Abstract
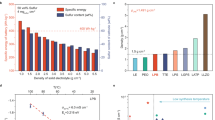
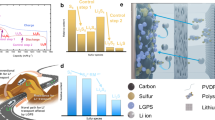
All-solid-state batteries using sulfur-based positive electrodes (cathodes) offer a cost-effective route to achieve high specific energy. However, low active material utilization and cycle life hinder performance. Here, we demonstrate a positive electrode design that employs sulfide solid-state electrolytes, where a high energy synthesis approach forms a metastable and ionically conductive interphase on the active material surface. This interphase facilitates high active material utilization and contributes capacity with cycling. We also show that tailoring active material particle sizes to the micron-scale improves rate performance and cycling stability. Structural analysis reveals that the substantial volume change of sulfur-based positive electrodes during operation can partially offset that of the negative electrodes, thereby mitigating internal mechanical stress. The combined design principles enable sulfur areal capacities up to 11 mAh cm-2 while maintaining stable cycling at 25 °C. We further demonstrate several specific-energy-focused cell architectures, particularly a Li2S anode-free pouch cell that operates under “low stack pressure” of 10 MPa. This work outlines practical design strategies for constructing high-specific-energy all-solid-state batteries for a broad range of emerging applications.
Similar content being viewed by others
Data availability
Source data are provided with this paper.
References
Marjolin, A. Lithium-ion battery capacity to grow steadily to 2030. S&P Global Market Intelligence (2023).
Adu-Gyamfi, B. A. & Good, C. Electric aviation: a review of concepts and enabling technologies. Transpor. Eng. 9, 100134 (2022).
Dornbusch, D. A., Viggiano, R. P., Connell, J. W., Lin, Y., Lvovich, V. F. Practical considerations in designing solid state Li-S cells for electric aviation. Electrochim Acta, 403, 139406 (2022).
Manthiram, A., Fu, Y. & Su, Y. S. Challenges and prospects of lithium-sulfur batteries. Acc. Chem. Res 46, 1125–1134 (2013).
Sun, J. et al. Will lithium-sulfur batteries be the next beyond-lithium-ion batteries and even much better? InfoMat https://doi.org/10.1002/inf2.12359 (2022).
Akridge, J. R. & Mikhaylik, Y. V. N. White, Li/S fundamental chemistry and application to high-performance rechargeable batteries. Solid State Ion. 175, 243–245 (2004).
Ren, W., Ma, W., Zhang, S. & Tang, B. Recent advances in shuttle effect inhibition for lithium sulfur batteries. Energy Storage Mater. 23, 707–732 (2019).
Ohno, S. & Zeier, W. G. Toward practical solid-state lithium-sulfur batteries: challenges and perspectives. Acc. Mater. Res. 2, 869–880 (2021).
Manthiram, A., Fu, Y., Chung, S. H., Zu, S. & Su, Y. S. Rechargeable lithium-sulfur batteries. Chem. Rev. 114, 11751–11787 (2014).
Lewis, J. A., Tippens, J., Cortes, F. J. Q. & McDowell, M. T. Chemo-mechanical challenges in solid-state batteries. Trends Chem. 1, 845–857 (2019).
Chen, M. & Adams, S. High performance all-solid-state lithium/sulfur batteries using lithium argyrodite electrolyte. J. Solid-State Electrochem. 19, 697–702 (2015).
Kinoshita, S., Okuda, K., Machida, N., Naito, M. & Sigematsu, T. All-solid-state lithium battery with sulfur/carbon composites as positive electrode materials. Solid State Ion. 256, 97–102 (2014).
Yu, C., van Eijck, L., Ganapathy, S. & Wagemaker, M. Synthesis, structure, and electrochemical performance of the argyrodite Li6PS5Cl solid electrolyte for Li-ion solid state batteries. Electrochim. Acta 215, 93–99 (2016).
Zhu, X. et al. Exploring the concordant solid-state electrolytes for all-solid-state lithium-sulfur batteries. Nano Energy 96, 107093 (2022).
Kim, H., Choi, H. N., Hwang, J. Y., Yoon, C. S., Sun, Y. K. Tailoring the interface between sulfur and sulfide solid electrolyte for high-areal-capacity all-solid-state lithium-sulfur batteries. ACS Energy Lett. 3971–3979 (2023).
Wang, S. et al. High-conductivity argyrodite Li6PS5Cl solid electrolytes prepared via optimized sintering processes for all-solid-state lithium-sulfur batteries. ACS Appl. Mater. Interfaces 10, 42279–42285 (2018).
Zhang, Y. et al. High-performance all-solid-state lithium-sulfur batteries with sulfur/carbon nano-hybrids in a composite cathode. J. Mater. Chem. A Mater. 6, 23345–23356 (2018).
Yao, X. et al. High-performance all-solid-state lithium–sulfur batteries enabled by amorphous sulfur-coated reduced graphene oxide cathodes. Adv. Energy Mater 7, 1602923 (2017).
Alzahrani, A. S. et al. Confining sulfur in porous carbon by vapor deposition to achieve high-performance cathode for all-solid-state lithium-sulfur batteries. ACS Energy Lett. 6, 413–418 (2021).
Zhou, J. et al. Healable and conductive sulfur iodide for solid-state Li–S batteries. Nature 627, 301–305 (2024).
Fu, K. et al. Three-dimensional bilayer garnet solid electrolyte based high energy density lithium metal-sulfur batteries. Energy Environ. Sci. 10, 1568–1575 (2017).
Yang, H. et al. Catalytic solid-state sulfur conversion confined in micropores toward superhigh coulombic efficiency lithium-sulfur batteries. Adv. Energy Mater https://doi.org/10.1002/aenm.202400249 (2024).
Liu, Y., Meng, X., Wang, Z. & Qiu, J. A Li2S-based all-solid-state battery with high energy and superior safety. Sci. Adv. 8, eabl8390 (2022).
Zhu, X., Wang, L., Bai, Z., Lu, J., Wu, T. Sulfide-based all-solid-state lithium–sulfur batteries: challenges and perspectives. Nanomicro. Lett. 15, 75 (2023).
Wang, D. et al. Realizing high-capacity all-solid-state lithium-sulfur batteries using a low-density inorganic solid-state electrolyte. Nat Commun. 14, 1895 (2023).
Tan, D. H. S. et al. Elucidating reversible electrochemical redox of Li6PS5Cl solid electrolyte. ACS Energy Lett. 4, 2418–2427 (2019).
Jang, J. et al. Enabling a co-free, high-voltage LiNi0.5Mn1.5O4 cathode in all-solid-state batteries with a halide electrolyte. ACS Energy Lett. 7, 2531–2539 (2022).
Cronk, A. et al. Overcoming the interfacial challenges of LiFePO4 in inorganic all-solid-state batteries. ACS Energy Lett. 8, 827–835 (2023).
Schwietert, T. et al. Clarifying the relationship between redox activity and electrochemical stability in solid electrolytes. Nat. Mater. 19, 428–435 (2020).
Ohno, S., Rosenbach, C., Dewald, G. F., Janek, J. & Zier, W. G. Linking solid electrolyte degradation to charge carrier transport in the thiophosphate-based composite cathode toward solid-state lithium-sulfur batteries. Adv. Funct. Mater. 31, 2010620 (2021).
Li, M. et al. Electrochemically primed functional redox mediator generator from the decomposition of solid state electrolyte. Nat. Comm. 10, 1890 (2019).
Lin, Z., Liu, Z., Fu, W., Dudney, N. J. & Liang, C. Lithium polysulfidophosphates: a family of lithium-conducting sulfur-rich compounds for lithium-sulfur batteries. Angew. Chem. Int. Ed. 52, 7460–7463 (2013).
Tanibata, N. et al. A novel discharge-charge mechanism of a S-P2S5 composite electrode without electrolytes in all-solid-state Li/S batteries. J. Mater. Chem. A 5, 11224 (2017).
Burmeister, C., Titscher, L., Breitung-Faes, S. & Kwade, A. Dry grinding in planetary ball mills: evaluation of a stressing model. Adv. Powder Technol. 29, 191–201 (2017).
Hofer, M. et al. Effective mechanochemical synthesis of sulfide solid electrolyte Li3PS4 in a high energy ball mill by process investigation. Adv. Powder Technol. 34, 104004 (2023).
Zheng, C. et al. High-performance all-solid-state lithium–sulfur batteries enabled by slurry-coated Li6PS5Cl/S/C composite electrodes. Front Energy Res. 8, 1602923 (2021).
Zhang, H. P. anM. et al. Carbon-free and binder-free Li-Al alloy anode enabling an all-solid-state Li-S battery with high energy and stability. Sci. Adv. 8, 4372 (2022).
Gao, X. et al. All-solid-state lithium-sulfur batteries enhanced by redox mediators. J. Am. Chem. Soc. 143, 18188–18195 (2021).
Ohno, S. et al. Observation of chemomechanical failure and the influence of cutoff potentials in all-solid-state Li-S batteries. Chem. Mater. 31, 2930–2940 (2019).
Gamo, H., Hikima, K. & Matsuda, A. Understanding decomposition of electrolytes in all-solid-state lithium-sulfur batteries. Chem. Mater. 34, 10952–10963 (2022).
Kim, J. T. et al. All-solid-state lithium–sulfur batteries through a reaction engineering lens. Nat. Chem. Eng. 1, 400–410 (2024).
Li, L., Ma, Z. & Li, Y. Accurate determination of optimal sulfur content in mesoporous carbon hosts for high-capacity stable lithium-sulfur batteries. Carbon 197, 200–208 (2022).
Ward, A. T. Raman spectroscopy of sulfur, sulfur-selenium, and sulfur-arsenic mixtures. J. Phys. Chem. 72, 4133–4139 (1968).
Kato, A., Yamamoto, M., Utsuno, F., Higuchi, H. & Takahashi, M. Lithium-ion-conductive sulfide polymer electrolyte with disulfide bond-linked PS4 tetrahedra for all-solid-state batteries. Commun. Mater. 2, 122 (2021).
Dominko, R. et al. Polysulfides formation in different electrolytes from the perspective of X-ray absorption spectroscopy. J. Electrochem. Soc. 165, A5014–A5019 (2018).
Tachez, M., Malugani, J.-P., Mercier, R. & Robert, G. Ionic Conductivity of and phase transition in lithium thiophosphate Li3PS4. Solid State Ion. 14, 181–185 (1984).
Hakari, T. et al. Structural and electronic-state changes of a sulfide solid electrolyte during the Li dinsertion-insertion processes. Chem. Mater. 29, 4768–4774 (2017).
Cuisinier, M. et al. Sulfur speciation in Li-S batteries determined by operando X-ray absorption spectroscopy. J. Phys. Chem. Lett. 4, 3227–3232 (2013).
Manceau, A. & Nagy, K. L. Quantitative analysis of sulfur functional groups in natural organic matter by XANES spectroscopy. Geochim. Cosmochim. Acta 99, 206–223 (2012).
Santhosha, A. L., Medenbach, L., Buchheim, J. R. & Adelhelm, P. The indium−lithium electrode in solid-state lithium-ion batteries: phase formation, redox potentials, and interface stability. Batteries Supercaps 2, 524–529 (2019).
Mei, B. A., Munteshari, O., Lau, J., Dunn, B. & Pilon, L. Physical interpretations of Nyquist plots for EDLC electrodes and devices. J. Phys. Chem. C. 122, 194–206 (2018).
Gu, J., Liang, Z., Shi, J. & Yang, Y. Electrochemo-mechanical stresses and their measurements in sulfide-based all-solid-state batteries: a review. Adv. Energy Mater. 13, 2203153 (2023).
Zhong, H. et al. Nano-scale interface engineering of sulfur cathode to enable high-performance all-solid-state Li–S batteries. Adv Funct. Mater. https://doi.org/10.1002/adfm.202315925 (2024).
Ji, X., Lee, K. T. & Nazar, L. F. A highly ordered nanostructured carbon-sulphur cathode for lithium-sulphur batteries. Nat. Mater. 8, 500–506 (2009).
Han, F. et al. High-performance all-solid-state lithium-sulfur battery enabled by a mixed-conductive Li2S nanocomposite. Nano Lett. 16, 4521–4527 (2016).
Schiele, A. et al. High-throughput in situ pressure analysis of lithium-ion batteries. Anal. Chem. 89, 8122–8128 (2017).
Deng, Z., Wang, Z., Chu, I.-H., Luo, J. & Ong, S. P. Elastic properties of alkali superionic conductor electrolytes from first principles calculations. J. Electrochem. Soc. 163, A67–A74 (2016).
Tan, D. H. S. et al. Carbon-free high-loading silicon anodes enabled by sulfide solid electrolytes. Science 373, 1494–1499 (2021).
Reimers, J. N. & Dahn, J. R. Electrochemical and in situ x-ray diffraction studies of lithium intercalation in LixCoO2. J. Electrochem. Soc. 139, 2091–2097 (1992).
Huo, H. et al. Chemo-mechanical failure mechanisms of the silicon anode in solid-state batteries. Nat. Mater. 23, 543–551 (2024).
Fujita, Y. et al. Dynamic volume change of Li2S-based active material and the influence of stacking pressure on capacity in all-solid-state batteries. Chem. Mater. 36, 7533–7540 (2024).
Koerver, R. et al. Chemo-mechanical expansion of lithium electrode materials-on the route to mechanically optimized all-solid-state batteries. Energy Environ. Sci. 11, 2142–2158 (2018).
Tan, D. H. S., Meng, Y. S. & Jang, J. Scaling up high-energy-density sulfidic solid-state batteries: a lab-to-pilot perspective. Joule 6, 1755–1769 (2022).
Ham, S. Y. et al. Overcoming low initial coulombic efficiencies of Si anodes through prelithiation in all-solid-state batteries. Nat. Commun. 15, 2991 (2024).
Lee, Y. G. et al. High-energy long-cycling all-solid-state lithium metal batteries enabled by silver–carbon composite anodes. Nat. Energy 5, 299–308 (2020).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Duquesnoy, M., Lombardo, T., Chouchane, M., Primo, E. N., Franco, A. A. Data-driven assessment of electrode calendering process by combining experimental results, in silico mesostructures generation and machine learning. J. Power Sources 480, 229103 (2020).
Cooper, S. J., Bertei, A., Shearing, P. R., Kilner, J. A. & Brandon, N. P. TauFactor: an open-source application for calculating tortuosity factors from tomographic data. SoftwareX 5, 203–210 (2016).
Tran, A. P., Yan, S. & Fang, Q. Improving model-based functional near-infrared spectroscopy analysis using mesh-based anatomical and light-transport models. Neurophotonics 7, 1 (2020).
Acknowledgements
This work was supported by the LG Energy Solution – U.C. San Diego Frontier Research Laboratory (FRL) Program (A.C., S.-Y.H., C.H., H.Y., M.V., C.L., D.L., M.-S.S., J.J., J.B.L., and Y.S.M.). A.C. acknowledges the National Science Foundation for having supported their Ph.D. research through the NSF Graduate Research Fellowship Program. The authors (A.C., B.S., and P.R.) would like to acknowledge the UCSD Crystallography Facility. This work was also performed in part at the San Diego Nanotechnology Infrastructure (SDNI) of UCSD, a member of the National Nanotechnology Coordinated Infrastructure, which is supported by the National Science Foundation (Grant ECCS−1542148, A.C., S.-Y.H., S.B., D.C., G.D., and M.V.), along with the use of facilities and instrumentation supported by NSF through the UC San Diego Materials Research Science and Engineering Center (UCSD MRSEC) (Grant DMR-201192). The authors (A.C., C.H., Y.S.M.) would like to acknowledge Prof. Bing Joe Hwang and Ms. Chia-Yu Chang for their help on XAS measurements at the Taiwan Light Source (TLS) beamline 16A1 of the National Synchrotron Radiation Research Center (NSRRC) in Hsinchu, Taiwan. The authors (A.C. and Y.S.M.) thank Dr. Jinkwan Jung for his assistance in sample preparation and cell fabrication.
Author information
Authors and Affiliations
Contributions
A.C. and Y.S.M. conceived the ideas for the study. A.C., J.B.L., and X.W. designed the experiments. B.S. and P.R. collected XRD measurements. S.B. contributed cryo-TEM characterization and investigation. C.H. performed XAS and analysis. A.C., M.V., and D.C. performed SEM-FIB. M.C. performed the electrode modeling and FEM simulations. C.L. and D.L. fabricated and evaluated the pouch cells. J.A.S.O., G.D., S.-Y.H., H.Y., M.V., J.J., M.-S.S., J.B.L., and Y.S.M. participated in the scientific discussion and data analysis. A.C. wrote the manuscript. Y.S.M. supervised the project. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Y.S.M., A.C., J.B.L., and M.-S.S. declare that two patents were filed for this work through UC San Diego’s Office of Innovation and Commercialization and LG Energy Solution, Ltd. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Daiwei Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. [A peer review file is available.]
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cronk, A., Wang, X., Oh, J.A.S. et al. A highly utilized and practical lithium-sulfur positive electrode enabled in all-solid-state batteries. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69750-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69750-0