Abstract
Animals must constantly scan their environment for imminent threats to their safety. However, they must also integrate their past experiences across long timescales to assess the potential recurrence of new threats. Though visual inputs are critical for the detection of environmental danger, whether and how visual information shapes an animal’s assessment of whether a new threat is likely to reappear in a given context is unknown. In this work, we developed a behavioral assessment of long-term threat avoidance behavior where animals will avoid a familiar location where they previously experienced a single threat exposure. This avoidance behavior is highly sensitive and lasts for multiple days. Intriguingly, we find that the melanopsin-expressing, intrinsically photosensitive retinal ganglion cells tune this behavior via a perihabenula-nucleus accumbens circuit distinct from canonical visual threat detection circuits in male mice. These findings define a long-term threat avoidance behavior that is shaped by a defined retinal cell type based on prior experience.
Similar content being viewed by others
Introduction
Detecting existing threats is key to animal survival, and organisms have evolved many mechanisms for threat detection and escape. However, animals entering each new or familiar environment must also constantly draw on past experience to calibrate their behavior based on the expectation that a new threat will appear. The ability to properly tune risk-taking versus avoidance behavior based on past experience has a clear evolutionary advantage for optimizing animal survival, foraging, and reproduction. The circuits underlying threat avoidance calculations must therefore be plastic, well-calibrated, and operate across varied contexts and timescales. Despite their importance for survival and decision making, these threat avoidance circuits are not well understood.
Visual information is a key sensory input for both threat detection and for providing contextual information about new and familiar environments to shape behavior. It is therefore likely that visual circuits tune threat avoidance behavior. In this work, to better define these key circuits, we report development of a behavioral paradigm to characterize and quantify threat avoidance based on past experience over long timescales, a behavior we have termed long-term threat avoidance (LTTA). Though escape circuits are often hardwired, we find that context-based decisions in the absence of the threat itself are tunable by visual input. Specifically, we find that avoidance is facilitated by input from the melanopsin-expressing, intrinsically photosensitive retinal ganglion cells via a retino-thalamic-limbic circuit. Thus, the LTTA paradigm has allowed us to define a previously unknown visual circuit key for shaping avoidance behavior based on past experience.
Results
A paradigm to measure long-term threat avoidance in mice
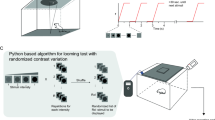
We sought to develop a paradigm to quantitatively assess threat avoidance over long timescales. To do this, we chose to test the impact of a single visual looming threat exposure on an animal’s future avoidance behavior in a previously neutral context (Fig. 1A). The visual looming stimulus has many features that make it ideal to probe the circuits and mechanisms underlying threat avoidance over long timescales because 1) a single exposure to a looming stimulus drives an innate fear response through well-characterized visual circuits, 2) it is highly ethologically relevant for the mouse, and 3) it does not rely on physically aversive stimuli, which make it difficult to distinguish visual from nociceptive contributions1,2,3,4. In this LTTA paradigm, dark-adapted male mice are placed in a square arena with a monitor overhead projecting a neutral gray background (see “Methods”). We track each animal’s exploratory behavior during this pre-exposure phase before any stimulus is presented. During Pre-exposure, we find that control animals readily explore the edges (80% of total time) and center (20% of total time) of the arena (Supplementary Fig. 1). Four minutes later, animals enter the Exposure phase, where they are exposed to a single looming stimulus in the center of the arena, which we refer to as the “threat zone” (TZ)5,6. Animals then remain in the arena for 1-min following Exposure, after which they are returned to their home cage. Two days later, we assess whether the prior exposure to that single looming stimulus drives avoidance of the TZ by placing animals back in the same arena for a 4-minute Test phase where they are allowed to freely explore the arena and no threat stimulus is presented (Fig. 1A).
A Schematic representation of LTTA paradigm. B Representative location paths of control male mice during the Pre-exposure (Pre-loom), Exposure and Test phases of the LTTA paradigm at 0, 6.6, and 36.5% Michelson contrasts. C Freezing time at 0 (n = 15 mice), 6.6 (n = 14 mice) and 36.5% (n = 8 mice) Michelson contrast during the Exposure phase of the LTTA paradigm in control male mice (0 vs. 6.6%, P = 0.999; 0 vs. 37.5%, P = 0.0001; 6.6 vs. 37.5%, P = 0.0001). D Time in threat zone (TZ) at 0 (n = 15 mice), 6.6 (n = 14 mice) and 36.5% (n = 8 mice) Michelson contrast during the Test phase normalized to time in TZ during the Pre-exposure phase in control male mice (0 vs. 6.6%, P = 0.003; 0 vs. 37.5%, P = 0.0001; 6.6 vs. 37.5%, P = 0.144). E Behavioral paradigm to test the role of light on LTTA (top) and representative animal position traces during the Test phase of the LTTA paradigm at 0 and 40 lux of control male mice (bottom). F Time in TZ during the Test phase normalized to time in TZ during the Pre-exposure phase at 0 (n = 6 mice), 10 (n = 6 mice) and 40 (n = 6 mice) lux in control male mice (0 vs. 10 lux, P = 0.232; 0 vs. 40 lux, P = 0.006; 10 vs. 40 lux, P = 0.161). G Behavioral paradigm to test anxiety in looming-exposed mice. H Time in open arms (left) (P = 0.964) and total distance (right) (P = 0.170) in the Elevated Zero Maze (EZM) assay of control male mice previously exposed to 0 (n = 7) or 6.6% (n = 6) looming stimulus. Source data are provided as a Source Data file. All data are Mean ± SEM, n.s. (not significant) P > 0.05; **P < 0.01; ***P < 0.001. Two-sided Student t- and one-way ANOVA with Tukey’s multiple comparisons test.
We first analyzed looming detection of expanding disks varying in contrast from low (6.6% Michelson contrast) to high (36.5%, maximum achievable contrast for a black disk on gray, see “Methods”) by quantifying freezing behavior during the Exposure phase (Fig. 1B, C and Supplementary Fig. 1). Mice showed detectable freezing behavior starting at 13.1% Exposure that increased with higher contrast. Animals showed no detectable freezing behavior at 6.6% Exposure (Fig. 1B, C and Supplementary Fig. 1). These results defined a subthreshold contrast (6.6%) for freezing behavior and support previous findings that increased looming stimulus contrast enhances saliency to drive increased freezing behavior5,6,7.
We then tested long-term avoidance behavior by quantifying and comparing time in the TZ during the Test versus Pre-exposure phases (Fig. 1A and see methods). Animals previously exposed to a looming stimulus of any contrast, even 6.6%, spent significantly less time in the TZ compared to 0% Exposure (i.e., no loom Exposure) controls (Fig. 1D, Supplementary Fig. 1), indicating that TZ avoidance is not simply due to habituation. We were surprised to observe robust TZ avoidance during the Test phase for this contrast, because freezing behavior was not detectable during the 6.6% looming Exposure itself (Fig. 1B–D, Supplementary Fig. 1). These findings indicate that the circuits underlying LTTA are more sensitive and likely distinct from the well-characterized, canonical, looming detection circuits5,8,9,10,11. Importantly, LTTA behavior requires visual input because animals placed in the dark during the Test phase following 6.6% Exposure fail to avoid the TZ, in stark contrast to animals in 10 lux and standard 40 lux illumination (Fig. 1E-F, Supplementary Fig. 1). Moreover, LTTA is not due to increased general anxiety because mice showed no differences in the Elevated Zero Maze 2 days post 0% (no stimulus control) or 6.6% Exposure (Fig. 1G-H). Thus, the circuits underlying LTTA are highly sensitive, long-lasting, and likely distinct from those driving anxiety or looming-dependent freezing responses.
ipRGCs tune long-term threat avoidance
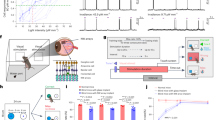
We next sought to identify the retinal cell types involved in LTTA. The melanopsin-(Opn4)-expressing intrinsically photosensitive retinal ganglion cells (ipRGCs) are a good candidate for tuning LTTA because melanopsin signaling within ipRGCs encodes environmental luminance over a large dynamic range and across long timescales12,13,14. To determine whether melanopsin signaling is required for LTTA, we compared TZ avoidance of melanopsin null (Opn4−/−) animals to littermate control animals following 6.6% or 0% Exposure. 6.6% looming contrast was chosen to isolate impacts on avoidance behaviors versus looming detection itself. Indeed, Opn4−/− and control littermates show no freezing behavior during Exposure at either contrast (Supplementary Fig. 2). However, during the Test phase, Opn4−/− animals spent significantly more time in TZ compared to controls following 6.6% Exposure, indicating that melanopsin signaling in ipRGCs is required for LTTA (Fig. 2A-C, Supplementary Fig. 2). Importantly, Opn4−/− mice begin to exhibit freezing behavior at the same contrast, 13.1%, as control animals, indicating that impacts on LTTA are not due to deficits in detecting the looming stimulus itself. Moreover, Opn4−/− mice spend similar time in TZ to controls in both the Pre-exposure and Test phases following 0% Exposure (Supplementary Fig. 2), indicating that differences in LTTA are not due to differences in baseline exploratory behavior or habituation (Fig. 2B, C, Supplementary Fig. 2). Additionally, these differences are unlikely due to altered anxiety because Opn4−/− mice showed no differences in anxiety-like behaviors compared to control littermates (Supplementary Fig. 3).
A Scheme of melanopsin expression in ipRGCs. B Representative animal position traces of control and melanopsin-null (Opn4−/−) male mice during the Test phase of the LTTA previously exposed to 0 or 6.6% Michelson contrast. C Time in threat zone (TZ) during the Test phase normalized to time in TZ during the Pre-exposure phase in control (black) and Opn4−/− (red) male mice exposed to 0 (n = 15 mice/group) (P = 0.162) or 6.6% (n = 14 control mice, n = 11 Opn4−/− mice) (P = 0.002) Michelson contrast. D Opn4 gene locus in WT (top), Opn4fl/fl before (middle) and after (bottom) Cre recombination. E Scheme of intravitreal injection of Vehicle, AAV2-hSyn-mCherry (control) or AAV2-hSyn-Cre-P2A-TdTomato in Opn4fl/fl male mice (left) melanopsin immunolabeling in flat-mount retinas (right). F Representative images of melanopsin immunolabeling in ipRGCs of Opn4fl mice followed by control or AAV2-hSyn-Cre-P2A-TdTomato injections. Arrowheads pointing ipRGC somas in retinas from AAV-Cre injected mice. G Representative animal position traces of Opn4fl/fl male mice during the Test phase of the LTTA previously exposed to 6.6% Michelson contrast. H Time in TZ during the Test phase normalized to time in TZ during the Pre-exposure phase in control (n = 5, gray) and AAV2-hSyn-Cre-P2A-TdTomato (n = 7, red) Opn4fl/fl mice exposed to 6.6% Michelson contrast (P = 0.010). I Scheme of glutamate release ablation specifically from ipRGCs. (J) Representative animal position traces of Vglut2cKO male mice during the Test phase of the LTTA previously exposed to 6.6% Michelson contrast. K Time in TZ during the Test phase normalized to time in TZ during the Pre-exposure phase in Vglut2cKO (n = 11, purple) and control (n = 5, gray) male mice exposed to 6.6% Michelson contrast (P = 0.020). L Schematic representations of Brn3b-positive ipRGC ablation in Brn3bDTA mice. M Representative animal position traces during the Test phase of the LTTA paradigm. N Time in TZ during the Test phase normalized to time in TZ during the Pre-exposure phase of Brn3bDTA (n = 11, pink) and control (n = 6, gray) littermate male mice (P = 0.038). Source data are provided as a Source Data file. All data are Mean ± SEM, n.s. (not significant) P > 0.05; *P < 0.05; **P < 0.01. Two-sided Student t and Mann-Whitney U tests.
Previous studies indicate that ipRGCs can influence brain development, which could lead to secondary impacts on LTTA15,16,17. If melanopsin itself is required for LTTA, then removal of melanopsin in adult animals should lead to LTTA deficits similar to those of Opn4−/− animals. To test this, we removed melanopsin in adult ipRGCs using a conditional knockout approach. Specifically, we used Opn4fl/fl littermate control (P60 intravitreal Vehicle or AAV-mCherry injection) or conditional knockout (Opn4cKO; P60 intravitreal AAV-Cre-TdTomato injection) (Fig. 2D, E). Mice were then exposed to the LTTA paradigm and the efficiency of Opn4 ablation was tested post-mortem. Opn4cKO mice injected with AAV-Cre-TdTomato, showed decreased LTTA compared to control littermates, similar to Opn4−/− animals (Fig. 2G, H, Supplementary Fig. 4). Post-mortem analysis of melanopsin expression showed a ~95% reduction in ipRGCs in Opn4cKO animals, demonstrating a high efficiency of ablation (Fig. 2F, Supplementary Fig. 4), These results indicate that it is melanopsin signaling itself that tunes LTTA, and not secondary developmental impacts on brain development in Opn4−/− animals.
If ipRGCs tune LTTA then disrupting ipRGC glutamate signaling should similarly alter LTTA. Indeed, conditional knockout of the vesicular glutamate transporter Vglut2 in ipRGCs (Vglut2cKO: Opn4Cre/+; Vglut2fl/fl)18 caused similar deficits in TZ avoidance to Opn4−/− animals despite similar baseline exploratory behavior to control littermates (Fig. 2I-K, Supplementary Fig. 4). Likewise, ablation of non-SCN-projecting Brn3b-positive ipRGCs using the Brn3bDTA line (Opn4Cre/+; Brn3bzDTA/+)19, resulted in similar TZ avoidance deficits (Fig. 2L-N, Supplementary Fig. 4). Combined, these results indicate that ipRGCs release glutamate at non-SCN targets to tune LTTA behavior.
The perihabenular nucleus is required for tuning LTTA
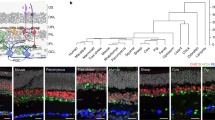
We next sought to identify the ipRGC-recipient target(s) required for LTTA using an unbiased cFos screen of brain regions differentially activated during the Test phase in control versus Opn4−/− animals (Fig. 3A). If a given ipRGC-recipient region is required for LTTA, then we would expect that target to show patterns in cFos activation that parallel differences in avoidance phenotypes (i.e., cFos levels in all groups are similar with the exception of 6.6% Exposed controls). Remarkably, the only ipRGC-recipient target showing the predicted differences in cFos labeling was the perihabenular nucleus (PHb) (Fig. 3B, C), suggesting an ipRGC-PHb circuit may underly LTTA. Notably, regions implicated in the canonical looming circuit such as medial superior colliculus (mSC) and the ventral lateral geniculate nucleus (vLGN)5,11,20 and ipRGC-recipient regions directly upstream of looming detection circuits, such as medial amygdala and periaqueductal gray, showed no differences in cFos expression following LTTA (Supplementary Fig. 5), again pointing to the possibility that the LTTA circuit architecture is distinct from the looming detection circuit.
A Schematic representation of experimental design for cFos staining. B Representative images of cFos staining in PHb from control and melanopsin-null (Opn4−/−) male mice exposed to 6.6% Michelson contrast. C cFos-positive cells quantification in the PHb from control and control and Opn4−/− male mice exposed to 0 (n = 4 control mice, black; n = 3 Opn4−/− mice, red) (P = 0.999) and 6.6% (n = 5 mice / group) (P = 0.014) Michelson contrast. D Schematic representation of potential ipRGC-PHb circuit driving LTTA. E Scheme of viral injection of Gi-DREADDs in PHb of Gad2Cre male mice. F Representative image of Gi-DREADDs expression in PHb. G Time of experimental design. H Percentage of time in center during the Pre-exposure phase in Vehicle (n = 12, blck) or CNO-injected (n = 11, blue) Gad2Cre male mice (P = 0.753). I Representative Gad2Cre animal position traces during the Test phase of the LTTA paradigm. J Time in threat zone (TZ) during the Test phase normalized to time in TZ during the Pre-exposure phase of Vehicle (n = 12, blue) and CNO-injected (n = 11, blue) Gad2Cre male mice (P = 0.037). K Scheme of viral injection of Gq-DREADDs in PHb of Vglut2Cre male mice. L Time of experimental design. M Representative image of Gq-DREADDs expression in PHb of Vglut2Cre male mice. N Percentage of time in center during the pre-exposure phase in Vehicle (n = 9) or CNO-injected (n = 12) Vglut2Cre male mice. O Representative Vglut2Cre animal position traces during the Test phase of the LTTA paradigm. P Time in threat zone (TZ) during the Test phase normalized to time in TZ during the Pre-exposure phase of Vehicle (n = 9, black) and CNO-injected (n = 12, pink) Vglut2Cre male mice (P = 0.010). Source data are provided as a Source Data file. All data are Mean ± SEM, n.s. (not significant) P > 0.05; *P < 0.05. Two-sided Student t and Mann-Whitney U tests.
ipRGCs primarily innervate GABAergic neurons in the caudal PHb (PHbGABA), which in turn make local connections with excitatory-relay PHb neurons (PHbGlu) and anterior PHbGABA neurons (Fig. 3D)21,22,23. If PHbGABA neurons are required for LTTA, then silencing that specific population should disrupt LTTA. To test this, we injected Cre-dependent Gi-DREADDs in the PHb of Gad2Cre mice to silence PHbGABA (Fig. 3E-F, see Supplementary Table 1) neurons. We then intraperitoneally (i.p.) injected either vehicle or the DREADD agonist clozapine-N-oxide (CNO) 30 min prior to both the Pre-exposure and Test phases of the LTTA paradigm (Fig. 3G). Importantly, i.p. CNO injection did not impact LTTA in naïve WT mice (Supplementary Fig. 6) and did not alter exploratory behavior during Pre-exposure phase compared to vehicle-injected mice, indicating that i.p. CNO itself does not alter baseline exploratory behavior (Fig. 3H). Silencing of PHbGABA (Fig. 3E–J), but not PHbGlu neurons (Supplementary Fig. 6), disrupted LTTA, causing animals to spend significantly more time in the TZ, confirming that the GABAergic neurons of the ipRGC-recipient PHb are required for LTTA.
If PHb-ipRGCs are required for LTTA, then activation of PHb-ipRGCs in Opn4−/− animals should rescue the observed deficits in LTTA. Light activates melanopsin in ipRGCs, triggering a Gq-mediated phototransduction cascade that ultimately depolarizes the cells and drives glutamate release onto downstream targets24. We have previously shown that Gq activation with Gq-DREADDs in ipRGCs ex vivo restores melanopsin-like photoresponses in Opn4−/− ipRGCs25. We therefore adapted this approach to our in vivo LTTA paradigm by bilaterally injecting retrograde (rg)AAV into the PHb of melanopsin null (Opn4Cre/Cre) animals to drive Cre-dependent expression of Gq-DREADDs (rgAAV-hSyn-DIO-hM3Gq-mCherry) selectively in PHb-ipRGCs (Fig. 4A, see Supplementary Table 1). We then intraperitoneally injected either vehicle or CNO 30 min prior to both the Pre-Exposure and Test phases (Fig. 4A). CNO-injected Opn4−/− animals avoided the TZ, spending significantly less time in the TZ than vehicle-injected Opn4−/− littermates. This indicates that Gq-DREADD activation of PHb-ipRGCs restores LTTA in Opn4−/− mice (Fig. 4B-D). CNO administration did not alter baseline exploratory behavior during the Pre-exposure phase compared to vehicle-injected mice (Supplementary Fig. 7), again suggesting that CNO does not affect exploratory behavior. These effects are not through collateral outputs of PHb-ipRGCs to the vLGN, a collateral target of PHb-ipRGCs (Supplementary Fig. 8, see Supplementary Table 1) previously implicated in looming-response behavior5,11, because chemogenetic silencing of the vLGN itself did not affect LTTA in WT mice (Supplementary Fig. 9, Supplementary Table 1 and see methods). Collectively, these findings indicate that PHb-ipRGCs are required for LTTA.
A Time of experimental design for Opn4−/− male mice. B Scheme of viral injection of rgGq-DREADDs in PHb of Opn4−/− male mice (left) and representative images of Gq-DREADDs expression in a PHb-ipRGC (right). C Representative Opn4−/− animal position traces during the Test phase of the LTTA paradigm. D Time in trheat zone (TZ) during the Test phase normalized to time in TZ during the Pre-exposure phase of Vehicle or CNO-injected Opn4−/− male mice (n = 14 Veh/Veh mice, red; n = 9 CNO/CNO mice, green; n = 6 CNO/Veh mice, green; n = 6 Veh/CNO mice, green) (Veh/Veh vs. CNO/CNO, P = 0.033; Veh/Veh vs. CNO/Veh, P = 0.999; Veh/Veh vs. Veh/CNO, P = 0.014). E Scheme of intravitreal injection of Gi-DREADDs and guide cannula implantation in the PHb of WT/Opn4Cre male mice. F Time of experimental design for WT/Opn4Cre/+ male mice. G Representative images of guide cannula implantation in PHb of WT/Opn4Cre/+ male mice. H Representative Vehicle and CNO cannula-infused in PHb of WT/Opn4Cre animal position traces during the Test phase of the LTTA paradigm. I Time in TZ during the Test phase normalized to time in TZ during the Pre-exposure phase of Vehicle (n = 6, black) and CNO (n = 5, green) cannula-infused WT/Opn4Cre male mice (P = 0.047). Source data are provided as a Source Data file. All data are Mean ± SEM, n.s. (not significant) P > 0.05; *P < 0.05. One-way ANOVA with Dunn’s multiple comparisons test and two-sided Student t test.
ipRGCs tune PHb neuron activity to facilitate recall of the threat-context association
To determine whether PHb-ipRGCs are important for LTTA during the formation of association between threat and context (i.e., at Pre-exposure / Exposure phases) or whether they facilitate the recall of this association upon re-exposure (i.e., at Test phase), we again bilaterally injected retrograde (rg)AAV-Gq-DREADDs into the PHb of melanopsin null (Opn4Cre/Cre) animals, but this time administered a single dose of i.p. CNO to Opn4−/− mice prior to either Pre-exposure (CNO/Veh group) or Test (Veh/CNO group) phases (Fig. 4A-D, see Supplementary Table 1). A third group received two vehicle injections (Veh/Veh group) as control. Opn4−/− animals that received CNO prior to the Test phase (Veh/CNO), but not those that received CNO prior to the Pre-exposure phase (CNO/Veh), showed rescued LTTA and decreased time in the TZ (Fig. 4D). These results were identical to those where animals received CNO in both the Pre-exposure and Test phases (Fig. 4B-D), indicating that PHb-ipRGC activity is required for LTTA only during the Test phase to facilitate the recall of the association between the threat and context.
To further test whether the PHb-ipRGC circuit is required for the recall of the threat–context association, we selectively inhibited PHb-projecting retinal axon terminals only during the Test phase. We intravitreally injected inhibitory Gi-DREADDs (AAV2-Syn-hM4Di-mCherry) into WT mice and implanted bilateral cannulas targeting the PHb for local infusion of Vehicle or CNO (Fig. 4E-G). After 4 weeks, vehicle was infused prior to the Exposure phase and either vehicle or CNO prior to the Test phase of the LTTA paradigm. CNO-infused mice spent significantly more time in the TZ compared to Vehicle-infused mice (Fig. 2H-I and Supplementary Fig. 7). These results demonstrate that inhibition of PHb-projecting ipRGC terminals during recall disrupts LTTA, confirming that PHb-ipRGC activity facilitates retrieval of the threat–context association. Moreover, this terminal-specific manipulation rules out confounding effects mediated by collateral ipRGC projections to the LGN (Supplementary Fig. 8).
Our data indicate that silencing PHbGlu neurons does not impact LTTA, in agreement with ipRGC synaptic input to these cells21. However, if PHbGlu neurons are downstream of PHbGABA neurons (ipRGC- PHbGABA-PHbGlu), it is possible that exciting, rather than inhibiting them, would drive deficits in LTTA. To test this, we next assessed the contribution of excitatory-relay PHbGlu neurons to the LTTA behavior during the Test phase. To do this, we bilaterally injected Cre-dependent Gq DREADDs in the PHb of Vglut2Cre mice to specifically excite PHbGlu neurons (Fig. 3K–M and Supplementary Fig. 6). We then intraperitoneally injected vehicle prior to Pre-exposure either vehicle or CNO prior to the Test phase of the LTTA paradigm (Fig. 3L and Supplementary Fig. 6). Chemogenetic excitation of PHbGlu neurons disrupted LTTA (Fig. 3N-P), indicating that PHbGlu neurons are likely inhibited by PHbGABA neurons during LTTA.
To understand PHb signaling dynamics and the impact of melanopsin signaling during LTTA, we next used fiber photometry (FP) to measure bulk Ca2+ activity in the PHb in vivo in Opn4−/− and control littermates at different phases of the LTTA paradigm. To do this, we injected an AAV containing GCamp6s (AAV1-CAG-GCaMP6s-WPRE-SV40) and implanted a FP canula unilaterally in the PHb of control and Opn4−/− littermates (Fig. 5A-B, see Supplementary Table 1 and methods). We then compared overall activity in the Pre-exposure versus Test phases of the LTTA paradigm using a 6.6% contrast looming stimulus. In agreement with cFos expression analysis (Fig. 3C), we found decreased PHb activity in Opn4−/− mice compared to control littermates during the Test, but not during the Pre-exposure phase of the LTTA paradigm (Fig. 5C-D). This indicates that ipRGC signaling during the Test phase serves to increase baseline PHb activity.
A Scheme of experimental design for fiber photometry (FP) in LTTA paradigm. B Representative image of GCaMPs expression and optic fiber implantation in the PHb. C Representative Ca2+ signal recordings from PHb in control and Opn4−/− male mice during Pre-exposure and Test phases of the LTTA paradigm. D Area under the curve (AUC) of calcium signal z-score during the Test phase normalized to AUC of calcium signal z-score during the Pre-exposure phase in control (n = 6, black) and Opn4−/− (n = 11, red) male mice (P = 0.017). Fiber photometry recording from PHb during Exposure. Schemes of recording at the Exposure phase (E) during the looming stimulus exposure (F). G Calcium signal z-score during the Exposure phase of the LTTA paradigm in control (n = 9) and Opn4−/− (n = 13) male mice. H Area under the curve (AUC) of calcium signal z-score during the Exposure phases of the LTTA paradigm in control (n = 9, black) and Opn4−/− (n = 13, red) male mice (P = 0.188). I Schematic representation of transitions from edge to TZ. Heatmaps (J), z-score of FP signal (K) and AUC of calcium signal z-score when mice were placed in TZ normalized to AUC of z-score of FP signal (L) when mice were placed in transitions from edge to TZ during the Pre-exposure phase of the LTTA paradigm in control (n = 16, black) and Opn4−/− (n = 25, red) male mice (P = 0.752). Heatmaps (M), z-score of FP signal (N) and AUC of calcium signal z-score(O) when mice were placed in TZ normalized to AUC of z-score of FP signal when mice were placed in transitions from edge to TZ during the Test phase of the LTTA paradigm in control (n = 12, black) and Opn4−/− (n = 17, red) male mice (P = 0.016). Source data are provided as a Source Data file. All data are Mean ± SEM, n.s. (not significant) P > 0.05; *P < 0.05. Two-sided unpaired and paired Student t tests.
We next examined how PHb neuron activity varied as animals exhibited certain behaviors during the LTTA paradigm. To identify key parameters for further analysis, we performed a principal component analysis (PCA) of bulk Ca2+ activity in the PHb and behavioral parameters data (Supplementary Fig. 10). We observed that the calcium signal z-score in the PHb was highly correlated with behavioral parameters when mice remained in the edges (i.e., increased avoidance) compared to those behavioral parameters when mice were in the TZ (Supplementary Fig. 10). To better understand this, we analyzed the calcium signal z-score in the PHb from control or Opn4−/− animals at different phases of the LTTA paradigm. We did not observe changes in calcium signal z-score in the PHb during the Exposure phase (Fig. 5E-H). Then, we analyzed the activity in the PHb while mice occasionally transitioned from the edge to the TZ during the Pre-exposure and the Test phases (Fig. 5I). In control mice, we found a significant decrease in the calcium signal z-score in the PHb when control mice transitioned from the edge to the TZ, but no significant change during transitions of Opn4−/− mice, indicating that the PHb may be particularly important for signaling entry into the TZ (Fig. 5M-O). These changes were not observed during the Pre-exposure phase (Fig. 5I-L), suggesting again that ipRGC to PHb signaling is required to facilitate the recall of the threat/context association. We did not observe changes when mice were transitioning from the TZ to the edges in the Test phase (Supplementary Fig. 10), indicating that the PHb nucleus is key for encoding environmental information while mice are entering the TZ and that this process requires melanopsin signaling within ipRGCs.
A PHb to nucleus accumbens (NAc) circuit is required for LTTA
The PHb nucleus is part of a thalamo-frontocortico-striatal loop projecting to the ventromedial prefrontal cortex (vmPFC) and the nucleus accumbens (NAc) (Fig. 6A-C)21,22,23. We therefore next tested whether PHb-NAc or PHb-vmPFC neurons are required for LTTA. If PHb-NAc or PHb-vmPFC neurons are required for LTTA, then activating them with Gq-DREADDs in Opn4−/− animals should rescue the LTTA deficits in these mice. To test this, we expressed excitatory Gq-DREADDs in vmPFC-, or NAc-projecting PHb neurons into the PHb of Opn4−/− mice (Fig. 6D, Supplementary Fig. 11, Supplementary Table 1 and see methods) Chemogenetic excitation of PHb-NAc circuit rescued LTTA deficits evidenced by a decreased time in the TZ (i.e., increased avoidance) in i.p. CNO-injected Opn4−/− mice compared to vehicle-injected Opn4−/− littermates (Fig. 6D-I). However, we did not observe differences in LTTA when PHb-vmPFC was chemogenetically excited in Opn4−/− mice (Supplementary Fig. 11). These results suggest that the PHb-NAc circuit, and not PHb-vmPFC, is required for LTTA.
A Schematic representation of PHb-downstream regions. B Scheme of viral injection to intersectionally trace the PHb-NAc circuit. C Representative image of PHb neuron axons innervating the NAc (n = 2 mice). D Scheme of experimental design. E Scheme of viral injection to interjectionally express Gq-DREADDs in PHb projecting to NAc in Opn4−/− male mice. F representative image of Gq-DREADD expression in PHb-NAc neurons. G Time in center in vehicle (n = 6, red) and CNO-injected (n = 7, green) Opn4−/− mice during the Pre-exposure phase of the LTTA paradigm (P = 0.569). H Representative Vehicle and CNO-injected Opn4−/− animal position traces during the Test phase of the LTTA paradigm. I Time in threat zone (TZ) during the Test phase normalized to time in TZ during the Pre-exposure phase of Vehicle (n = 6, red) and CNO-injected (n = 7, green) Opn4−/− male mice (P = 0.019). J Scheme of injection of Gi-DREADDs in the PHb and guide cannula implantation in NAc of WT/Opn4Cre male mice. K Representative images of guide cannula implantation in NAc of WT/Opn4Cre/+ male mice. L Time of experimental design for WT/Opn4Cre/+ male mice. M Representative Vehicle and CNO cannula-infused in NAc of WT/Opn4Cre animal position traces during the Test phase of the LTTA paradigm. N Time in TZ during the Test phase normalized to time in TZ during the Pre-exposure phase of Vehicle (n = 5, black) and CNO (n = 4, green) cannula-infused in NAc of WT/Opn4Cre male mice (P = 0.032). Source data are provided as a Source Data file. All data are Mean ± SEM, n.s. (not significant) P > 0.05; *P < 0.05. Two-sided Student t and Mann-Whitney U tests.
To further test whether the PHb-NAc circuit is required for the recall of the threat–context association, we selectively, locally inhibited NAc-projecting PHb axon terminals only during the Test phase. If PHb-NAc circuit is required for LTTA, then silencing PHb neuron terminals in the NAc in WT animals should cause LTTA deficits in these mice. To test this, we bilaterally injected inhibitory Gi-DREADDs (AAV2-Syn-hM4Di-mCherry) into the PHb of WT mice and implanted bilateral cannulas targeting the NAc for local infusion of Vehicle or CNO (Fig. 6J, K and Supplementary Fig. 11). After 4 weeks, vehicle was infused prior to the Exposure phase and either vehicle or CNO prior to the Test phase of the LTTA paradigm. CNO-infused mice spent significantly more time in the TZ compared to Vehicle-infused mice (Fig. 6L, N and Supplementary Fig. 11). These results demonstrate that inhibition of NAc-projecting PHb terminals during recall disrupts LTTA, confirming that ipRGC-PHb-NAc neurons are required. Moreover, this terminal-specific manipulation rules out any other potential outcome effects mediated by the PHb nucleus22,23.
Discussion
The ability to balance and calibrate behaviors that promote risk versus safety are key to animal survival. Here we describe LTTA, a visual, ethologically relevant, long-term avoidance behavior in which animals exposed to a single, low contrast looming stimulus, showed robust avoidance of the TZ days following threat exposure. The circuit underlying LTTA is distinct from the looming detection or anxiety circuits, and requires ipRGC input to the recently identified perihabenular-nucleus accumbens circuit. Unlike classical contextual fear conditioning that pairs a foot shock with a context, LTTA relies on a purely visual, non-painful threat; allows animals to actively choose whether to revisit the threat-associated zone during testing; and engages circuitry not implicated in shock-based learning.
Though threat detection circuits are often hard-wired, we find that these threat avoidance behaviors are plastic and calibrated based on prior experience. Excitingly, we identified a previously unappreciated role of ipRGCs in facilitating avoidance behavior during the recall of the threat-context association. ipRGCs achieve this facilitation by increasing baseline PHb activity during the Test phase days after the threat exposure. This role for ipRGCs is supported by 1) a correlation between threat avoidance behavior and cFos expression patterns in the PHb during the Test phase; 2) the restoration of the LTTA phenotype following chemogenetic manipulation of PHb-ipRGCs in Opn4−/− mice, but only prior to the Test phase, not before the Exposure; 3) the inhibition of PHb-projecting retinal axon terminals in WT mice only during the Test phase; and 4) in vivo recordings of PHb calcium signals. Moreover, we find that ipRGC inputs to the PHb are particularly crucial for signaling arena location during edge-TZ transitions, only in the Test phase of the LTTA paradigm.
The PHb then relays signals to the NAc, the next node in the LTTA circuit. Opn4−/− deficits in LTTA can be rescued by exciting PHb-NAc projections, and locally inhibiting NAc-projecting PHb axon terminals cause LTTA disruptions in WT mice. Thus, not only do these findings define the NAc as a key node in LTTA, but they also identify a robust, ipRGC-dependent modulation of NAc-associated circuits. Together, these findings support a working model in which ipRGC inputs activate PHbGABA, which in turn modulate PHbGlu neuron projections to the NAc (Supplementary Fig. 12). This raises the exciting possibility that ipRGCs broadly modulate other NAc-dependent behaviors and defines a circuit architecture by which light input through ipRGCs could tune behaviors related to reward, addiction, or motivation, among others.
Calculations involving risk versus avoidance are key to animal and human survival. This work defines a tractable paradigm for mechanistic investigation of these circuits and defines circuits and cell types originating in the retina that are routed through the thalamus and limbic areas to influence behavioral decisions. Previous studies linking ipRGCs to learning or mood modulation have described a role for ipRGCs only under prolonged, severely aberrant lighting conditions22,26. However, our findings reveal an active role for ipRGCs in shaping learned risk-avoidance behaviors under standard environmental conditions, highlighting their critical involvement in daily survival decisions. Collectively, these findings define new avenues by which ipRGCs may broadly modulate ethologically relevant behaviors in previously unappreciated ways.
Methods
Animals
All procedures were approved by the Animal Care and Use Committee at Northwestern University (Protocol number: IS00003845). Animals were housed in vivarium under 12:12 light/dark cycle conditions with ad libitum access to food and water. Temperature ranged from 21 to 23 °C and humidity ranged from 30% to 70%. P60-P120 male mice were used for this study. For circuit tracing we used WT and Opn4Cre (RRID:IMSR_JAX:035925) mice. For behavior, chemogenetic and histological studies, we used WT, Opn4Cre, Opn4fl (C57BL/6JCya-Opn4em1flox/Cya, ID: S-CKO-21264), Opn4Cre/+; Vglut2fl/fl (MGI:4879031), Brn3bDTA (RRID:IMSR_JAX:038952), Gad2Cre (IMSR_JAX:028867) and Vglut2Cre (RRID:IMSR_JAX:016963) male mice. For fiber photometry experiments we used Opn4Cre male mice. Opn4fl mouse was generated by mating “knockout-first” allele Opn4tm1a(KOMP)Wtsi (UCDAVIS KOMP Repository) to FLPO line (C57BL/6N-Tg(CAG-Flpo)1Afst/Mmucd, MMRRC.
Long-term threat anticipation paradigm
The experimental arena consisted of a 40 × 40 × 40 cm3 opaque white foamboard Plexiglas box with a screen embedded in the ceiling. Throughout the experimental sessions, the monitor displayed a dim gray background (40 lux), on which a dark expanding disk was presented as the looming stimulus during the Exposure phase (Fig. 1A). We used Adobe Flash (RRID:SCR_017258) to design the looming stimuli and VLC Media Player (RRID:SCR_003070) to trigger the stimuli. Each stimulus consisted of ten expansions of a dark disk projected onto the gray background of the monitor. A single disc expansion consisted of two phases: 1) the disk expanded over a duration of 0.5 s, 2) the disk then remained static at its maximum expansion for 0.5 s before disappearing. An interval of 0.5-s was left before the next disk expansion; thus, each stimulus presentation lasted 10 s. Each mouse was exposed once, to one unique condition: 0, 6.6, 13.1, 27.5, or 36.5% Michelson contrast. Michelson contrast was calculated based in arena illumination levels measured using a XL-500 BLE Spectroradiometer (nanoLambda Korea Co., Ltd).
Mice were dark-adapted for 20–30 min and then placed in the LTTA arena for 4 min (Pre-exposure, Fig. 1A). A single contrast of looming stimulus of contrasts ranging from 0 to 36.5% Michelson contrast and then recorded in the arena for 1 min following the Exposure phase. After a retention period of 2 days in the home cage, 30-min dark-adapted animals were then placed in the same arena for 4 min with no stimulus triggered (Test phase, Fig. 1A) and behavior was recorded using two cameras (C920x HD, Logitech and ELP Wide Angle Infrared Webcam).
For chemogenetic experiments, 4 weeks after viral infections (see below), mice were dark-adapted and 30 min before the Pre-exposure and Test phases of the LTTA paradigm, animals were i.p. injected with saline (Vehicle, 0.9 % NaCl) or Clonazepine N-Oxide (1 mg/kg) (CNO, Tocris Bioscience Cat# 6329). After i.p. injections behavioral assay was performed as described above. For local axon-terminal manipulations, 4 weeks after viral infections and cannula implantations (see below), mice were dark adapted and 20 min before the Pre-exposure and Test phases of the LTTA paradigm, animals were infused with saline (Vehicle, 0.9 % NaCl) or CNO (0.3 mg/kg). For Vehicle or CNO cannula infusions, bulk catheter (Cat#: BC-22, RWD Life Science Inc.) were attached to a Hamilton Syringe (Cat # 88500) to infuse a total volume of 0.3 μL at a rate of 0.1 μL/min. After infusions, behavioral assay was performed as described above. Mice with mistargeted viral expression or cannula implantation, confirmed by post hoc histology, were excluded from all analyses.
Behavior analysis
For behavior analysis, we used eZtrack (RRID:SCR_021496) automated tracking software27 to analyze IR videos and quantify freezing behavior during Exposure phase of the LTTA paradigm at different looming stimuli contrasts. Freezing behavior was computed when mice were tracked immobile for at least 1 second. To quantify avoidance behavior, we used eZtrack software to analyze IR and regular light videos and quantify the time spent in the center of the arena during the Test phase (TZtest) of the LTTA paradigm. This parameter was normalized to the time spent in the center of the arena in the Pre-exposure phase for each mouse (TZpre-exp).
Viral infection and guide cannula implantations
Mice were anesthetized with isoflurane (Kent Scientific VetFlo system) and then unilaterally or bilaterally injected with 150 nl of AAVs (see Supplementary Table 1) in the perihabenular nucleus (PHb) (AP:−1.60, ML: ± 0.55, DV:2.65 mm), ventral lateral geniculated nucleus (vLGN) (AP:−2.15, ML: ± 2.65, DV:3.60 mm), nucleus accumbens (NAc) (AP:1.42, ML: ± 0.80, DV:4.00 mm) or ventromedial prefrontal cortex (vmPFC) (AP:1.94, ML: ± 0.40, DV:2.82 mm) using a stereotaxic injector (Neurostar) controlled by the software Stereodrive at an injection rate of 30 nl/min. For intravitreal injections, mice were anesthetized by intraperitoneal (IP) injection of Avertin (2,2,2-Tribromoethanol) and a 30-gauge needle was used to open a hole in the ora serrata. Each eye was intravitreally injected with 1 μL of AAVs (see Supplementary Table 1) using a custom Hamilton syringe (Borghuis Instruments) with a 33-gauge needle (Hamilton). After the surgery procedures, mice were subcutaneously injected with 20 mg/kg of meloxicam (Covetrus). Efficiency of the viral injections was tested postmortem.
In some cases, bilateral guide cannulas in the PHb or NAc were implanted followed by viral infection. For the PHb, Series I bilateral cannula (RWD Life Science Inc.) were implanted with a center-to-center (C.C) distance of 1.2 mm, and a length of 3.65 mm (DV-PHb coordinate + 1 mm, Model No. 62025; O.D.0.48 mm −26G/C.C1.2/B7.8/M3.5) guide cannula with an internal cannula of 0.5 mm projection length (O.D.0.30 mm −30G/C.C1.2/Mates with M3.5) and a dummy cannula of 0 mm projection (O.D.0.30 mm/C.C1.2/Mates with M3.5). For the NAc, Series I bilateral cannula (RWD Life Science Inc.) were implanted with a C.C distance of 1.6 mm, and a length of 5.00 mm (DV-NAc coordinate +1 mm, Model No. 62029; O.D.0.48 mm −26G/C.C1.6/B7.8/M3.5) guide cannula with an internal cannula of 0.5 mm projection length (O.D.0.30 mm −30G/C.C1.6/Mates with M3.5) and a dummy cannula of 0 mm projection (O.D.0.30 mm/C.C1.6/Mates with M3.5). For both cannula sets, dust caps (O.D.5.5 mm/L7.0 mm/M3.5) were attached until the day of behavioral test.
Immunohistochemical procedures
For immunological studies in brain sections, mice were anesthetized by IP injection of Avertin and intracardially perfused with saline solution (PBS) followed by PFA 4% in PBS. Brians were dissected and post-fixated in PFA 4% in PBS overnight at 4 °C. For cryoprotection, brains were incubated in sucrose 30% for 3 days, embedded in OCT and then sectioned at 40 µm using a Leica CM1950 cryostat. For immunolabeling studies in retinas, animals were anesthetized by IP injection of Avertin and sacrificed by cervical dislocation. Eyecups were enucleated and retinas were dissected and then fixed in PFA 4% in PBS for 30–60 min at RT. Brain slices and retinas were washed and then blocked at 4 °C overnight in 6% normal goat serum in 0.3% Triton PBS prior to incubating in primary antibody solution for 2–3 nights at 4 °C. Then, tissue was washed and incubated in secondary antibody solution for 2 hours at RT (Supplementary Table 2). The tissue was finally mounted using Fluoromount (Sigma) and imaged using a Leica SP5 confocal microscope. Primary and secondary antibody solutions were made in 3% normal goat serum in 0.3% Triton PBS. For melanopsin and PHb-ipRGC mCherry expression analysis, high magnification images (pixel size of 0.36 µm) with a z-stack size of 1 µm were taken. For viral expression in brain regions, high-magnification images (pixel size of 0.72 µm) with a z-stack size of 2 µm were taken. All images were processed and analyzed using ImageJ.
Fiber photometry
Mice were anaesthetized with isoflurane (Kent Scientific VetFlo system) and then unilaterally injected with 150 nl of AAV1-CAG-GCaMP6s-WPRE-SV40 ( ~ 8 ×1012 GC/mL, see Supplementary Table 1) in the PHb using a stereotaxic injector (Neurostar) controlled by the software Stereodrive at an injection rate of 30 nl/min. A FP cannula (MFC_200/230-0.48_3mm_MF1.25_FLT, DORIC) was then implanted 150 µm above the site of injection. After surgery procedures, mice were subcutaneously injected with 2 mg/kg of meloxicam (Covetrus). Efficiency of the brain injection and fiber implantation was tested postmortem.
Behavioral assay and FP recordings were made 4 weeks post-surgery. For that, dark-adapted mice were exposed to the LTTA paradigm as described above. For FP recording, an optic fiber (MFP_400/430/1100-0.57_1m_FC-ZF1.25(F)_LAF, DORIC) was attached to the head of the animals and then were placed to the LTTA arena. FP signal was collected using an assisted electrical rotary system (AERJ_24_FMC, DORIC), Amplifier (Doric Fluorescence Detector, DORIC), LED driver (Laser Diode Fiber Light Source, DORIC) and processor (RZ5P Base Processor, Tucker-Davis Technologies). FP data were extracted using pMAT (RRID:SCR_022570) software28. All FP data processing and behavior correlation analyses were made using eZtrack, Microsoft Excel (RRID:SCR_016137) and Graph Pad Prism 10.1.2 (RRID: SCR_002798).
Statistical comparisons
All graphs and statistical analyses were performed using Graph Pad Prism 10.1.2. Normal distribution of data was tested using Shapiro-Wilk test. Data points were excluded only if identified as statistical outliers using the ROUT method (Q = 1%), which was pre-established prior to data analysis. No other data were excluded from the analyses. Two-sided Student’s t or Mann Whitney U tests were used to compare two experimental groups. For multiple statistical comparisons, we performed one-way ANOVA followed by Tukey’s and Dunn’s tests. Significance was concluded when P < 0.05.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data are provided with this paper.
References
Gross, C. T. & Canteras, N. S. The many paths to fear. Nat. Rev. Neurosci. 13, 651–658 (2012).
LeDoux, J. E. Emotion circuits in the brain. Annu. Rev. Neurosci. 23, 155–184 (2000).
Tovote, P., Fadok, J. P. & Luthi, A. Neuronal circuits for fear and anxiety. Nat. Rev. Neurosci. 16, 317–331 (2015).
Yilmaz, M. & Meister, M. Rapid innate defensive responses of mice to looming visual stimuli. Curr. Biol. 23, 2011–2015 (2013).
Fratzl, A. et al. Flexible inhibitory control of visually evoked defensive behavior by the ventral lateral geniculate nucleus. Neuron 109, 3810–3822 e3819 (2021).
Evans, D. A. et al. A synaptic threshold mechanism for computing escape decisions. Nature 558, 590–594 (2018).
Calanni, J. S. et al. An ethologically relevant paradigm to assess defensive response to looming visual contrast stimuli. Sci. Rep. 14, 12499 (2024).
Shang, C. et al. Divergent midbrain circuits orchestrate escape and freezing responses to looming stimuli in mice. Nat. Commun. 9, 1232 (2018).
Wei, P. et al. Processing of visually evoked innate fear by a non-canonical thalamic pathway. Nat. Commun. 6, 6756 (2015).
Wang, F., Li, E., De, L., Wu, Q. & Zhang, Y. OFF-transient alpha RGCs mediate looming-triggered innate defensive response. Curr. Biol. 31, 2263–2273 e2263 (2021).
Salay, L. D. & Huberman, A. D. Divergent outputs of the ventral lateral geniculate nucleus mediate visually evoked defensive behaviors. Cell Rep. 37, 109792 (2021).
Aranda, M. L. & Schmidt, T. M. Diversity of intrinsically photosensitive retinal ganglion cells: circuits and functions. Cell Mol. Life Sci. https://doi.org/10.1007/s00018-020-03641-5 (2020).
Berson, D. M., Dunn, F. A. & Takao, M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 295, 1070–1073 (2002).
Hattar, S., Liao, H. W., Takao, M., Berson, D. M. & Yau, K. W. Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science 295, 1065–1070 (2002).
Chew, K. S. et al. A subset of ipRGCs regulates both maturation of the circadian clock and segregation of retinogeniculate projections in mice. Elife 6, https://doi.org/10.7554/eLife.22861 (2017).
Lazzerini Ospri, L. et al. Light affects the prefrontal cortex via intrinsically photosensitive retinal ganglion cells. Sci. Adv. 10, eadh9251 (2024).
Hu, J. et al. Melanopsin retinal ganglion cells mediate light-promoted brain development. Cell 185, 3124–3137 e3115 (2022).
Keenan, W. T. et al. A visual circuit uses complementary mechanisms to support transient and sustained pupil constriction. Elife 5, https://doi.org/10.7554/eLife.15392 (2016).
Chen, S. K., Badea, T. C. & Hattar, S. Photoentrainment and pupillary light reflex are mediated by distinct populations of ipRGCs. Nature 476, 92–95 (2011).
Li, C. et al. Pathway-specific inputs to the superior colliculus support flexible responses to visual threat. Sci. Adv. 9, eade3874 (2023).
Weil, T. et al. Daily changes in light influence mood via inhibitory networks within the thalamic perihabenular nucleus. Sci. Adv. 8, eabn3567 (2022).
Fernandez, D. C. et al. Light Affects Mood and Learning through Distinct Retina-Brain Pathways. Cell 175, 71–84 e18 (2018).
An, K. et al. A circadian rhythm-gated subcortical pathway for nighttime-light-induced depressive-like behaviors in mice. Nat. Neurosci. 23, 869–880 (2020).
Contreras, E., Nobleman, A. P., Robinson, P. R. & Schmidt, T. M. Melanopsin phototransduction: beyond canonical cascades. J. Exp. Biol. 224, https://doi.org/10.1242/jeb.226522 (2021).
Sonoda, T., Lee, S. K., Birnbaumer, L. & Schmidt, T. M. Melanopsin phototransduction is repurposed by ipRGC subtypes to shape the function of distinct visual circuits. Neuron 99, 754–767 e754 (2018).
LeGates, T. A. et al. Aberrant light directly impairs mood and learning through melanopsin-expressing neurons. Nature 491, 594–598 (2012).
Pennington, Z. T. et al. ezTrack: An open-source video analysis pipeline for the investigation of animal behavior. Sci. Rep. 9, 19979 (2019).
Bruno, C. A. et al. pMAT: An open-source software suite for the analysis of fiber photometry data. Pharmacol. Biochem. Behav. 201, 173093 (2021).
Acknowledgements
National Institutes of Health grant R01 EY030565 and National Institutes of Health grant DP2 EY022584 (TMS). We thank Dr. Samer Hattar for the gift of Opn4Cre/+mice, Maria Syeda for her contributions to obtaining the Opn4fl mice and Dr. William Klein for the gift of Brn3bDTA mice.
Author information
Authors and Affiliations
Contributions
Conceptualization: M.L.A. and T.M.S. Methodology: M.L.A., E.M., L.L., A.E.S., H.W. Investigation: M.L.A., E.M., L.L., A.E.S., H.W. and T.M.S. Visualization: M.L.A. and T.M.S. Funding acquisition: T.M.S. Project administration: T.M.S. Supervision: M.L.A and T.M.S. Writing—original draft: M.L.A. and T.M.S. Writing—review & editing: M.L.A and T.M.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Aranda, M.L., Min, E., Liu, L.T. et al. Light tunes long-term threat avoidance behavior in male mice. Nat Commun 17, 2728 (2026). https://doi.org/10.1038/s41467-026-69564-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69564-0