Abstract
2D covalent organic frameworks (COFs) usually possess a polycrystalline nature as well as lower porosity and surface area than 3D counterparts, restraining their exploration over gas storage applications. Herein, a substituent strategy has been proposed and employed to generate three robust single-crystal 2D COFs isomers with atom-resolution structures determined by 3D electron diffraction. Among three isomers, a precise engineering of their interlayer distance affords the highest Brunauer−Emmett−Teller surface area of ~2100 m2 g−1 and the largest pore volume of 1.40 cm3 g−1 for the desolvated GZU-1. This COF shows the highest total volumetric methane uptake of 240 cm3 (STP) cm−3 at 273 K and 100 bar among 2D COFs, even comparable with those for excellent 3D MOFs. This work not only delivers unique insight into the design of 2D single-crystal COFs by interlayer stacking regulation, but also promotes the application of highly porous 2D COFs in gas storage.
Similar content being viewed by others
Introduction
Natural gas (NG) with about 95% methane (CH4) component is a clean and widely available energy source with relatively low carbon emission and economic advantage1,2,3,4,5. The low energy density of CH4, 0.04 MJ L−1, retards the application of NG in onboard transportation fuel2,3,4,5,6. Among potential materials and technologies for high methane storage, porous sorbents have shown bright promise, particularly under moderate pressure and ambient temperature, attributed to their extraordinarily high porosities and variable pore structures1,7,8,9,10. Accordingly, a number of three-dimensional metal-organic frameworks (3D MOFs) have been reported to exhibit high performance for methane storage3,4,5,6,7. MOFs are generally robust and can sustain thermal/vacuum activation to keep their high porosities3,11. Meanwhile, covalent organic frameworks (COFs) can also exhibit high porosities and stability2,12,13,14. However, the covalent bonds in COFs are much more directional and less flexible than the coordination bonds in MOFs, meaning that single-crystal COFs are much more difficult to synthesize than MOFs15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31 and remain scarcely explored for methane storage to date2,10,12,32.
Different from their MOFs counterparts dominated by the three-dimensional (3D) extended frameworks, most of the reported COFs are of two-dimensional (2D) layered structures1,33,34,35,36. This seems to retard the exploration of their methane storage application due to the still lack of reports on high methane storage performance for 2D MOFs associated with the low porosities and instability of the latter materials. Fortunately, the late construction of highly mesoporous 2D COFs with pore volume as high 2.7 cm3 g−1 motivates us to explore 2D COFs for their methane storage functionality35. Additionally, the significant progress achieved on the single-crystal growth of COFs and single-crystal structure determination technologies over the past few years also motivates us to reveal the fundamental science of 2D/3D COFs in a more comprehensive manner16,17,18,19. However, it is still challenging to prepare 2D single-crystal COFs due to the uncontrollable nucleation and complicated crystallization processes. Only three examples, including Py-1P37,38,39, COF-40036, and oDMPA-Py-COF40 with atom-resolution 2D single-crystal structures have been directly obtained from monomer polymerization thus far. In-depth exploration of the growth and structure of 2D COFs single crystals is very difficult, further restraining the structure-originated property and functionality investigation. Previous investigations have revealed that changing the planarity of building blocks through substituents could lead to the preparation of high-quality 3D COFs single crystals16,41.
Herein, along the substituent strategy, we report three highly porous, robust 2D single-crystal COF isomers for their methane storage (Fig. 1), reaching total volumetric methane storage up to 240 cm3 (STP) cm−3 at 273 K and 100 bar, and in particular gravimetric methane delivery working capacity of 213 mg g−1 (5 to 65 bar at 298 K). Detailed crystal structure and molecular modeling studies show that multiple weak intermolecular interactions, such as C–H···π interactions, can collaboratively enforce the COF framework stability and thus maximize their porosities by interlayer packing regulation. This work paves the way towards a systematic development of highly porous 2D COFs for gas storage in the future.
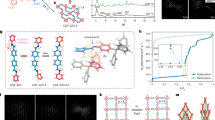
a Schematic assembly of single-crystal GZU-1, GZU-2 and GZU-3 2D COFs. b A unit cell of GZU-1 containing ABCDEF 6-fold alternating stacking (AAl6) structure with multiple C-H···π interactions between the substituents and frameworks to stabilize its permanent porosity.
Results and Discussion
Design and synthesis of single-crystal GZU-1, GZU-2, and GZU-3
In the present case, the methyl or methoxy substituents were introduced onto the positions between the two neighboring phenyl moieties of 1,3,5-tri(phenyl)benzene to afford 1,3,5-trimethoxy-2,4,6-tris(4-aminophenyl)benzene (TOAB), 1,3,5-trimethyl-2,4,6-tris(4-formylphenyl)benzene (TTFB), 1,3,5-trimethyl-2,4,6-tris(4-aminophenyl)benzene (TTAB), and 1,3,5-trimethoxy-2,4,6-tris(4-formylphenyl)benzene (TOFB) (Supplementary Figs. 1–6). Subsequently, [3 + 3] imine polymerization reaction at 298 K in different solvents with the assistance of aniline and acetic acid afforded three novel 2D single-crystal COFs, GZU-1, GZU-2, and GZU-3 (Fig. 1). GZU-1 was made from TOAB and TTFB in a mixed solution of mesitylene (Mes) and 1,4-dioxane (Diox) for 15 days with a yield of 71%. In contrast, isomeric GZU-2 and GZU-3 were obtained by TTAB and TOFB in trichloromethane (TCM) and mixed Mes/1,4-dioxane, respectively, with the yield of 52 and 63%, respectively. Comparison in FT-IR data of these single-crystal COFs and building blocks indicates the disappearance of N−H stretching bonds (3339 and 3420 cm−1 for TOAB and 3338 and 3410 cm−1 for TTAB) and C = O stretching bonds (1702 cm−1 for TTFB and 1698 cm−1 for TOFB). Moreover, the presence of the C=N stretching bonds (1625 cm−1 for GZU-1, 1628 cm−1 for GZU-2, and 1629 cm−1 for GZU-3) suggests the successful imine condensation (Supplementary Figs. 7–9). This is further confirmed by the observation of the resonance peak due to the carbon from the C=N bonds at 160 ppm for GZU-1 and 157 ppm for GZU-2 and GZU-3 in their solid-state 13C cross-polarization magic-angle-spinning (CP/MAS) NMR spectra (Supplementary Figs. 10–12). Scanning electron microscopy (SEM) images reveal a rod-shaped morphology of GZU-1 with a length size up to 10 μm and hexagonal prism morphologies of GZU-2 and GZU-3 with a length size up to 5 μm (Fig. 2a–c and Supplementary Figs. 13–15). The more than 10 intense, sharp diffraction peaks for these three COFs highlight their high-crystallinity nature (Fig. 2d–f). In particular, GZU-2 and GZU-3, composed of the same building units, display obviously different PXRD patterns, indicating their isomerism nature (Fig. 2e, f and Supplementary Figs. 16–19).
a–c The SEM images of single-crystal GZU-1, GZU-2, and GZU-3, respectively. d–f The Le Bail fitting (red lines) of experimental PXRD patterns (black circles) for activated GZU-1, GZU-2, and GZU-3, respectively.
Structural determination of GZU-1, GZU-2, and GZU-3
Single-crystal structural analyses of the three COFs isomers were carried out by 3D electron diffraction (ED) at 77 K and resolutions of 0.8 ~ 1.0 Å, which show that they have essentially isomeric imine-linked honeycomb layered architectures with the hcb-b topology (Supplementary Figs. 20–22 and Supplementary Table 1–3). They possess similar porous frameworks with the 1D channels (16 ~ 17 Å) along the crystallographic c-axis (Fig. 1). GZU-1, GZU-2, and GZU-3 crystallize in distinct space groups, i.e. P32, P-1, P65, respectively. There are different numbers of independent imine-bonded 1,3,5-tris(phenyl)benzene-based building blocks in corresponding asymmetric units of the three COFs.
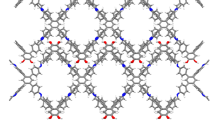
For GZU-1 derived from TOAB and TTFB, its unit cell parameters are a = b = 25.600(4) Å, c = 27.550(6) Å, α = β = 90°, γ = 120°, and V = 15636(6) Å3 (Fig. 3a and Supplementary Table 1). In the asymmetric unit of GZU-1, there are two crystallographically independent TTFB and two TOAB moieties linked through imine bonds (Fig. 3a and Supplementary Fig. 23). For the TTFB units, the dihedral angles between the central benzene ring and the three peripheral benzene rings are 77.94 ~ 82.19°, respectively, disrupting the planarity of this module. This is also true for TOAB units with the dihedral angles of 48.78 ~ 56.33° between central and three peripheral benzene rings (Supplementary Fig. 23). The TOAB and TTFB building blocks are interconnected into a 2D covalent honeycomb-like layered structure (Fig. 3b and Supplementary Figs. 24, 25). Moreover, the alternating arrangement of TTAB and TOFB is observed in the neighboring layers. In this compound, there are multiple weak C−H···π interactions between adjacent layers with short H···C distances of 2.71 ~ 2.85 Å (Supplementary Fig. 26). Different from the previous 2D COFs with normal AA-, AB-, and ABC-packing structures, GZU-1 shows the unknown ABCDEF 6-fold alternating stacking (AAl6) (Figs. 1, c–e and Supplementary Fig. 27). The neighboring layers crossing the centers of TTFB and TOAB moieties are 4.58 and 4.60 Å (Fig. 3d, e and Supplementary Fig. 27). Density Functional Theory (DFT) calculations explored the relative energy of the different stacking patterns of GZU-1. The results demonstrate that the AAl6 form is the most energetically stable one, being approximately 62, 73, and 122–141 kJ mol−1 per sheet more stable than the slip AAl, the eclipsed AA, and the staggered AB patterns, respectively, as detailed in Supplementary Fig. 28 and Supplementary Table 4. All DFT-optimized geometries and details of the calculations are provided in the Supplementary Information.
a Imine-bonded TOAB and TTFB units in single-crystal GZU-1. b A 2D honeycomb layer (the thick grey bond represents hcb-b topology). c Packing structure of GZU-1 along c axis showing the pore surfaces of 1D channels indicated by yellow/gray (inner/outer) curved planes. d and e The side view of the packing structure along a and b axis, respectively.
For GZU-2 and GZU-3, these two COF isomers are prepared from the same building blocks of TTAB and TOFB, possessing different imine-orientation from that of GZU-1 (Supplementary Figs. 29, 30). The slight difference in the molecular configuration of 1,3,5-tris(phenyl)benzene-based building blocks with the same substituents is displayed for these three COFs (Supplementary Fig. 31). Similar to the dihedral angles between central and three peripheral benzene rings in TTFB for GZU-1, these values for TTAB unit are in a range of 78.11 ~ 87.45° for GZU-2 and 82.49 ~ 89.19° for GZU-3 (Supplementary Figs. 29 and 30). In contrast, corresponding dihedral angles of TOFB vary in 47.18 ~ 57.52° for GZU-2 and 53.34 ~ 60.42° for GZU-3. However, the numbers and strength of weak C−H···π interactions among three neighboring layers in GZU-2 and GZU-3 are different from those of GZU-1. In contrast to GZU-1, there are fewer C−H···π weak interactions between adjacent layers in GZU-2 (2.82~2.93 Å) and GZU-3 (2.58 ~ 3.03 Å) (Supplementary Figs. 32 and 33). The distances of neighboring layers crossing the centers of TTAB and TOFB units are 4.37/4.47 Å and 4.35/4.44 Å (Supplementary Figs. 34 and 35). It is worth noting that the gradual decrease of interlayered distance from GZU-1, GZU-2 to GZU-3 slightly changes their pore volume from of 1.41, 1.40, to 1.34 cm3 g−1 derived from the single-crystal structures. Furthermore, GZU-2 and GZU-3 exhibit distinctly different packing forms, namely a unidirectional ABCD 4-fold tilted AA stacking (AAt4) structure and AAl6 architecture, respectively (Supplementary Figs. 28, 34, 35). DFT calculations show that these stacking patterns are the most stable among all possible arrangements, with GZU-2 being slightly more stable than GZU-3 by 5 kJ mol−1 per sheet. Notably, the preferential packing in GZU-2 is more stable by 66, 74, and 102–135 kJ mol−1 per sheet than the AAl slip, AA eclipsed, and the staggered AB patterns, respectively, as shown in Supplementary Fig. 28 and Table 4 (see Supplementary Information for more details of the distinct geometries). Interestingly, this energetic stabilization is very similar to that discussed above for GZU-1.
The experimental PXRD patterns of these three COFs align with those derived from the single-crystal structures (Supplementary Figs. 36–38). Following the dynamic vacuum activation, the three activated COFs (GZU-1a, GZU-2a, GZU-3a) exhibit analogous PXRD patterns with parent materials, suggesting a rather rigid behavior of this family of COFs upon desolvation (Supplementary Figs. 36–38). Additionally, the phase purity of all activated GZU-1, GZU-2, and GZU-3 is also demonstrated using whole profile pattern matching by the Le Bail refinement, giving the ultimate Rp and Rwp values of 4.49 and 6.66% for GZU-1a, 4.54 and 8.25% for GZU-2a, and 6.48 and 8.76% for GZU-3a, respectively (Fig. 2d–f).
To further elucidate the underlying reasons for the synthesizability and high crystallinity of these single-crystal COFs, DFT calculations were further conducted to assess the relative stability of the structures compared to other similar COFs reported in the literature, such as DISTAP-142, TPB-DMTP24, COF-70143, TTI-COF44, and HHTP-DPB45. The data presented in Fig. 4 show that GZU-2 exhibits the most favorable formation energy, at −172 kJ mol−1 per sheet, followed by GZU-3 and GZU-1 with −167 and −165 kJ mol−1 per sheet, respectively. Interestingly, these values are substantially more favorable (i.e., more negative) than those presented by the other COFs, which show values of −113, −106, −103, −71, and −59 kJ mol−1 per sheet for TPB-DMTP, DISTAP-1, COF-701, TTI-COF, and HHTP-DPB, respectively. This more favorable formation energy and consequently predicted greater stability of the resulting crystal structures can directly relate to the higher incidence of C−H⋯π interactions between the sheets of the GZU COFs. These interactions arise from the relative displacement presented by the special stacking pattern of these structures. These C−H,...,π interactions combined, which generally exhibit greater stability than the π⋯π eclipsed interactions, confer to the GZU-1, GZU-2, and GZU-3 a greater overall crystal stability46. This is ultimately associated with the high-quality single crystals obtained for these materials, a task notably difficult for 2D COFs47,48. To further explore the nature of the interaction between the COF layers, we conducted a non-covalent interaction (NCI) analysis based on the DFT electronic density. The result shown in Supplementary Fig. 39 for the GZU-1 indicates that there are two main types of interactions between the adjacent COF layers. The first one is the CH3,...,π interaction between the OMe groups and the aromatic ring from the upper (or lower) layer, with the NCI surface highlighted in green in Supplementary Fig. 39a. The second interaction is the T-shaped benzene-benzene interaction, with the NCI surface highlighted in yellow. Both interactions that can only occur due to the relative shift between the layers are individually stronger than the simple π···π benzene interaction and act as conformational lockers on the crystal structure, playing a decisive role in dictating the relative orientation of the building blocks during crystallization. This indicates the representative structural motif nature of the combination of CH₃,...,π and T–shaped benzene interactions within this family of materials.
a Formation energies in kJ mol−1 per COF sheet and b atomic representation of the structures explored.
Permanent porosity of activated single-crystal 2D COFs
The permanent porosities of the activated COFs (GZU-1a, GZU-2a, and GZU-3a) were revealed using N2 adsorption and desorption measurements at 77 K (Fig. 5a and Supplementary Figs. 40 and 41). The N₂ adsorption isotherms of these three COFs display a pronounced two-step uptake behavior. A sharp increase in adsorption isotherms is observed at very low relative pressures (P/P₀ <0.01), followed by a more gradual uptake in the range of P/P₀ ≈ 0.01–0.05. Upon further increasing the relative pressure, the other sharp increase of adsorption appears in the range of P/P₀ ≈ 0.05–0.1, achieving a saturated adsorption. For GZU-1a with the largest interlayer distance among the three COFs, it also displays the largest N2 uptake of 917 cm3 g−1 at 1.0 bar with the highest BET area of 2100 m2 g−1, in line with the calculated value from the crystal structure of 2414 m2 g−1 (Supplementary Fig. 42).
a The N2 adsorption (solid) and desorption (hollow) isotherms of GZU-1a at 77 K. b The high-pressure total methane uptake of GZU-1a. c The methane gravimetric and volumetric working capacities of GZU-1a, GZU-2a, GZU-3a in comparison to other 2D COFs reported to date. d The relationship between the gravimetric methane storage capacities of the 2D COFs (grey dots) and the gravimetric BET surface area.
In contrast, GZU-2a with the second interlayered distance adsorbs smaller N2 volume of 836 cm3 g−1 at 1.0 bar with a slightly lower BET area of 1965 m2 g−1, close to its simulated value of 2396 m2 g−1 (Supplementary Figs. 43 and 44). Its packing isomer, GZU-3a, with the smallest average interlayered distance, accommodates the least N2 amount of 820 cm3 g−1 at 1.0 bar with the smallest BET surface area of 1897 m2 g−1, consistent with the simulated value of 2326 m2 g−1. Interestingly, there exists an overall linear relationship between the average interlayered distance and experimental BET surface area for the three COFs, indicating the capability of the packing mode and imine orientation of 2D COFs isomers in finely engineering the interlayered distance and in turn the pore surface area (Supplementary Fig. 45). It is worth noting that the present single-crystal COFs possess much higher BET areas (1897~2100 m2 g−1) than their unsubstituted COF counterpart TAPB-TFPB (~224 m2 g−1)49, further confirming the advantage of substituent strategy in stabilizing the permanent porosity. Following the increased interlayer distance from GZU-3a, GZU-2a, and GZU-1a, their experimental pore volume at P/P0 at 1.0 bar also gets increased in a gradual manner from 1.23, 1.28, to 1.40 cm3 g−1, overall consistent with the calculated values from the simulated structures of 1.34, 1.40, of 1.41 cm3 g−1 (Fig. 5a and Supplementary Figs. 40 and 41). In addition, the pore-size distribution curves of these three single-crystal COFs have been studied by adsorption isotherms employing the density functional theory method, displaying pores centered at 17.3 Å for GZU-1a, 17.0 Å for GZU-2a, and 16.1 Å for GZU-3a, Supplementary Figs. 46–48, aligning with the pore sizes obtained from the single-crystal structures (17.1 Å for GZU-1a, 16.9 Å for GZU-2a, and 16.2 Å for GZU-3a), Supplementary Figs. 49–51. These activated COFs afford moderate CO2 absorption of 20, 25, and 26 cm3 g−1 at room temperature and 1 bar for GZU-1a, GZU-2a, and GZU-3a due to the weak interactions between CO2 molecules and COFs (Supplementary Figs. 52–54). These results further prove their permanent porosity.
High-pressure methane storage of activated COFs
Considering the high permanent porosity and large pore volume of these activated COFs, high-pressure methane adsorption experiments were performed at 273 and 298 K (Fig. 5b and Supplementary Figs. 55, 56). The total gravimetric methane uptake of the activated GZU-1a amounts to 299 mg g−1 (418 cm3 (STP) g−1) at 298 K and 100 bar. While GZU-2a and GZU-3a with different packing mode exhibit total gravimetric methane uptake of 289 mg g−1 (405 cm3 (STP) g−1) and 264 mg g−1 (370 cm3 (STP) g−1), respectively, under the same conditions. The gravimetrically higher methane uptake for GZU-1a than GZU-2a and GZU-3a is associated with its highest gravimetric BET surface area and pore volume among the three COFs. Force field grand Canonical Monte Carlo (GCMC) simulations confirm the attractive methane sorption performance of these three COFs as highlighted in Fig. 5b and Supplementary Figs. 57–59. These simulations reveal that the high methane uptake is associated with the high accessible surface exhibited by the single-crystal COFs, with the periodic and multidirectional layer slipping introducing indentations between the COF layers that favor methane adsorption (Supplementary Fig. 60). Compared with the traditional AA and AB stacking models of 2D COFs, this special layer slipping mode of these three single-crystal COFs can increase the number and cooperative strength of C–H···π interactions between adjacent layers, which help maintain the framework rigidity during activation and high-pressure CH4 adsorption.
On the basis of the crystal densities of 0.523, 0.518, and 0.537 cm g−1 for GZU-1a, GZU-2a, and GZU-3a without guest molecules, the corresponding total volumetric methane uptakes are calculated as 218, 210, and 198 cm3 (STP) cm−3 at 298 K and 100 bar (Supplementary Tables 1–3). In particular for GZU-1a, its total volumetric methane uptake is 240 cm3 (STP) cm−3 at 273 K and 100 bar. After the high-pressure CH4 sorption experiment, three COFs display similar PXRD patterns to the as-prepared species (Supplementary Figs. 61–63), further disclosing the rigid structures of these three COFs with multiple weak C−H···π interactions.
According to the methane isotherms obtained at 273 and 298 K, the adsorption isosteric heat of GZU-1a is calculated as 13.2 kJ mol−1 (Supplementary Figs. 55, 56, 64), in agreement with the simulated value of 11.4 kJ mol−1 and indicating a weak binding interaction between methane and the COF framework4,6. This is also true for GZU-2a and GZU-3a, with the adsorption isosteric heats of 12.3 and 11.8 kJ mol−1 (Supplementary Fig. 64), also showing good consistency with the simulated values of 12.6 and 12.7 kJ mol−1, respectively. This leads to the reduced CH4 capacities at 5 bar and 298 K for GZU-1a (32 mg g−1), GZU-2a (29 mg g−1), GZU-3a (30 mg g−1) (Fig. 5b and Supplementary Fig. 65). This, in combination with the high gravimetric methane uptake of GZU-1a (245 mg g−1), GZU-2a (227 mg g−1), and GZU-3a (215 mg g−1) at 65 bar and 298 K, results in the usable methane gravimetric working capacities of 213, 198, and 185 mg g−1, respectively. It is worth noting that the total gravimetric methane capacity of GZU-1 at 65 bar and 298 K is lower than that for many excellent MOFs absorbents such as UTSA-76a (263 mg g−1)7, NOTT-101a (247 mg g−1)50, MAF-38 (247 mg g−1)11, and NJU-Bai 42 (254 mg g−1)11 (Supplementary Figs. 65–67)9,10,11. However, its lower adsorption capacity at 5 bar than the above-mentioned MOFs remarkably induces high gravimetric working capacity (Fig. 5c and Supplementary Fig. 67). Noticeably, introduction of substituents onto the COFs frameworks seems to be able to reduce the CH4 affinity at low pressure to enhance the gravimetric working capacity. Indeed, it must be pointed out that thus far, an increasing number of COFs have been synthesized and structurally characterized. However, high-pressure methane storage studies over COFs have been rarely explored due to the lack of single-crystal structural information to evaluate their performances. On the basis of the present result with the help of those reported previously, herein we established the correlation between total gravimetric methane uptake (Ctotal, cm3 (STP) g−1) at 65 bar and 298 K and experimental gravimetric BET area (S, m2 g−1), showing an overall linear relationship with an empirical formula of Ctotal = 0.13383 × S + 5.73085 (R = 0.93), similar to that for MOFs6 (Fig. 5d).
According to the crystal densities of COFs, the methane volumetric working capacities (5 to 65 bar) of GZU-1a, GZU-2a, and GZU-3a at 298 K are calculated as 156, 143, and 139 cm3 (STP) cm−3, respectively, (Fig. 5c). It is noteworthy that both methane volumetric and gravimetric working capacities (5 to 65 bar and 298 K) of these three COFs are significantly superior to those for the thus far reported 2D COFs (Fig. 5c). Moreover, both the methane volumetric and gravimetric working capacities (5 to 65 bar and 298 K) of GZU-1a are higher than those for GZU-2a and GZU-3a due to its larger experimentally revealed BET area and pore volume than the latter two species. In addition, the methane volumetric working capacities (5 to 65 bar and 298 K) of GZU-1a (156 cm3 (STP) cm−3) are even comparable with those for 3D COFs like COF-103 (165 cm3 (STP) cm−3)10 and TAM-TFS-COF (157 cm3 (STP) cm−3)12 (Supplementary Fig. 68). The above phenomena clearly indicate the bright perspective of 2D COFs in the methane storage since the further enlargement of pore size of hcb-b topological COFs enables the enhanced BET area and pore volume.
In summary, a substituent strategy has been proposed to design and prepare three single-crystal 2D hcb-b COFs isomers with different imine orientation and stacking modes. The atom-resolution crystal structures of these three COFs have been determined using 3D ED. Among the three compounds, GZU-1 and GZU-3 show an unprecedented ABCDEF 6-fold alternating stacking mode (AAl6). Moreover, GZU-2 and GZU-3, with different imine orientations from GZU-1, exhibit the first example of stacking isomers with atom-resolution structures in single-crystal 2D COFs, and GZU-2 depicts the first unidirectional ABCD 4-fold tilted stacking structure (AAt4). Moreover, fine adjustment of interlayered distance of COFs at atomic precision results in the highest total volumetric methane uptake of 240 cm3 (STP) cm−3 at 273 K and 100 bar for the activated sample. In particular, GZU-1a possesses the highest methane gravimetric working capacity (5 to 65 bar at 298 K) of 213 mg g−1, outperforming many state-of-the-art porous materials. The exceptional methane storage capacity of 2D single-crystal COFs demonstrates that the present structure-dependent property is originated from their precisely engineered interlayer packing. These findings will stimulate an improved understanding and leveraging of 2D covalent porous materials in multifunctional applications.
Methods
Synthesis of GZU-1
A 20 mL vial was loaded with TTFB (112.5 mg, 0.26 mmol), aniline (4.0 mL), and dry mesitylene (10.0 mL). The mixture was subjected to ultrasonication until complete dissolution of the solid components. This was followed by the addition of glacial acetic acid (2.0 mL). Subsequently, a solution of TOAB (114.8 mg, 0.26 mmol) in dry 1,4-dioxane (5.0 mL) was introduced into this reaction system. The resulting mixture was left undisturbed at 25 °C for 15 days, affording a large quantity of GZU-1 single crystals with sizes up to 10 μm. The pristine GZU-1 sample was successively solvent-exchanged with 1,4-dioxane, acetone, and n-hexane for 24 h, with the solvent renewed every 8 h. After this process, the material was collected by filtration and desolvated under dynamic vacuum at 100 °C for 12 h, yielding the activated material (147.5 mg, 71%).
Synthesis of GZU-2
A 20 mL vial was loaded with TOFB (124.9 mg, 0.26 mmol), aniline (1.5 mL), and dry TCM (5.0 mL). The mixture was subjected to ultrasonication until complete dissolution of the solid components, then glacial acetic acid (2.0 mL) was added. Subsequently, a solution of TTAB (102.3 mg, 0.26 mmol) in dry TCM (5.0 mL) was immediately introduced into this reaction system. The resulting mixture was left undisturbed at 25 °C for 10 days, affording a large quantity of GZU-2 single crystals with sizes up to 5 μm. The pristine GZU-2 sample was successively solvent-exchanged with TCM and n-hexane for 24 h, with the solvent renewed every 8 h. The solvent-exchanged sample was then collected by filtration and desolvated under dynamic vacuum at 100 °C for 12 h, yielding the activated material (110.1 mg, 52%).
Synthesis of GZU-3
A 20 mL vial was loaded with TOFB (124.9 mg, 0.26 mmol), aniline (4.0 mL), and dry mesitylene (10.0 mL). The mixture was subjected to ultrasonication until complete dissolution of the solid components. This was followed by the addition of glacial acetic acid (2.0 mL). Subsequently, a solution of TTAB (102.3 mg, 0.26 mmol) in dry 1,4-dioxane (5.0 mL) was immediately introduced into this reaction system. The resulting mixture was left undisturbed at 25 °C for 15 days, yielding a large quantity of GZU-3 single crystals with sizes up to 5 μm. The pristine GZU-3 sample was successively solvent-exchanged with 1,4-dioxane, acetone, and n-hexane for 24 h, with the solvent renewed every 8 h. The solvent-exchanged sample was then collected by filtration and desolvated under dynamic vacuum at 100 °C for 12 h, affording the activated material (132.9 mg, 63%).
Density functional theory calculations
All Density functional theory (DFT) calculations were carried out using the Perdew–Burke–Ernzerhof (PBE) exchange–correlation functional51 with DFT-D3(BJ)52,53 dispersion corrections using the Quickstep module implemented in the CP2K package (version 2024.3)54. GTH pseudopotentials55,56 were employed in conjunction with triple-ζ Gaussian basis sets augmented with one set of polarization functions, together with an auxiliary plane-wave basis set using a cutoff energy of 1200 Ry mapped onto a five-level multigrid. The orbital transformation (OT) method was applied for electronic structure optimization of all models57. To assess the relative stability of different stacking isomers of the GZU COFs, various stacking configurations were constructed using the pyCOFBuilder package58, as illustrated in Supplementary Fig. 26, and systematically compared with the experimentally determined structures.
Grand canonical Monte Carlo (GCMC) simulations
All methane adsorption isotherms were simulated using the RASPA2 package59,60. For each pressure point of the isotherm, 50000 Monte Carlo cycles were conducted, and the equilibrium adsorption uptake was determined by averaging over the equilibrated portion of the trajectory, as identified using the pyMSER package61. Methane molecules were described using Lennard–Jones parameters from the TraPPE-UA force field62, while COF framework atoms were modeled with the DREIDING force field63. Cross-interaction parameters were derived using the Lorentz–Berthelot mixing rules, and all Lennard–Jones interactions were truncated and shifted to zero at a cutoff distance of 12.8 Å. All simulations were performed using supercells large enough to ensure that each lattice vector exceeds the short-range interaction cutoff, thereby eliminating interactions between periodic images. Adsorption enthalpy was evaluated based on the fluctuations of energy and particle number in the grand canonical ensemble.
Textural properties assessment
Pore size distribution, pore volume, and gravimetric surface area were calculated using the Zeo++ software (v0.3)64,65. A probe molecule with a radius of 1.86 Å was employed, and all calculations were performed in the High Accuracy mode with 100000 sampling cycles.
Data availability
Crystallographic data reported in this paper are provided in the supplementary materials and archived at the Cambridge Crystallographic Data Centre under reference numbers 2455725 (GZU-1), 2415331 (GZU-2), and 2415329 (GZU-3). Copies of the data can be obtained free of charge from https://www.ccdc.cam.ac.uk/structures/. All other data are available in the main text or the supplementary materials. In addition, the data that support the findings of this study and the raw data for all the figures have been uploaded to Figshare at https://doi.org/10.6084/m9.figshare.30576989. Source data are provided with this paper. All data are available from the corresponding author upon request. Source data are provided with this paper.
References
Yaghi, O. M., Kalmutzki, M. J. & Diercks, C. S. Introduction to reticular chemistry: Metal–organic frameworks and covalent organic frameworks. Vol. John Wiley & Sons (2019).
Yin, Y. et al. Ultrahigh–surface area covalent organic frameworks for methane adsorption. Science 386, 693–696 (2024).
Chen, Z. et al. Balancing volumetric and gravimetric uptake in highly porous materials for clean energy. Science 368, 297–303 (2020).
Zhou, J. et al. High-connected ternary metal–organic framework platform: synthesis, structure, and methane storage capacity. J. Am. Chem. Soc. 147, 21811–21817 (2025).
Mason, J. A. et al. Methane storage in flexible metal–organic frameworks with intrinsic thermal management. Nature 527, 357–361 (2015).
Li, B., Wen, H.-M., Zhou, W., Xu, J. Q. & Chen, B. Porous metal–organic frameworks: promising materials for methane storage. Chem 1, 557–580 (2016).
Wen, H.-M. et al. A metal–organic framework with optimized porosity and functional sites for high gravimetric and volumetric methane storage working capacities. Adv. Mater. 30, 1704792 (2018).
Wang, J.-X. et al. Construction of highly porous and robust hydrogen-bonded organic framework for high-capacity clean energy gas storage. Angew. Chem., Int. Ed. 63, e202411753 (2024).
Li, M. et al. Construction of porous aromatic frameworks with exceptional porosity via building unit engineering. Adv. Mater. 30, 1804169 (2018).
Furukawa, H. & Yaghi, O. M. Storage of hydrogen, methane, and carbon dioxide in highly porous covalent organic frameworks for clean energy applications. J. Am. Chem. Soc. 131, 8875–8883 (2009).
Zhang, M. et al. Fine tuning of mof–505 analogues to reduce low–pressure methane uptake and enhance methane working capacity. Angew. Chem., Int. Ed. 129, 11584–11588 (2017).
Yu, B. et al. Single-crystalline 3D covalent organic frameworks with exceptionally high specific surface areas and gas storage capacities. J. Am. Chem. Soc. 146, 28932–28940 (2024).
Chang, J. et al. Synthesis of three-dimensional covalent organic frameworks through a symmetry reduction strategy. Nat. Chem. 17, 571–581 (2025).
El-Kaderi, H. M. et al. Designed Synthesis of 3D covalent organic frameworks. Science 316, 268–272 (2007).
Gong, C. et al. Selective electrocatalytic synthesis of urea using entangled iron porphyrins in covalent organic frameworks. Nat. Synth. 4, 720–729 (2025).
Gao, C., Li, J., Yin, S., Sun, J. & Wang, C. Twist building blocks from planar to tetrahedral for the synthesis of covalent organic frameworks. J. Am. Chem. Soc. 142, 3718–3723 (2020).
Han, J. et al. Fast growth of single-crystal covalent organic frameworks for laboratory X-ray diffraction. Science 383, 1014–1019 (2024).
Ma, T. et al. Single-crystal x-ray diffraction structures of covalent organic frameworks. Science 361, 48–52 (2018).
Evans, A. M. et al. Seeded growth of single-crystal two-dimensional covalent organic frameworks. Science 361, 52–57 (2018).
Wang, M., Jin, Y., Zhang, W. & Zhao, Y. Single-crystal polymers (SCPs): from 1D to 3D architectures. Chem. Soc. Rev. 52, 8165–8193 (2023).
Pelkowski, C. E. et al. Tuning crystallinity and stacking of two-dimensional covalent organic frameworks through side-chain interactions. J. Am. Chem. Soc. 145, 21798–21806 (2023).
Auras, F. et al. Dynamic two-dimensional covalent organic frameworks. Nat. Chem. 16, 1373–1380 (2024).
Kang, C. et al. Covalent organic framework atropisomers with multiple gas-triggered structural flexibilities. Nat. Mater. 22, 636–643 (2023).
Deng, L. et al. Synthesis of single-crystal two-dimensional covalent organic frameworks and uncovering their hidden structural features by three-dimensional electron diffraction. J. Am. Chem. Soc. 146, 35427–35437 (2024).
Endo, K. et al. Crystalline porous frameworks based on double extension of metal–organic and covalent organic linkages. Nat. Synth. 4, 603–613 (2025).
Wang, Z. et al. On-water surface synthesis of charged two-dimensional polymer single crystals via the irreversible Katritzky reaction. Nat. Synth. 1, 69–76 (2022).
Zhao, W. et al. Using sound to synthesize covalent organic frameworks in water. Nat. Synth. 1, 87–95 (2022).
Li, X. et al. Constructing ambivalent imidazopyridinium-linked covalent organic frameworks. Nat. Synth. 1, 382–392 (2022).
Haase, F. & Lotsch, B. V. Solving the COF trilemma: towards crystalline, stable and functional covalent organic frameworks. Chem. Soc. Rev. 49, 8469–8500 (2020).
Zhou, Z. et al. Growth of single-crystal imine-linked covalent organic frameworks using amphiphilic amino-acid derivatives in water. Nat. Chem. 15, 841–847 (2023).
Yu, B. et al. Linkage conversions in single-crystalline covalent organic frameworks. Nat. Chem. 16, 114–121 (2024).
Zeng, T. et al. Atomic observation and structural evolution of covalent organic framework rotamers. Proc. Natl. Acad. Sci. USA. 121, e2320237121 (2024).
Alahakoon, S. B., Diwakara, S. D., Thompson, C. M. & Smaldone, R. A. Supramolecular design in 2D covalent organic frameworks. Chem. Soc. Rev. 49, 1344–1356 (2020).
She, P., Qin, Y., Wang, X. & Zhang, Q. Recent progress in external-stimulus-responsive 2D covalent organic frameworks. Adv. Mater. 34, 2101175 (2022).
Feriante, C. H. et al. Rapid synthesis of high surface area imine-linked 2D covalent organic frameworks by avoiding pore collapse during isolation. Adv. Mater. 32, 1905776 (2020).
Yi, L. et al. Moiré Patterns of Two-dimensional covalent organic framework single crystals created by superstacking. J. Am. Chem. Soc. 147, 32145–32156 (2025).
Yi, L., Gao, Y., Luo, S., Wang, T. & Deng, H. Structure Evolution of 2D covalent organic frameworks unveiled by single-crystal x-ray diffraction. J. Am. Chem. Soc. 146, 19643–19648 (2024).
Kang, C. et al. Growing single crystals of two-dimensional covalent organic frameworks enabled by intermediate tracing study. Nat. Commun. 13, 1370 (2022).
Peng, L. et al. Ultra-fast single-crystal polymerization of large-sized covalent organic frameworks. Nat. Commun. 12, 5077 (2021).
Wei, L. et al. Dimensionality and molecular packing control of covalent organic frameworks through pendant group design. J. Am. Chem. Soc. 146, 31384–31390 (2024).
Xiao, Y. et al. Constructing a 3D covalent organic framework from 2d hcb nets through inclined interpenetration. J. Am. Chem. Soc. 145, 13537–13541 (2023).
Wang, S. et al. Single-crystal 2D covalent organic frameworks for plant biotechnology. J. Am. Chem. Soc. 145, 12155–12163 (2023).
Lyu, H., Diercks, C. S., Zhu, C. & Yaghi, O. M. Porous crystalline olefin-linked covalent organic frameworks. J. Am. Chem. Soc. 141, 6848–6852 (2019).
Pütz, A. M. et al. Total scattering reveals the hidden stacking disorder in a 2D covalent organic framework. Chem. Sci. 11, 12647–12654 (2020).
Spitler, E. L. et al. A 2D covalent organic framework with 4.7-nm pores and insight into its interlayer stacking. J. Am. Chem. Soc. 133, 19416–19421 (2011).
Sinnokrot, M. O., Valeev, E. F. & Sherrill, C. D. Estimates of the ab initio limit for π−π interactions: the benzene dimer. J. Am. Chem. Soc. 124, 10887–10893 (2002).
Castano, I. et al. Mapping grains, boundaries, and defects in 2D covalent organic framework thin films. Chem. Mater. 33, 1341–1352 (2021).
Nguyen, V. & Grünwald, M. Microscopic origins of poor crystallinity in the synthesis of covalent organic framework COF-5. J. Am. Chem. Soc. 140, 3306–3311 (2018).
Kaleeswaran, D., Vishnoi, P. & Murugavel, R. [3+3] Imine and β-ketoenamine tethered fluorescent covalent-organic frameworks for CO2 uptake and nitroaromatic sensing. J. Mater. Chem. C. 3, 7159–7171 (2015).
Li, B. et al. Porous metal–organic frameworks with Lewis basic nitrogen sites for high-capacity methane storage. Energy Environ. Sci. 8, 2504–2511 (2015).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Kühne, T. D. et al. CP2K: An electronic structure and molecular dynamics software package - Quickstep: efficient and accurate electronic structure calculations. J. Chem. Phys. 152, 194103 (2020).
Goedecker, S., Teter, M. & Hutter, J. Separable dual-space gaussian pseudopotentials. Phys. Rev. B 54, 1703–1710 (1996).
Hartwigsen, C., Goedecker, S. & Hutter, J. Relativistic separable dual-space gaussian pseudopotentials from H to Rn. Phys. Rev. B 58, 3641–3662 (1998).
VandeVondele, J. & Hutter, J. An efficient orbital transformation method for electronic structure calculations. J. Chem. Phys. 118, 4365–4369 (2003).
Oliveira, F. L. & Esteves, P. M. pyCOFBuilder: A Python package for automated creation of covalent organic framework models based on the reticular approach. J. Chem. Inf. Model. 64, 3278–3289 (2024).
Dubbeldam, D., Torres-Knoop, A. & Walton, K. S. On the inner workings of Monte Carlo codes. Mol. Simul. 39, 1253–1292 (2013).
Dubbeldam, D., Calero, S., Ellis, D. E. & Snurr, R. Q. RASPA: molecular simulation software for adsorption and diffusion in flexible nanoporous materials. Mol. Simul. 42, 81–101 (2016).
Oliveira, F. L., Luan, B., Esteves, P. M., Steiner, M. & Neumann Barros Ferreira, R. pyMSER─an open-source library for automatic equilibration detection in molecular simulations. J. Chem. Theory Comput. 20, 8559–8568 (2024).
Martin, M. G. & Siepmann, J. I. Transferable potentials for phase equilibria. 1. united-atom description of n-alkanes. J. Phys. Chem. B 102, 2569–2577 (1998).
Mayo, S. L., Olafson, B. D. & Goddard, W. A. DREIDING: a generic force field for molecular simulations. J. Phys. Chem. 94, 8897–8909 (1990).
Willems, T. F., Rycroft, C. H., Kazi, M., Meza, J. C. & Haranczyk, M. Algorithms and tools for high-throughput geometry-based analysis of crystalline porous materials. Microporous Mesoporous Mater. 149, 134–141 (2012).
Ongari, D. et al. Accurate characterization of the pore volume in microporous crystalline materials. Langmuir 33, 14529–14538 (2017).
Acknowledgements
This work was financially supported by the Natural Science Foundation of China (Nos. 22235001 [J.J.], 22175020 [J.J.], 22131005 [H.W.], 22505047 [B.Y.], W2431013 [B.C.], and 22261132512 [H.W.]), Supported by the Postdoctoral Fellowship Program and China Postdoctoral Science Foundation (BX20250107 [B.Y.]), Xiaomi Young Scholar Program, the Fundamental Research Funds for the Central Universities (FRF-KST-25-006 [H.W.]), the Guizhou Provincial Key Laboratory Platform Project (ZSYS [2025] 008 [X.X.]), Talent Program of Guizhou University (LJ-2024-03) [J.J.], Guizhou University, and University of Science and Technology Beijing. We also thank the staff from the BL17B1 beamline of the National Facility for Protein Science in Shanghai (NFPS) at Shanghai Synchrotron Radiation Facility, for assistance during data collection. In addition, we acknowledge ReadCrystal Technology Co. for their prominent works in MicroED analysis. We are also very grateful for the help and discussion from Prof. Rui-Biao Lin at the Sun Yat-Sen University.
Author information
Authors and Affiliations
Contributions
B.Y., H.W., X.X., B.C., and J.J. conceived the subject and designed the experiments. B.Y. synthesized the materials and performed most of the characterization experiments. B.Y. measured the SEM of the COFs under the supervision of Q.X and X.D. B.Y., H.W., H.L., S.Y., and Y.J. solved and analyzed the crystal structures. F.L.O., W.L, J.Z, and G.M. carried out the theoretical calculation of 2D COFs. B.Y., F.L.O., H.W., X.X., G.M., B.C., and J.J. interpreted the results and wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ramanathan Vaidhyanathan and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yu, B., Oliveira, F.L., Li, W. et al. Single-crystal 2D covalent organic frameworks for high-capacity methane storage. Nat Commun 17, 2740 (2026). https://doi.org/10.1038/s41467-026-69614-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69614-7