture that concentrates mechanical stress at the tip and synergizes the three effects along the skin-normal direction (Fig. 1b, Supplementary Figs. 1 and 2). This design also amplifies stress-induced ion polarization in the hydrogel, resulting in a significant increase in output current (from 0.7 to 100 μA) and mechanical sensitivity (GF > 18.7).
a Schematic illustration of the hydrogel e-skin and its multimodal sensing mechanisms compared to those of human skin: (i) thermogalvanic effect for temperature sensing mimics the ionic response of free nerve endings to temperature through thermosensitive ion channels (e.g., TRP channels); (ii) piezoionic sensing reflects the mechanosensitive behavior of Merkel cells and Ruffini endings through mechanically gated ion channels; and (iii) diffusion-based electrolyte sensing emulates the chemo-sensitive ion channels of free nerve endings (e.g., TRPV1, ASIC, etc.) b Synergistic integration of thermogalvanic, piezoionic, and diffusion effects within a prismatic structural design. c A fully integrated AMSG wristband and its structural overview, comprising a signal generation unit, a signal reproduction unit, and signal processing and transmission circuitry. The AMSG unit (right) includes a hydrogel-based sensor array and multiple encapsulation layers. d Schematic illustrations of the interface multifunctionality, including real-time monitoring of skin temperature, pulse waveform, and sweat Na+ concentration, together with human-machine interactions through gesture-based robotic control and haptic feedback reproduction.
Building on the PVA hydrogel e-skin, an active multimodal signal generator (AMSG) wristband interface is developed for multifunctional human-machine interactions. In addition to self-powered multimodal sensing, the interface enables active closed-loop interactions by enabling gesture-based robot control and haptic feedback reproduction. The AMSG wristband comprises a signal generation unit based on the hydrogel e-skin, a signal reproduction unit, and signal processing and transmission circuitry (Fig. 1c). All electronic components, including the flexible printed circuit board, the signal reproduction unit, the sensor array and their connections are fully encapsulated into a flexible wristband, providing a wearable and comfortable interface for human users. Using machine learning (ML) algorithms, including long short-term memory (LSTM) and convolutional neural network (CNN) for signal processing, this interface is capable of high-accuracy classification and recognition of complex stimuli, high-reliability response to hand gestures, and reproduction of tactile sensations from robotic fingers (Fig. 1d). Hence, the hydrogel e-skin offers a promising all-round toolkit for real-time multi-stimulus interaction in complex human-machine systems, with potential applications in intelligent prosthetics, soft robotics, and immersive virtual interfaces.
Results
Design principle
Figure 2a illustrates the preparation process of the PVA hydrogel e-skin. Through freeze-thawing and solvent substitution, nanocrystalline formation and structural densification are achieved, yielding hydrogels with high strength, high stretchability, and skin-like modulus. Specifically, PVA powder was first dissolved in dimethyl sulfoxide (DMSO) at a concentration of 15 wt.% (Supplementary Fig. 3) to obtain a homogenous solution. Upon freeze-thawing, PVA macromolecular chain conformations pre-assembled into a loosely interwoven network, while some non-covalent interactions within and between the polymer chains were disrupted. DMSO was subsequently substituted by glycerol, a poor solvent, to further enhance PVA intermolecular interactions over polymer-solvent interactions, facilitating their transition into a compact network via hydrogen bonding (Supplementary Fig. 4). However, excessive stiffness of the organogel at this point could cause nonconformal skin contact, reducing practical sensitivity and stability due to stress concentration and poor mechanical coupling. To address this, glycerol was finally replaced by an aqueous ferric solution, forming a hydrogel with preserved chain entanglement and crystallinity but reduced stiffness. Nuclear magnetic resonance (1H-NMR) spectroscopy was used to validate the completeness of solvent substitution. The characteristic hydroxyl peaks of glycerol (ca. 4.41 and 3.35) were observed only after glycerol substitution and disappeared upon replacement with the ionic solution in the final hydrogel e-skin (Fig. 2b and Supplementary Fig. 5).
a Schematic illustration of the fabrication process, including freeze-thawing (FT), glycerol substitution (GS), and ionic substitution (IS), leading to progressively enhanced hydrogel networks. b 1H-NMR spectra of FT, GS, and IS in DMSO-d6 at 295 K. c Tensile stress–strain curves of PVA hydrogels after different solvent substitution treatments, compared to the range of human skin (orange shaded region). d Toughness and stiffness of the hydrogels across different treatments. e Schematic of the mechanical enhancement mechanism involving dense crystalline and ionic crystalline regions. f DSC thermograms of FT, GS, and IS. g XRD profiles of FT, GS, and IS. h WAXS patterns showing the crystallinity evolution among FT, GS, and IS. i Synchronized and asynchronized 2D-COS maps of FT, GS, and IS, where red and blue indicate positive and negative intensity, respectively. j Comparison of hydrogel toughness achieved in this study with that of various reported hydrogels and biological tissues.
The sequential solvent substitution strategy offers a distinct advantage in preparing strong yet soft PVA hydrogels (Fig. 2c). To track mechanical changes throughout the process, PVA gels at each step after freeze-thawing, glycerol substitution, and final ionic solution substitution are designated as FT, GS, and IS, respectively. Notably, the fracture energy of GS (78.9 kJ m−2) and IS (50.4 kJ m−2) were 14 and 9 times that of FT (5.6 kJ m−2), respectively (Supplementary Fig. 6). Moreover, the final PVA hydrogel IS not only maintained the high stretchability of GS (>830%) but also reduced the effective modulus of elasticity from 2.73 MPa to 300 kPa (Fig. 2d), which is within the typical range of human skin.
The mechanical enhancement observed after glycerol substitution is attributed to the densified network structure, where PVA chains become more entangled and cross-linked (Fig. 2e). The nanocrystalline domains formed can arrest microcracks and hinder crack propagation via a pinning effect, thereby enhancing toughness. In amorphous regions, less entangled PVA chains act as sliding elements that distribute stress across the network, contributing to the overall strength and elasticity under deformation. Upon further stretching, the breaking of covalent bonds in individual chains releases not only their own elastic energy but also that stored in interconnected chains, enabling efficient energy dissipation. When glycerol is substituted by the ionic solution, Fe3+ ions can bind to the carboxyl oxygens of PVA chains and enhance cross-linking, but the aqueous environment also loosens the PVA network. Some strong interchain hydrogen bonds are thereby disrupted, reducing the energy dissipation from covalent bond breakage and the elastic energy stored in interconnected chains upon stretching. This effectively lowers the stiffness of the PVA hydrogel, despite the reduced stress response and overall toughness.
To validate and reveal the molecular mechanisms underlying the mechanical properties of PVA gels, a series of molecular-level characterizations were conducted. Differential scanning calorimetry (DSC) was performed to characterize the crystallinity of PVA gels. As shown in Fig. 2f, all samples exhibited a small endothermic peak around 170 °C, attributed to the melting of imperfect or loosely packed crystalline domains. In contrast, a prominent melting peak at 245 °C, especially in GS and IS, indicates the presence of more thermally stable, well-ordered crystalline domains. Using the melting enthalpy of 100% crystallized PVA as a reference, FT showed the lowest crystallinity of 7.92%, which increased to 30.2% in GS. IS exhibited a slightly lower crystallinity of 25.2% than GS, which is consistent with the observed decline in mechanical properties. As the hydrogen bonds between PVA chains can be partially disrupted by the aqueous solution and replaced by Fe3+ ionic coordination, the total number of hydrogen bonds is reduced in IS, thereby lowering the thermal stability of PVA molecular chains in its crystalline regions. This trend is also supported by Fourier-transform infrared (FTIR) spectroscopy (Supplementary Fig. 7). The stretching peak of the hydrogen bond O–H at ~3300 cm−1 exhibited a red shift in GS and a blue shift in IS, indicating enhanced hydrogen bonding with glycerol substitution and its reduction after ionic replacement.
X-ray diffraction (XRD) analysis was performed to further investigate the crystalline structures of the gels (Fig. 2g). All PVA gels exhibited a characteristic diffraction peak at 2θ = 19.3°, corresponding to the (\(10\bar{1}\)) plane of semi-crystalline PVA35. The peak was relatively weak in FT but became more pronounced in both GS and IS, indicating increased crystallinity following glycerol and ionic solution substitution. Wide-angle X-ray scattering (WAXS) further revealed the primary diffraction peaks of FT, GS, and IS at 1.32, 1.37, and 1.36 Å−1, respectively (Fig. 2h and Supplementary Fig. 8). According to the Bragg equation L = 2π/qmax, the average distance between crystalline domains decreased slightly from 0.48 nm in FT to 0.46 nm in GS and IS, indicating tighter packing of the crystalline domains. The comparable peak intensities and inter-crystalline distances in GS and IS suggest that the slight reduction in the crystallinity of IS is mainly due to water-induced loosening of the polymer network, while the crystalline domains formed during glycerol substitution are mostly preserved even after ionic solution substitution.
Two-dimensional correlation spectroscopy (2D-COS) can reveal more subtle changes in the molecular interactions of PVA gels (Fig. 2i)36. The synchronous and asynchronous spectra capture simultaneous and sequential variations in spectral intensity at specific wave numbers37, with red indicating positive correlations and blue indicating negative correlations. Five auto peaks were identified in the synchronous spectrum, centered at 1149 cm−1, 1143 cm−1, 1118 cm−1, 1084 cm−1, and 1013 cm−1. Based on Noda’s rule, the sequence of spectral changes was determined as 1084 cm−1 → 1143 cm−1 → 1118 cm−1 → 1149 cm−1 → 1013 cm−1, corresponding to the vibrations of C–O stretching (hydrogen-bonded) → C–C skeleton vibrations → C–C/C–O stretching in the main chain → C–C stretching in the main chain → C–O stretching (non-hydrogen-bonded). This sequence suggests that the introduction of Fe³⁺ in the ionic solution preferentially disrupts the original hydrogen-bonded hydroxyl network structure, destabilizing the interchain hydrogen bonding and the hydroxyl-rich PVA backbone. As a result, the PVA chain segments gradually rearrange into a gently softened cross-linked structure.
These characterization results reveal that while the stiffness of IS is significantly reduced, its crystallinity remains relatively high. This decoupling of crystallinity and stiffness is primarily attributed to the partial disruption of strong interchain hydrogen bonds and the formation of weaker Fe³⁺-mediated ionic interactions. Therefore, the sequential solvent substitution strategy shows clear advantages over conventional methods such as freeze-thawing, wet annealing, salt precipitation, and tensile-assisted processing38,39,40,41,42,43,44,45. Unlike these approaches that typically increased toughness at the expense of higher stiffness, this method enables the fabrication of soft, elastic PVA hydrogels (Fig. 2j, Supplementary Tables 2–4). The controlled stiffness allows the hydrogel to better conform to the skin and match the modulus of human tissue, thereby improving comfort and wearability.
Device characteristics
Leveraging its high stretchability, low Young’s modulus, and good biocompatibility, the PVA hydrogel e-skin shows strong potential as a flexible sensor material that can be conformally attached to human skin. More importantly, through the Fe³⁺ ionic solvent substitution, the hydrogel e-skin is ingeniously engineered for multimodal sensing of skin temperature, pressure, and electrolyte (Na⁺) concentration. Temperature sensing is achieved via the thermogalvanic effect, where a temperature gradient leads to redox imbalances. Pressure sensing relies on the piezoionic effect, in which mechanical stress induces directional migration of unipolar ions. Electrolyte sensing is enabled by the diffusion effect, where ion concentration gradients lead to differential ion diffusion. As these mechanisms operate through distinct ion-driven processes, they function independently in the same system with minimal interference, allowing for synergistic multimodal sensing.
Temperature sensing performance
First, the hydrogel e-skin functions as a self-powered temperature sensor, leveraging the natural temperature difference between human skin and the ambient environment to spontaneously generate a thermoelectric voltage. Under a temperature gradient, the Fe²⁺/Fe³⁺ redox equilibrium within the hydrogel e-skin shifts asymmetrically, with the reversible reaction favoring the lower-temperature direction. This generates a potential difference between the electrodes and drives a current through the external circuit (Fig. 3a). The reversibility of the redox reaction is confirmed by cyclic voltammetry (CV) tests (Supplementary Figs. 9 and 10), where the CV curves remained highly overlapping in 10 cycles at different scan rates and temperatures. This indicates that the oxidized species (Fe3+) formed at the hot side and the reduced species (Fe2+) at the cold side migrate efficiently to opposite electrodes under the concentration gradient, sustaining continuous current output.
a Schematic illustration of the thermogalvanic effect as the temperature sensing mechanism. b MD simulation of the PVA chain structure and its solvation behavior around Fe²⁺/Fe³⁺ ions, with inset images showing the first solvation shell. c Radial distribution function g(r) (solid lines) and coordination number N(r) (dotted lines) of oxygen atoms surrounding Fe3+ and Fe2+ ions in the PVA hydrogel. d Output voltage-current curves and corresponding power densities under various temperature gradients. e Output current and current density as a function of temperature difference. f Schematic of the piezoionic effect for pressure sensing. g Enhancement of ionic piezoelectric output in d33 mode using a prismatic architecture. h COMSOL simulation of ion concentration gradients and potential distributions under pressure. i COMSOL simulation of stress distributions in PVA gels at increasing deformation. j Relative current change as a function of applied pressure. k Waveform signals measured from radial artery pulses. l Contact angle measurement of various carbon paper surfaces: Pristine, PDMS-coated, Perforated, and Hydrochloric acid soaked. m Top view of the sweat transport electrode and its transport capacity (scale bar: 1 mm). n Schematic of the diffusion effect for sweat detection. o Current responses to different sweat Na+ concentrations. p Repeated current responses over 10 alternating cycles of NaCl addition and water dilution.
Across different Fe²⁺/Fe³⁺ concentrations in the PVA hydrogels (Supplementary Fig. 11a), the highest Seebeck coefficient (\({S}_{e}\)) of 1.6 mV K−1 was found at a 0.05 M concentration, slightly lower than that in pure Fe²⁺/Fe³⁺ aqueous solutions (1.7 mV K−1). The high water content of the hydrogel also facilitates the thermogalvanic effect, as \({S}_{e}\) would rapidly drop at lower water content (Supplementary Fig. 11b). According to the liquid thermoelectric theory46,47,48, \({S}_{e}\) is the ratio of the temperature difference (\(\Delta T\)) to the potential difference (\(\Delta E\)) and directly related to the entropy change of the redox reaction (\(\Delta {S}_{{\mathrm{rc}}}\)) by the equation \({S}_{e}=\Delta E/\Delta T=\Delta {S}_{{\mathrm{rc}}}/{nF}\), where n is the number of electrons transferred and F is Faraday’s constant. As described by Powell49, the molar entropy of ions is influenced by both their charge and effective ionic radius. Therefore, molecular dynamics (MD)50,51,52 simulation is used to estimate the solvation radii of Fe²⁺/Fe³⁺ ions in PVA hydrogels (Fig. 3b and Supplementary Note 1). The radial distribution function (RDF) and coordinated number distribution of oxygen atoms around Fe²⁺/Fe³⁺ ions were calculated, with the distance to the first minimum in the Fe–O RDF taken as the radius of the first solvation shell (R1). Both Fe3+ and Fe2+ exhibited a value of 6 for N(r) in r = R1, indicating that they are bonded to 6 aqueous molecules in the first solvation shell (Supplementary Tables 5 and 6)53. The R1 of Fe3+ and Fe2+ ions were 2.25 and 2.33 Å, respectively, with a difference of 0.08 Å, smaller than the interpolated value of 0.1 Å in pure water (Fig. 3c). While this slight reduction is attributed to the tighter binding of water molecules to Fe3+and Fe2+ ions caused by PVA polymerization, the overall entropy change remains close to that in pure water. This indicates that the polymer network has minimal impact on the ionic thermoelectric effect, which is consistent with the measured Seebeck coefficients.
The thermoelectrical performance of the PVA hydrogel was evaluated by its electric outputs under various temperature gradients (Fig. 3d). As ΔT rose from 8 to 24 K, the short-circuit current (Isc) increased from 5.2 to 14.5 μA, and the open-circuit voltage (Voc) from 15 to 37 mV. Based on these measurements, the maximum output power density (\({P}_{\max }\)) was calculated as 11 mW m−2, with the normalized power density (\({P}_{\max }{\Delta T}^{-2}\)) reaching 19 μW m−2 K−2. Moreover, the hydrogel demonstrates sensitivity to temperature changes as small as 0.1 K (Supplementary Fig. 12). Since the distance from the heat source can also affect output, the highest current density was found at a 3-mm distance, with lower performance observed at both closer and further distances due to a reduced temperature gradient (Supplementary Fig. 13).
To achieve stable and reliable target signals, the PVA hydrogel was fabricated into multiple four-pronged modular units and assembled into an array. At ΔT = 10 K, Voc stabilized at 16 mV, while Isc increased to 20 μA when scaled to 20 parallel units, demonstrating a highly linear relationship between current output and number of units (Supplementary Fig. 14). At increasing ΔT, both Isc and Voc showed a positive correlation, consistent with the COMSOL simulation results (Fig. 3e, Supplementary Figs. 15 and 16). In addition, the effective thermal conductivity (keff) of the hydrogel remained stable at ~0.45 W m−1 K−1 at temperatures from 295 to 320 K (Supplementary Fig. 17), while electrochemical impedance spectroscopy showed negligible variation in internal resistance (Supplementary Fig. 18). Although the final prismatic structure may slightly reduce thermoelectric currents compared with simple cubic blocks—due to the smaller electrode contact area at the apexes and higher overall resistance from the reduced volume—it still maintains a robust linear response (Supplementary Fig. 19). Together, these properties ensure stable thermal sensing performance of the hydrogel over a wide operating temperature window.
Pressure sensing performance
The piezoionic effect in the hydrogel e-skin arises from the charge imbalance induced by deformation-driven ionic flux (Fig. 3f). Within the hydrogel, the diffusion of anions is partially hindered by the polymer network due to their relatively large size, leading to localized ionic enrichment under deformation. As anions and cations are driven at different rates by stress concentration, their distinct distribution gradients generate a transient potential difference and a net current along the deformation direction. Further leveraging this, the prismatic structure design is introduced to amplify the vertical stress concentration, generating an electric field along the force direction (d33 mode). Compared to the cubic structure, this design achieved over a hundredfold increase in output current under identical stress conditions (Fig. 3g and Supplementary Fig. 20). Using COMSOL simulations, the ion movement and distribution in the prism were revealed (Fig. 3h). Ions were initially assumed to be distributed uniformly with no potential difference across the hydrogel. Upon loading, stress concentration and deformation at the prism tip led to changes in contact area and surface charge, resulting in high sensitivity to even low pressures (Supplementary Fig. 21).
Theoretically, the relationship between output current and applied pressure is derived in Supplementary Note 2 as
where \({n}_{+/-}\) are the concentration of cations and anions, \({Z}_{+/-}\) are their respective charges, \(e\) is the elementary charge (1.602 × 10−19 C), κ is permeability, η is viscosity, h, p, and q are prism height, bottom side length, and top side length, respectively, \(k\) is the mechanical transfer efficiency, N is the number of prisms, \(E\) is the effective Young’s modulus, and b is the prism height at steady state under pressure. This formula describes a linear relationship between output current and applied pressure. Meanwhile, the sensitivity (S) of the hydrogel e-skin was determined experimentally as the slope of the relative current change versus pressure, defined by \(S=\delta (\Delta I/{I}_{0})/\delta P\) 54\(,\) where \(\Delta I=I-{I}_{0}\) is the change in current, \({I}_{0}\) is the initial current, and \(P\) is the applied pressure. As shown in Fig. 3i, the hydrogel exhibited a continuous current-pressure response, showing a higher sensitivity of 1.94 kPa−1 at pressures below 2 MPa and 0.4 kPa−1 above 2 MPa. While the linearity aligns with theoretical derivations, the sensitivity change is attributed to the prismatic structure. At low pressures, the prism tip deforms more readily, resulting in higher sensitivity. As pressure increases, the base of the prisms is stiffer and more resistant to deformation, thereby reducing sensitivity. This stiffness effect is also captured in Eq. (2) and supported by COMSOL simulations of the three-dimensional structure, which reveal that higher pressures are required to achieve deformation near the prism bases (Fig. 3j and Supplementary Fig. 22).
The repeatability of the hydrogel e-skin was evaluated under cyclic mechanical loading. While repeated loading typically leads to energy dissipation and irreversible structural changes in hydrogels, the hydrogel e-skin showed minimal long-term performance loss. Mechanical hysteresis and the Mullins effect were most evident only during the first loading-unloading cycle and quickly stabilized after two cycles (Supplementary Fig. 23). The hysteresis loops were mostly overlapping over 20 cycles, further indicating the hydrogel’s highly elastic behavior (Supplementary Figs. 24 and 25). In addition, across various loading frequencies, it exhibited stable current responses under both repeated stretching and compression (Supplementary Fig. 26). No significant decline in current output was observed even after over 500 cycles (Supplementary Fig. 27).
Moreover, the low-modulus prismatic hydrogel e-skin showcases high sensitivity to pressures as low as 60 Pa (Supplementary Fig. 28). Its dynamic response features a rapid pressure response time of only ~37 ms (Supplementary Fig. 29), which is significantly faster than the temperature response time (15.2 s) and comparable to that of human skin (30–50 ms). The hydrogel e-skin is also capable of generating distinct current response waveforms at different compression rates, such as 1 and 10 mm s−1, indicating its ability to dynamically track varying loading speeds (Supplementary Fig. 30). With its high sensitivity and repeatability to pressure stimuli, the hydrogel e-skin shows strong potential for physiological monitoring. Notably, it can capture high-resolution pulse waveforms (Fig. 3k and Supplementary Fig. 31), with clearly identifiable Percussion (P), Tidal (T), and Dicrotic (D) waves, demonstrating its suitability for real-time pulse and blood pressure monitoring.
Electrolyte sensing performance
In addition to temperature and pressure sensing, the hydrogel e-skin is also designed for continuous, non-invasive monitoring of sweat electrolytes, another key indicator of physiological health. To this end, the polydimethylsiloxane (PDMS) substrate in contact with the skin is engineered with interfacial hydrophilic pores (Supplementary Fig. 32). Although PDMS is widely used as a substrate material in wearable and microfluidic devices due to its flexibility and physicochemical stability, its inherent hydrophobicity greatly limits its effectiveness in liquid transport applications14. To overcome this, the PDMS surface is modified through hydrochloric acid treatment to achieve durable hydrophilicity. The improved surface wettability, as evidenced by a reduced contact angle (Fig. 3l and Supplementary Fig. 33), significantly enhances sweat transport efficiency, resulting in a transport rate of 3.3 ml min−1 upon initial surface moisturization by sweat (Fig. 3m).
Through the engineered hydrophilic channels, sweat is captured by the hydrogel e-skin and transported across the concentration gradient, generating a potential difference and a measurable current change (Fig. 3n). In addition, the ionic diffusion is further enhanced by the Soret effect, which arises from the temperature gradient between the skin and the ambient environment and leads to differential ion mobility. Using NaCl solution to emulate human sweat, the hydrogel e-skin demonstrates excellent linearity and reusability. In Fig. 3o, the output current gradually increased to 2 μA as the ion accumulated during sweat uptake, whereas adding pure water diluted ion concentration, leading to a significant decrease in current. Moreover, the hydrogel maintained a stable current response over 10 cycles of alternating NaCl solution and pure water stimulation, demonstrating reusability for practical applications (Fig. 3p). Compared with cubic shapes, the prismatic structure showed no significant difference in ion diffusion currents, as the two ion transport distances were comparable in solvated environments (Supplementary Fig. 34).
Together, these results demonstrate the strong performance of the hydrogel e-skin, with its desirable sensitivity, stability, and detection range to temperature, pressure, and electrolyte stimuli. Notably, its self-powered multimodal sensing marks a significant advantage over previously reported sensors that relied on external power sources or unsustainable self-powered systems.
Active multimodal signal generator wristband interface
Building upon the self-powered multimodal sensing capability of the hydrogel e-skin, an AMSG interface is developed. The hydrogel-based AMSG unit is assembled with a signal replication module, signal processing and transmission circuits, and a flexible encapsulation layer into a highly integrated, multifunctional wristband. This interface not only enables reliable, real-time, and non-invasive monitoring of the aforementioned key physiological signals but also supports comprehensive human-machine interactions through gesture command control, remote temperature alerts, and haptic feedback.
Machine learning-based multimode sensing applications
The human body, as a natural heat source, typically maintains a skin temperature around 36 °C, which may vary with health conditions such as fever. Simultaneously, arterial pulsations and spontaneous perspiration produce continuous pressure and ionic stimuli, respectively, as key physiological inputs for the AMSG. To replicate these stimuli in a controlled laboratory setting, a custom multimodal stimulation platform is developed (Fig. 4a). It simulates body temperature using a heated Peltier element at the base, emulates arterial pulses via applied pressure from above, and replicates sweat secretion by dispensing NaCl solution. Notably, the AMSG exhibits distinct current patterns in response to each stimulus: with temperature changes, the current increases and decreases gradually; under applied pressure, it shows a sharp rise followed by a slow recovery; and with varying electrolyte concentrations, the current magnitude changes proportionally.
a Schematic illustration of the AMSG wristband interface for multimodal sensing of body temperature, pulse rate, and sweat secretion, simulated by individually applying temperature, pressure, and Na+ electrolyte stimuli to obtain their characteristic signals. b Architecture of the machine learning-based signal decoupling model, featuring three LSTM layers with local attention and three fully connected layers for extracting and decoupling the signal features of each stimulus independently. c Internal structure of the LSTM network. d Comparison of compound signals under multiple simultaneous stimuli and their decoupled components. e Multimodal monitoring of body temperature, pulse rate, and sweat Na+ concentration across various activity states (relax, walk, run, sleep, fever), benchmarked against commercial devices. f User interface displaying the real-time waveforms of multimodal signals and corresponding monitoring outputs.
To investigate potential crosstalk between the signals, we applied repeated pressure stimuli of 2, 4, 6, and 8 kPa under different temperature gradients (Supplementary Fig. 35a) and different Na+ concentrations (Supplementary Fig. 35b). The piezoionic current increments remained nearly identical in all cases, indicating that the pressure-induced ionic signals are not affected by variations in temperature nor Na⁺ concentration. This independence arises from the distinct physical origins of the signals: the piezoionic current is generated by mechanically induced, transient ionic polarization, whereas the thermoelectric and diffusion currents are driven by inherently slow, steady-state processes. A minor interaction was observed between the temperature and Na⁺ concentration signals: a larger temperature gradient produced higher diffusion currents because temperature not only drove the thermogalvanic potential but also triggered the Soret effect, causing additional Na+ migration along the thermal gradient (Supplementary Fig. 36). Nevertheless, within the temperature range investigated in this work, both temperature- and concentration-driven diffusion remained in the linear regime, which allows their contributions to superimpose proportionally and produce a stable, predictable current response.
It is also worth noting that the evolution of the current response typically exhibits a higher-order Markov property. Therefore, the output at any given time depends primarily on the present and recent stimulus states rather than on distant past or future inputs55. This localized dependence and stationary behavior provides a theoretical basis for real-time decoupling of multiple simultaneous physiological inputs. To implement this, an ML-based decoupling model is developed (Fig. 4b), featuring three stacked LSTM layers with a local attention mechanism, followed by three fully connected layers. As a variant of recurrent neural networks (RNNs), LSTM integrates a forget gate, input gate, and output gate to adaptively regulate information flow and efficiently capture long-term dependencies56,57. The attention mechanism is mathematically defined as:
where \({e}_{{{\rm{t}}},{{\rm{j}}}}\) is the attention weight, \({s}_{{{\rm{t}}}}\) is the input signal at the current time step, \({s}_{{{\rm{j}}}}\) is the historical signal within a certain window, and \({c}_{{{\rm{t}}}}\) is the context vector. \({c}_{{{\rm{t}}}}\) and the current signal \({s}_{{{\rm{t}}}}\) over the latest 10 s are concatenated to obtain \({x}_{{{\rm{t}}}}\) for each LSTM time step. Given that a single stimulus typically generates a signal lasting about 40 s, the local attention window is set to [t−40, t + 40] to capture the most relevant temporal context. This localized attention not only enhances feature extraction from critical regions but also substantially reduces computational complexity by minimizing the influence of noise from distant time points58. Based on this attention mechanism, the internal model of LSTM is further constructed (Fig. 4c), with its internal computational flow defined by the following equations:
where \({f}_{{{\rm{t}}}}\) is the forget gate output, which retains a portion of the historical state, \({i}_{{{\rm{t}}}}\) is the input gate modulating the contribution of new input, \(\widetilde{{C}_{{{\rm{t}}}}}\) is the candidate cell state, representing a nonlinear transformation from the current input to previous hidden states, \({C}_{{{\rm{t}}}}\) is the cell state that stores long-time memory, \({O}_{{{\rm{t}}}}\) is the output gate that determines the output from the cell state, and \({h}_{{{\rm{t}}}}\) is the final output of the LSTM. Subsequently, a three-layer fully connected network is appended to the final LSTM layer to map the stimulus state vector \({C}_{{{\rm{t}}}}\) at every time step, thereby decoupling the real-time signals of temperature, pressure, and Na⁺ electrolyte concentration. This design enables the AMSG to accurately perceive and differentiate multimodal stimulus responses.
When temperature, pressure, and Na⁺ electrolyte stimuli in their characteristic patterns are simultaneously applied to the AMSG, the resulting compound current signal can be accurately decoupled into distinct components corresponding to each stimulus (Fig. 4d and Supplementary Fig. 37). In particular, over the entire test duration of 3000 time steps (sampling rate: 10 Hz), the LSTM model achieved mean absolute errors below 2% for temperature and pressure sensing. Despite the relatively weak current signals from the Na⁺ electrolyte stimulus, the model maintained an estimation error within 30%.
To further validate its performance, five representative physiological states—relaxing, walking, running, sleeping, and fever—are simulated with controlled variations in body temperature (36–41 °C), heart rate (60–130 bpm), and sweat Na+ concentrations (10–90 mmol L−1) (Fig. 4e and Supplementary Movie 1). The output signals closely matched those from commercial sensors, confirming the accuracy and reliability of the AMSG and decoupling model. Integrated with a multimodal monitoring module and a user interface, the system can display real-time variations in the physiological signals. For example, during running, the monitored pulse rate elevated to 133 bpm, while the Na+ concentration accumulated to 90 mmol L−1 due to increased perspiration. During sleep, on the other hand, the pulse rate dropped to 62 bpm, while sweat secretion was negligible with Na+ concentration approaching zero. In addition, these data can be transmitted via cloud connectivity to remote health management systems for continuous monitoring and feedback (Fig. 4f).
Machine control and haptic reproduction
Beyond multimodal physiological monitoring, the AMSG is further integrated into a human-machine interface with closed-loop control and feedback capabilities. As finger and wrist movements activate certain superficial forearm muscles, the resulting muscle contractions generate pressure signals. These subtle signals can be detected by the AMSG with the aid of a woven wristband that applies gentle back pressure to ensure firm skin contact (Supplementary Figs. 38 and 39). Combined with deep learning, the signals associated with finger and wrist motions are decoded into gesture commands for real-time robotic arm control.
As shown in Fig. 5a, the AMSG wristband functions as a self-powered wireless human-machine interface (Supplementary Movie 2). Characteristic current signals are generated by various gestures, such as grasping, flexing, extending, and rotating (Supplementary Fig. 40), and transmitted via Bluetooth to a processing workstation. To extract relevant features and accurately recognize gesture commands, a ResNet-34 deep learning framework based on a one-dimensional CNN is constructed. The framework comprises a convolutional layer, a max-pooling layer, and a fully connected layer for automated feature extraction and classification, translating the user gesture inputs into robotic arm movements. The dataset is randomly divided into a training set (75%) and a test set (25%) for model training and validation. As a result, the AMSG wristband interface achieved an average recognition accuracy of 97.57% across 10 wrist gestures in 200 sign language tests (Fig. 5b). Furthermore, principal component analysis (PCA) was performed to project high-dimensional signal sequences into a 2D feature space for visualizing the model’s classification performance (Fig. 5c). The PCA distribution revealed distinct class boundaries with signals from the same gesture clustering together and those from different gestures well separated. Finally, through the mechanical control module, the user interface can provide real-time visual feedback of gesture commands and corresponding robotic arm actions (Fig. 5d). These capabilities demonstrate significant potential for digital twin applications.
a Real-time control of a robotic arm using hand gestures captured by the AMSG wristband and interpreted via a deep learning model. b Confusion matrix showing the classification accuracy of 10 distinct gestures, with each based on the current signal sequences of 200 test samples. c Principal component analysis of 10 gestures and their cluster differentiation. d User interface displaying the current signals of gesture commands and corresponding robotic motion outputs. e Illustration of a use-case scenario where the temperature and pressure detected by the AMSG on the robotic hand are reproduced on the user’s wristband. f Modulation of temperature and vibration responses according to the current magnitude. g Current signals of the AMSG as the robotic hand approaches or retreats from heat or cold sources, illustrated by optical and infrared images. h Current signals when gripping a paper cup, where variations in the amplitude and tangent angle are used to determine appropriate force levels. i User interface displaying real-time haptic signal waveforms and haptic reproduction parameters.
Furthermore, two-way communication between the user and the robotic system is achieved by applying the AMSG at both ends: while the wristband captures user gestures to control robotic motion, the AMSG on the robotic hand can also sense temperature and pressure variations during interaction and relay this information back to the user (Fig. 5e). To complete this feedback loop, a thermal actuator and a haptic feedback module are integrated into the user wristband, combining both sensory input and haptic feedback within a single wearable platform (Supplementary Movies 3 and 4). When the robotic hand grasps an object, the AMSG at its fingertip detects temperature and pressure variations, generating corresponding signal outputs. These signals are then processed by a control module, which linearly maps them to the thermal output of a micro-Peltier device and the vibration frequency of a linear motor (Fig. 5f). As a result, the user can perceive the object’s temperature and contact forces in real time, enabling haptic reproduction without direct physical interaction. The integration of deep learning algorithms and processing modules further enables a range of intelligent features. For example, a thermal safeguard function is built into the AMSG wristband interface. By analyzing current responses as the robotic hand approaches hot or cold surfaces, the interface is able to distinguish between safe and hazardous temperatures. When the surface temperature exceeds 80 °C or drops below −20 °C, thermal and vibrational warnings can be delivered to alert the user in real time (Fig. 5g).
Figure 5h demonstrates a representative application scenario showcasing the capability of the AMSG wristband interface, including sensing, haptic feedback, as well as safety alert functions. The entire workflow produced a three-stage current profile: (1) non-contact temperature sensing, (2) contact-based accurate temperature sensing, and (3) grip-force sensing (Supplementary Movie 5). As the robotic hand approached a paper cup filled with hot water, the current signal gradually increased to 14 μA. If the detected temperature remained within a safe range, the grasping action would proceed; otherwise, robotic motion would be stopped. Upon contact, the current rose to 30 μA due to the sudden temperature increase. Meanwhile, the thermal actuator at the user end was triggered to reproduce the detected temperature. A similar response can also be achieved for cold objects, enabled by the polarity of the thermoelectric signal. In addition, the safety feature is integrated into the pressure-sensing feedback. For specific objects, an adaptive grasping action generates a piezoelectric signal below a set threshold. By comparing the current amplitude (C1) and slope angle (θ1) with the set threshold, the interface detects excessive gripping force. If the current amplitude (C2) and slope angle (θ2) exceed their limits, grasping is stopped to prevent potential damage to the object. In this process, real-time pressure and temperature information is transmitted via Wi-Fi and displayed on the user interface (Fig. 5i). Together, these demonstrations highlight the potential of AMSG as a self-powered, multifunctional human-machine interface.
Discussion
In summary, this work developed a self-powered, single-component hydrogel e-skin that effectively emulated the key functions of human skin, integrating thermogalvanic, piezoionic, and diffusion effects for multimodal sensing of temperature, pressure, and sweat electrolyte, respectively. Composed of a highly stretchable (>800% strain), low-modulus (<300 kPa) PVA hydrogel and engineered with a prismatic architecture, the e-skin demonstrated desirable skin conformity, sensitivity, and repeatability for reliable, non-invasive physiological monitoring. Its further integration into an AMSG wristband, combined with ML algorithms for signal decoupling, achieved both real-time multimodal health monitoring and closed-loop human-machine interaction. These functions included gesture-based command control, haptic feedback reproduction, and additional safety functions. Therefore, this study presented a versatile material platform for self-powered, multimodal sensing by synergizing thermogalvanic, piezoionic, and diffusion effects within a single, architecturally engineered hydrogel matrix. Enabled by ML-based signal decoupling and high-level system integration into wearable electronics, the interface demonstrated intuitive and multifunctional human-machine interaction for potential applications in health monitoring, soft robotics, and immersive virtual environments.
Methods
Materials
PVA (Mw = 89,000–98,000, 99% hydrolyzed), FeCl3·6H2O (Mw = 270.3, ≥99%), and FeCl2·4H2O (Mw = 198.81, ≥99%) were purchased from Macklin Industries Corporation. Hydrochloric acid (37%), Glycerol (AR, ≥99%), NaCl (Mw=58.44, Spectral level), and DMSO (Mw = 78.13, ≥99.5%) were purchased from Aladdin Industrial Corporation. All chemicals were used as received. Graphite papers of 0.1 mm thickness were used as electrodes.
Fabrication of freeze-thawed PVA hydrogels
PVA powder was dissolved in deionized water under vigorous stirring at 95 °C for 3 h to form a homogeneous solution (15 wt.%). The solution was cast into Teflon molds and then frozen at −20 °C for 8 h, followed by thawing at 25 °C for 2 h. The freeze-thawing cycle was repeated three times to obtain the FT hydrogels.
Fabrication of GS PVA organogels
PVA powder was dissolved in DMSO under vigorous stirring at 95 °C to form a 15 wt.% solution. After degassing via sonication for 2.5 h, the obtained clear solution was cast into 3D-printed molds and frozen at −80 °C for 2 h. The frozen gels were then immersed in glycerol at room temperature for 24 h to initiate substitution between DMSO and glycerol, with the glycerol replaced at least twice to ensure thorough exchange. The resulting organogel was designated as GS.
Fabrication of IS PVA hydrogels
Different concentrations of FeCl2/ FeCl3 solutions (0.01 M, 0.02 M, 0.05 M, 0.1 M) were prepared by dissolving FeCl2·4H2O and FeCl3·6H2O powder in deionized water, followed by sonication for 10 min to obtain clear solutions. GS PVA hydrogels were then immersed in the ionic solutions for 48 h at room temperature. The resulting hydrogels were designated as IS.
Preparation of porous electrodes
PDMS was spin-coated onto graphite paper at 400 rpm for 50 s and then cured at 80 °C for 20 min. The coated layer was patterned into porous structures (500 μm diameter, 1 mm spacing) using laser cutting, followed by immersion in 37% hydrochloric acid for 1 h to obtain the final porous electrodes.
Water content measurement
The hydrogel was immersed in 0.05 M FeCl2/FeCl3 solution for 1–30 days. The initial weight and the weight after immersion (\({m}_{{{\rm{a}}}}\)) were recorded, and the water content was calculated as
Mechanical test
All mechanical tests were conducted using a universal tensile testing machine at room temperature. For tensile tests, PVA gels were cut into dog-bone-shaped specimens with a gauge width of 3 mm and a thickness of 2 mm. Tensile tests were conducted at a loading rate of 100 mm min−1, and the integrated area under the stress–strain curve was calculated as toughness. Compression tests were carried out at a rate of 50 mm min−1. For each group, at least three independent specimens were tested.
Pure shear test
Hydrogels were cut into rectangular specimens with a gauge length of 40 mm, a width of 3 mm, and a thickness of 2 mm as measured using a caliper. To obtain the fracture energy, two identical samples, one notched and one unnotched, were tested as a pair26. For the notched sample, a 1.5-mm straight cut was introduced at the midpoint of the gauge length. An initial clamp distance of 1 or 2 mm was used for every pair of specimens, which were loaded at a rate of 100 mm min−1. The fracture energy was calculated by:
where \(A\) is the cross-sectional area (width multiplied by thickness), \(\Delta {L}_{{{\rm{c}}}}\) is the critical elongation upon crack initiation in the notched sample, and \(U(\Delta {L}_{{{\rm{c}}}})\) is the mechanical work done to stretch the unnotched sample to \(\Delta {L}_{{{\rm{c}}}}\).
1H-NMR characterization
Gel samples were first dissolved in DMSO-d6, and the mixtures were analyzed using 1H nuclear magnetic resonance (NMR) spectroscopy (Bruker Plus 600 MHz, Germany).
ATR-FTIR characterization
FTIR spectroscopy coupled with an attenuated total reflectance module (ATR-FTIR, Cary610/670, Varian, USA) was used to characterize the functional groups of the hydrogels.
XRD characterization
XRD spectra of hydrogels were obtained using an X-ray diffractometer (Rigaku Smart Lab SE) with a Cu Kα radiation source with a scanning range from 5° to 90° and a step size of 0.02°.
DSC characterization
The crystallinity of PVA gels was measured by DSC at a heating rate of 20 °C min−1 from 50 to 250 °C under argon atmosphere at a flow rate of 50 ml min−1. The hydrogel slices were first immersed in a chemical cross-linking agent (consisting of 10 ml of 50 vol% glutaraldehyde, 1 mL of 36.5 to 38 wt.% hydrochloric acid, and 100 ml of ultrapure water) to stabilize the amorphous chains. They were then washed thoroughly with ultrapure water for 2 h to remove excess hydrochloric acid and dried in a blast oven at 40 °C for 24 h. The melting enthalpy of the hydrogel was calculated by integrating the area of the melting heat absorption transition peak between 200 °C and 250 °C. Its ratio to the melting enthalpy of 100% crystallized PVA (138.6 J g−1) was used to determine the degree of crystallinity.
Theoretical calculation of the maximum output power density (\({P}_{\max }\)) and normalized power density (\({P}_{\max }{{\mathrm{\varDelta T}}}^{-2}\))
For the theoretical calculation of \({P}_{\max }\):
in which
where \({V}_{{{\rm{oc}}}}\) is the open-circuit voltage, \(I\) is the current density, \({I}_{{{\rm{SC}}}}\) is the short-circuit current, and \(A\) is the cross-sectional area.
With Pmax, the normalized power density \(({P}_{\max }{\Delta T}^{-2})\) can be calculated as
where \(\Delta T\) is the temperature difference between hot and cold ends.
Ethics oversight
All procedures involving human research participants were conducted in accordance with the experimental protocol approved by the Ethics Committee of Taiyuan University of Technology (TYUT2026012301). All participants were informed with written consent.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data generated in this study are provided in the main text and the Supplementary Information. Additional data are available from the corresponding author upon request. Source data are provided with this paper.
References
Li, D. et al. Touch IoT enabled by wireless self-sensing and haptic-reproducing electronic skin. Sci. Adv. 8, eade2450 (2022).
Sun, Y. et al. Stretchable and smart wettable sensing patch with guided liquid flow for multiplexed in situ perspiration analysis. ACS Nano 18, 2335–2345 (2024).
Ma, X. et al. A monolithically integrated in-textile wristband for wireless epidermal biosensing. Sci. Adv. 9, eadj2763 (2023).
Zhang, L. et al. Cellulose nanofiber-mediated manifold dynamic synergy enabling adhesive and photo-detachable hydrogel for self-powered E-skin. Nat. Commun. 15, 3859 (2024).
Ha, K.-H. et al. Full freedom-of-motion actuators as advanced haptic interfaces. Science 387, 1383–1390 (2025).
Cheng, A. et al. An intelligent hybrid-fabric wristband system enabled by thermal encapsulation for ergonomic human-machine interaction. Nat. Commun. 16, 591 (2025).
Zhang, Z. et al. Active mechanical haptics with high-fidelity perceptions for immersive virtual reality. Nat. Mach. Intell. 5, 643–655 (2023).
Lyu, X. et al. Multi-modal sensing ionogels with tunable mechanical properties and environmental stability for aquatic and atmospheric environments. Adv. Mater. 36, 2410572 (2024).
Zhang, X. et al. Flexible temperature sensor with high reproducibility and wireless closed-loop system for decoupled multimodal health monitoring and personalized thermoregulation. Adv. Mater. 36, 2407859 (2024).
Zhao, C., Park, J., Root, S. E. & Bao, Z. Skin-inspired soft bioelectronic materials, devices and systems. Nat. Rev. Bioeng. 2, 671–690 (2024).
He, J. et al. Contactless user-interactive sensing display for human–human and human–machine interactions. Adv. Mater. 36, 2401931 (2024).
Song, Y. et al. 3D-printed epifluidic electronic skin for machine learning–powered multimodal health surveillance. Sci. Adv. 9, eadi6492 (2023).
Wang, L., Qi, X., Li, C. & Wang, Y. Multifunctional tactile sensors for object recognition. Adv. Funct. Mater. 34, 2409358 (2024).
Liu, Z. et al. A three-dimensionally architected electronic skin mimicking human mechanosensation. Science 384, 987–994 (2024).
Zhang, B. et al. A three-dimensional liquid diode for soft, integrated permeable electronics. Nature 628, 84–92 (2024).
Zhao, W., Wu, B., Lei, Z. & Wu, P. Hydrogels with differentiated hydrogen-bonding networks for bioinspired stress response. Angew. Chem. Int. Ed. 63, e202400531 (2024).
Xie, L. et al. Ultrasensitive wearable pressure sensors with stress-concentrated tip-array design for long-term bimodal identification. Adv. Mater. 36, 2406235 (2024).
Zhou, N., Cui, T., Lei, Z. & Wu, P. Bioinspired learning and memory in ionogels through fast response and slow relaxation dynamics of ions. Nat. Commun. 16, 4573 (2025).
Zhang, Y. et al. Ultrafast piezocapacitive soft pressure sensors with over 10 kHz bandwidth via bonded microstructured interfaces. Nat. Commun. 15, 3048 (2024).
Chen, R. et al. Nonlinearity synergy: an elegant strategy for realizing high-sensitivity and wide-linear-range pressure sensing. Nat. Commun. 14, 6641 (2023).
Liu, Q. et al. 3D printable strong and tough composite organo-hydrogels inspired by natural hierarchical composite design principles. Nat. Commun. 15, 3237 (2024).
Liu, Q., Hong, L., Dong, X. & Zhai, W. 3D printed hierarchical interpenetrating phase composites with multi-scale mechanical energy absorption mechanisms. Compos. Part B Eng. 264, 110911 (2023).
Wang, J. et al. A metalgel with liquid metal continuum immobilized in polymer network. Adv. Mater. 36, 2409137 (2024).
Tan, P. et al. Self-powered gesture recognition wristband enabled by machine learning for full keyboard and multicommand input. Adv. Mater. 34, 2200793 (2022).
Liu, Q. & Zhai, W. Hierarchical porous ceramics with distinctive microstructures by emulsion-based direct ink writing. ACS Appl. Mater. Interfaces 14, 32196–32205 (2022).
Dong, X. et al. Strong and tough conductive organo-hydrogels via freeze-casting assisted solution substitution. Adv. Funct. Mater. 32, 2203610 (2022).
Xu, C. et al. A Physicochemical-sensing electronic skin for stress response monitoring. Nat. Electron. 7, 168–179 (2024).
Lu, C., Chen, X. & Zhang, X. Highly sensitive artificial skin perception enabled by a bio-inspired interface. ACS Sens. 8, 1624–1629 (2023).
Liu, L. et al. Strong tough thermogalvanic hydrogel thermocell with extraordinarily high thermoelectric performance. Adv. Mater. 35, 2300696 (2023).
Kong, D. et al. Super-resolution tactile sensor arrays with sparse units enabled by deep learning. Sci. Adv. 11, eadv2124 (2025).
Wang, L. et al. Ionic potential relaxation effect in a hydrogel enabling synapse-like information processing. ACS Nano 18, 29704–29714 (2024).
Dobashi, Y. et al. Piezoionic mechanoreceptors: Force-induced current generation in hydrogels. Science 376, 502–507 (2022).
Han, C.-G. et al. Giant thermopower of ionic gelatin near room temperature. Science 368, 1091–1098 (2020).
Liu, K. et al. Self-powered multimodal temperature and force sensor based-on a liquid drople. Angew. Chem. Int. Ed. 55, 15864–15868 (2016).
Zhuo, H. et al. Bamboo-inspired ultra-strong nanofiber-reinforced composite hydrogels. Nat. Commun. 16, 980 (2025).
Park, Y., Noda, I. & Jung, Y. M. Diverse applications of two-dimensional correlation spectroscopy (2D-COS). Appl. Spectrosc. 79, 36–68 (2025).
Li, X., Wu, B., Sun, S. & Wu, P. Making sticky-slippery switchable fluorogels through self-adaptive bicontinuous phase separation. Adv. Mater. 36, 2411273 (2024).
Sang, S. et al. Finger temperature-driven thermogalvainc gel-based smart pen: Utilized for identity recognition, stroke analysis, and grip posture assessment. Nano Energy 123, 109366 (2024).
Tian, C., Khan, S. A., Zhang, Z., Cui, X. & Zhang, H. Thermoelectric hydrogel electronic skin for passive multimodal physiological perception. ACS Sens 9, 840–848 (2024).
Wu, X. et al. Ultra-robust and sensitive flexible strain sensor for real-time and wearable sign language translation. Adv. Funct. Mater. 33, 2303504 (2023).
Hua, M. et al. Strong tough hydrogels via the synergy of freeze-casting and salting out. Nature 590, 594–599 (2021).
Li, N. et al. Deep-learning-assisted thermogalvanic hydrogel E-skin for self-powered signature recognition and biometric authentication. Adv. Funct. Mater. 34, 2314419 (2024).
Wu, Y. et al. Solvent-exchange-assisted wet annealing: a new strategy for superstrong, tough, stretchable, and anti-fatigue hydrogels. Adv. Mater. 35, e2210624 (2023).
Guo, X., Dong, X., Zou, G., Gao, H. & Zhai, W. Strong and tough fibrous hydrogels reinforced by multiscale hierarchical structures with multimechanisms. Sci. Adv. 9, eadf7075 (2023).
Zhang, M. et al. Toughening double-network hydrogels by polyelectrolytes. Adv. Mater. 35, 2301551 (2023).
Liu, Y. et al. Solvent effect on the Seebeck coefficient of Fe2+ /Fe3+ hydrogel thermogalvanic cells. J. Mater. Chem. A 10, 19690–19698 (2022).
Inoue, H., Zhou, H., Ando, H., Nakagawa, S. & Yamada, T. Exploring the local solvation structure of redox molecules in a mixed solvent for increasing the Seebeck coefficient of thermocells. Chem. Sci. 15, 146–153 (2024).
Alberti, L., Carnevali, E., Colombo, A. & Crivellini, A. An entropy conserving/stable discontinuous Galerkin solver in entropy variables based on the direct enforcement of entropy balance. J. Comput. Phys. 508, 113007 (2024).
Qian, X., Ma, Z., Huang, Q., Jiang, H. & Yang, R. Thermodynamics of ionic thermoelectrics for low-grade heat harvesting. ACS Energy Lett. 9, 679–706 (2024).
Martínez, L., Andrade, R., Birgin, E. G., Martínez, J. M. & ACKMOL, P. A package for building initial configurations for molecular dynamics simulations. J. Comput. Chem. 30, 2157–2164 (2009).
Sambasivarao, S. V. & Acevedo, O. Development of OPLS-AA force field parameters for 68 unique ionic liquids. J. Chem. Theory Comput. 5, 1038–1050 (2009).
Everaers, R. & Kremer, K. A fast grid search algorithm for molecular dynamics simulations with short-range interactions. Comput. Phys. Commun. 81, 19–55 (1994).
Jin, Y., Gao, N. & Zhu, T. Effect of resistive load characteristics on the performance of Organic Rankine cycle (ORC). Energy 246, 123407 (2022).
Zhang, D. et al. Highly antifreezing thermogalvanic hydrogels for human heat harvesting in ultralow temperature environments. Nano Lett. 23, 11272–11279 (2023).
Yang, C. et al. A machine-learning-enhanced simultaneous and multimodal sensor based on moist-electric powered graphene oxide. Adv. Mater. 34, 2205249 (2022).
Roy, S. S., Awad, A. I., Amare, L. A., Erkihun, M. T. & Anas, M. Multimodel phishing URL detection using LSTM, bidirectional LSTM, and GRU models. Future Internet 14, 340 (2022).
Jin, Y. & Lee, S. M. Sampled-data state estimation for LSTM. IEEE Trans. Neural Netw. Learn. Syst. 36, 2300–2313 (2025).
Casabianca, P., Zhang, Y., Martínez-García, M. & Wan, J. Vehicle destination prediction using bidirectional LSTM with attention mechanism. Sensors 21, 8443 (2021).
Acknowledgements
This work is supported by the RIE2025 Green Buildings Innovation Cluster 2.0 Funding Initiative (Award GBIC R&D/2025/2), administered by Building and Construction Authority and supported by the National Research Foundation Singapore, and the Singapore Ministry of Education Academic Research Fund Tier 2 Grant (grant number MOE-T2EP50122-0007). It is also funded by the National Natural Science Foundation of China (52475600) and the Special Project of Science and Technology Cooperation and Exchange of Shanxi Province (202304041101021). The authors gratefully acknowledge the theoretical and computational chemistry team at Scientific Compass (www.shiyanjia.com) for their valuable technical support and guidance.
Author information
Authors and Affiliations
Contributions
W.Z. and H.Z. supervised the research. C.B., X.D., and Q.L. conceived the research. C.B. designed the experiments. X.D. prepared the samples. M.Z., Y.N., and K.Y. carried out the experiments. C.B., X.D., M.Z., and Q.L. analyzed the data. C.B. and X.D. wrote the original manuscript. Q.L., W.Z., and H.Z. revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Geng Yang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bai, C., Dong, X., Liu, Q. et al. A self-powered hydrogel electronic skin with decoupled multimodal sensing for closed-loop human-machine interactions. Nat Commun 17, 2675 (2026). https://doi.org/10.1038/s41467-026-69450-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69450-9