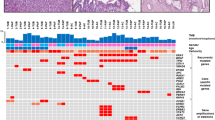
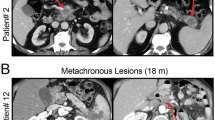
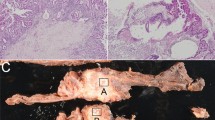
Abstract
Intraductal papillary mucinous neoplasms (IPMNs) are critical precursors to pancreatic ductal adenocarcinoma, a highly lethal cancer characterized by late detection and rapid progression. Here we integrate multi-region whole-genome and transcriptome sequencing to trace the evolution of IPMN, constructing detailed phylogenetic trees to provide insights into subclonal architectures and progression pathways. Our analysis identifies two distinct evolutionary trajectories: one driven by a single ancestral clone, and another involving multiple independent ancestral clones. We further explore the roles of mutational signatures and structural variants in promoting clonal evolution and the emergence of new subclones. Complementing these genomic findings, our transcriptomic analysis reveals unique gene expression profiles and variations in the immune landscape that correlate with different progression stages of IPMN. These insights reveal the complex molecular dynamics of IPMN heterogeneity and progression, highlighting the need to refine early detection and treatment strategies.
Similar content being viewed by others
Data availability
Whole-genome sequencing (WGS) and RNA-seq raw data have been deposited in the European Genome-phenome Archive (EGA) under accession numbers EGAS50000001182 and EGAS50000001540 respectively. Processed datasets supporting the findings of this study are available at the following repositories: https://github.com/Wedge-lab/IPMNPDACpaperArchive/tree/main/IPMNPDAC_WGS/Data, https://github.com/Wedge-lab/IPMNPDACpaperArchive/tree/main/IPMNPDAC_WGS/Data/sigDPC, https://github.com/Wedge-lab/IPMNPDACpaperArchive/tree/main/IPMNPDAC_WGS/Data/svDriverCluster, https://github.com/Wedge-lab/IPMNPDACpaperArchive/tree/main/IPMNPDAC_WGS/Data/cnvDriverCluster Source data are provided with this paper.
Code availability
The Code and data used for the analysis are available at: https://gitfront.io/r/xtgitfhe2/8f2rTdyZs1S9/IPMNPDACpaperArchive/ [gitfront.io].
References
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Ohtsuka, T. et al. International evidence-based Kyoto guidelines for the management of intraductal papillary mucinous neoplasm of the pancreas. Pancreatology 24, 255–270 (2024).
Notta, F. et al. A renewed model of pancreatic cancer evolution based on genomic rearrangement patterns. Nature 538, 378–382 (2016).
Waddell, N. et al. Whole genomes redefine the mutational landscape of pancreatic cancer. Nature 518, 495–501 (2015).
Sakamoto, H. et al. The evolutionary origins of recurrent pancreatic cancer. Cancer Discov. 10, 792–805 (2020).
Chan-Seng-Yue, M. et al. Transcription phenotypes of pancreatic cancer are driven by genomic events during tumor evolution. Nat. Genet. 52, 231–240 (2020).
Pea, A. et al. Targeted DNA sequencing reveals patterns of local progression in the pancreatic remnant following resection of intraductal papillary mucinous neoplasm (IPMN) of the pancreas. Ann. Surg. 266, 133–141 (2017).
Noe, M. et al. Genomic characterization of malignant progression in neoplastic pancreatic cysts. Nat. Commun. 11, 4085 (2020).
Fischer, C. G. et al. Intraductal papillary mucinous neoplasms arise from multiple independent clones, each with distinct mutations. Gastroenterology 157, 1123–1137 e22 (2019).
Basturk, O. et al. A revised classification system and recommendations from the baltimore consensus meeting for neoplastic precursor lesions in the pancreas. Am. J. Surg. Pathol. 39, 1730–1741 (2015).
Makohon-Moore, A. P. et al. Limited heterogeneity of known driver gene mutations among the metastases of individual patients with pancreatic cancer. Nat. Genet. 49, 358–366 (2017).
Luchini, C. et al. Comprehensive characterisation of pancreatic ductal adenocarcinoma with microsatellite instability: histology, molecular pathology and clinical implications. Gut 70, 148–156 (2021).
Gerstung, M. et al. The evolutionary history of 2,658 cancers. Nature 578, 122–128 (2020).
Cancer Genome Atlas Research Network Electronic address, a.a.d.h.e. & Cancer Genome Atlas Research, N. Integrated genomic characterization of pancreatic ductal adenocarcinoma. Cancer Cell 32, 185–203 e13 (2017).
Liberzon, A. et al. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 1, 417–425 (2015).
Bailey, P. et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 531, 47–52 (2016).
Collisson, E. A., Bailey, P., Chang, D. K. & Biankin, A. V. Molecular subtypes of pancreatic cancer. Nat. Rev. Gastroenterol. Hepatol. 16, 207–220 (2019).
Brunton, H. et al. HNF4A and GATA6 loss reveals therapeutically actionable subtypes in pancreatic cancer. Cell Rep. 31, 107625 (2020).
Sturm, G., Finotello, F. & List, M. Immunedeconv: an R package for unified access to computational methods for estimating immune cell fractions from bulk RNA-sequencing data. Methods Mol. Biol. 2120, 223–232 (2020).
Yoshihara, K. et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat. Commun. 4, 2612 (2013).
Witkiewicz, A. K. et al. Whole-exome sequencing of pancreatic cancer defines genetic diversity and therapeutic targets. Nat. Commun. 6, 6744 (2015).
Omori, Y. et al. Pathways of progression from intraductal papillary mucinous neoplasm to pancreatic ductal adenocarcinoma based on molecular features. Gastroenterology 156, 647–661 e2 (2019).
Connor, A. A. & Gallinger, S. Pancreatic cancer evolution and heterogeneity: integrating omics and clinical data. Nat. Rev. Cancer 22, 131–142 (2022).
Fujikura, K. et al. Multiregion whole-exome sequencing of intraductal papillary mucinous neoplasms reveals frequent somatic KLF4 mutations predominantly in low-grade regions. Gut 70, 928–939 (2021).
Wood, L. D., Canto, M. I., Jaffee, E. M. & Simeone, D. M. Pancreatic cancer: pathogenesis, screening, diagnosis, and treatment. Gastroenterology 163, 386–402 e1 (2022).
Braxton, A. M. et al. 3D genomic mapping reveals multifocality of human pancreatic precancers. Nature 629, 679–687 (2024).
Habib, J. R. et al. Informing decision-making for transected margin reresection in intraductal papillary mucinous neoplasm-derived PDAC: an international multicenter study. Ann. Surg., ahead of print, (2024).
Jamouss, K. T. et al. Tumor immune microenvironment alterations associated with progression in human intraductal papillary mucinous neoplasms. J. Pathol. 266, 40–50 (2025).
Pea, A. & Luchini, C. The importance of tumor microenvironment modulations in the progression of pancreatic intraductal papillary mucinous neoplasms(dagger). J. Pathol. 269, 126-129 (2025).
McGranahan, N. et al. Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science 351, 1463–1469 (2016).
Baldwin, L. A. et al. DNA barcoding reveals ongoing immunoediting of clonal cancer populations during metastatic progression and immunotherapy response. Nat. Commun. 13, 6539 (2022).
Wolf, Y. & Samuels, Y. Intratumor heterogeneity and antitumor immunity shape one another bidirectionally. Clin. Cancer Res. 28, 2994–3001 (2022).
Connor, A. A. et al. Association of distinct mutational signatures with correlates of increased immune activity in pancreatic ductal adenocarcinoma. JAMA Oncol. 3, 774–783 (2017).
Grant, R. C. et al. Clinical and genomic characterisation of mismatch repair deficient pancreatic adenocarcinoma. Gut 70, 1894–1903 (2021).
Petljak, M. et al. Characterizing mutational signatures in human cancer cell lines reveals episodic APOBEC mutagenesis. Cell 176, 1282–1294 e20 (2019).
Alexandrov, L. B. et al. The repertoire of mutational signatures in human cancer. Nature 578, 94–101 (2020).
Yoshida, K. et al. Tobacco smoking and somatic mutations in human bronchial epithelium. Nature 578, 266–272 (2020).
Brown, L. C. et al. LRP1B mutations are associated with favorable outcomes to immune checkpoint inhibitors across multiple cancer types. J. Immunother. Cancer 9, e001792 (2021).
He, Z. et al. LRP1B mutation is associated with tumor immune microenvironment and progression-free survival in lung adenocarcinoma treated with immune checkpoint inhibitors. Transl. Lung Cancer Res. 12, 510–529 (2023).
Holm, B., Barsuhn, S., Behrens, H. M., Kruger, S. & Rocken, C. The tumor biological significance of RNF43 and LRP1B in gastric cancer is complex and context-dependent. Sci. Rep. 13, 3191 (2023).
Tanaka, M. et al. Revisions of international consensus Fukuoka guidelines for the management of IPMN of the pancreas. Pancreatology 17, 738–753 (2017).
Pulvirenti, A. et al. Intraductal papillary mucinous neoplasms: have IAP consensus guidelines changed our approach?: results from a multi-institutional study. Ann. Surg. 274, e980–e987 (2021).
Nik-Zainal, S. et al. The life history of 21 breast cancers. Cell 149, 994–1007 (2012).
Mermel, C. H. et al. GISTIC2.0 facilitates sensitive and confident localization of the targets of focal somatic copy-number alteration in human cancers. Genome Biol. 12, R41 (2011).
Steele, C. D. et al. Signatures of copy number alterations in human cancer. Nature 606, 984–991 (2022).
Ansari-Pour, N. et al. Whole-genome analysis of Nigerian patients with breast cancer reveals ethnic-driven somatic evolution and distinct genomic subtypes. Nat. Commun. 12, 6946 (2021).
Dentro, S. C., Wedge, D. C. & Van Loo, P. Principles of reconstructing the subclonal architecture of cancers. Cold Spring Harb Perspect. Med. 7, a026625 (2017).
Collisson, E. A. et al. Subtypes of pancreatic ductal adenocarcinoma and their differing responses to therapy. Nat. Med 17, 500–503 (2011).
Moffitt, R. A. et al. Virtual microdissection identifies distinct tumor- and stroma-specific subtypes of pancreatic ductal adenocarcinoma. Nat. Genet. 47, 1168–1178 (2015).
Acknowledgements
This work was performed under the frame of the Scottish Genome Partnership. This study was funded by the Scottish Genome Partnership and, in part, by Associazione Italiana Ricerca Cancro (AIRC IG n. 26343), Italian Ministry of Health through Fondazione Italiana Malattie Pancreas (FIMP_CUP J37G22000230001), Fondazione Cariverona: Oncology Biobank Project “Antonio Schiavi” (prot. 203885/2017), European Union - NextGenerationEU through the Italian Ministry of University and Research under PNRR - M4C2-I1.3 Project PE_00000019 “HEAL ITALIA” CUP: B33C22001030006, European Union - NextGenerationEU through Italian Ministry of Health, PNRR HUB Diagnostica Avanzata PNC-E3-2022-23683266 PNCHLS-DA. DCW carried out this research at the National Institute for Health and Care Research (NIHR) Manchester Biomedical Research Centre (BRC) (NIHR203308). XH was funded by Cancer Research UK RadNet Manchester (C1994/A28701).
Author information
Authors and Affiliations
Contributions
A.P. and D.K.C. designed the study. A.P., R.U.G., C.L., S.D., F.D., N.B.J., C.J.M., E.D., S.R., P.P., N.S., R.T.L., R.S., and A.S. analyzed tissue samples. A.P., R.U.G., C.L., S.D., F.D., N.B.J., C.J.M., E.D., S.R., P.P., N.S., R.T.L., R.S., A.S., and D.K.C. acquired data. A.P., X.H., R.U.G., C.L., L.P.S.S., A.V.B., D.C.W., and D.K.C. analyzed data. A.P., X.H., R.U.G., C.L., L.P.S.S., A.PU., F.E.M.F., M.M., R.S., A.S., A.V.B., D.C.W., and D.K.C. interpreted data. A.P., X.H., D.C.W., and D.K.C. wrote the paper. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
D.K.C. has received consulting and lecture fees from Immodulon Therapeutics and Mylan, research funding from Astrazeneca, BMS GmbH & Co. KG, Merck, and Immodulon Therapeutics. MM reports honoraria from AstraZeneca, MSD Oncology, Ipsen, Hippocrates, Viatris, and Servier, and reports consulting or advisory role for AstraZeneca, MSD Oncology, and Janssen Oncology; research funding from Roche and other relationships (participation to protocol Steering Committees and Independent Data Monitoring Committee) with Novartis and OncoSil. FEMF has received research funding from Astrazeneca and Sierra Oncology, speakers fees from Servier Oncology, Viatris travel support from Viatris, and is in the advisory board of Astellas and Abbott. NBJ has received honorariums and delivered talks for Nanostring, 10x genomics, APkoya, and is the scientific advisory board for Galvani. C.L. has received funds from NTP (scientific advisor), MSD (speaker bureau), and Medica srl (speaker bureau). No potential conflicts of interest were disclosed by the other authors.
Peer review
Peer review information
Nature Communications thanks anonymous reviewers for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pea, A., He, X., Upstill-Goddard, R. et al. Clonal evolutionary analysis reveals patterns of malignant transformation of Intraductal Papillary Mucinous Neoplasms of the pancreas. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69762-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69762-w