Abstract
Light-mediated electronic spin modulation possesses intriguing potentials for photochemistry, enabling on-demand customization of catalysts towards distinct reactions and catalytic requirements. However, the significant photobleaching of the transient spin transitions as well as their temporal mismatch with slower chemical reaction dynamics substantially hinders its applicability. Herein, we demonstrate light-driven steady-state and on-demand catalyst spin modulation that effectively activates plasmonic catalysis. The rapidly oscillating plasmonic electromagnetic near-field spin-polarizes a low-spin CoFe2O4 catalyst and overcomes the photobleaching to produce stable high-spin states with astounding spin lifetimes >60 μs. The high-spin plasmonic catalyst effectively balances the tradeoff between spin polarization and carrier dynamics. For benchmark light-driven nitrate reduction catalysis, it achieves substantial photo-enhancement in ammonia production rate and selectivity as well as photocatalytic performance driven by sunlight, benefiting from polarization activation of nitrate reactant and preferential reaction pathway modulation. The highly generalized light-mediated strategy opens intriguing new avenues for on-demand and steady-state electronic spin engineering with profound implications for distinct disciplines.
Similar content being viewed by others
Introduction
Light-mediated electronic spin modulation represents an emerging strategy to engineer the optical and electric responses of nanomaterials in the presence of external electromagnetic (EM) stimuli1,2,3. It has, thus, demonstrated intriguing potentials in distinct disciplines, including quantum communications4, optoelectronic devices5, paramagnetic sensors6, and catalysis7,8. In particular, nanocatalysts could benefit from effective modulation of their spin states, factoring in facilitated charge transfer dynamics9,10, modulated electronic d band center11,12, and/or activated surface reactive sites13,14. Compared to conventional spin modulation approaches based on crystal structure engineering strategies or external (electro)magnetic fields8,11,15, light modulation is distincted by its in-situ, on-demand and non-invasive nature without jeopardizing additional intrinsic properties of the catalysts, such as their charge carrier mobility16,17, which is exceptionally valuable for light-driven catalytic applications. However, the temporal mismatch between transient spin transitions and slower chemical reaction dynamics significantly hinders its applicability, as the rapid, sub-nanosecond relaxation of the photoexcited high-spin state(s) back to steady-state low-spin counterpart16,18 substantially limits the availability of the former for key chemical conversion steps, including adsorption and diffusion (micro-to-milliseconds). This issue is unlikely to be alleviated even with constant illumination, as the limited absorption cross-section of the catalysts and significant photobleaching effects of spin transitions prevent their continuous photoexcitation.
Localized surface plasmon resonance (LSPR) could provide a plausible solution to the above-mentioned challenge. It is signified by the collective charge oscillation upon resonant photoexcitation19,20, creating rapidly alternating EM near-fields at pico-to-femtohertz frequencies21,22, presenting intriguing possibilities for spin polarization. In this work, we achieve light-driven and surface plasmon-mediated steady-state electronic spin modulation that stabilizes the otherwise transient high-spin states in situ to effectively activate plasmonic catalytic performance. The photoexcited and rapidly oscillating plasmonic EM near-field spin-polarizes low-spin CoFe2O4 and effectively overcomes the photobleaching in its spin transition, producing stable high-spin form with astounding lifetimes as long as ~60 μs. Nitrate reduction reaction is used as a benchmark for the light-driven catalytic performance. Benefiting from the polarization activation of nitrate reactant, preferential modulation of ammonia production pathways, and facilitated ammonia desorption, the high-spin plasmonic catalyst balances the tradeoff between spin polarization and carrier dynamics, and achieves considerable photo-enhancement in ammonia production rate and selectivity as well as photocatalytic nitrate reduction performance driven solely by light. The highly generalized light-mediated strategy opens intriguing new avenues for on-demand and steady-state electronic spin engineering with profound implications for light-driven catalysis and beyond.
Results
Design principle of the surface plasmon-mediated spin modulation strategy
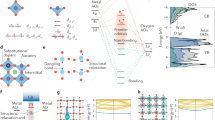
The key to the light-induced and surface plasmon-mediated catalyst spin modulation lies in the exploitation of the resonant plasmonic EM near-field to induce a stable magnetic dipole for the duration of plasmonic excitation, facilitating strong dipole interactions towards activated light-driven catalysis (Fig. 1a). The increased optical cross-sections and the rapidly oscillating field directionality is able to conserve the spin-multiplicity after initial polarization, enforcing spin-selectivity in the charge relaxation process, and thus overcoming the significant down-transition and photobleaching to achieve steady-state spin modulation concurrent to the plasmonic near-field. As depicted in our finite-difference time domain (FDTD) simulations, the plasmonic near field contains intense electric as well as magnetic components perpendicular to each other (Fig. 1a, insets). The role of electric field component in induced dipole interactions has been previously demonstrated for resonant plasmonic energy transfers in the form of “forced” plasmons22,23. On this basis, it is hypothesized that the synergy between the oscillating near-field magnetic and electric components could respectively induce stable spin polarization and carrier dynamics modulation.
Schematic illustrations of the spin modulation strategy (a) and the Au-CoFe2O4@SiO2 spin-active plasmonic catalyst (b). Red spheres: Oxygen; Brown: Fe; Blue: Co; Yellow arrow: surface Fe unit cell; Blue arrow: bulk Fe unit cell; green: Co unit cell. (c) TEM and HRTEM micrographs of Au-CoFe2O4@SiO2. Raman (d) and single-particle dark-field scattering (e) spectra of Au-CoFe2O4@SiO2 and controls. Au-CoFe2O4@SiO2 Fe 2p (f) and Co 2p (g) ISI-XPS spectra with light or in dark.
As the resonant plasmonic near-field is highly intense at nanoplasmonic interfaces but decays exponentially with distance, it is essential to localize the spin-active component within the direct vicinity of the plasmonic nanoantenna. To this end, a prototypical plasmonic antenna-reactor (A-R) catalyst architecture is constructed using gold nanoantenna and cobalt ferrite (CoFe2O4) catalyst (Fig. 1a, see also Fig. S1). As elaborated in the next section, the CoFe2O4 nanocrystals contains predominantly (~72%) normal spinel structure, which possesses Fe(III) species (3d5) within octahedral interstices (Fig. 1b). In accordance with the crystal field theory11,15, the octahedral crystal field could then split the degenerate d orbitals into lower-energy t2g and higher-energy eg bands with different filling profiles at high-spin (HS) (eg2 t2g3) or low-spin (LS) (eg0 t2g5), rendering the Fe(III)oct highly spin-“active”. For surface ·Fe species, the absence of an axial lattice oxygen leads to a square pyramidal crystal field, producing additional elevations in the orbital energies of dxy and dx²−y² orbitals. This is corroborated by our density-functional theory (DFT) calculations of the electron configurations and orbital energies (Supplementary Data 1 and Fig. S2). On the other hand, the tetrahedral Co(II) (3d7) would exhibit identical electron configuration of eg4t2g3 and is, thus, spin-“inactive”.
The CoFe2O4 nanocrystals are encapsulated within a silica nanoshell to insulate possible plasmonic hot carrier or heat transfer processes for close examination of the plasmon-mediated spin modulation. The nanoshell is highly permeable by the near field as well as molecular reactants in later catalytic measurements. CoFe2O4@SiO2 was tethered to Au nanoantenna via a thiolated silane linker to form the spin-active plasmonic nanocatalyst termed Au-CoFe2O4@SiO2 (Fig. 1a, see also Fig. S1). The distance between the antenna and reactor components were controlled to be 3–5 nm for optimal near field-enhancement24. Transmission electron microscopy (TEM) images (Fig. 1c) and element mapping (Fig. S3) indicate that the CoFe2O4 nanocrystals (10.1 ± 0.9 nm in diameter) are evenly dispersed around the Au nanoantenna (60.1 ± 2.9 nm in diameter). High-Resolution TEM (HRTEM) image exhibits lattice spacings of 0.252 and 0.219 nm in CoFe2O4 as well as 0.236 nm in Au NPs (Fig. 1c), respectively corresponding to the CoFe2O4 (311), (400) and Au (111) crystal facets, consistent with the dominant peaks from X-ray diffraction (XRD) spectra (Fig. S4). Raman spectra of Au-CoFe2O4@SiO2 display clear signatures for CoFe2O4 (A1g at 627 and 684 cm−1, T2g at 442 cm−1, and Eg at around 300 cm−1)25 as well as for Au-linker26 (Fig. 1d), further corroborating its successful formation.
The resonant constructive enhancement between Au plasmon resonance and CoFe2O4 interband transitions24 leads to a prominent increase in the visible light absorbance of Au-CoFe2O4@SiO2 (Fig. S5). Notably, the enhancement in absorbance exceeds a physical mixture control containing identical concentration of Au and CoFe2O4 colloids, which signifies the importance of localizing the spin-active component within the plasmonic near-field for efficient resonant enhancement. As a consequence of the strong resonance, significant energy transfer from the Au nanoantenna towards CoFe2O4 could be mediated by the resonant plasmonic near-field21,27. Single-particle dark-field hyperspectral imaging and scattering spectra depict an obvious red-shift as well as an increase in FWHM for Au-CoFe2O4@SiO2 in comparison to Au nanoparticle (Figs. 1e and S6), which indicates the dampening of the energies associated with the excited plasmons and energy transfer towards the CoFe2O4 nanocrystals owing to an increase in dielectric function in the surrounding environment of Au antenna in the presence of CoFe2O428.
To further characterize the oxidation state and photo-response of Au-CoFe2O4@SiO2, in-situ irradiated X-ray photoelectron spectroscopy (ISI-XPS) was next performed. Fe and Co 2p XPS spectra respectively exhibit predominant feature peaks for Fe3+ and Co2+ (Fig. 1f, g), consistent with the prevalence of normal spinel structure and a chemical stoichiometry of around 2:1 (Table S1). When illuminated with Xe lamp, the Au 4f5/2 and 4f7/2 peaks for both Au-CoFe2O4@SiO2 and a Au NPs-only control both depict a uniform shift towards lower binding energies (BE) (Fig. S7), consistent with the resonant excitation of the localized surface plasmons29,30. Notably, the Fe3+ 2p3/2 and 2p1/2 features31,32 of Au-CoFe2O4@SiO2 depict apparent shifts towards lower BE (Δshift = 0.3 eV) when illuminated (Fig. 1f), which is not observed for the Co species (Fig. 1g) or with the CoFe2O4-only control (Fig. S8). These observations are highly consistent with the spin polarization of the spin-active Fe species, but not the spin-inactive Co in Au-CoFe2O4@SiO2, from LS to HS, leading to lone-pair HS electrons having lower apparent binding energies33.
Evidence for the surface plasmon-mediated spin modulation
At room temperature and when not exposed to external stimuli such as EM field or illumination, the Fe species preferentially occur in its LS state in the CoFe2O4 nanocrystal9,34. To corroborate this phenomenon, the magnetic susceptibility (χ) of CoFe2O4 was first measured as a function of temperature (Fig. S9), where very low χ on the order of 10−2 is obtained, consistent with its superparamagnetic nature35. Compositing the nanocrystal with Au antenna further decreased the overall χ to 3.3 × 10−3 (Fig. S9). As expected, both CoFe2O4 and Au-CoFe2O4@SiO2 exhibit no evident hysteresis behavior (Fig. S10). The spin state of the nanocrystal was next analyzed based on the Curie-Weiss law36,37, taking into consideration the magnetic moments of both Co and Fe cations. A Curie–Weiss constant (C) of 0.821 was fitted from the magnetic susceptibility profile (Fig. S9b), which corresponds to a HS/LS ratio of around 0.08 (~7.4% HS + 92.6% LS), confirming the predominance of LS state without external stimuli.
To corroborate the light-induced and surface plasmon-mediated spin modulation, operando X-ray emission spectroscopy (XES) was first performed for Au-CoFe2O4@SiO2 and controls (Fig. 2a). XES represents a benchmark to probe the spin states of molecular or nanoscale systems, where the Kβ emission lines of spin-active elements are highly sensitive to the effective number of unpaired 3 d electrons18,38,39, and the Kβ’ satellite could also be correlated with local 3d spin moments40. In our measurements, a main Fe3+ Kβ1,3 emission peak as well as a pronounced Kβ’ satellite line could be observed, respectively at ~7058 and ~7044 eV. Importantly, when Au-CoFe2O4@SiO2 is illuminated with Xe lamp (1-sun), both Kβ1,3 and Kβ’ lines demonstrate an apparent redshift towards lower emission energies (Δshift = 0.4 eV and 1.1 eV, respectively), consistent with the lower binding energies of the HS eg electron. Furthermore, an observable increase in the relative intensity of the Kβ’ satellite peak is observed with illumination (relative area ratio of 0.20) compared to in dark (0.17) (Fig. S11 and Table S2). These phenomena are highly consistent with light-induced spin polarization, as HS electronic states favor the 3p-3d spin-orbit coupling and could lead to the redshift of Kβ emission as well as favor the Kβ’ satellite feature39. In contrast, the CoFe2O4-only control showed no evident changes with illumination (Fig. 2a, inset), which underscores the necessity of surface plasmon in spin modulation.
a XES spectra of Au-CoFe2O4@SiO2 and CoFe2O4-only control (inset) with/without illumination. b Raman spectra of Au-CoFe2O4@SiO2 under non-resonant (top) and resonant (bottom) incident wavelengths. c–e Au-CoFe2O4@SiO2 Fe K-edge XANES spectra (Inset: Magnification of the absorption rising edge and pre-edge peak) (c), Fourier transforms of k2-weighted EXAFS in R-space (d) and wavelet transformed-EXAFS (e) with/without illumination. f Calculated magnetic moment, Fe–O bond length and O–Fe–O bond angles at LS and HS. Red spheres: Oxygen; brown: Fe; yellow: lone-pair electron.
Spin polarization has also been shown to induce distinct alterations to Raman vibrational features41,42. In accordance, surface-enhanced Raman spectroscopy was carried out for Au-CoFe2O4@SiO2 at on-resonance and off-resonance excitation wavelengths (Fig. S12). Consistent with our measurements in Fig. 1d, non-resonant 633-nm excitation led to two obvious A1g Raman features at 627 and 684 cm−1 (Fig. 2b). Importantly, with resonant 785-nm excitation, a prominent new peak appears at higher frequencies (709 cm−1), indicating a higher-energy vibrational mode corresponding to HS-CoFe2O4. This observation is consistent with previous reports that spin polarization into the HS state typically leads to blueshift of the vibrational modes or new signatures at larger frequencies41. As expected, this wavelength-dependent new peak is not seen with any excitation conditions for CoFe2O4-only (Fig. S13). Notably, the Raman spectra were also measured for Au-CoFe2O4@SiO2 with in situ heating up to 40 °C, where no evident changes to the two A1g peaks were detected (Fig. S14), providing direct proof that it is through the resonant plasmonic near-field, rather than plasmonic heating, that the spin modulation is achieved.
To obtain in-depth understandings on the coordination and electronic structure, X-ray absorption spectroscopy (XAS) was next performed43,44. The average coordination number of Fe was first analyzed from the X-ray absorption near-edge structure (XANES) spectra as well as Fourier transformation (FT) of k2-weighted Extended X-ray Absorption Fine Structure (EXAFS) spectra (Fig. S15 and Table S3). A predominant proportion of 86% of Fe cations with a coordination number of 6 (positioning in octahedral interstices) is calculated, translating to a ratio of 0.72:0.28 between regular and inverse spinel structures, with clear prevalence of the former. This confirms the crystal and electronic structure outlined in the previous section that octahedral Fe(III) represents a most dominant spin-active component.
To illustrate the light-induced alterations to the electronic structure, in situ illumination was next applied in XAS measurements. When illuminated, the Au-CoFe2O4@SiO2 Fe K-edge XANES spectrum exhibits a conspicuous shift of the absorption rising edge towards lower absorption energies, as well as increases in the white-line intensity and width (Figs. 2c and S16). These observations are highly consistent with the lower binding energies as well as the higher coordination number associated with the unpaired 3d electrons at HS43. Transport of plasmonic charge carriers from Au towards CoFe2O4 could, in theory, also lead to similar shifts towards lower energies; yet it is effectively excluded by the silica nanoshell and the XPS peak shift upon illumination (Fig. S8). Moreover, the pre-edge absorption peak around 7115 eV is also correlated with the spin configuration, particularly for surface Fe(III) in square pyramidal crystal fields. This peak is derived from the Fe 1s-3d spin transition, whose intensity is inversely related to the degree of centrosymmetry of the local coordination environment45,46. The lone-pair electrons in HS Fe could induce moderate distortion of the equatorial O ligands owing to steric repulsion, and thus decrease its degree of centrosymmetry. As expected, the Fe pre-edge intensity in Au-CoFe2O4@SiO2 increased with illumination (Fig. 2c, inset), consistent with the LS-to-HS spin transition.
Fourier transformation of the Fe K-edge EXAFS spectra (Fig. 2d) as well as wavelet-transform analysis (Fig. 2e) indicate that when illuminated, the fitted Fe–O scattering in Au-CoFe2O4@SiO2 exhibits significant lengthening, which can typically be correlated with elongated bond length associated with the HS population owing to its abundant lone-pair electrons47,48. In addition, evident increase in R-space signal intensity is also observed with illumination, which can be accounted for by the increase in local degree of disorder caused by the spin polarization into HS (Table S4).
The observed changes in coordination structure as well as magnetic properties of the plasmonic spin-active catalyst are further confirmed by our DFT calculations (Fig. 2f). For fully LS surface Fe in square pyramidal crystal field, a net average number of lone-pair electron of 1.50 (effective magnetic moment μeff = 2.28) is calculated, consistent with our Curie-Weiss analyses (Fig. S9b). As anticipated, increasing magnetic moment and bond lengths are found to be associated with the high-spin catalyst, consistent with our EXAFS analyses above. Calculations of the O–Fe–O bond angles shed further light on the spin modulation effect for surface Fe(III). For LS CoFe2O4, the equatorial O ligands primarily assume a planar configuration, with the sum of adjacent O–Fe–O bond angles (α + β) close to 180°. Importantly, with illumination, the HS CoFe2O4 shows an increase in the sum of adjacent O–Fe–O bond angle, exceeding 180° (Fig. 2f). This agrees well with the strong steric repulsion of HS lone-pair electron that pushes the equatorial oxygen ligands out-of-plane.
Photochemical implications of plasmon-mediated spin modulation
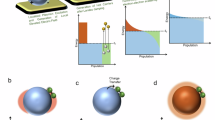
The rapidly oscillating plasmonic near-field could not only achieve spin polarization to facilitate HS production, but also exert spin-selectivity to the relaxation processes, overcoming photobleaching and maintaining spin multiplicity. This could, in turn, significantly prolong the lifetime of HS excited states, thus bridging the temporal mismatch between the transient light-induced spin transitions and chemical reactivity. To obtain insights on the relaxation kinetics, transient absorption (TA) spectroscopy was next performed. After excitation with resonant pump laser (515 nm), significant ground state bleaching (GSB) signal could be observed for Au-CoFe2O4@SiO2 at 550 nm due to the rapid relaxation of the excited plasmons49 (Fig. 3a), which is also observed in the absorption pattern of the Au NP-only control (Fig. S17). Importantly, for Au-CoFe2O4@SiO2, a strong broadband photo-induced absorption (PIA) feature is observed in the 650–950 nm range (Fig. 3a, b), which is much weaker in the CoFe2O4-only control sample owing to the latter’s rapid photobleaching of the light-induced spin transition (Fig. S18). The PIA persists far beyond the time window of our TAS measurements, as evidenced by a significant non-zero background signal before time zero. By comparing the amplitudes before and after time zero (see SI), we estimate its lifetime to be as long as ~60 μs (Fig. 3c). Such a long-lived species is consistent with a Fe HS excited state that relaxes slowly to the ground state. Notably, Au-CoFe2O4@SiO2 exhibits a slow buildup time of ~62 ps. This feature corresponds to the plasmonic near-field-mediated energy transfer from Au nanoantenna to CoFe2O4, leading to an order-of-magnitude enhancement of the long-lived HS population compared to the CoFe2O4-only control, in which the slow buildup feature is not present. Time-resolved circular dichroism further reveals that the spin polarization of Au-CoFe2O4@SiO2 decays with a time constant of ~6.8 ps (Fig. 3d), much slower than the sub-picosecond spin-flip times typically observed50. This suggests that the spin polarization is likely preserved via resonant plasmonic energy transfer concurrent with illumination following the initial spin transition.
Transient absorption spectra (a), pseudo-color map (b), fitting of the absorption feature at 700 nm (c) and time-resolved circular dichroism spectrum (d) for Au-CoFe2O4@SiO2. e DFT calculations of the PDOS for HS and LS Au-CoFe2O4@SiO2. f Pulsed chemisorption for Au-CoFe2O4@SiO2 with/without illumination using CO as a molecular marker. Inset: saturated quantity of adsorption.
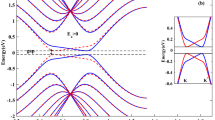
Light-induced spin polarization into high-spin Fe states can significantly modulate the local density of EM states as well as the band structure of the plasmonic catalyst11,12. In this term, DFT calculations of the projected density of states (PDOS) was performed for the illuminated and non-illuminated conditions (Figs. 3e and S19). Upon resonant irradiation, the light-induced spin polarization leads to noticeable dichotomy in state population distribution in HS CoFe2O4 owing to the spin transition and the difference in energy between different spin states, leading to an overall lower d-band center and wider distribution deviation. HS contains substantially increased and more localized state population at higher energy levels, which could present significant changes to its adsorption capability towards distinct reaction intermediates51. In the HS configuration, characterized by weaker crystal field splitting47, unpaired electrons occupy both t2g and eg orbitals. This results in a high magnetic moment and significant contributions from eg orbitals near the Fermi level (EFermi) (dz² and dx²−y²). Conversely, the LS configuration, stabilized by stronger crystal field splitting, pairs electrons in t2g orbitals, reducing the magnetic moment.
Stable HS catalysts have been previously shown to be associated with increased surface available active site for chemical reaction as well as modulated adsorption capabilities52,53. To demonstrate theses effects, pulsed chemisorption (PC) experiments were performed for Au-CoFe2O4@SiO2 with or without illumination (Fig. 3f), where a CO molecular marker was exposed to the catalyst surface in a pulsed manner to determine chemical adsorption capability. The rate of adsorption (slope) for non-diffusion-controlled region can be considered to reflect the intrinsic adsorption capability of individual active sites, while the time (or number of pulses) at which saturation adsorption is reached is considered to reflect the number of active sites available for adsorption. As expected, upon resonant irradiation, the HS Au-CoFe2O4@SiO2 exhibited both larger slope and much later saturation than the LS counterpart in dark, pointing at both improved adsorption capability and increased number of available sites for adsorption (Fig. 3f, inset).
Spin-modulated plasmonic catalytic performance
To investigate the effect of surface plasmon-mediated spin polarization on light-driven plasmonic catalytic performance, we next utilized the photoelectrochemical nitrate reduction reaction (NO3RR) into ammonia as a benchmark. NO3RR involves the relatively non-polar nitrate reactant molecules in the catalytic process, which could benefit from polarization activation by spin-modulated nanocatalysts54,55. NO3RR has also been shown to be sensitive to catalysts’ electronic spin state56,57, which renders it a meaningful platform to test the effect of the plasmon-mediated spin modulation. To characterize the light-driven catalytic performance, linear sweep voltammetry (LSV) was first performed in 1 M KOH and 0.1 M NaNO3 in dark or under 1-sun (100 mW/cm2) Xe-lamp illumination (Figs. 4a and S20). When illuminated, the LSV curves for Au-CoFe2O4@SiO2 exhibited a tremendous gain in photocurrent density (Jphoto) (106.8 mA/cm2), which corroborates the distinct light-driven catalytic reactivity. A similar Jphoto (91.4 mA/cm2) was obtained with 590 nm LED irradiation, which resonates with the red-edge of Au surface plasmons (Fig. 1e). Considering the extremely low current density in dark, the plasmon-mediated spin modulation effectively activates plasmonic catalysis for light-mediated NO3RR. To probe the effect of surface plasmons in promoting the reaction kinetics, the kinetic order (n) for nitrate58 was determined by varying its concentrations in LSV measurements (Fig. S21). Notably, higher kinetic orders are calculated with illumination (n = 2.83) than in dark (n = 1.59) for Au-CoFe2O4@SiO2.
a Photoelectrochemical nitrate reduction LSV curves for Au-CoFe2O4@SiO2 and controls. b, c Incident wavelength and power dependence of photocurrent. d Light-modulated photocurrent measurement with alternating lights on/off cycles (100 s-on/100 s-off) (Inset: Magnification of the photocurrent response). e Chronoamperometric I-t curves at −1.5 V with light or in dark. f Photoelectrochemical ammonia production rate (Column chart) and selectivity (Line graph).
The role of photo-illumination on light-driven NO3RR catalysis is next probed by monitoring the photocurrent as a function of incident wavelength and light power. In wavelength-dependence tests, maximum Jphoto is attained with 590 nm illumination (Fig. 4b), which matches the plasmon resonance maximum of the nanocatalyst. For measurement of the catalytic photocurrent on illumination light power, it has been previously demonstrated that a linear dependence indicates a photochemistry-dominating process; whereas non-linear, exponential profile is typically considered a signature of the photothermal effect22. As expected, both 590 nm and polychromatic Xe lamp illumination depicted a linear dependence profile (Fig. 4c), consistent with the photochemical origin of the enhanced photocatalytic performance driven by the HS lone-pair electrons. This is further evidenced by our light-modulated photocurrent measurements59, where illumination led to evident surges in the absolute value of the NO3RR photocurrent (Fig. 4d). The increase in photocurrent upon irradiation contains primarily a fast-response region with rapid increase in photocurrent owing to the dominant photochemical effects. The moderate slow-response region with more attenuated increase due to photothermal effect is much less pronounced60.
To quantify the contribution of photochemical and photothermal effects to light-driven catalytic performance, the temperature of Au-CoFe2O4@SiO2 was monitored using infrared (IR) camera in situ. A 16 °C increase in catalyst surface temperature was observed under identical illumination and aqueous electrolyte conditions, which led to very minor changes to the electrolyte temperature (<2 °C) (Fig. S22). As demonstrated above, such extent of temperature increase does not have significant impact on the spin state of the plasmonic catalyst (Fig. S14). Its promotion role on catalytic performance is also moderate, as indicated by the much lower thermal current (11.2 mA/cm2) obtained with in situ external heating compared to the photocurrent (Jphoto = 106.8 mA/cm2) (Figs. S23 and S24). These results further illustrate that it is the surface plasmon-mediated spin modulation that effectively activates light-driven catalytic performance.
The catalytic stability was next evaluated through chronoamperometric I-t measurements with illumination (Fig. 4e, see also Fig. S25), where the photocurrent density remains steady over a measurement period of 10 hours. This is further evidenced by the TEM images and Raman spectra for “spent” catalyst after the light-driven catalytic reaction, which depict no noticeable changes (Fig. S26). As a result of the stable photo-response, robust and highly selective ammonia production was achieved under photoelectrochemical catalytic conditions (Fig. 4f, see also Fig. S27). A highest NH3 yield of 1.135 mmol mgcat.−1 h−1 is attained by Au-CoFe2O4@SiO2 with illumination. The selectivity of ammonia depicts a parabolic dependence pattern on applied bias, peaking at −1.6 V (87.3%) (Fig. 4f, see also Fig. S28), consistent with the increased water splitting at higher biases. These findings further corroborate the substantial light-mediated catalytic modulation of the spin-active plasmonic catalyst.
Catalytic mechanism and applicability
To illustrate the spin-modulated catalytic pathway, in situ Raman spectroscopy was utilized to probe the catalytic intermediates for Au-CoFe2O4@SiO2 during operando photoelectrochemical NO3RR measurements (Figs. 5a and S29). For all spectra obtained, two strong peaks from the D (~1313 cm−1) and G (~1587 cm−1) bands of the substrate as well as a clear band between 1000 and 1050 cm−1 from the NO3− reactant are present61. No additional peaks could be observed at time 0 (Fig. 5a, bottom). According to the previous reports, the reduction and hydrogenation of the nitrogen species would lead to sequential production of *NH, *NH2, and ammonia species62. Consistent with these reports, as the catalytic reaction progressed with illumination and −1.5 V bias, the signal of *NH at 1507 cm−1 gradually emerged (30 min) but soon disappeared, accompanied by the appearance of distinct bands at 1130, 1240, 1380, and 1570 cm−1 at 40 min, which are respectively assigned to the *NH2 intermediate and ammonia products63. These features became more pronounced as the reaction further proceeded. For the measurement with illumination at −1.6 V, evident acceleration of the reaction could be seen, where signature peaks for the ammonia products appeared in the spectrum obtained at 30 min (Fig. S29). In contrast, no noticeable intermediate/product signals could be observed under identical illumination conditions for CoFe2O4-only (Fig. S30), due to the lack of plasmonic near-field-mediated spin modulation.
a In-situ Raman spectra for photoelectrochemical NO3RR by Au-CoFe2O4@SiO2. b DFT calculation of the free energy diagrams for NO3RR with the HS (red) and LS (blue) plasmonic catalyst. c In-situ Raman investigation of the nitrate activation behavior. d ICOHP calculations of ammonia energy levels adsorbed on HS or LS plasmonic catalyst. e Radar map of the light-driven plasmonic catalytic performance for Au-CoFe2O4@SiO2 with/without illumination (red/black) and CoFe2O4-only in dark (blue). f Purely photocatalytic nitrate reduction performance for Au-CoFe2O4@SiO2. Retest: 3 times. g NO3RR catalytic performance as a function of external field intensity. h Generality of the plasmon-mediated spin modulation strategy.
To corroborate the experimental findings on reaction pathway, DFT calculations of the Gibbs free energy diagram were performed with the HS and LS plasmonic catalyst (Fig. 5b). The effect of excited Au plasmons on the Fe species was modeled by altering the magnetic moment of Fe active sites. Gibbs free energy diagram of the NO3RR reaction indicates that the adsorbed *NO3 is reduced to *NO, and then hydrogenated sequentially to *NH3 via *NH, *NH2 intermediates, consistent with our in-situ Raman results. For HS Au-CoFe2O4@SiO2, in general, lower free energy barriers were calculated compared to the LS counterpart, indicating the facilitated adsorption and formation of the key reaction intermediates. For instance, the transition from *NO3 to *HNO3 requires only 0.28 eV in the HS state, compared to 0.44 eV in the LS state. Similarly, intermediate steps such as *NHO to *NH3 exhibit significantly reduced Gibbs free energy changes in the HS state. The lower energy barriers are attributed to the unpaired eg electrons, which enhance bonding interactions with adsorbed species and facilitate critical bond-breaking and forming processes that underpin catalytic efficiency. Notably, the HS state achieves a final Gibbs free energy of −0.40 eV during NH3 formation, in contrast to 0.82 eV observed for the LS state, underscoring its thermodynamic favorability. Therefore, desorption of the *NH3 into ammonium product is apparently facilitated by the HS nanocatalyst.
The role of light-induced HS Au-CoFe2O4@SiO2 plasmonic catalyst in activating the nitrate reactant is highly consistent with our PDOS calculation and pulsed chemisorption results (Fig. 3e, f). The plasmon-mediated spin modulation creates substantially increased population of state at higher energy sections above the vacuum level, which demonstrates significant overlap with the LUMO of nitrate that facilitate their bonding interactions. This is corroborated by our calculation of the nitrate adsorption energy of the CoFe2O4, where the (400) crystal facet, calculated to be the predominant active facet, demonstrates significant enhancement in nitrate adsorption capability at light-induced HS state (Fig. S31a). In addition, the (311) crystal facet, containing the spin-active Fe(III)oct species, is also found to be catalytically active (Fig. S31b). Furthermore, the plasmonic near-field as well as HS electrons effectively polarize the relatively non-polar N–O bondage, as indicated by our further in-situ Raman investigations, where the stretching frequencies of free-stranding and adsorbed nitrate are compared (Fig. 5c). Significant shift of N–O vibration towards lower frequencies are observed for adsorbed nitrate on Au-CoFe2O4@SiO2 when exposed to resonant illumination, indicative of bond elongation through polarization activation. This change is not seen with non-resonant illumination, or with control groups missing in one or more of the active components (Fig. S32).
HS Au-CoFe2O4@SiO2 could also modulate the reaction pathway for ammonia production by inhibiting side reactions including nitrogen and hydrogen production. HS Au-CoFe2O4@SiO2 strongly disfavors formation of *N, which is crucial for forming nitrogen side product (Fig. 5b). As a result, no obvious production of N2 gas or participation of atmospheric nitrogen are detected in our 15N isotope-labeling experiments (Fig. S33). On the other hand, light illumination considerably increases the proton adsorption energy barrier for HS species, inhibiting hydrogen production (Fig. S34). Once produced, desorption of the *NH3 products are facilitated by the HS Au-CoFe2O4@SiO2. The total Integrated Crystal Orbital Hamiltonian Population (ICOHP) of the Fe–NH3 coordination in the HS configuration is −1.50 eV, which is substantially less negative than that in the LS counterpart (−2.14 eV), indicating reduced bonding strength of the former (Fig. 5d). The COHP spectrum of the HS configuration exhibits pronounced anti-bonding states near the Fermi level, which diminish orbital overlap and destabilize the Fe–NH3 interaction, thereby facilitating *NH3 desorption. In contrast, the LS state displays a more stabilized bonding landscape, with fully occupied bonding orbitals and negligible anti-bonding character, accounting for the stronger NH3 retention and higher desorption barrier. This is consistent with our observation from the final step of ammonia desorption in the free energy diagram, and is also experimentally supported by our in-situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) measurements, where a light-induced repeatable change in the N–H bending vibration intensity at 1625 cm−1 is observed (Fig. S35).
The improved polarization activation of nitrate reactant, modulated reaction pathway and facilitated ammonia desorption collectively activates the favorable light-driven plasmonic catalytic performance for the plasmonic spin-active catalyst (Fig. 5e). To shed further light on the unique role of light illumination, purely photocatalytic nitrate reduction was next performed for Au-CoFe2O4@SiO2 under 1-sun illumination only, and the ammonia product was monitored over time (Fig. 5f). Remarkable photocatalytic properties were observed over 1.5-hour constant measurement, and an ammonia production rate of 0.041 mmol mgcat.−1 h−1 was recorded, which presents intriguing potentials for sunlight-driven reactivity.
The unique feature of the surface plasmon-mediated strategy to simultaneously achieve in situ steady-state spin modulation while promoting charge carrier dynamics is extremely valuable and unlikely to be achieved with external fields. To demonstrate this aspect, we compared the spin state and carrier dynamics of Au-CoFe2O4@SiO2 in the presence of external magnetic fields. Whilst external magnetic fields ≥100 Oe can also induce HS Raman features (Figs. S36 and S37), they could typically also bring about increased electron transport resistances (Rct) as well as considerably reduced charge carrier population according to our photoelectrochemical impedance and Mott-Schottky measurements (Fig. S38). These results are in sharp contrast with the effect of illumination, where reduced Rct and promoted carrier transport behaviors are consistent observed. As a result of the tradeoff between the spin polarization and carrier dynamics, the external field-mediated catalytic performance manifests a parabolic dependence pattern on field intensity (Fig. 5g). Even the highest magneto-current obtained (86 mA/cm2) is far lower than the photocurrent obtained with illumination (150 mA/cm2). For the illuminated condition, the localized plasmonic near-field guarantee effective spin polarization for long-lasting magnetic moments, while their pico-to-femtohertz oscillation frequency exerts spin-selectivity in the recombination and facilitates carrier extraction64,65.
Another important advantage associated with the surface plasmon-mediated spin modulation strategy is the highly general applicability towards different catalyst configuration as well as plasmonic and spin-active components. To demonstrate the generality, plasmonic Ag and Cu nanoantenna, as well as CoO nanocrystals (having spin-active 3d7 octahedral Co2+) were utilized in place of the Au antenna and/or the CoFe2O4 catalyst. With their respective resonant excitation wavelengths, the Ag and Cu antenna have also been shown to induce effective spin modulation, as evidenced by the noticeable HS Raman features (Fig. 5h), as well as significant activation in light-driven catalytic performance (Fig. S39). The generalized applicability is likely to enable the on-site and on-demand customization of spin modulation towards distinct types of catalytic reactions.
Discussion
In summary, we develop in this work an effective and generalized strategy for in situ, steady-state and on-demand electronic spin modulation through resonant light excitation of localized surface plasmons. The rapidly oscillating plasmonic EM near-field spin-polarizes low-spin spinel CoFe2O4, and effectively overcomes its photobleaching to produce stable high-spin states with an astounding lifetime of ~60 μs, thus overcoming the temporal mismatch between transient spin transitions and slower chemical reaction kinetics, activating its light-driven catalytic performance. Benefiting from the polarization activation of nitrate reactant, modulated reaction pathways and facilitated ammonia desorption, the plasmonic high-spin catalyst balances the tradeoff between spin polarization and carrier dynamics, and achieves considerable photo-enhancement in ammonia production rate and selectivity as well as photocatalytic nitrate reduction performance. Our strategy provides valuable and generalized insights for on-demand and steady-state electronic spin engineering, and opens new avenues for light-induced spin modulation towards distinct fields of applications.
Methods
Materials
All chemicals were used as raw material without further purification. Gold acid chloride trihydrate (HAuCl4·3H2O, ≥99.9%), oleylamine (OAm, 80–90%), iron acetylacetonate (Fe(acac)3, >98%), cobalt acetylacetonate (Co(acac)2, >97%) and (3-Mercaptopropyl)trimethoxysilane were purchased from Aladdin. Trisodium citrate dihydrate (C6H5Na3O7·2H2O) was purchased from Enox. Sodium borohydride (NaBH4, 99%) was purchased from Thermo Scientific. L-Ascorbic acid (AA, 99.99%) and polyvinylpyrrolidone (PVP, average mol wt 10,000) were purchased from Macklin. The silica precursor tetraethyl orthosilicate (TEOS) was obtained from TCI (Shanghai). Sigma-Aldrich supplied potassium iodide (KI, 99%), ammonium hydroxide (28 wt%) and the surfactant Igepal CO-520. All solvents (ethanol, acetone, and cyclohexane) were supplied by Sinopharm Chemical Reagent Co., Ltd.
Preparation of Au NPs
Au NPs were synthesized through a modified seed-mediated growth approach. Seed solution was first prepared by mixing water solutions of HAuCl4 (5 mM, 1 mL) and C6H5Na3O7 (1 mL, 5 mM), and diluting to a total volume of 18 mL DI water. Then, a reducing agent containing 10 mM NaBH4 was added under vigorous stirring. The color of the seed solution turned dark brown after adding the reducing agent. For NPs growth, a growth solution containing aqueous solutions of PVP (1 wt%, 1.25 mL) and HAuCl4 (0.25 M, 300 µL) was prepared. Then, the mixed solution containing 1.25 mL AA (0.1 M) and 1.25 mL KI (0.2 M) was added into the growth solution. Finally, 30 µL seed solution was added into the growth mixture with violent stirring for 20 min, then washed three times with deionized water, and stored at 4 °C for further use.
Preparation of CoFe2O4 nanocrystals
The synthesis of CoFe2O4 nanocrystals followed a modified solvothermal protocol. Specifically, a precursor solution was prepared by dissolving 3.6 mM Fe(acac)3 and 1.8 mM Co(acac)2 in 32 mL of oleylamine under vigorous stirring for 10 min at room temperature. This solution was then sealed in a 100 mL autoclave and heated to 200 °C for a duration of 12 h. Following the reaction, the autoclave was cooled to room temperature. The solid product was isolated via centrifugation, purified through sequential washing with acetone and ethanol, and finally dispersed in acetone for future use.
Preparation of CoFe2O4@SiO2 Nanocrystals
A typical synthesis began with dissolving 1 g of the surfactant Igepal CO-520 in 11 mL of cyclohexane via 30 min of ultrasonication. Subsequently, 1 mL of a pre-prepared cyclohexane dispersion containing CoFe2O4 nanocrystals (10 mg) was introduced to this mixture under stirring. After the combined solution was stirred for 5 h, tetraethyl orthosilicate (TEOS, 0.05 mL) and aqueous ammonia (28 wt%, 0.1 mL) were added sequentially. The reaction was allowed to proceed for another 5 h under continuous stirring. The final core-shell nanoparticles were isolated by centrifugation, purified through multiple ethanol washes, and ultimately stored in ethanol.
Preparation of Au-CoFe2O4@SiO2 Nanocrystals
Au NPs and CoFe2O4@SiO2 nanocrystals were linked by the thiolated silane linker. For the thiolated silane linking with Au NPs, 30 mg of Au NPs were dissolved in 30 mL of ethanol under ultrasonication for 10 min and then mixed with 0.75 mL of (3-Mercaptopropyl)trimethoxysilane. After stirring for 12 h at room temperature, the mixed solution was washed three times with ethanol and stored in ethanol. Next, 20 mg of the above solution (Au NPs with the thiolated silane linker) were dissolved in 10 mL ethanol and 15 mL DI water. Then, 0.6 mg of CoFe2O4@SiO2 was added in three times with a 10-min interval under continuous ultrasonication. Finally, the products were collected by centrifugation, cleaned with ethanol for several times and stored in ethanol.
Structural characterizations
Transmission electron microscopy (TEM) images and Energy-Dispersive X-Ray Spectroscopy (EDS) mappings were obtained with an FEI Talos F200X S/TEM at 200 kV with a field-emission gun. Samples were prepared by casting several drops of 0.1 mg/mL solutions onto copper-grid mounted “holey” carbon films, dried at room temperature. The Au, Fe and Co contents were measured by inductively coupled plasma-optical emission spectroscopy (ICP-OES) (Avio200, PerkinElmer).
Measurement of UV-vis absorbance spectra
UV-vis absorbance spectra were obtained using a Lambda 750 UV/VIS/NIR spectrometer from PerkinElmer at a scan rate of 120 nm min−1 over the range 200–800 nm. Two microliters solution of Au-CoFe2O4@SiO2 and other controls were separately recorded in a standard quartz cuvette with path length of 1 cm, volume of 3.5 cm. The blank control group ethanol solvent was performed in the same condition with the solvent above supplying the testing background.
Measurement of Raman spectroscopy
Raman spectroscopic measurements were performed using a Renishaw InVia confocal Raman Microscope equipped with a Leica 100× air objective (Numerical Aperture = 1.25). Samples, including Au-CoFe2O4@SiO2 and controls prepared as drop-casted thin films on silicon wafers (<100>, Ferrotec Shanghai), were analyzed either on the substrate or within an in-situ electrolysis cell. The measurements employed 633 nm (1800 l/mm grating) and 785 nm (1200 l/mm grating) excitation lasers under the following parameters: 14 mW power, 60 s exposure time, and 2 accumulations.
Measurement of single-particle scattering spectroscopy
The scattered light from individual particles was first collected using a Zeiss 50× air-spaced objective (numerical aperture = 0.8). This signal was then directed through a hyperspectral imaging spectrograph (Princeton Instruments, Acton SpectraPro 2156) equipped with a slit aperture. The dispersed spectrum was ultimately detected by a thermoelectrically cooled, back-illuminated CCD camera (Pixis 400BR, Princeton Instruments). The entire detection assembly was mounted on a computer-controlled translation stage (Newport, LTA-HL).
In-situ irradiated X-ray photoelectron spectroscopy (ISI-XPS) characterization
ISI-XPS measurements were conducted using a Thermo Escalab 250Xi photoelectron spectrometer. A non-monochromatized Al Kα X-ray source (1486.6 eV) was employed for excitation. A full-spectrum light source (HF-GHX-XE-300) at an intensity of 100 mW/cm² was used as the illumination source. The light was introduced from the top of the analysis chamber through a quartz window. To probe spin polarization behavior, then the XPS spectra (specifically Au 4f, Fe 2p, and Co 2p) were acquired and compared under both dark and illuminated conditions.
X-ray absorption spectroscopy (XAS)/X-ray emission spectroscopy (XES) characterization
Fe K-edge XAFS and XES spectra were performed using a lab-based instrument (easyXAFS300+ from easyXAFS, LLC) with an industrial Pd tube source (VAREX) and a silicon drift diode detector (Amptek SDD-X123). Data merging, background subtraction, normalization, and fitting of the XANES and EXAFS spectra were modeled using Artemis and Athena software, respectively. The k2-weighted Fourier transforms for all the EXAFS data were conducted in a Hanning-shaped window. The amplitude-reduction factor S02 was defined at 0.7. The R-ranges for the fitting of all the EXAFS data were set as 1–4.5 Å. The coordination numbers (CN), distances (R), and Debye–Waller factors (σ) were determined independently. The quality of the fitting was evaluated by the R-factor. We performed EXAFS curve fitting for the spinel oxides by referencing previous benchmark.
Measurement of transient absorption (TA) spectroscopy
The TA measurements were performed on a custom-built setup. A 1030 nm femtosecond output from a laser (BFL-1030-20BS, BWT) was split into two beams in an 80:20 intensity ratio. The more intense (80%) beam served as the pump. It was frequency-doubled to 515 nm, modulated by a 500 Hz optical chopper, and then focused onto the sample using a convex lens. The weaker (20%) beam functioned as the probe. It was focused into a 5 mm YAG crystal to generate a supercontinuum white-light spectrum. The transmitted probe light was collected by a fast fiber-optic spectrometer, whose acquisition was synchronized with the chopper frequency for lock-in detection.
Photoelectrochemical characterizations
Using a three-electrode setup on a CHI 660E electrochemical workstation, we performed LSV, chronoamperometry (i-t), EIS and Mott-Schottky measurements. The working electrode was a 5 mm Glassy Carbon (GC) disk, modified by drop-casting 5 μL of the catalyst colloid (Au-CoFe2O4@SiO2 or controls). An Ag/AgCl electrode and a Pt wire served as the reference and counter electrodes, respectively. The potential of −1.2 V, −1.5 V and −1.6 V with a similar collection rate of 1 Hz were adopted for the chronoamperometric i-t measurement. The wavelength dependence test used monochromatic-light LEDs with wavelengths of 420, 470, 520, 590, 620, 740 nm (MC-LED-M-λ, Beijing MerryChange Technology Co., Ltd.) with the light intensity of 100 mW/cm2. The intensity-dependent photoelectrochemical test also used the full-spectrum Xe-lamp and 590 nm LED illumination by adjusting the light intensity of illumination from 50 to 200 mW/cm2, respectively. EIS Nyquist plots were collected at an open circuit −1.6 V, with a frequency range from 100,000 to 10 Hz.
Current density (mA/cm2) of Au-CoFe2O4@SiO2 and other control samples was calculated from the measured current and the total geometric surface area (SA) of different samples on the working electrode. For the particulate photocatalyst, the calculated geometric surface area gives a conservative estimation of the current densities and photocurrent densities. Five microliters Au-CoFe2O4@SiO2 colloid with an Au concentration of 175.05 ppm calculated from ICP-OES measurements (Table S1), was dropcasted on GC electrodes for PEC measurements, which gives a mass of 8.925 × 10−7 g Au. Based on the mass density of 19.32 g/cm3, the total particle volume and the volume of an individual Au NP can be calculated. The number of Au-CoFe2O4@SiO2 NPs is calculated as 1.5 × 108, SA amounts to 0.033 cm2.
Photocurrent densities (Jphoto, mA/cm2) were calculated by subtracting the current densities in dark from those measured with illumination. Conversion between Ag/AgCl reference electrode and Reversible Hydrogen Electrodes is based on: E (vs RHE) = E(vs Ag/AgCl) + 0.0591 pH + Eө(vs Ag/AgCl), where the Eө(vs Ag/AgCl) = 0.1976, the pH = 14.
Catalytic mechanism characterizations
The NH3-desorption in situ DRIFTS experiments were performed using an FTIR spectrometer (Thermo Nicolet iS50). The CO pulse chemisorption experiments were performed in the flow reactor with gas chromatography (GC).
Density functional theory (DFT) calculations
All spin-polarized DFT computations were performed with the Vienna Ab initio Simulation Package (VASP) code. The computational setup utilized the projector-augmented wave (PAW) pseudopotentials and the Perdew–Burke–Ernzerhof (PBE) generalized gradient approximation (GGA) exchange-correlation functional. A uniform 3 × 3 × 3 k-mesh grid was used to optimize the crystal structure of bulk CoFe2O4, with lattice parameter a = b = c = 8.45 Å. For the slab models, the high-spin (HS) CoFe2O4 structure was constructed with lattice parameters of a = b = 8.53 Å, c = 21.80 Å, and α = β = γ = 90°. Pending inevitable variations in the actual crystal lattice, this model provides a closest replication of the nanocatalyst to the best of our capabilities. The low-spin (LS) CoFe2O4 slab dimensions were reduced to 89% of the original values. Both HS and LS CoFe2O4 slab models consisted of five atomic layers with (001) exposed surface separated by ~16 Å of vacuum. For the HS structures, only the bottom three atomic layers were fixed during optimizations, while all other atoms were allowed to relax. For the LS structures, both sides of the slabs were fixed to maintain the single eg orbital occupation. The first Brillourin zone was sampled with a gamma-center 3 × 3 × 1 k-point grid for the slab models. The kinetic energy cutoff for the wavefunctions was set at 450 eV for the bulk and slab structures without and with molecular adsorption. The structural optimizations were considered convergent when the force on each atom was less than 0.01 eV/Å. The density of states (DOS) calculations were performed using the DFT + U approach, with U values of 3.5 and 4.0 eV applied to Fe and Co, respectively, in both HS and LS CoFe2O4 models. Following the previous study, which indicates that adsorption energies are independent of the U value, the U correction was only applied for electronic structure calculations.
The Gibbs free energy values were calculated using the following equations:
The terms in equations are defined as follows: EDFT stands for the DFT-computed energy, EZPE signifies the zero-point vibrational energy, \({\int }_{0}^{298.15}{C}_{v}{dT}\) corresponds to the integrated heat capacity and S is the entropy. The constants ℎ (Planck’s constant) and kB (Boltzmann constant) are used alongside the vibrational frequency. For entropy, only vibrational contributions were considered due to the insignificance of translational and rotational terms.
Data availability
The data related to the figures in the paper are provided as Excel files in Source data. Source data are provided with this paper.
References
Wang, X. et al. Light-induced ferromagnetism in moiré superlattices. Nature 604, 468–473 (2022).
Shim, J. & Sung, Y.-E. Unlocking iron spin states for oxygen reduction: electrocatalysis. Nat. Catal. 8, 417–419 (2025).
Cheng, O. H.-C., Son, D. H. & Sheldon, M. Light-induced magnetism in plasmonic gold nanoparticles. Nat. Photonics. 14, 365–368 (2020).
Bhaskar, M. K. et al. Experimental demonstration of memory-enhanced quantum communication. Nature 580, 60–64 (2020).
Marchukov, O. V., Volosniev, A. G., Valiente, M., Petrosyan, D. & Zinner, N. Quantum spin transistor with a Heisenberg spin chain. Nat. Commun. 7, 13070 (2016).
Yan, S. et al. Nonlocally sensing the magnetic states of nanoscale antiferromagnets with an atomic spin sensor. Sci. Adv. 3, e1603137 (2017).
Zhao, K.-M. et al. Identifying high-spin hydroxyl-coordinated Fe3+ N4 as the active centre for acidic oxygen reduction using molecular model catalysts. Nat. Catal. 8, 422–435 (2025).
Garcés-Pineda, F. A., Blasco-Ahicart, M., Nieto-Castro, D., López, N. & Galán-Mascarós, J. R. Direct magnetic enhancement of electrocatalytic water oxidation in alkaline media. Nat. Energy 4, 519–525 (2019).
Hao, C. et al. Engineering magnetic heterostructures with synergistic regulation of charge-transfer and spin-ordering for enhanced water oxidation. Adv. Sci. 12, 2409842 (2025).
Chen, Z. et al. Molecular triplet generation enabled by adjacent metal nanoparticles. J. Am.Chem. Soc. 146, 19360–19368 (2024).
Zhang, Z. et al. Regulating spin states in oxygen electrocatalysis. Angew. Chem. Int. Ed. 62, e202216837 (2023).
Pan, L. et al. Manipulating spin polarization of titanium dioxide for efficient photocatalysis. Nat. Commun. 11, 418 (2020).
Wu, T. et al. Spin pinning effect to reconstructed oxyhydroxide layer on ferromagnetic oxides for enhanced water oxidation. Nat. Commun. 12, 3634 (2021).
Sun, Z. et al. Regulating the spin state of FeIII enhances the magnetic effect of the molecular catalysis mechanism. J. Am. Chem. Soc. 144, 8204–8213 (2022).
Wang, Z., Li, Y., Wu, C. & Tsang, S. C. E. Electric-/magnetic-field-assisted photocatalysis: mechanisms and design strategies. Joule 6, 1798–1825 (2022).
Auböck, G. & Chergui, M. Sub-50-fs photoinduced spin crossover in [Fe(bpy)3]2+. Nat. Chem. 7, 629–633 (2015).
Oppermann, M., Zinna, F., Lacour, J. & Chergui, M. Chiral control of spin-crossover dynamics in Fe (II) complexes. Nat. Chem. 14, 739–745 (2022).
Kinschel, D. et al. Femtosecond X-ray emission study of the spin cross-over dynamics in haem proteins. Nat. Commun. 11, 4145 (2020).
Linic, S., Chavez, S. & Elias, R. Flow and extraction of energy and charge carriers in hybrid plasmonic nanostructures. Nat. Mater. 20, 916–924 (2021).
Zhou, L. et al. Quantifying hot carrier and thermal contributions in plasmonic photocatalysis. Science 362, 69–72 (2018).
Li, J. et al. Plasmon-induced resonance energy transfer for solar energy conversion. Nat. Photonics. 9, 601–607 (2015).
Zhu, Z., Tang, R., Li, C., An, X. & He, L. Promises of plasmonic antenna-reactor systems in gas-phase CO2 photocatalysis. Adv. Sci. 10, 2302568 (2023).
Swearer, D. F. et al. Heterometallic antenna-reactor complexes for photocatalysis. Proc. Natl. Acad. Sci. USA 113, 8916–8920 (2016).
An, X., Stelter, D., Keyes, T. & Reinhard, B. M. Plasmonic photocatalysis of urea oxidation and visible-light fuel cells. Chem 5, 2228–2242 (2019).
Singh, S. & Khare, N. Defects/strain influenced magnetic properties and inverse of surface spin canting effect in single domain CoFe2O4 nanoparticles. Appl. Surf. Sci. 364, 783–788 (2016).
Butcher, D. P. Jr, Boulos, S. P., Murphy, C. J., Ambrosio, R. C. & Gewirth, A. A. Face-dependent shell-isolated nanoparticle enhanced Raman spectroscopy of 2, 2′-bipyridine on Au (100) and Au (111). J. Phys. Chem. C 116, 5128–5140 (2012).
Hsu, L.-Y., Ding, W. & Schatz, G. C. Plasmon-coupled resonance energy transfer. J. Phys. Chem. Lett. 8, 2357–2367 (2017).
Forcherio, G. T. et al. Single-particle insights into plasmonic hot carrier separation augmenting photoelectrochemical ethanol oxidation with photocatalytically synthesized Pd–Au bimetallic nanorods. ACS Nano. 16, 12377–12389 (2022).
Shao, T. et al. A stacked plasmonic metamaterial with strong localized electric field enables highly efficient broadband light-driven CO2 hydrogenation. Adv. Mater. 34, 2202367 (2022).
Ramalingam, V. et al. Heteroatom-mediated interactions between ruthenium single atoms and an MXene support for efficient hydrogen evolution. Adv. Mater. 31, 1903841 (2019).
Gerber, S. J. & Erasmus, E. Electronic effects of metal hexacyanoferrates: an XPS and FTIR study. Mater. Chem. Phys. 203, 73–81 (2018).
Grosvenor, A., Kobe, B., Biesinger, M. C. & McIntyre, N. Investigation of multiplet splitting of Fe 2p XPS spectra and bonding in iron compounds. Surf. Interface Anal. 36, 1564–1574 (2004).
Huang, G.-Z. et al. On–off switching of a photocatalytic overall reaction through dynamic spin-state transition in a Hofmann clathrate system. J. Am. Chem. Soc. 145, 26863–26870 (2023).
Liu, J. et al. Achieving high-performance for catalytic epoxidation of styrene with uniform magnetically separable CoFe2O4 nanoparticles. Appl. Catal. B Environ. 254, 214–222 (2019).
Sun, Q.-C., Birkel, C. S., Cao, J., Tremel, W. & Musfeldt, J. L. Spectroscopic signature of the superparamagnetic transition and surface spin disorder in CoFe2O4 nanoparticles. ACS Nano 6, 4876–4883 (2012).
Wang, Y. et al. Low-spin Fe3+ evoked by multiple defects with optimal intermediate adsorption attaining unparalleled performance in water oxidation. Adv. Mater. 36, 2412598 (2024).
Sun, K. et al. Manipulating the spin state of Co sites in metal–organic frameworks for boosting CO2 photoreduction. J. Am. Chem. Soc. 146, 3241–3249 (2024).
Hou, K. et al. Reactive high-spin iron (IV)-oxo sites through dioxygen activation in a metal-organic framework. Science 382, 547–553 (2023).
Saveleva, V. A. et al. Potential-induced spin changes in Fe/N/C electrocatalysts assessed by in situ X-ray emission spectroscopy. Angew. Chem. Int. Ed. 60, 11707–11712 (2021).
Chen, J.-M. et al. A complete high-to-low spin state transition of trivalent cobalt ion in octahedral symmetry in SrCo0.5Ru0.5O3-δ. J. Am. Chem. Soc. 136, 1514–1519 (2014).
Smeigh, A. L., Creelman, M., Mathies, R. A. & McCusker, J. K. Femtosecond time-resolved optical and Raman spectroscopy of photoinduced spin crossover: temporal resolution of low-to-high spin optical switching. J. Am. Chem. Soc. 130, 14105–14107 (2008).
Kudlacik, D. et al. Single and double electron spin-flip Raman scattering in CdSe colloidal nanoplatelets. Nano Lett. 20, 517–525 (2019).
Bressler, C. et al. Femtosecond XANES study of the light-induced spin crossover dynamics in an iron (II) complex. Science 323, 489–492 (2009).
Nozawa, S. et al. Direct probing of spin state dynamics coupled with electronic and structural modifications by picosecond time-resolved XAFS. J. Am. Chem. Soc. 132, 61–63 (2010).
Wang, X. et al. Pivotal role of reversible NiO6 geometric conversion in oxygen evolution. Nature 611, 702–708 (2022).
Zhang, X. et al. High-spin Co3+ in cobalt oxyhydroxide for efficient water oxidation. Nat. Commun. 15, 1383 (2024).
Shen, G. et al. Low-spin-state hematite with superior adsorption of anionic contaminations for water purification. Adv. Mater. 32, 1905988 (2020).
Decker, A. et al. Spectroscopic and quantum chemical studies on low-spin FeIV=O complexes: Fe–O bonding and its contributions to reactivity. J. Am. Chem. Soc. 129, 15983–15996 (2007).
Zhang, C. et al. Modulating the plasmon-mediated oxidation of p-aminothiophenol with asymmetrically grafted thiol molecules. J. Phys. Chem. Lett. 11, 7650–7656 (2020).
Bühlmann, K., Saerens, G., Vaterlaus, A. & Acremann, Y. Detection of femtosecond spin injection into a thin gold layer by time and spin resolved photoemission. Sci. Rep. 10, 12632 (2020).
Suntivich, J., May, K. J., Gasteiger, H. A., Goodenough, J. B. & Shao-Horn, Y. A perovskite oxide optimized for oxygen evolution catalysis from molecular orbital principles. Science 334, 1383–1385 (2011).
Blackwell, R. E. et al. Spin splitting of dopant edge state in magnetic zigzag graphene nanoribbons. Nature 600, 647–652 (2021).
Ren, X. et al. Spin-polarized oxygen evolution reaction under magnetic field. Nat. Commun. 12, 2608 (2021).
Gao, Y. et al. The polarization effect in surface-plasmon-induced photocatalysis on Au/TiO2 nanoparticles. Angew. Chem. Int. Ed 132, 18375–18380 (2020).
Liang, X. et al. Adsorption of gaseous ethylene via induced polarization on plasmonic photocatalyst Ag/AgCl/TiO2 and subsequent photodegradation. Appl. Catal. B Environ. 220, 356–361 (2018).
Dai, J. et al. Spin polarized Fe1-Ti pairs for highly efficient electroreduction nitrate to ammonia. Nat. Commun. 15, 88 (2024).
Wang, J. et al. Ferromagnetic Fe–TiO2 spin catalysts for enhanced ammonia electrosynthesis. Nat.Commun. 16, 1129 (2025).
Li, J. et al. Reaction kinetics and interplay of two different surface states on hematite photoanodes for water oxidation. Nat. Commun. 12, 255 (2021).
Sekar, P. et al. Decoupling plasmonic hot carrier from thermal catalysis via electrode engineering. Nano Lett. 24, 8619–8625 (2024).
Zhan, C. et al. Disentangling charge carrier from photothermal effects in plasmonic metal nanostructures. Nat. Commun. 10, 2671 (2019).
Hager, T. J. & Sadergaski, L. R. Measuring Hydroxylammonium, Nitrate, and Nitrite Concentration with Raman Spectroscopy for 238Pu Supply Program (Oak Ridge National Laboratory (ORNL), 2021).
John, J., MacFarlane, D. R. & Simonov, A. N. The why and how of NOx electroreduction to ammonia. Nat. Catal. 6, 1125–1130 (2023).
Liu, Y. et al. Facile all-optical method for in situ detection of low amounts of ammonia. Iscience 23, 101757 (2020).
Sun, T. et al. Ferromagnetic single-atom spin catalyst for boosting water splitting. Nat. Nanotechnol. 18, 763–771 (2023).
Hu, C., Dong, Y., Shi, Q., Long, R. & Xiong, Y. Catalysis under electric-/magnetic-/electromagnetic-field coupling. Chem. Soc. Rev. 54, 524–559 (2025).
Acknowledgements
We acknowledge the support from the National Natural Science Foundation of China (22302137, X.A.; W2412063, L.H.; 62422512, J.Y.; 52572255, X.A.), the National Key R&D Program of China (2024YFA1509300, X.A. and C.L.), Jiangsu Key Laboratory for Carbon-Based Functional Materials and Devices (ZZ2201), Collaborative Innovation Center of Suzhou Nano Science & Technology, and Suzhou Key Laboratory of Advanced Photonic Materials. J.Y. acknowledges financial support from the Hong Kong Polytechnic University (P0055295 and P0053682), and the Research Grants Council of the Hong Kong Special Administrative (SAR) Region, China (Project No. PolyU 15300724). The authors thank the BL11B and BL16U1 beamlines of the Shanghai Synchrotron Radiation Facility for providing the XAFS beamtime as well as Prof. Rujun Tang for measurements of the magnetic susceptibilities.
Author information
Authors and Affiliations
Contributions
L.H., X.Z., X.A., J.Y., and B.W. conceived and directed the study; X. Hu and Z.Z. carried out the preparation of materials; J.L. and J.Y. performed DFT calculations; X.Hu and S.L. carried out the photoelectrochemical measurements and the TEM characterization; L.W. and L.Z. performed the XAFS experiments; J.C., X. Huang, and B.W. performed TA spectra measurements; C.L., K.F., and Y.Z. performed the FDTD simulation; Y.X. and Q.F. performed XRD and DRIFTS measurements; B.Z. and Q.Z. performed Single particle Scattering Spectroscopy (SCA) experiments; X.C. performed ISI-XPS experiments. J.Y., B.W., and X.Z. supervised result analyses; X.A. and L.H. co-wrote the manuscript with contributions from all the authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Peng Chen and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.”
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hu, X., Liu, J., Zhu, Z. et al. Activating plasmonic catalysis through light-mediated steady-state spin modulation. Nat Commun 17, 2849 (2026). https://doi.org/10.1038/s41467-026-69577-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69577-9