Abstract
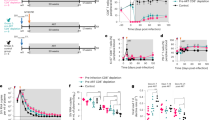
Mechanisms underlying durable control of HIV after antiretroviral therapy interruption remain poorly understood. Here we provide a comprehensive longitudinal analysis in a non-human primate model of post-treatment control using SIVmac251-infected male cynomolgus macaques (pVISCONTI study). Controllers exhibit lower levels of SIV DNA, intact proviruses, transcriptional activity, and viral evolution compared to non-controllers in blood and tissues long after therapy interruption. Before interruption, controllers already have fewer intact proviruses in lymph nodes, and this difference persists in blood shortly after interruption, prior to viral rebound. Intact provirus levels in lymph nodes before interruption negatively correlate with CD8⁺ T-cell capacity to suppress SIV and reflect rebound magnitude. The study demonstrates that markers of post-treatment control are detectable in lymph nodes before therapy interruption and in blood shortly after, and suggests that host immune responses may shape intact provirus profiles during treatment.
Similar content being viewed by others
Data availability
Sequencing data generated in this study (proviral DNA sequences from tissue samples collected at euthanasia and plasma viral RNA sequences from longitudinal timepoints) have been deposited in the Sequence Read Archive (SRA) under BioProject accession code PRJNA1256680 [http://www.ncbi.nlm.nih.gov/bioproject/1256680]. Basecalled FASTQ files are provided for all runs. Metadata for each macaque, tissue type and timepoints are included in the associated BioSamples. Additionnaly, the data that support the findings of this study are presented in the main figures and Supplementary Information/Source Data files. Source data are provided with this paper. Further information and requests for resources and reagents should be directed to the lead contact. Request for biological resources will be fulfilled based on availability and upon the establishment of an MTA. Source data are provided with this paper.
References
Avettand-Fènoël, V. et al. Total HIV-1 DNA, a marker of viral reservoir dynamics with clinical implications. Clin. Microbiol. Rev. 29, 859–880 (2016).
Calin, R. Treatment interruption in chronically HIV-infected patients with an ultralow HIV reservoir. AIDS 30, 761–769 (2016).
Li, J. Z. Time to viral rebound after interruption of modern antiretroviral therapies. Clin. Infect. Dis. 74, 865–870 (2022).
Li, J. Z. The size of the expressed HIV reservoir predicts timing of viral rebound after treatment interruption. AIDS 30, 343–353 (2016).
Wong, J. K. Recovery of replication-competent HIV despite prolonged suppression of plasma viremia. Science 278, 1291–1295 (1997).
Chomont, N. HIV reservoir size and persistence are driven by T cell survival and homeostatic proliferation. Nat. Med. 15, 893–900 (2009).
Hiener, B. et al. Identification of genetically intact HIV-1 proviruses in specific CD4+ T cells from effectively treated participants. Cell Rep. 21, 813–822 (2017).
Chun, T.-W. et al. Quantification of latent tissue reservoirs and total body viral load in HIV-1 infection. Nature 387, 183–188 (1997).
Chun, T.-W. et al. In vivo fate of HIV-1-infected T cells: quantitative analysis of the transition to stable latency. Nat. Med. 1, 1284–1290 (1995).
Chun, T. W. Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc. Natl. Acad. Sci. USA 94, 13193–13197 (1997).
Lee, G. Q. et al. Clonal expansion of genome-intact HIV-1 in functionally polarized Th1 CD4+ T cells. J. Clin. Investig. 127, 2689–2696 (2017).
Ho, Y. C. Replication-competent noninduced proviruses in the latent reservoir increase barrier to HIV-1 cure. Cell 155, 540–551 (2013).
Finzi, D. et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science 278, 1295–1300 (1997).
Charre, C., Merad, Y. & Avettand-Fenoel, V. HIV-1 reservoir landscape of post-treatment control. Curr. Opin. HIV AIDS 20, 99–108 (2025).
Sáez-Cirión, A. et al. Post-treatment HIV-1 controllers with a long-term virological remission after the interruption of early initiated antiretroviral therapy ANRS VISCONTI Study. PLoS Pathog 9, e1003211 (2013).
Gunst, J. D. et al. Time to HIV viral rebound and frequency of post-treatment control after analytical interruption of antiretroviral therapy: an individual data-based meta-analysis of 24 prospective studies. Nat. Commun. 16, 906 (2025).
Namazi, G. et al. The control of HIV after antiretroviral medication pause (CHAMP) study: Posttreatment Controllers Identified From 14 Clinical Studies. J. Infect. Dis. 218, 1954–1963 (2018).
Murray, J. M. Latent HIV dynamics and implications for sustained viral suppression in the absence of antiretroviral therapy. J. Virus Erad. 4, 91–98 (2018).
Passaes, C. et al. Early antiretroviral therapy favors post-treatment SIV control associated with the expansion of enhanced memory CD8+ T-cells. Nat. Commun. 15, 178 (2024).
Daly, M. B. et al. SHIV remission in macaques with early treatment initiation and ultra long-lasting antiviral activity. Nat. Commun. 15, 10550 (2024).
Hocqueloux, L. et al. Long-term antiretroviral therapy initiated during primary HIV-1 infection is key to achieving both low HIV reservoirs and normal T cell counts. J. Antimicrob. Chemother. 68, 1169–1178 (2013).
Colby, D. J. Rapid HIV RNA rebound after antiretroviral treatment interruption in persons durably suppressed in Fiebig I acute HIV infection. Nat. Med. 24, 923–926 (2018).
Assoumou, L. A low HIV-DNA level in peripheral blood mononuclear cells at antiretroviral treatment interruption predicts a higher probability of maintaining viral control. AIDS 29, 2003–2007 (2015).
Sáez-Cirión, A. HIV controllers exhibit potent CD8 T cell capacity to suppress HIV infection ex vivo and peculiar cytotoxic T lymphocyte activation phenotype. Proc. Natl. Acad. Sci. USA 104, 6776–6781 (2007).
Etemad, B. HIV post-treatment controllers have distinct immunological and virological features. Proc. Natl. Acad. Sci. USA 120, e2218960120 (2023).
Hurst, J. Immunological biomarkers predict HIV-1 viral rebound after treatment interruption. Nat. Commun. 6, 8495 (2015).
Avettand-Fenoel, V. et al. HIV-DNA in rectal cells is well correlated with HIV-DNA in blood in different groups of patients, including long-term non-progressors. AIDS Lond. Engl. 22, 1880–1882 (2008).
Rose, R. et al. HIV maintains an evolving and dispersed population in multiple tissues during suppressive combined antiretroviral therapy in individuals with cancer. J. Virol. 90, 8984–8993 (2016).
Lamers, S. L. et al. HIV DNA is frequently present within pathologic tissues evaluated at autopsy from combined antiretroviral therapy-treated patients with undetectable viral loads. J. Virol. 90, 8968–8983 (2016).
De Scheerder, M.-A. et al. HIV rebound is predominantly fueled by genetically identical viral expansions from diverse reservoirs. Cell Host Microbe. 26, 347–358.e7 (2019).
Chaillon, A. et al. HIV persists throughout deep tissues with repopulation from multiple anatomical sources. J. Clin. Investig. 130, 1699–1712 (2020).
Dufour, C. et al. Near full-length HIV sequencing in multiple tissues collected postmortem reveals shared clonal expansions across distinct reservoirs during ART. Cell Rep. 42, 113053 (2023).
Ganusov, V. V. & De Boer, R. J. Do most lymphocytes in humans really reside in the gut?. Trends Immunol. 28, 514–518 (2007).
Chun, T.-W. et al. Persistence of HIV in gut-associated lymphoid tissue despite long-term antiretroviral therapy. J. Infect. Dis. 197, 714–720 (2008).
Yukl, S. A. et al. The distribution of HIV DNA and RNA in cell subsets differs in gut and blood of HIV-positive patients on ART: implications for viral persistence. J. Infect. Dis. 208, 1212–1220 (2013).
Estes, J. D. et al. Defining total-body AIDS-virus burden with implications for curative strategies. Nat. Med. 23, 1271–1276 (2017).
Leyre, L. et al. Abundant HIV-infected cells in blood and tissues are rapidly cleared upon ART initiation during acute HIV infection. Sci. Transl. Med. 12, eaav3491 (2020).
Benlhassan-Chahour, K. Kinetics of lymphocyte proliferation during primary immune response in macaques infected with pathogenic simian immunodeficiency virus SIVmac251: preliminary report of the effect of early antiviral therapy. J Virol 77, 12479–12493 (2003).
Fray, E. J. et al. Antiretroviral therapy reveals triphasic decay of intact SIV genomes and persistence of ancestral variants. Cell Host Microbe. 31, 356–372.e5 (2023).
Mannioui, A. Dynamics of viral replication in blood and lymphoid tissues during SIVmac251 infection of macaques. Retrovirology 6, 106, (2009).
Policicchio, B. B., Pandrea, I. & Apetrei, C. Animal models for HIV cure research. Front. Immunol. 7, 12 (2016).
Goulder, P. & Deeks, S. G. HIV control: Is getting there the same as staying there?. PLOS Pathog. 14, e1007222 (2018).
Mastrangelo, A. et al. Anti-HIV antibodies are representative of the latent reservoir but do not correlate with viral control in people with long-lasting virological suppression undergoing analytical treatment interruption (APACHE study). J. Antimicrob. Chemother. 76, 1646–1648 (2021).
Jambo, K. C. et al. Small alveolar macrophages are infected preferentially by HIV and exhibit impaired phagocytic function. Mucosal Immunol. 7, 1116–1126 (2014).
Clarke, J. R. et al. HIV-1 proviral DNA copy number in peripheral blood leucocytes and bronchoalveolar lavage cells of AIDS patients. Clin. Exp. Immunol. 96, 182–186 (1994).
Costiniuk, C. T. et al. HIV persistence in mucosal CD4+ T cells within the lungs of adults receiving long-term suppressive antiretroviral therapy. AIDS Lond. Engl. 32, 2279–2289 (2018).
Canaud, G. et al. The kidney as a reservoir for HIV-1 after renal transplantation. J. Am. Soc. Nephrol. 25, 407–419 (2014).
Hughes, K. et al. HIV-1 infection of the kidney: mechanisms and implications. AIDS 35, 359–367 (2021).
Baker, E. J., Hughes, K., Travieso, T., Klotman, M. E. & Blasi, M. Establishment, persistence, and reactivation of latent HIV-1 infection in renal epithelial cells. J. Virol. 96, e00624–22 (2022).
Trémeaux, P. et al. In-depth characterization of full-length archived viral genomes after nine years of posttreatment HIV control. Microbiol. Spectr. 11, e0326722 (2023).
Abrahams, M.-R. et al. The replication-competent HIV-1 latent reservoir is primarily established near the time of therapy initiation. Sci. Transl. Med. 11, eaaw5589 (2019).
Lichterfeld, M., Gao, C. & Yu, X. G. An ordeal that does not heal: understanding barriers to a cure for HIV-1 infection. Trends Immunol 43, 608–616 (2022).
Cadena, A. M. et al. Persistence of viral RNA in lymph nodes in ART-suppressed SIV/SHIV-infected Rhesus Macaques. Nat. Commun. 12, 1474 (2021).
Solis-Leal, A. et al. Lymphoid tissues contribute to plasma viral clonotypes early after antiretroviral therapy interruption in SIV-infected rhesus macaques. Sci. Transl. Med. 15, eadi9867 (2023).
Passaes, C. Optimal maturation of the SIV-specific CD8+ T cell response after primary infection is associated with natural control of SIV: ANRS SIC study. Cell Rep 32, 108174 (2020).
Harper, J. IL-21 and IFNalpha therapy rescues terminally differentiated NK cells and limits SIV reservoir in ART-treated macaques. Nat Commun. 12, 2866 (2021).
Climent, N. et al. Immunological and virological findings in a patient with exceptional post-treatment control: a case report. Lancet HIV 10, e42–e51 (2023).
Essat, A. et al. A genetic fingerprint associated with durable HIV remission after interruption of antiretroviral treatment: ANRS VISCONTI/PRIMO. Med 100670 https://doi.org/10.1016/j.medj.2025.100670 (2025).
Armani-Tourret, M. et al. Immune targeting of HIV-1 reservoir cells: a path to elimination strategies and cure. Nat. Rev. Microbiol. 22, 328–344 (2024).
Lian, X. et al. Progressive transformation of the HIV-1 reservoir cell profile over two decades of antiviral therapy. Cell Host Microbe. 31, 83–96.e5 (2023).
Li, J. Z. et al. Predictors of HIV rebound differ by timing of antiretroviral therapy initiation. JCI Insight 9, e173864 (2024).
Mesquita, F. S., Li, Y. & Li, J. Z. Viral and immune predictors of HIV posttreatment control. Curr. Opin. HIV AIDS 20, 54–60 (2025).
Giron, L. B., Pasternak, A. O. & Abdel-Mohsen, M. Soluble markers of viral rebound and post-treatment HIV control. Curr. Opin. HIV AIDS 20, 61–69 (2025).
Wedrychowski, A. Transcriptomic signatures of human immunodeficiency virus post-treatment control. J. Virol. 97, 0125422 (2023).
Hofmann-Lehmann, R. Sensitive and robust one-tube real-time reverse transcriptase-polymerase chain reaction to quantify SIV RNA load: comparison of one- versus two-enzyme systems. AIDS Res. Hum. Retroviruses. 16, 1247–1257 (2000).
Bruner, K. M. A quantitative approach for measuring the reservoir of latent HIV-1 proviruses. Nature 566, 120–125 (2019).
Bender, A. M. et al. The landscape of persistent viral genomes in ART-Treated SIV, SHIV, and HIV-2 infections. Cell Host Microbe. 26, 73–85.e4 (2019).
Policicchio, B. B. et al. Dynamics of simian immunodeficiency virus two-long-terminal-repeat circles in the presence and absence of CD8+ cells. J. Virol. 92, e02100–e02117 (2018).
Levy, C. N. et al. A highly multiplexed droplet digital PCR assay to measure the intact HIV-1 proviral reservoir. Cell Rep. Med. 2, 100243 (2021).
Cai, D. & Sun, Y. Reconstructing viral haplotypes using long reads. Bioinform. Oxf. Engl. 38, 2127–2134 (2022).
Pinzone, M. R. et al. Longitudinal HIV sequencing reveals reservoir expression leading to decay which is obscured by clonal expansion. Nat. Commun. 10, 728 (2019).
Tamura, K. & Nei, M. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol.https://doi.org/10.1093/oxfordjournals.molbev.a040023 (1993).
Nguyen, L.-T., Schmidt, H. A., von Haeseler, A. & Minh, B. Q. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274 (2015).
Sáez-Cirión, A., Shin, S. Y., Versmisse, P., Barré-Sinoussi, F. & Pancino, G. Ex vivo T cell–based HIV suppression assay to evaluate HIV-specific CD8+ T-cell responses. Nat. Protoc. 5, 1033–1041 (2010).
Acknowledgements
This study was funded by MSDAvenir though a research grant to the ANRS-RHIVIERA consortium, and the ANRS | Emerging infectious diseases French agency. A.M., E.G., and M.F. were supported by ANRS. IDMIT was supported by ANR French agency under references ANR-11-INBS-0008 and ANR-10-EQPX-02-01. We thank animal care workers, in particular Sebastien Langlois, Benoit Delache, Clare-Maelle Fovet, Maxime Pottier, Jean-Marie Robert as well as Julie Morin, Laetitia Bossevot, Brice Targat, Wesley Gros, Marco Leonec for expert technical assistance and Isabelle Mangeot-Méderlé for helpful project management at IDMIT. FTC, DTG, and TDF were obtained from Gilead and ViiV Healthcare through the “IAS Towards an HIV Cure” common Material Transfer Agreement. The SIV1C cell line was kindly provided by François Villinger.
Author information
Authors and Affiliations
Contributions
C.R., R.L.G., and A.S.C. designed the pVISCONTI program with the contribution of D.D., O.L., M.M.T., N.D.B., and V.A.F. C.R. and V.A.F. designed the virological study of this program. C.R., R.L.G, A.S.C., and V.A.F. obtained funding. C.C., A.M, A.Mi., and V.A.F. designed the experiments. C.C., A.M., E.G., D.D., C.P., A.Mi., M.F., N.D., V.M., and N.D.B. carried out the experiments. C.C., A.M., A.C., E.G., D.D., C.P., A.Mi, M.F., N.D., V.M., N.D.B., R.L.G., A.S.C., and V.A.F. analyzed the data. C.C., A.M., A.C., M.M.T., C.R., R.L.G., A.S.C., and V.A.F. interpreted the results. V.A.F. supervised the study. C.C. and V.A.F. wrote the manuscript with assistance from A.M. All authors critically reviewed the manuscript and contributed to the final version.
Corresponding authors
Ethics declarations
Competing interests
C.C. honoraria and travel grants from MSD, ViiV Healthcare and Gilead Sciences for participation in educational programs and conferences. A.S.C. has received speaker fees from MSD, ViiV Healthcare, Gilead, Janssen. V.A.F. has received grants (to her institution) from ViiV Healthcare and honoraria and travel grants from ViiV Healthcare and Gilead Sciences for participation in educational programs and conferences. The other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ole Søgaard, who co-reviewed with Katie Fisher and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Charre, C., Melard, A., Chaillon, A. et al. Post-treatment SIV control is associated with specific features of viral persistence before and after treatment interruption. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69720-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69720-6