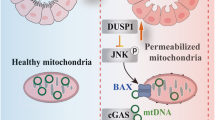
Abstract
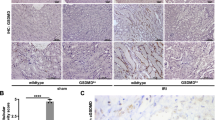
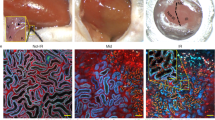
Acute kidney injury (AKI) remains a leading cause of morbidity and mortality, yet the molecular pathways driving kidney tubule damage in AKI are not fully understood. Here, we report dual-specificity phosphatase 26 (DUSP26) as a critical regulator of kidney tubule injury in AKI. In our study, DUSP26 expression was markedly reduced in kidney biopsies from AKI patients of both sexes and in male murine models of cisplatin nephrotoxic and ischemic AKI. This down-regulation was driven by hypermethylation of the gene promoter of DUSP26 in kidney tubular cells. Loss of DUSP26 exacerbated tubular damage, whereas knock-in of DUSP26 specifically in kidney proximal tubule cells conferred protection. Mechanistically, DUSP26 directly bound to p53 to dephosphorylate it at serine 312, dampening the transcriptional activity of p53 towards cell death genes. Pharmacologic inhibition of DUSP26 sensitized kidneys to AKI, whereas DUSP26 overexpression was protective. Pharmacologic inhibition of DUSP26 also exacerbated ischemia-reperfusion injury in the liver. These findings uncover DUSP26 as a key phosphatase guarding against tissue injury by dephosphorylating p53 at serine 312, and highlight the DUSP26-p53 axis as a promising therapeutic target.
Similar content being viewed by others
Data availability
The transcriptomic datasets used in this study are available in the Gene Expression Omnibus (GEO) database under accession codes GSE21374, GSE30718 and GSE43974. The mass spectrometry proteomics data generated in this study have been deposited in the ProteomeXchange Consortium via the PRIDE partner repository under dataset identifier PXD070789. The bisulfite sequencing data generated in this study have been deposited in the NCBI BioProject database under accession code PRJNA1365351. All datasets generated or analyzed in this study will be available without access restrictions upon publication. Source data are provided with this paper.
Code availability
No custom code was used in this study. All data analyses were performed using standard packages in R (version 4.4.0) and GraphPad Prism (version 10.0).
References
Ostermann, M. et al. Acute kidney injury. Lancet 405, 241–256 (2025).
Vijayan, A. Tackling AKI: prevention, timing of dialysis and follow-up. Nat. Rev. Nephrol. 17, 87–88 (2021).
Reid, S. & Scholey, J. W. Recent approaches to targeting canonical NFκB signaling in the early inflammatory response to renal IRI. J. Am. Soc. Nephrol. 32, 2117–2124 (2021).
Lameire, N. H. et al. Acute kidney injury: an increasing global concern. Lancet 382, 170–179 (2013).
Sanz, A. B., Sanchez-Niño, M. D., Ramos, A. M. & Ortiz, A. Regulated cell death pathways in kidney disease. Nat. Rev. Nephrol. 19, 281–299 (2023).
Tang, C., Livingston, M. J., Safirstein, R. & Dong, Z. Cisplatin nephrotoxicity: new insights and therapeutic implications. Nat. Rev. Nephrol. 19, 53–72 (2023).
Agarwal, A. et al. Cellular and molecular mechanisms of AKI. J. Am. Soc. Nephrol. 27, 1288–1299 (2016).
Ikizler, T. A. et al. A prospective cohort study of acute kidney injury and kidney outcomes, cardiovascular events, and death. Kidney Int 99, 456–465 (2021).
Lake, B. B. et al. An atlas of healthy and injured cell states and niches in the human kidney. Nature 619, 585–594 (2023).
Guo, C., Dong, G., Liang, X. & Dong, Z. Epigenetic regulation in AKI and kidney repair: mechanisms and therapeutic implications. Nat. Rev. Nephrol. 15, 220–239 (2019).
Fu, Y., Xiang, Y., Zha, J., Chen, G. & Dong, Z. Enhanced STAT3/PIK3R1/mTOR signaling triggers tubular cell inflammation and apoptosis in septic-induced acute kidney injury: implications for therapeutic intervention. Clin. Sci. 138, 351–369 (2024).
Park, J. Y. et al. Blockade of STAT3 signaling alleviates the progression of acute kidney injury to chronic kidney disease through antiapoptosis. Am. J. Physiol. Ren. Physiol. 322, F553–f572 (2022).
Fu, Y. et al. The STAT1/HMGB1/NF-κB pathway in chronic inflammation and kidney injury after cisplatin exposure. Theranostics 13, 2757–2773 (2023).
Livingston, M. J. et al. Autophagy activates EGR1 via MAPK/ERK to induce FGF2 in renal tubular cells for fibroblast activation and fibrosis during maladaptive kidney repair. Autophagy 20, 1032–1053 (2024).
An, J. N. et al. Periostin induces kidney fibrosis after acute kidney injury via the p38 MAPK pathway. Am. J. Physiol. Ren. Physiol. 316, F426–f437 (2019).
Xiong, J. et al. DUSP2-mediated inhibition of tubular epithelial cell pyroptosis confers nephroprotection in acute kidney injury. Theranostics 12, 5069–5085 (2022).
Shi, L. et al. DUSP1 protects against ischemic acute kidney injury through stabilizing mtDNA via interaction with JNK. Cell Death Dis. 14, 724 (2023).
Bai, F. et al. DUSP5 deficiency suppresses the progression of acute kidney injury by enhancing autophagy through AMPK/ULK1 pathway. Transl. Res. 274, 1–9 (2024).
Thompson, E. M. & Stoker, A. W. A review of DUSP26: structure, regulation and relevance in human disease. Int. J. Mol. Sci. 22 (2021).
Patterson, K. I., Brummer, T., Daly, R. J. & O’Brien, P. M. DUSP26 negatively affects the proliferation of epithelial cells, an effect not mediated by dephosphorylation of MAPKs. Biochim. Biophys. Acta 1803, 1003–1012 (2010).
Boutros, T., Chevet, E. & Metrakos, P. Mitogen-activated protein (MAP) kinase/MAP kinase phosphatase regulation: roles in cell growth, death, and cancer. Pharm. Rev. 60, 261–310 (2008).
Shi, Y. et al. NSC-87877 inhibits DUSP26 function in neuroblastoma resulting in p53-mediated apoptosis. Cell Death Dis. 6, e1841 (2015).
Eroglu, B. et al. Dusp26 phosphatase regulates mitochondrial respiration and oxidative stress and protects neuronal cell death. Cell Mol. Life Sci. 79, 198 (2022).
Gómez-Banoy, N. et al. Adipsin preserves beta cells in diabetic mice and associates with protection from type 2 diabetes in humans. Nat. Med. 25, 1739–1747 (2019).
Liu, C. et al. Targeting DUSP26 to drive cardiac mitochondrial dynamics via FAK-ERK signaling in diabetic cardiomyopathy. Free Radic. Biol. Med. 225, 856–870 (2024).
Mimura, I., Chen, Z. & Natarajan, R. Epigenetic alterations and memory: key players in the development/progression of chronic kidney disease promoted by acute kidney injury and diabetes. Kidney Int. 107, 434–456 (2025).
Jung, S. et al. Dual-specificity phosphatase 26 (DUSP26) stimulates Aβ42 generation by promoting amyloid precursor protein axonal transport during hypoxia. J. Neurochem. 137, 770–781 (2016).
Fogal, V., Hsieh, J. K., Royer, C., Zhong, S. & Lu, X. Cell cycle-dependent nuclear retention of p53 by E2F1 requires phosphorylation of p53 at Ser315. EMBO J. 24, 2768–2782 (2005).
Katayama, H. et al. Phosphorylation by aurora kinase A induces Mdm2-mediated destabilization and inhibition of p53. Nat. Genet. 36, 55–62 (2004).
Slee, E. A. et al. Phosphorylation of Ser312 contributes to tumor suppression by p53 in vivo. Proc. Natl. Acad. Sci. USA 107, 19479–19484 (2010).
Kellum, J. A. et al. Acute kidney injury. Nat. Rev. Dis. Prim. 7, 52 (2021).
Taguchi, K. et al. IL-22 is secreted by proximal tubule cells and regulates DNA damage response and cell death in acute kidney injury. Kidney Int. 105, 99–114 (2024).
Baker, M. L. & Cantley, L. G. Adding insult to injury: the spectrum of tubulointerstitial responses in acute kidney injury. J. Clin. Investig. 135 (2025).
Siew, E. D. et al. Genome-wide association study of hospitalized patients and acute kidney injury. Kidney Int. 106, 291–301 (2024).
Kelly, K. J., Plotkin, Z., Vulgamott, S. L. & Dagher, P. C. P53 mediates the apoptotic response to GTP depletion after renal ischemia-reperfusion: protective role of a p53 inhibitor. J. Am. Soc. Nephrol. 14, 128–138 (2003).
Wei, Q. et al. Activation and involvement of p53 in cisplatin-induced nephrotoxicity. Am. J. Physiol. Ren. Physiol. 293, F1282–F1291 (2007).
Zhang, D. et al. Tubular p53 regulates multiple genes to mediate AKI. J. Am. Soc. Nephrol. 25, 2278–2289 (2014).
Ma, Z. et al. p53/microRNA-214/ULK1 axis impairs renal tubular autophagy in diabetic kidney disease. J. Clin. Investig. 130, 5011–5026 (2020).
Blaydes, J. P. et al. Stoichiometric phosphorylation of human p53 at Ser315 stimulates p53-dependent transcription. J. Biol. Chem. 276, 4699–4708 (2001).
Lee, M. K., Tong, W. M., Wang, Z. Q. & Sabapathy, K. Serine 312 phosphorylation is dispensable for wild-type p53 functions in vivo. Cell Death Differ. 18, 214–221 (2011).
Slee, E. A. & Lu, X. Requirement for phosphorylation of P53 at Ser312 in suppression of chemical carcinogenesis. Sci. Rep. 3, 3105 (2013).
Slee, E. A. & Lu, X. In the right place at the right time: analysis of p53 serine 312 phosphorylation in vivo. Cell Cycle 10, 1345–1346 (2011).
Kruse, J. P. & Gu, W. Modes of p53 regulation. Cell 137, 609–622 (2009).
Lavin, M. F. & Gueven, N. The complexity of p53 stabilization and activation. Cell Death Differ. 13, 941–950 (2006).
Shieh, S. Y., Taya, Y. & Prives, C. DNA damage-inducible phosphorylation of p53 at N-terminal sites including a novel site, Ser20, requires tetramerization. EMBO J. 18, 1815–1823 (1999).
Seternes, O. M., Kidger, A. M. & Keyse, S. M. Dual-specificity MAP kinase phosphatases in health and disease. Biochim. Biophys. Acta Mol. Cell Res. 1866, 124–143 (2019).
Jeffrey, K. L., Camps, M., Rommel, C. & Mackay, C. R. Targeting dual-specificity phosphatases: manipulating MAP kinase signalling and immune responses. Nat. Rev. Drug Discov. 6, 391–403 (2007).
Xiang, H. et al. Dual-specificity phosphatase 26 protects against kidney injury caused by ischaemia-reperfusion through restraint of TAK1-JNK/p38-mediated apoptosis and inflammation of renal tubular epithelial cells. Toxicol. Appl. Pharm. 487, 116954 (2024).
Yu, W. et al. A novel amplification target, DUSP26, promotes anaplastic thyroid cancer cell growth by inhibiting p38 MAPK activity. Oncogene 26, 1178–1187 (2007).
Kim, H. et al. The DUSP26 phosphatase activator adenylate kinase 2 regulates FADD phosphorylation and cell growth. Nat. Commun. 5, 3351 (2014).
Xiao, X. et al. Hypermethylation leads to the loss of HOXA5, resulting in JAG1 expression and NOTCH signaling contributing to kidney fibrosis. Kidney Int. 106, 98–114 (2024).
Guo, C. et al. DNA methylation protects against cisplatin-induced kidney injury by regulating specific genes, including interferon regulatory factor 8. Kidney Int. 92, 1194–1205 (2017).
Funahashi, Y. et al. Nanotherapeutic kidney cell-specific targeting to ameliorate acute kidney injury. Kidney Int. 106, 597–610 (2024).
Guo, Q. et al. Lysozyme-targeted liposomes for enhanced tubular targeting in the treatment of acute kidney injury. Acta Biomater. 192, 394–408 (2025).
Zhang, K. et al. Neuronal dual-specificity phosphatase 26 inhibition via reactive-oxygen-species responsive mesoporous-silica-loaded hydrogel for spinal cord injury repair. ACS Nano 19, 4942–4958 (2025).
Zhao, M. et al. Protein tyrosine phosphatases: emerging role in cancer therapy resistance. Cancer Commun. 44, 637–653 (2024).
Hung, V. et al. Proteomic mapping of the human mitochondrial intermembrane space in live cells via ratiometric APEX tagging. Mol. Cell 55, 332–341 (2014).
Wang, Y. et al. Repression of peroxisome proliferation-activated receptor γ coactivator-1α by p53 after kidney injury promotes mitochondrial damage and maladaptive kidney repair. Kidney Int. 107, 869–887 (2025).
Acknowledgements
This work was supported by the Postdoctoral Fellowship Program and China Postdoctoral Science Foundation (grant no. GZB20250487 to Y.F.), the Scientific Research Project of the Hunan Provincial Health Commission (grant no. 20254762 to Y.F.), the National Natural Science Foundation of China (grant nos. 82222013 and 82470731 to A.C.), the Key Program of the Natural Science Foundation of Hunan Province (grant no. 2025JJ30048 to A.C.), the National Key Clinical Specialty Scientific Research Project of the Second Xiangya Hospital, Central South University (grant no. 02201322 to A.C.), the U.S. Department of Veterans Affairs (grant nos. 1TK6BX005236 and I01BX000319 to Z.D.), and the U.S. National Institutes of Health (grant nos. 5R01DK058831 and 5R01DK087843 to Z.D.).
Author information
Authors and Affiliations
Contributions
Y.F. and Z.D. conceived the project and designed the experiments. Y.F. performed most of the experiments, analyzed the data, and wrote the manuscript. Y.X. and Y.H. contributed to the generation and analysis of the genetically modified mouse models. Y.F. and Y.X. performed bioinformatics analysis and supported statistical interpretation. J.C. and S.D. assisted with histological evaluation and clinical sample acquisition. A.C. and Z.D. supervised the entire project and revised the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Akihito Hishikawa, Kaori Hayashi, Ling Zheng, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fu, Y., Xiang, Y., Han, Y. et al. DUSP26 protects against acute kidney injury by dephosphorylating p53 at serine 312. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69688-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69688-3