Abstract
Methane dry reforming offers a promising approach for converting CH4 and CO2 into valuable syngas, while its application is restricted by catalyst deactivation and carbon deposition. Here, we report a well-designed heterostructured plasmonic photocatalyst consisting of a plasmonically active Ag core and a catalytically active Ir cage selectively grown on the vertices and edges of the Ag core, which preserves strong plasmonic absorption and enables the significant concentration of electromagnetic energy on the Ir cage, as well as the selective dissipation of that energy to generate hot carriers. This heterostructured plasmonic photocatalyst demonstrates long-term stability (300 h), high selectivity (>97%), and much enhanced H2 and CO production in light-driven methane dry reforming. We demonstrate that light-excited hot carriers, coupled with electron-enriched Ir sites, enhance the activation of CO2 and CH4, and facilitate the conversion of *CH intermediates to *CHO, thereby preventing coke formation and contributing to the high catalytic performance.
Similar content being viewed by others
Introduction
Dry reforming of methane (DRM; CH4 + CO2 = 2H2 + 2CO) offers a promising pathway to achieve a carbon-cycle economy by converting two potent greenhouse gases, methane (CH4) and carbon dioxide (CO2), into syngas, a highly versatile feedstock for Fischer-Tropsch synthesis to produce value-added lower olefins, gasoline, diesel, and other chemicals, and therefore has attracted increasing attention in recent years1,2,3,4. However, the high energy barrier for activating CH4 and CO2 typically requires DRM to operate under high-temperature conditions (700–1000 °C)5,6. This not only results in significant energy consumption and substantial CO2 emissions but also leads to the rapid deactivation of the catalysts due to coking or sintering, consequently hindering the industrialization of DRM7. Therefore, the development of DRM reaction systems that can operate at low temperatures while maintaining high activity and stability remains highly urgent yet continues to face significant challenges8,9.
Plasmonic photocatalysis, typically demonstrated on noble metals such as Au10,11, Ag12,13, and Cu14,15, provides an efficient pathway for light-driven reactions due to the unique localized surface plasmon resonance (LSPR) effect16,17,18. Noble metal based plasmonic nanostructures, such as Ag nanostars and Au nanotriangles, can localize incident light energy at sharp vertices or edges, forming high-intensity electromagnetic hot-spots19,20. These hot-spots can significantly enhance light absorption, stimulate energetic hot-carrier generation, and induce localized high-temperatures in their vicinity21. As a result, they enable light-driven reactions under considerably milder conditions compared to traditional thermocatalytic processes, using light as the sole energy input22. However, the limited catalytic reactivity of plasmonic noble metals in activating CH4 and CO2 has restricted their applications in light-driven DRM reactions. To address this issue, hybrid plasmonic metal nanostructures that integrate plasmonically active noble metal antenna nanoparticles with catalytically active reactor materials have been developed and demonstrate high photocatalytic efficiency and selectivity for DRM23. For example, Zhou et al. reported a plasmonic photocatalyst consisting of Cu nanoparticle antenna with single-Ru atomic reactor sites on surface which achieved a long-term stability (50 h) and high selectivity (>99%) for DRM under illumination at room temperature24. In light of this inspiring antenna-reactor configuration, to further enhance the photocatalytic efficiency and stability in DRM, controlling and concentrating the LSPR energy flow is critical. Hence, we hypothesized that a hybrid plasmonic antenna-reactor nanostructure that would meet this critical requirement should contain a core plasmonic metal nanoparticle as antenna with a low imaginary dielectric function at the LSPR frequency and the catalytically active metal sites as reactor featured by a high imaginary dielectric function at the LSPR frequency, in which the reactor sites should be selectively located at the hot-spots (typically like vertices or edges) of the core plasmonic antenna nanoparticle25. Such an antenna-reactor nanostructure would have following desired features. (1) The selective growth of catalytically active metal on the vertices or edges of the plasmonic metal core—resulting in a core-cage heterostructure—can effectively avoid the shielding effect commonly observed in conventional fully coated core-shell structures and thus preserve the strong LSPR absorption of the plasmonic metal core; (2) Since the energy dissipation of LSPR through the formation of energetic charge carriers is larger in the material with a larger imaginary dielectric function compared to that with a lower imaginary dielectric function, the above as-proposed antenna-reactor structure—comprising a core with a low imaginary dielectric function and a surrounding cage with a high imaginary dielectric function—would allow for the selective dissipation of the light energy stored in LSPR modes of plasmonic core to generate energetic charge carriers in the cage; (3) Due to the lightning rod effect as well as the near field coupling between cage metal and core metal, the distribution of catalytically active metal at the vertices or edges of the plasmonic metal core would promote the concentration of the electromagnetic energy and enhance the intensity of electromagnetic field at the hot-spots, thereby facilitating molecule activation and chemical transformations.
In this work, we present such an antenna-reactor plasmonic photocatalyst in form of core-cage heterostructure that contains an Ag nanocrystal core serving as the antenna and a Ir cage functioning as the reactor that is grown selectively on the vertices and edges of the Ag core (denoted as Ag@Ir-cage). This plasmonic Ag@Ir-cage heterostructure exhibits strong light absorption and enables efficient directional electron transfer from Ag to Ir. More importantly, it allows for a significant concentration of electromagnetic energy on the surface of the Ir cage and meanwhile facilitates the selective dissipation of the energy for generating hot carriers. As a result, the Ag@Ir-cage achieves a long-term stability exceeding 300 h, and demonstrates high selectivity (>97%) along with favorable H2 and CO production rates (2080 and 2145 mmol h−1 g−1) in photocatalytic methane reforming under the light irradiation with no external heating. With the aid of in-situ characterizations and theoretical calculations, we propose that the enhanced energy efficiency and high coking resistance of the Ag@Ir-cage can be attributed to the combination of the Ag-Ir heterostructure and the efficient generation of hot carriers. This study may offer valuable insights for the rational design of plasmonic photocatalysts, enabling more energy-efficient DRM and other relevant catalysis reactions through the effective utilization of LSPR energy.
Results
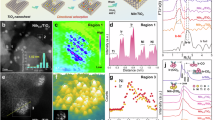
Plasmonic Ag@Ir-cages were synthesized by a one-pot thermal decomposition approach using silver nitrate (AgNO3) and iridium acetylacetonate (Ir(acac)3) as Ag and Ir sources, respectively, during which Ag nanocrystals (Ag NCs) were firstly formed and subsequently, Ir atoms were selectively grown on the vertices and edges of Ag NCs to form the Ir cage structure (Fig. 1a, preparation details please see Methods). The observation by transmission electron microscopy (TEM) and scanning transmission electron microscopy (STEM) shows the uniform dispersion of Ag@Ir-cages with an average size of 8.9 ± 1.2 nm in diameter (Fig. 1b, and Supplementary Figs. 1, 2). As displayed in Supplementary Fig. 3a, the high-resolution transmission electron microscopy (HRTEM) image confirms the Ag cores within Ag@Ir-cages have a high crystallinity, which is also demonstrated by the selected-area electron diffraction (SAED) pattern (Supplementary Fig. 3b). It should be noted that the absence of signal attributable to Ir-based species in the SAED pattern may be related to the ultrathin structure of Ir cages. Figure 1c and Supplementary Fig. 3c, d show the atomic resolution high-angle annular dark-field scanning transmission electron microscopy (AR-HAADF-STEM) images acquired from different individual Ag@Ir-cages. Due to a lager atomic number of Ir (Z = 77) than Ag (Z = 47), Ir atoms would present brighter contrasts than Ag atoms in AR-HAADF-STEM images. It is evident that Ir atoms with brighter contrasts are observed to be selectively distributed on the vertices and edges of Ag NC cores, confirming the formation of the Ir cage structure. This conclusion can also be verified by the intensity profile curves (Fig. 1d) and the atomic overlap Gaussian function fitting mapping (Fig. 1e), in which Ir atoms with higher surface intensity are enriched on edges and vertices of the Ag NC. To further confirm the formation of Ir cage structure, acid etching was used to remove Ag cores from Ag@Ir-cages. As shown in Supplementary Fig. 4, hollow Ir cages with an edge thickness of ~1 nm are presented. Figure 1f shows a HAADF-STEM image and the corresponding elemental dispersive X-ray spectroscopy (EDS) mappings of an individual Ag@Ir-cage. The spatial distribution of the Ir signal surrounding the Ag core further confirms the formation of Ir cage encapsulating the Ag core. Inductively coupled plasma optical emission spectroscopy (ICP-OES) was employed to determine the weight ratio of Ir:Ag as 1:3.6 for Ag@Ir-cages (Supplementary Table 1). It is worthy of mentioning that such unique Ag@Ir-cage heterostructures with Ir atoms selectively located on vertices and edges of Ag cores would be advantageous for synergistically enhancing plasmon-driven catalysis because the vertices and edges of plasmonic nanostructures typically act as localized hotspots capable of concentrating and coupling hot-carriers and thermal energy to drive chemical transformations26.
a Schematic illustration of the synthesis of Ag@Ir-cages. b TEM and c HAADF-STEM images of Ag@Ir-cages. d The line intensity profiles of the regions labeled by red dashed boxes in (c). The atomic distances are determined along the lattice according to the arrow direction shown in (c). e The intensity image of the region marked by the red solid square in (c). f HAADF-STEM image and the corresponding EDS elemental mappings of an individual Ag@Ir-cage.
Figure 2a shows the X-ray diffraction (XRD) patterns of Ag@Ir-cages and Ag NCs, in which characteristic diffraction peaks corresponding to metallic silver (JCPDS, 04-0783) can be clearly observed. Furthermore, a small peak at around 40.5 degree is observed in the XRD pattern of Ag@Ir-cages, which could be attributed to the diffraction of (111) plane of metallic Ir (JCPDS, 01-88-2342). X-ray absorption spectroscopy (XAS) measurements were employed to investigate the electronic structure and local atomic environment of Ir in Ag@Ir-cages. The Ir L3-edge X-ray absorption near edge structure (XANES) spectra of Ag@Ir-cages, Ir foil, and IrO2 are displayed in Fig. 2b, which shows that the intensity of the white line is ordered as IrO2 > Ag@Ir-cages > Ir foil. This indicates the average valence states of Ir in Ag@Ir-cages are between 0 and +4, suggesting the surface oxidation of Ir cages27. Extended X-ray absorption fine structure (EXAFS) was employed to study the local coordination environment of Ir atoms in Ag@Ir-cages. As shown in Fig. 2c, the Fourier-transformed (FT) Ir L3-edge EXAFS spectrum of Ag@Ir-cages exhibits a dominant peak at ~1.5 Å, which could be attributed to the first-shell Ir−O coordination, further confirming the surface oxidation of Ir cages28. Particularly, a distinct shoulder peak appears between 2.0–3.2 Å, which can be attributed to the combined scattering contributions from higher coordination shells, specifically Ir−Ag and Ir−Ir bonds29. To further resolve the atomic contributions in different coordination shells, wavelet transform (WT) analysis was performed. This approach can provide enhanced resolution by simultaneously visualizing the scattering contributions in both R-space and k-space, allowing for a better distinguishment between the Ir−O, Ir−Ag, and Ir−Ir coordination. As shown in Fig. 2d-f, the WT analysis of Ag@Ir-cages reveals a distinct intensity maximum around 7 Å−1, which could be ascribed to the Ir−O scattering, aligning with the FT-EXAFS results. Furthermore, the WT analysis identifies a scattering signature at R ≈ 2.56 Å for Ag@Ir-cages, which can be attributed to the combined action of Ir−Ir and Ir−Ag coordination. This attribution is grounded in the observation that the peak demonstrates substantial broadening relative to the Ir−Ir scattering peak of Ir metal, which suggests the existence of multiple scattering paths. The presence of Ir−Ag bonds demonstrates the strong electronic interaction between Ag core and Ir cage in Ag@Ir-cages. To further elucidate the quantitative coordination environment of Ir atoms in Ag@Ir-cages, the Ir L3-edge EXAFS spectrum of Ag@Ir-cages was analyzed through multi-shell fitting (Fig. 2g, Supplementary Fig. 5 and Supplementary Table 3). The central Ir atoms exhibit primary coordination with 2.36 oxygen atoms in the first shell with an average bond distance of 2.05 Å. Notably, the second coordination shell demonstrates heterometallic interactions, where each Ir atom coordinates with 1.85 Ir and 1.18 Ag atoms, corresponding to the average bond lengths of 2.73 and 3.09 Å for Ir−Ir and Ir−Ag, respectively. These atomic-level coordination metrics confirm the presence of Ir−Ag coordination motifs, indicative of the interfacial interaction between Ag and Ir.
a XRD patterns of Ag@Ir-cages and Ag NCs. b Ir L3-edge XANES spectra of Ag@Ir-cages and the references. c FT k3-weighted Ir L3-edge EXAFS spectra of Ag@Ir-cages and the references; WT-EXAFS plots of (d) Ag@Ir-cages, (e) IrO2, and (f) Ir foil. g EXAFS fitting curve of Ag@Ir-cages at Ir L3-edge. h XPS spectra of Ag 3d measured under dark and light conditions for Ag@Ir-cages and Ag NCs. i Charge density difference analysis at the interface of Ag and Ir in Ag@Ir-cages. Gray and light-yellow balls correspond to Ag and Ir atoms, respectively.
To further confirm the electronic interaction between Ag and Ir in Ag@Ir-cages, X-ray photoelectron spectroscopy (XPS) was performed. As shown in Fig. 2h and Supplementary Fig. 6, the Ag 3d spectrum of Ag NCs presents two peaks at around 367.7 and 373.7 eV, corresponding to the binding energies for Ag3d5/2 and Ag3d3/2 of metallic Ag30. Compared with Ag NCs, the Ag 3d signal of Ag@Ir-cages exhibits a shift toward higher binding energies. This result implies the electron transfer from Ag core to Ir cage at the interface, highlighting the interfacial electronic interaction between them. The electron transfer from Ag to Ir may be related to the higher electronegativity of Ir compared to Ag. This interfacial electronic interaction would be beneficial for the separation and transfer of charge carriers generated via the LSPR excitation of Ag. Theoretical calculations on charge density difference also demonstrate that, there is a significant electron redistribution at the interface of Ag and Ir (Fig. 2i), in which the electron density on Ag atoms exhibits a localized dissipation, while Ir atoms show an obvious electron enrichment feature, suggesting a directional charge migration from Ag to Ir. This is in good line with the XPS results.
As shown in Fig. 3a, Ag NCs exhibit a strong absorption peak ranging from 300 to 600 nm, which can be attributed to the LSPR excitation of Ag. Meanwhile, Ir-cages display a broad absorption in the visible and UV ranges, which is dominated by the interband transitions31,32,33. Notably, Ag@Ir-cages combine the LSPR absorption from the Ag core and the interband absorption from the Ir cage, thereby demonstrating significantly enhanced light absorption spanning both the UV and visible ranges. Meanwhile, compared with that of Ag NCs, the LSPR peak of Ag@Ir-cages is slightly shifted and broadened, which is probably due to the change in the dielectric environment of Ag cores caused by the coating of Ir cages25,34. Since the surface plasmons on Ag NCs could decay radiatively by emission of luminescence, the photoluminescence (PL) measurements were conducted to investigate the hot-electron transfer behavior within Ag@Ir-cages. As shown in Fig. 3b, Ag NCs exhibit a strong PL spectrum under 400-nm light excitation. By comparison, the PL signal of Ag@Ir-cages is much quenched. This observation implies that hot electrons generated by plasmonic excitation of Ag can be transferred from Ag core to Ir cage, thereby suppressing the recombination of hot carriers and resulting in diminished light emission.
a UV-visible absorption spectra of Ag@Ir-cages, Ag NCs and Ir-cages. b PL spectra of Ag@Ir-cages and Ag NCs. 2D pseudo-color maps of TA spectra of (c) Ag NCs and (d) Ag@Ir-cages recorded after the excitation of a 400 nm laser. e Normalized decay kinetic curves probed at 650 nm for Ag NCs and Ag@Ir-cages. f XPS spectra of Ir 4f measured under dark and light conditions for Ag@Ir-cages. g Simulated electromagnetic field distribution and enhancement effect over Ag NCs (top), Ir-cages (middle) and Ag@Ir-cages (bottom) under 400-nm light excitation. The color scale shows the electromagnetic field enhancement.
To evaluate the hot-electron-transfer behavior more accurately, femtosecond time-resolved transient (TA) absorption measurements were further performed, in which a 400-nm laser pump was used to ensure the LSPR excitation of Ag. As illustrated in Fig. 3c, d and Supplementary Figs. 7 and 8, the TA spectra of Ag@Ir-cages and Ag NCs display exhibit similar characteristics, including a strong and broad plasmon-bleaching (PB) signal ranging from 460 to 640 nm, as well as two exciton absorption signals located at λ = 450 nm and λ = 650 nm, respectively35. These observations suggest that hot carriers are generated within Ag@Ir-cages and Ag NCs through plasmonic excitation of Ag under 400-nm light irradiation36. Fig. 3e presents the kinetics probed at 650 nm (corresponding to the exciton absorption) for Ag@Ir-cages, which can be fitted by a bi-exponential function. The τ1 time constant is attributed to electron-phonon scattering that relates to the generation rate of local heat, whereas the τ2 time constant originates from the process of charge recombination that is indicative of the lifetime of hot electrons37. Compared with Ag NCs (τ1 = 5.56 ps, and τ2 = 371.19 ps), Ag@Ir-cages exhibit significantly longer τ1 (9.01 ps) and τ2 (882.99 ps). The prolonged τ1 indicates a reduced photothermal conversion efficiency, reflecting that Ag@Ir-cages enable the preferential conversion of light energy harvested through LSPR excitation to hot carriers. Meanwhile, the extended τ2 suggests that the generated hot electrons can be effectively transferred from Ag to Ir within Ag@Ir-cages and therefore affords an increased lifetime.
To shed light on the concentration behavior of electromagnetic energy over Ag NCs, Ir-cages and Ag@Ir-cages, we performed the three-dimensional finite-difference time-domain (FDTD) simulations. The models used for the simulations were built according to the TEM observations. Figure 3g depict the comparative electromagnetic field profiles under 400 nm illumination, where both Ag NC and Ag@Ir-cage exhibit significant near-field enhancement, while the electromagnetic field intensity of Ir-cage is quite weak. As for the Ag NC, photoexcitation induces collective oscillation of conduction electrons, generating localized surface plasmon resonances that amplify the electromagnetic field intensity. Notably, the maximum field enhancement occurs at the vertices of the Ag NC, a characteristic manifestation of the lightning rod effect arising from geometric field focusing. In stark contrast, Ag@Ir-cage demonstrates significantly augmented electromagnetic field intensity, particularly at edge and vertex regions, leading to more plasmonic hotspot generation. Specifically, the electromagnetic field intensity at the vertices of the Ag@Ir-cage is more than 3 and 6 times those of the Ag NC and the Ir-cage, respectively. This significant amplification in electromagnetic field originates from the enhanced lightning rod effect and the near field coupling between Ag and Ir38, where the selective growth of Ir on the edges and vertices of Ag NCs leads to the formation of sharper tips on the nanoparticle and modifies the localized dielectric environment of Ag.
The LSPR excitation-induced hot carrier transfer behavior within Ag@Ir-cage heterostructures was further dynamically investigated by in-situ XPS characterization under light irradiation (380 nm < λ < 800 nm). As shown in Supplementary Fig. 9, the Ag 3d peaks of Ag NCs exhibit a shift toward lower binding energy under light irradiation. This phenomenon can be attributed to the dynamic enrichment of hot electrons on the surface induced by LSPR excitation. The in-situ Ag 3d XPS spectra of Ag@Ir-cages are presented in Fig. 2h, which demonstrate a shift of the signal to lower binding energy upon light irradiation. This finding further corroborates the surface electron enrichment resulting from LSPR excitation of Ag cores. More importantly, the light irradiation also leads to the negative shift of Ir 4f XPS peaks of Ag@Ir-cages (Fig. 3f). This potentially indicates the occurrence of hot-electron transfer from Ag core to Ir cage. Based on the above experimental results and theoretical simulations, we propose that the growth of the Ir cage surrounding the Ag core introduces a plasmon decay pathway through the Ir cage. Furthermore, a significant portion of the electromagnetic energy concentrated within Ag@Ir-cages is dissipated through the thin Ir cage via the generation of energetic hot-carriers, in which hot-electrons are transferred from Ag core to Ir cage.
The performance of Ag@Ir-cages for plasmonic photocatalytic DRM was evaluated under the focused irradiation (18.5 W cm−2) from a 300 W Xe lamp without external heating. For comparison, the performance of Ir-cages and Ag NCs was also tested. Before the measurements, MgAl layered double hydroxide (MgAl-LDH), which is inert for DRM under the present reaction conditions, was employed as a support to disperse the catalysts. As depicted in Fig. 4a, Ir-cages exhibit substantial deactivation of catalytic activity during the 30 h measurement period. This is evidenced by a pronounced decline in H2 production rate from 1741 to 1253 mmol g−1 h−1 and a corresponding decrease in CO production rate from 1785 to 1474 mmol g−1 h−1. Additionally, there is a notable reduction in the H2/CO ratio (selectivity), which decreases from 0.97 to 0.85. The observed reduction in activity agrees with the evidence of coke deposition, as Raman analysis confirms the significant formation of graphitic carbon on the surface of the post-reaction Ir-cages (Fig. 4b). Ag NCs are found to be inert under light irradiation toward DRM, with no detectable products formed (Supplementary Fig. 10), likely attributed to their limited ability to activate CH4. In stark contrast, Ag@Ir-cages demonstrate much higher photocatalytic activity and stability for DRM. Specifically, they achieve initial production rates of 2080 mmol g−1 h−1 for H2 and 2145 mmol g−1 h−1 for CO, respectively, with a H2/CO ratio of 0.97. After 300 h of reaction, only a 12% reduction in the H2 production rate is observed, which could be potentially attributed to the gradually reduced light intensity caused by the Xe lamp aging during the long-term operation (Supplementary Fig. 11). The markedly improved photocatalytic stability of Ag@Ir-cages in comparison to Ir-cages might be related to the Ag-Ir heterostructure and the LSPR induced hot carrier generation, which effectively inhibits the coke deposition: Raman spectroscopy analysis verifies the absence of coke formation on Ag@Ir-cages following 300 h of reaction (Fig. 4b), and meanwhile a carbon balance close to unity is demonstrated (Supplementary Fig. 12). Characterization on the post-reaction Ag@Ir-cages confirms the reduction of Ir species to metallic Ir during the DRM reaction (Supplementary Figs. 13-15). In addition, it should be noted that the H2/CO ratio being slightly less than 1 suggests that the DRM process may involve reverse water-gas shift reactions and other side reactions. Supplementary Table 4 compares the catalytic performance of Ag@Ir-cages with that of some recently reported state-of-the-art catalysts. The data indicate that Ag@Ir-cages exhibit competitive catalytic performance among them.
a Long-term stability and selectivity of Ag@Ir-cages for DRM under light illumination (18.5 W cm−2, Xe lamp). The performance of Ir-cages is also provided for comparison, which was measured under the identical reaction conditions to Ag@Ir-cages. b Raman spectra of Ir-cages and Ag@Ir-cages collected following the 30-h and 300-h DRM reactions, respectively. The G and D bands are assigned to the deposited graphitic carbon and active carbon, respectively. c H2 production rate and d CH4 conversion rate for DRM over Ag@Ir-cages as a function of light intensity. e Light intensity dependence on the reaction selectivity over Ag@Ir-cages. f Dependence of catalyst surface temperature on light intensity over Ag@Ir-cages. g Production rates of H2 and CO in DRM over Ag@Ir-cages under both light irradiation and external heating alone. h Normalized absorption spectrum of Ag@Ir-cages plotted as a function of the wavelength-dependent DRM reaction rate. In the measurements, due to the intensity of monochromatic light is too low to initiate the reaction, an external heating (550 °C) was employed. Reaction conditions: 5 mg catalyst, CH4/CO2 = 1/1, a total flow rate of 20 mL min−1. Vertical error bars represent the standard deviation (s.d.) from three independent experiments; horizontal error bars represent the wavelength range.
In the plasmonic photocatalytic process, both photoinduced heating and photoinduced hot-carriers might contribute to surface chemical reactions. To clarify the role of light energy toward DRM, a series of experiments were performed. Firstly, we measured the catalytic activity of Ag@Ir-cages at varying light intensities. As shown in Fig. 4c, d, both the production rate of H2 and the conversion rate of CH4 display a nearly linear relationship with the light intensity. This characteristic is a typical indicator of a photocatalytic process governed by photogenerated charge carriers39,40. However, if the reaction were to proceed solely through photocatalysis without any thermal contribution, this linear relationship would extrapolate to the origin, while such feature is absence in the present reaction system. This implies that, besides the photocatalysis process governing the reaction rate, there is an activation step associated with thermal process in the reaction41. Fig. 4e illustrates the selectivity of DRM over Ag@Ir-cages as a function of light intensity. The selectivity exhibits a monotonic increase with light intensity and approaches approximately 100% at light intensities reaching approximately 18.5 W cm−2. This behavior supports the hot-carrier-mediated mechanism24. Then, we evaluate and compare the thermocatalytic and photocatalytic activities under identical surface temperature conditions, where the temperatures are respectively achieved through external heating and light illumination. Under light illumination of 18.5 W cm−2 without external heating (the surface temperature of the catalyst was measured to be around 425 °C (Fig. 4f)), the photocatalytic production rates of H2 and CO achieve 2080 and 2145 mmol g−1 h−1, respectively. In contrast, as depicted in Fig. 4g, the thermocatalytic DRM process is scarcely initiated at 425 °C and further increasing the reaction temperature even to 580 °C leads to only a negligible improvement in reaction rate. Based on the above results, we conclude that the LSPR-induced hot-carrier generation is the predominant mechanism in light-driven DRM over Ag@Ir-cages, while the photothermal conversion induced local high temperature is also necessary for initiating the reaction. Figure 4h shows the variation of the normalized photocatalytic rate over Ag@Ir-cages as a function of light wavelength. The one-to-one correspondence between the normalized photocatalytic rate and the LSPR absorption of Ag@Ir-cages provides additional evidence supporting that LSPR excitation induced hot-carriers are responsible for the observed catalytic activity of Ag@Ir-cages toward DRM.
To illustrate the CO2 adsorption and activation capacity of MgAl-LDH supported Ag@Ir-cages, CO2 temperature-programmed desorption (CO2-TPD) measurements were performed. As depicted in the CO2-TPD spectra presented in Fig. 5a, both MgAl-LDH supported Ag@Ir-cages and MgAl-LDH supported Ir-cages demonstrate three distinct desorption peaks with the temperature ranges of 50–150 °C, 200–350 °C and 400–650 °C, corresponding to the weak, medium, and strong basic sites on their surface, respectively. It is noteworthy that the strong basic site peak of MgAl-LDH supported Ag@Ir-cages is shifted to a higher temperature by ~13 °C as compared with that of MgAl-LDH supported Ir-cages. This result indicates that MgAl-LDH supported Ag@Ir-cages exhibit a stronger affinity for CO2 adsorption than MgAl-LDH supported Ir-cages. This is likely due to that the electron-enriched Ir atoms on the surface of Ag@Ir-cages provide stronger Lewis-base sites for CO2 adsorption42,43. The strong adsorption of CO2 would facilitate its activation, thereby contributing to the improvement in DRM performance.
a CO2-TPD spectra of MgAl-LDH supported Ag@Ir-cages and Ir-cages. b In-situ FTIR spectra of MgAl-LDH supported Ag@Ir-cages collected at different irradiation time. c Enlarged in-situ FTIR spectra of MgAl-LDH supported Ag@Ir-cages corresponding to the region highlighted in blue in (b). d In-situ FTIR peak intensity for *CHxO intermediates over MgAl-LDH supported Ag@Ir-cages as a function of irradiation time. Reaction pathways of DRM on the surface of (e, f) Ag@Ir(111) and (g, h) Ir(111). Yellow, blue, red, gray and white balls correspond to Ag, Ir, O, C and H atoms, respectively.
To get an in-depth understanding of the DRM process, in-situ Fourier transform infrared (FTIR) measurements were carried out. As shown in Fig. 5b, c and Supplementary Fig. 16, the bands centered at ca. 3011 and 2347 cm−1 can be ascribed to the C−H deformation vibration of CH4 and C−O stretching vibrations of CO2, respectively43,44. Upon exposure to irradiation, a new absorption band corresponding to *CHxO species emerges at 1072 cm−1 45,46 and its intensity increases progressively with prolonged irradiation time (Fig. 5d). This finding indicates the dissociation of CH4 into *CHxO species. It is noteworthy that the reaction pathway involving *CHxO species can effectively mitigate coking, thereby enhancing the stability. This is evidenced by the stable catalytic performance of Ag@Ir-cages (Fig. 4a) and the absence of detectable coke formation after a 300 h activity measurement (Fig. 4b). Furthermore, a band at 2160 cm−1 is also observed upon irradiation (Fig. 5b, c), which can be attributed to the stretching vibration of linearly bound CO, suggesting the formation of CO in the reaction47,48. No characteristic peaks belong to *COOH species are detected during the measurement. In addition, in the in-situ FTIR spectra of Ir-cages (Supplementary Fig. 17), a band centered at 1624 cm−1 appears and grows gradually under light irradiation, which could be attributed to the stretching vibrations of C=C bonds49,50,51, suggesting the formation of carbon species and the fast coke deposition on the catalyst surface. Concurrently, no signals at 1072 cm−1, characteristic of *CHxO species, are detected. These observations are consistent with the significant coke formation observed on Ir-cages during the DRM reaction (Fig. 4b) and their rapid deactivation in catalytic performance (Fig. 4a). Based on the above results, it can be reasonably inferred that the DRM pathway over Ag@Ir-cages proceeds through the following steps: 1) CH4 and CO2 are dissociated into (*CHx + *H) and (*CO + *O), respectively; 2) *CHx subsequently reacts with *O to form *CHxO, which further dissociates into (*CO + *H); 3) *CO desorbs to produce CO, while *H species couple to form H2.
Density functional theory (DFT) calculations were further conducted to offer qualitative insights into the high photocatalytic DRM performance of Ag@Ir-cages, although a complete description of the real catalytic conditions is beyond the present models. Two models, including Ir(111) and Ag-supported Ir(111) (denoted as Ag@Ir(111)), were built based on experimental characterization results (Supplementary Fig. 18) and then the energetics of the elementary steps of the DRM reaction were calculated on the surface of both models (Fig. 5e-h). As shown in Fig. 5e, on the surface of Ag@Ir(111), the change of Gibbs free energy (ΔG) required for the oxidation of *CH to *CHO (1.24 eV) is much less than that for *CH cleavage to *C (1.86 eV). This suggests the *CH → *CHO pathway dominates on Ag@Ir(111), which is consistent with the in-situ FTIR results that shows evident signals corresponding to *CHxO species during the DRM reaction (Fig. 5b, d). In contrast, *CH would prefer to be converted into *C rather than *CHO on the surface of Ir(111), as evidenced by the greater energetic favorability of the *CH → *C pathway relative to the *CH → *CHO pathway (Fig. 5g). Furthermore, the subsequent oxidation of *C to *CO on Ir(111) is severely impeded due to a high thermodynamic barrier of 3.24 eV, suggesting the occurrence of carbon deposition. These findings are highly consistent with the in-situ FTIR results, which show evident signals corresponding to carbon species but no detectable signals for *CHxO species, thereby providing a rational explanation for the significant coke deposition and the rapid deactivation observed over Ir-cages during the DRM reaction (Fig. 4a, b). In addition, regarding the dissociation of CO2 on both Ag@Ir(111) and Ir(111), the reaction pathway *CO2 → (*CO + *O) → *CO is energetically more favorable compared to the pathway *CO2 → *COOH → (*CO + *OH) (Fig. 5f, h). This indicates that the former pathway is predominant on both surfaces, which is also supported by the in-situ FTIR results. Considering the more significant reduction in free energy, Ag@Ir(111) demonstrates a greater propensity for driving the conversion of *CO2 to *CO compared to Ir(111). This provides an additional contribution to the significantly enhanced catalytic performance observed for Ag@Ir-cages over Ir-cages (Fig. 4a).
Discussion
In summary, we report a well-designed Ag@Ir-cage plasmonic photocatalyst consisting of a plasmonically active Ag core and a catalytically active Ir cage that is grown selectively on the vertices and edges of the Ag core. These plasmonic Ag@Ir-cage heterostructures not only retain significant LSPR absorption but also allow for a significant concentration of electromagnetic energy on the surface of the Ir cage and meanwhile, enables the selective dissipation of that energy for generating hot carriers to drive chemical transformations. In photocatalytic DRM applications, these Ag@Ir-cages effectively prevent coke formation and exhibit exceptional stability (300 h), high product selectivity (>97%), and significantly enhanced production of H2 and CO. Such high catalytic performance could be attributed to the efficient generation of hot carriers and the unique Ag-Ir heterostructure within Ag@Ir-cages. We believe that the well-defined plasmonic photocatalyst model presented in this work will motivate the rational design of other heterostructured plasmonic photocatalysts to control energy flow toward more-efficient DRM and other related photocatalytic applications.
Methods
Chemicals
Sliver nitrate (AgNO3) and butylamine were purchased from Sinopharm Chemical Reagent. Iridium (III) acetylacetonate (Ir(acac)3, 97%) and triphenylphosphine (TPP, 99%) were purchased from Sigma-Aldrich Technology Co., Ltd. Oleylamine (OAm, 80–90%), magnesium (II) nitrate hexahydrate (Mg(NO3)2·6H2O), aluminum nitrate nonahydrate (Al(NO3)3·9H2O) and formamide (CH3NO, 99%) were purchased from Shanghai Aladdin Bio-Chem Technology Co., Ltd. Sodium hydroxide (NaOH, 97%), hexane and ethanol were purchased from Shanghai Macklin Biochemical Technology Co. Ltd. All chemicals were used as received without further purifications. Deionized (DI) water (18.2 MΩ·cm at 25 °C) was used in all experiments.
Synthesis of Ag@Ir-cages
In a typical process, 34 mg of AgNO3, 20 mg of Ir(acac)3 and 52 mg of TPP were sequentially added to a three-neck flask containing 6 mL of OAm and heated to 120 °C for 10 min under vacuum. Subsequently, the flask was purged with N2 and the mixture solution was heated to 170 °C for 60 min, after which the solution was further heated to 210 °C for 240 min. After being cooled to room temperature, the product was collected by centrifugation and washed several times with hexane and ethanol for further characterizations.
Synthesis of Ag NCs
Typically, 34 mg of AgNO3 and 52 mg of TPP were sequentially added to a three-neck flask containing 6 mL of OAm and heated to 120 °C for 10 min under vacuum. Subsequently, the flask was purged with N2 and the mixture solution was heated to 170 °C for 60 min, after which the solution was further heated to 210 °C for 240 min. After being cooled to room temperature, the product was collected by centrifugation and washed several times with hexane and ethanol for further characterizations.
Synthesis of Ir-cages
Typically, the as-prepared Ag@Ir-cages were dispersed in 20 mL of 0.1 mol/L HNO3 solution via sonication, followed by stirring at room temperature for three days. Subsequently, the Ir-cages were collected by centrifugation and thoroughly washed with DI water for further use.
Synthesis of MgAl-LDH
MgAl-LDH was prepared using a modified method reported previously52. In a typical process, 19 mg of Mg(NO3)2·6H2O and 9 mg of Al(NO3)3·9H2O were added to a beaker containing 20 mL of formamide solution (23 vol.%) and heated to 80 °C for 10 min. During this period, 1.0 M NaOH solution was gradually added to the flask to maintain the pH of about 10 (totally, 12.5 mL of NaOH solution was added). The product was collected by centrifugation, washed repeatedly with DI water and ethanol, and kept in 10 mL of ethanol for subsequent use.
Preparation of MgAl-LDH supported Ag@Ir-cages
The as-prepared Ag@Ir-cage and MgAl-LDH were dispersed in 25 mL of n-butylamine to form a homogeneous suspension. After being stirred at room temperature for 24 h, the catalyst was collected by centrifugation and washed several times with hexane and ethanol to remove excess n-butylamine. Finally, the catalyst was dried overnight at 80 °C under vacuum conditions. The loading amount of Ag and Ir were determined to be 15.4 and 4.1 wt%, respectively, by inductively coupled plasma optical emission spectroscopy (ICP-OES).
Preparation of MgAl-LDH supported Ir-cages
The as-prepared Ir-cages and MgAl-LDH were dispersed in 25 mL of n-butylamine to form a homogeneous suspension. After being stirred at room temperature for 24 h, the catalyst was collected by centrifugation and washed several times with hexane and ethanol to remove excess n-butylamine. Finally, the catalyst was dried overnight at 80 °C under vacuum conditions. The loading amount of Ir was determined to be 5.0 wt% by ICP-OES.
Preparation of MgAl-LDH supported Ag NCs
The as-prepared Ag NCs and MgAl-LDH were dispersed in 25 mL of n-butylamine to form a homogeneous suspension. After being stirred at room temperature for 24 h, the catalyst was collected by centrifugation and washed several times with hexane and ethanol to remove excess n-butylamine. Finally, the catalyst was dried overnight at 80 °C under vacuum conditions.
Characterizations
Powder X-ray diffraction (XRD) was performed on a Bruker D8 Discover X-ray diffractometer. A JEOL-2100F transmission electron microscope (TEM) and a JEOL JEM-ARM200F scanning transmission electron microscope (STEM) were employed to conduct TEM imaging and energy-dispersive X-ray spectroscopy (EDS) elemental mapping on the samples. The surface intensity image was generated by ImageJ (version 1.53k). In-situ X-ray photoelectron spectroscopy (XPS) analysis was performed under light irradiation (380 < λ < 800 nm) and in the dark using a Thermo Fisher Scientific ESCALAB 250Xi photoelectron spectrometer equipped with a 300 W Xe lamp as simulated solar light source. During the XPS measurements conducted under light irradiation, the XPS spectra of the sample were recorded while the sample was simultaneously exposed to the visible light irradiation (Supplementary Fig. 19a). During the XPS measurements performed in the dark, the XPS spectra were collected without the visible light irradiation on the sample (Supplementary Fig. 19b). The UV-visible absorption spectra were measured on an UV-Vis spectrophotometer (UV-1800; Shimadzu, Japan). The steady-state photoluminescence (PL) spectra were obtained at room temperature using a FLSP920 fluorescence spectrometer (Edinburgh Instruments Ltd, UK). ICP-OES measurements were conducted on an Agilent 5110 system ICP-OES analyzer. Raman spectra were recorded on a LabRAM HR Evolution (HORIBA) spectrometer. Thermogravimetric Analysis (TGA) was performed on a METTLER TOLEDO thermal analysis system (TGA/DSC 3+ ). X-ray absorption fine structure (XAFS) measurements were conducted at the BL14W1 beamline of the Shanghai Synchrotron Radiation Facility (SSRF). The XAFS data were analyzed with the ATHENA and ARTEMIS code according to the standard procedures, including normalization, background removal and Fourier transformation.
Catalytic performance measurements
The methane dry reforming (DRM) performance was measured at ambient pressure in a flow reactor with a quartz window at the top of the reactor for light exposure (Supplementary Figs. 20 and 21). Prior to each measurement, 5.0 mg of the MgAl-LDH supported catalyst was uniformly placed in the sample holder of the reactor with a diameter of 6 mm. Then, reaction gas (50% CH4/50% CO2) was continuously fed into the reactor at a flow rate of 20 mL min−1. A 300 W Xe lamp (MC-PF300C, Merry Change light) equipped with a focusing lens was used as a light source to drive the DRM reaction. The quantities of CO and H2 were determined by an on-line gas chromatography (GC, Scion 456i, Techcomp Co., LTD) equipped with thermal conductivity (TCD) and flame ionization detectors. The surface temperatures of the catalyst under illumination were recorded using a K-type thermocouple. For wavelength-dependent experiments, the reaction temperature was maintained at 550 °C using a temperature controller and the monochromatic light of 380, 405, 420, 480, 532, 600, and 660 nm wavelengths, with a bandwidth of ±10 nm, were obtained using a set of band pass filters.
The production rates of H2 (\({r}_{{H}_{2}}\)) and CO (\({r}_{{CO}}\)) and the reaction rates of CH4 (\({r}_{{{CH}}_{4}}\)) and CO2 (\({r}_{{{CO}}_{2}}\)) were defined as follows:
where, \({C}_{C{H}_{4}}^{{in}}\) and \({C}_{C{O}_{2}}^{{in}}\) are the concentrations (vol%) of CH4 and CO2 in the stream of inlet, respectively; \({C}_{C{H}_{4}}^{{out}}\) and \({C}_{C{O}_{2}}^{{out}}\) are the concentrations (vol%) of CH4 and CO2 in the stream of outlet, respectively; \({F}_{{out}}\) and \({F}_{{in}}\) indicate the gas flow rates (ml min−1) of inlet and outlet, respectively; \({V}_{m}\) means the gas molar volume under standard conditions; and m represents the mass of catalyst.
The reaction selectivity was defined as follows:
The carbon balance rate was calculated by the Eq. (6):
CO2-temperature-programmed desorption measurements (CO2-TPD)
CO2-TPD experiments were conducted on a temperature-programmed desorption instrument (JW-HX100, Beijing JWGB Instrument Co., Ltd.) with a TCD detector. Firstly, 50 mg sample was treated under a He atmosphere at a flow rate of 30 mL min−1 at 400 °C for 1 h. After the sample was cooled to 50 °C, the gas was switched to 10% CO2/He for 1 h and then the sample was purged with pure He for 1 h at the same temperature. Finally, the sample was heated from 50 to 700 °C at 10 °C min−1 under a He atmosphere at a flow rate of 30 mL min−1 and the gas signal was detected by a TCD detector.
In-situ Fourier transform infrared measurements (In-situ FTIR)
In-situ FTIR were conducted on a Bruker Invenio-S FTIR spectrometer equipped with a Mercury Cadmium Telluride (MCT) detector. Xe lamp was employed as the light source. Before the measurement, the fresh catalyst was first purged with N2 at 600 °C for 60 min to remove impurities. Then, the fresh catalyst was placed in a 50% CH4/50% CO2 gas stream to collect FTIR spectra for CO2 and CH4 adsorption. After saturation of adsorption, the catalyst was exposed to illumination and the FTIR spectra were recorded at 0, 1, 3, 5, 10, 20, 30, 40, 50, and 60 min. The spectrum detected before illumination was used as the background.
Transient absorption (TA) measurements
Femtosecond transient absorption measurements were performed following a similar procedure reported previously53. A femtosecond laser (1030 nm wavelength, 50 kHz, pulse width <190 fs, Pharos, Light Conversion) was used as the source beam, 90% of which was converted by an optical parametric amplifier (OPA, Orpheus-HP, Light Conversion) to a 400 nm pump excitation (pulse duration of ~120 fs), while the rest was served as the probe beam for transient absorption measurements. In the TA spectrometer TA100 (Time-Tech Spectra), both the pump beam and the probe beam were fed into the optical bench. At 380 to 720 nm, the probe beam was focused onto the sapphire crystal, producing continuous white light. As for the pump beam, the frequency of the chopper was set to 2.5 kHz and the energy attenuation of the pump pulse at 400 nm excitation was about 64 μJ/cm2. The time delay between pump-probe pulses was regulated by a computer-controlled optical delay stage. Both pump and probe pulses were focused and overlapped on the sample layer, which was spin-coated on a 1 mm thick quartz glass. Transient absorption spectra were obtained by the software by collecting and analyzing the white light spectrum of the probe passing through the sample with and without the pump beam.
Finite-difference time-domain (FDTD) simulations
Computational simulations were performed by using the FDTD method with perfectly matched layer boundary conditions. In the simulations, a plane wave with a wavelength of 400 nm was incident perpendicular to the surface of the nanostructures. In addition, the external region was set as an absorbing boundary condition to avoid boundary reflection. Mesh with sufficient accuracy was applied to ensure that reliable computational results were obtained. Finally, the electric field distribution of the nanoparticles under light irradiation was calculated by using the frequency domain solver.
Density functional theory (DFT) calculations
Density functional theory with D3 dispersion correction (DFT + D3) calculations were carried out using the Vienna Ab-initio Software Package (VASP) with Perdew-Burke-Emzerhof (PBE) exchange-correlation functional, following a protocol similar to that reported previously54. According to our experimental observations, Ag (111) and Ir (111) surfaces were constructed using 4×4×1 supercells and they were combined to model the Ag-Ir heterostructure, where Ir atoms served as the surface active sites. For comparison, a pristine Ir (111) surface was considered as the reference catalytic surface. The Brillouin zone was sampled using a 4 × 4 × 1 Monkhorst-Pack k-point mesh. Adsorption configurations of reaction intermediates were optimized on the constructed surfaces to evaluate adsorption energies. A vacuum layer of 20 Å was introduced along the c-direction to eliminate interactions between periodic images. The plane-wave cutoff energy was set to 400 eV and the convergence criteria for energy and force were 10−5 eV and 10−2 eV Å−1, respectively.
The adsorption energy (Eads) was calculated using the following equation:
where Esubstrate+adsorbate is the total energy of the (adsorbate+substrate) system, and Esubstrate and Eadsorbate represent the energies of the clean surface and isolated adsorbate, respectively.
The Gibbs free energy change (ΔG) for each elementary reaction step was determined by:
Where ΔE is the adsorption energy, ΔEZPE and ΔS denote the zero-point energy and entropy corrections, respectively, and T is the temperature (300 K).
The atomic coordinate information is available in Supplementary Data 1.
Data availability
All data generated in this study are provided in the article and its Supplementary Information. Source data are provided with this paper.
References
Henrici-Olivé, G. & Olivé, S. The Fischer-Tropsch synthesis: molecular weight distribution of primary products and reaction mechanism. Angew. Chem. Int. Ed. 15, 136–141 (2003).
Chen, Y. et al. Carbon-based catalysts for Fischer-Tropsch synthesis. Chem. Soc. Rev. 50, 2337–2366 (2021).
Li, X., Wang, C. & Tang, J. Methane transformation by photocatalysis. Nat. Rev. Mater. 7, 617–632 (2022).
Ding, X. et al. Engineering a nickel-oxygen vacancy interface for enhanced dry reforming of methane: a promoted effect of CeO2 introduction into Ni/MgO. ACS Catal. 13, 15535–15545 (2023).
Fan, M. S., Abdullah, A. Z. & Bhatia, S. Catalytic technology for carbon dioxide reforming of methane to synthesis gas. ChemCatChem 1, 192–208 (2009).
Pakhare, D. & Spivey, J. A review of dry (CO2) reforming of methane over noble metal catalysts. Chem. Soc. Rev. 43, 7813–7837 (2014).
Palmer, C. et al. Dry reforming of methane catalysed by molten metal alloys. Nat. Catal. 3, 83–89 (2020).
Liang, Z., Li, T., Kim, M., Asthagiri, A. & Weaver, J. F. Low-temperature activation of methane on the IrO2(110) surface. Science 356, 298–301 (2017).
Wang, Y. et al. Low-temperature catalytic CO2 dry reforming of methane on Ni-Si/ZrO2 catalyst. ACS Catal. 8, 6495–6506 (2018).
Mubeen, S. et al. An autonomous photosynthetic device in which all charge carriers derive from surface plasmons. Nat. Nanotechnol. 8, 247–251 (2013).
Mukherjee, S. et al. Hot-electron-induced dissociation of H2 on gold nanoparticles supported on SiO2. J. Am. Chem. Soc. 136, 64–67 (2013).
Christopher, P., Xin, H. & Linic, S. Visible-light-enhanced catalytic oxidation reactions on plasmonic silver nanostructures. Nat. Chem. 3, 467–472 (2011).
Xie, W. & Schlücker, S. Hot electron-induced reduction of small molecules on photorecycling metal surfaces. Nat. Commun. 6, 7570 (2015).
Marimuthu, A., Zhang, J. W. & Linic, S. Tuning selectivity in propylene epoxidation by plasmon mediated photo-switching of Cu oxidation state. Science 339, 1590–1593 (2013).
Wang, Z.-J. et al. Photo-assisted methanol synthesis via CO2 reduction under ambient pressure over plasmonic Cu/ZnO catalysts. Appl. Catal. B 250, 10–16 (2019).
Hutter, E. & Fendler, J. H. Exploitation of localized surface plasmon resonance. Adv. Mater. 16, 1685–1706 (2004).
Narang, P., Sundararaman, R. & Atwater, H. A. Plasmonic hot carrier dynamics in solid-state and chemical systems for energy conversion. Nanophotonics 5, 96–111 (2016).
Aslam, U., Rao, V. G., Chavez, S. & Linic, S. Catalytic conversion of solar to chemical energy on plasmonic metal nanostructures. Nat. Catal. 1, 656–665 (2018).
Garcia-Leis, A., Torreggiani, A., Garcia-Ramos, J. V. & Sanchez-Cortes, S. Hollow Au/Ag nanostars displaying broad plasmonic resonance and high surface-enhanced Raman sensitivity. Nanoscale 7, 13629–13637 (2015).
Lee, S. et al. Hot nanogap networks-in-triangular nanoframes: a strategy for positioning adsorbates near hot spots. Small 21, 2409814 (2025).
Harutyunyan, H. et al. Anomalous ultrafast dynamics of hot plasmonic electrons in nanostructures with hot spots. Nat. Nanotechnol. 10, 770–774 (2015).
Christopher, P., Xin, H., Marimuthu, A. & Linic, S. Singular characteristics and unique chemical bond activation mechanisms of photocatalytic reactions on plasmonic nanostructures. Nat. Mater. 11, 1044–1050 (2012).
Zhu, Z., Tang, R., Li, C., An, X. & He, L. Promises of plasmonic antenna-reactor systems in gas-phase CO2 photocatalysis. Adv. Sci. 10, 2302568 (2023).
Zhou, L. et al. Light-driven methane dry reforming with single atomic site antenna-reactor plasmonic photocatalysts. Nat. Energy 5, 61–70 (2020).
Aslam, U., Chavez, S. & Linic, S. Controlling energy flow in multimetallic nanostructures for plasmonic catalysis. Nat. Nanotechnol. 12, 1000–1005 (2017).
Linic, S., Christopher, P. & Ingram, D. B. Plasmonic-metal nanostructures for efficient conversion of solar to chemical energy. Nat. Mater. 10, 911–921 (2011).
Wang, H. et al. Significantly enhanced overall water splitting performance by partial oxidation of Ir through Au modification in core-shell alloy structure. J. Am. Chem. Soc. 143, 4639–4645 (2021).
Gao, J. et al. Breaking long-range order in iridium oxide by alkali ion for efficient water oxidation. J. Am. Chem. Soc. 141, 3014–3023 (2019).
Qin, Y. et al. Interface-engineering strategy for boosting low-ir catalytic water oxidation using a conductive Ti4O7 support. ACS Catal. 14, 13915–13926 (2024).
Chala, S. A. et al. Site activity and population engineering of NiRu-layered double hydroxide nanosheets decorated with silver nanoparticles for oxygen evolution and reduction reactions. ACS Catal. 9, 117–129 (2018).
Weaver, J. H., Olson, C. G. & Lynch, D. W. Optical investigation of the electronic structure of bulk Rh and Ir. Phys. Rev. B 15, 4115–4118 (1977).
Yamada, K., Miyajima, K. & Mafuné, F. Thermionic emission of electrons from gold nanoparticles by nanosecond pulse-laser excitation of interband. J. Phys. Chem. C. 111, 11246–11251 (2007).
Sarina, S. et al. Viable photocatalysts under solar-spectrum irradiation: nonplasmonic metal nanoparticles. Angew. Chem. Int. Ed. 53, 2935–2940 (2014).
Zheng, Z., Tachikawa, T. & Majima, T. Plasmon-enhanced formic acid dehydrogenation using anisotropic Pd-Au nanorods studied at the single-particle level. J. Am. Chem. Soc. 137, 948–957 (2015).
Wu, K., Rodríguez-Córdoba, W. E., Yang, Y. & Lian, T. Plasmon-induced hot electron transfer from the Au Tip to CdS Rod in CdS-Au nanoheterostructures. Nano Lett. 13, 5255–5263 (2013).
Liu, G. et al. Promoting active species generation by plasmon-induced hot-electron excitation for efficient electrocatalytic oxygen evolution. J. Am. Chem. Soc. 138, 9128–9136 (2016).
Huang, H. et al. Unraveling surface plasmon decay in core-shell nanostructures toward broadband light-driven catalytic organic synthesis. J. Am. Chem. Soc. 138, 6822–6828 (2016).
Song, H. et al. Light-enhanced carbon dioxide activation and conversion by effective plasmonic coupling effect of Pt and Au nanoparticles. ACS Appl. Mater. Interfaces 10, 408–416 (2017).
Zhang, Z. & Yates, J. T. Direct observation of surface-mediated electron-hole pair recombination in TiO2(110). J. Phys. Chem. C. 114, 3098–3101 (2010).
Ingram, D. B. & Linic, S. Water splitting on composite plasmonic-metal/semiconductor photoelectrodes: evidence for selective plasmon-induced formation of charge carriers near the semiconductor surface. J. Am. Chem. Soc. 133, 5202–5205 (2011).
Kushida, M., Yamaguchi, A. & Miyauchi, M. Photocatalytic dry reforming of methane by rhodium supported monoclinic TiO2-B nanobelts. J. Energy Chem. 71, 562–571 (2022).
Liu, B. et al. Oxygen vacancy promoting dimethyl carbonate synthesis from CO2 and methanol over Zr-Doped CeO2 Nanorods. ACS Catal. 8, 10446–10456 (2018).
Xiong, H. et al. Highly efficient and selective light-driven dry reforming of methane by a carbon exchange mechanism. J. Am. Chem. Soc. 146, 9465–9475 (2024).
Rao, Z. et al. Light-reinforced key intermediate for anticoking to boost highly durable methane dry reforming over single atom Ni active sites on CeO2. J. Am. Chem. Soc. 145, 24625–24635 (2023).
Kattel, S., Yan, B., Yang, Y., Chen, J. G. & Liu, P. Optimizing binding energies of key intermediates for CO2 hydrogenation to methanol over oxide-supported copper. J. Am. Chem. Soc. 138, 12440–12450 (2016).
Solis-Garcia, A., Louvier-Hernandez, J. F., Almendarez-Camarillo, A. & Fierro-Gonzalez, J. C. Participation of surface bicarbonate, formate and methoxy species in the carbon dioxide methanation catalyzed by ZrO2-supported Ni. Appl. Catal., B 218, 611–620 (2017).
Zhang, J. et al. Photoinducing different mechanisms on a Co-Ni bimetallic alloy in catalytic dry reforming of methane. ACS Catal. 13, 10855–10865 (2023).
Hu, Q. et al. Extraordinary catalytic performance of nickel half-metal clusters for light-driven dry reforming of methane. Adv. Energy Mater. 13, 2300071 (2023).
Bagley, K., Dollinger, G., Eisenstein, L., Singh, A. K. & Zimányi, L. Fourier transform infrared difference spectroscopy of bacteriorhodopsin and its photoproducts. Proc. Natl. Acad. Sci. USA 79, 4972–4976 (1982).
Guo, H.-L., Wang, X.-F., Qian, Q.-Y., Wang, F.-B. & Xia, X.-H. A green approach to the synthesis of graphene nanosheets. ACS Nano 3, 2653–2659 (2009).
Zhang, L. et al. Deprotonated 2-thiolimidazole serves as a metal-free electrocatalyst for selective acetylene hydrogenation. Nat. Chem. 16, 893–900 (2024).
Xu, Y. N. et al. Tuning the microenvironment in monolayer MgAl layered double hydroxide for CO2-to-ethylene electrocatalysis in neutral media. Angew. Chem. Int. Ed. 62, e202217296 (2023).
Xu, D. et al. Versatile synthesis of nano-icosapods via cation exchange for effective photocatalytic conversion of biomass-relevant alcohols. Chem. Sci. 14, 10167–10175 (2023).
Ouyang, B. et al. Facet control of nickel nitride nano-framework for efficient hydrogen evolution electrocatalysis via auxiliary cooling assisted plasma engineering. Small 18, 2204634 (2022).
Acknowledgements
G.L. thanks the financial support from National Natural Science Foundation of China (Project No. 22275088, 52101260), the Project of Shuangchuang Scholar of Jiangsu Province (Project No. JSSCBS20210212), the Fundamental Research Funds for the Central Universities (Project No. 30921011203) and the Start-Up Grant (Project No. AE89991/340) from Nanjing University of Science and Technology. W.J. thanks the financial support from the Joint Funds of the National Natural Science Foundation of China (Project No. U2141202). B.O. thanks the financial support from National Natural Science Foundation of China (Project No. 12304020), Natural Science Foundation of Jiangsu Province (Project No. BK20230909) and Fundamental Research Funds for the Central Universities (Project No. 30923011013). We thank the Shanghai Synchrotron Radiation Facility of BL14W1 for the assistance on XAFS measurements. We express our sincere gratitude to Prof. Kuo Yuan from Tianjin University of Technology for his assistance in the FDTD simulations.
Author information
Authors and Affiliations
Contributions
G.L. conceived the idea and designed this study. T.Y. synthesized the materials, conducted characterization, catalytic performance measurements and data analysis. B.O. conducted DFT calculations. H.Y., Q.W., Z.S., S.Y., F.W., E.K., and W.J. provided constructive suggestions on the manuscript. G.L. and T.Y. wrote the manuscript. All authors checked the manuscript and agreed with its content and have given approval to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interest.
Peer review
Peer review information
Nature Communications thanks Ruiyi Wang and the other anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yin, T., Yuan, H., Wang, Q. et al. Concentrating and directing energy flow in plasmonic heterostructures for stable and efficient light-driven methane dry reforming. Nat Commun 17, 2672 (2026). https://doi.org/10.1038/s41467-026-69581-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69581-z