Abstract
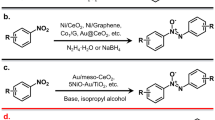
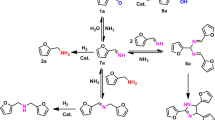
Single-atom catalysts have found widespread application in selective hydrogenation reactions partially due to their well-defined active site structures, which ensure exceptional chemical selectivity. However, the limited binding sites on single-atom catalysts hinder their application in hydrogenating larger multidentate substrates (e.g., benzonitrile). In this work, we introduced a heteronuclear Rh-Co dual-atom catalyst stabilized on the defective graphene supports (Rh1Co1/ND@G), which resolves the activity-selectivity trade-off in nitrile hydrogenation reaction. The Rh1 site primarily activates H2, whereas the Co1 site synergistically optimizes the adsorption of benzonitrile. The cooperative interaction between Rh-Co dual sites enhances the activation of the C ≡ N bond, significantly reducing the apparent activation energy compared to Rh SACs. The Rh1Co1/ND@G catalyst achieves exceptional performance under mild reaction conditions, delivering a TOF of 4068 h-1 with >98% dibenzylamine selectivity, surpassing all previous reported heterogeneous catalysts. Remarkably, the Rh1Co1/ND@G catalyst still maintains robust catalytic performance even after 12 cycles. This work not only presents a breakthrough in dual-atom catalyst design for nitrile hydrogenation, but also opens an avenue for developing high-performance industrial hydrogenation catalysts.
Similar content being viewed by others
Data availability
The data supporting this study and other findings are available from the corresponding authors upon request. Source data are provided with this paper.
References
Leatherwood HM. Top 200 Small Molecule Drugs by Retail Sales in 2024. (The Njarðarson Group, The University of Arizona, 2025); https://bpb-us-e2.wpmucdn.com/sites.arizona.edu/dist/9/130/files/2025/05/Top200smallMoleculeDrugs2024.pdf.pdf.
Hadjipavlou-Litina, D. J. & Geronikaki, A. A. Thiazolyl and benzothiazolyl Schiff bases as novel possible lipoxygenase inhibitors and anti inflammatory agents. Synthesis and biological evaluation. Drug Des. Discov. 15, 199–206 (1998).
Adams, J. P. Imines, enamines and oximes. J. Chem. Soc., Perkin Trans. 1, 125–139 (2000).
Gawronski, J., Wascinska, N. & Gajewy, J. Recent progress in lewis base activation and control of stereoselectivity in the additions of trimethylsilyl nucleophiles. Chem. Rev. 108, 5227–5252 (2008).
Murugesan, K. et al. Catalytic reductive aminations using molecular hydrogen for synthesis of different kinds of amines. Chem. Soc. Rev. 49, 6273–6328 (2020).
Sabatier, P. & Senderens, J. Application aux nitriles de la methode d’hydrogenation directe par catalyse: synthese d’amines primaires, secondaires et tertiaires. CR Hebd. Séances Acad. Sci. 140, 482–484 (1905).
Hegedűs, L. & Máthé, T. Selective heterogeneous catalytic hydrogenation of nitriles to primary amines in liquid phase. Appl. Catal. A: Gen. 296, 209–215 (2005).
De Bellefon, C. & Fouilloux, P. Homogeneous and heterogeneous hydrogenation of nitriles in a liquid phase: chemical, mechanistic, and catalytic aspects. Catal. Rev. 36, 459–506 (1994).
Blaser, H.-U., Malan, C., Pugin, B., Spindler, F., Steiner, H. & Studer, M. Selective hydrogenation for fine chemicals: recent trends and new developments. Adv. Synth. Catal. 345, 103–151 (2003).
Braun, J. V., Blessing, G. & Zobel, F. Katalytische Hydrierungen unter Druck bei Gegenwart von Nickelsalzen, VI.: Nitrile. Ber. der Dtsch. chemischen Ges. (A B Ser.) 56, 1988–2001 (1923).
Freifelder, M. A low pressure process for the reduction of nitriles. use of rhodium catalyst1. J. Am. Chem. Soc. 82, 2386–2389 (1960).
Vilches-Herrera M., Werkmeister S., Junge K., Börner A., Beller M. Selective catalytic transfer hydrogenation of nitriles to primary amines using Pd/C. Catalysis Sci. Technol. 4, https://pubs.rsc.org/en/content/articlelanding/2014/cy/c3cy00854a (2014).
Nait Ajjou, A. & Robichaud, A. Chemoselective hydrogenation of nitriles to secondary or tertiary amines catalyzed by aqueous-phase catalysts supported on hexagonal mesoporous silica. Appl.Organometallic Chem. 32, https://doi.org/10.1002/aoc.4547 (2018).
Jiao, Z.-F. et al. Turning the product selectivity of nitrile hydrogenation from primary to secondary amines by precise modification of Pd/SiC catalysts using NiO nanodots. Catal. Sci. Technol. 9, 2266–2272 (2019).
Wang, H. et al. Quasi Pd1Ni single-atom surface alloy catalyst enables hydrogenation of nitriles to secondary amines. Nat. Commun. 10, 4998 (2019).
Bhosale, A., Yoshida, H., Fujita, S. -i & Arai, M. Carbon dioxide and water: An effective multiphase medium for selective hydrogenation of nitriles with a Pd/Al2O3 catalyst. J. CO2 Utilization 16, 371–374 (2016).
Saito, Y., Ishitani, H., Ueno, M. & Kobayashi, S. Selective hydrogenation of nitriles to primary amines catalyzed by a polysilane/sio2-supported palladium catalyst under continuous-flow conditions. ChemistryOpen 6, 211–215 (2017).
Yamada, T. et al. Highly selective hydrogenative conversion of nitriles into tertiary, secondary, and primary amines under flow reaction conditions. ChemSusChem, 10.1002/cssc.202102138 (2021).
Chatterjee, M., Kawanami, H., Sato, M., Ishizaka, T., Yokoyama, T. & Suzuki, T. Hydrogenation of nitrile in supercritical carbon dioxide: a tunable approach to amine selectivity. Green. Chem. 12, 87–93 (2010).
Liu, L. et al. A ppm level Rh-based composite as an ecofriendly catalyst for transfer hydrogenation of nitriles: triple guarantee of selectivity for primary amines. Green. Chem. 21, 1390–1395 (2019).
Zhang, S., Xia, Z., Zou, Y., Zhang, M. & Qu, Y. Spatial intimacy of binary active-sites for selective sequential hydrogenation-condensation of nitriles into secondary imines. Nat. Commun.12, 3382 (2021).
Chen, J. et al. Atomic-dispersed rh enables efficient catalytic nitrile hydrogenation: size effect and metal-dependent effect. ACS Catalysis, 8354–8361, https://pubs.acs.org/doi/10.1021/acscatal.3c01562 (2023).
Liu, Y. et al. Polyoxometalate-based metal-organic framework as molecular sieve for highly selective semi-hydrogenation of acetylene on isolated single pd atom sites. Angew. Chem. Int Ed. Engl. 60, 22522–22528 (2021).
Zhao, L. et al. A magnetically separable pd single-atom catalyst for efficient selective hydrogenation of phenylacetylene. Adv. Mater. 34, e2110455 (2022).
Stambula, S. et al. Chemical structure of nitrogen-doped graphene with single platinum atoms and atomic clusters as a platform for the pemfc electrode. J. Phys. Chem. C. 118, 3890–3900 (2014).
Kwon, Y., Kim, T. Y., Kwon, G., Yi, J. & Lee, H. Selective activation of methane on single-atom catalyst of rhodium dispersed on zirconia for direct conversion. J. Am. Chem. Soc. 139, 17694–17699 (2017).
Kumar, N., Jothimurugesan, K., Stanley, G. G., Schwartz, V. & Spivey, J. J. In situ FT-IR study on the effect of cobalt precursors on CO adsorption behavior. J. Phys. Chem. C. 115, 990–998 (2011).
Wang, L. et al. Catalysis and in situ studies of rh1/co3o4 nanorods in reduction of NO with H2. ACS Catal. 3, 1011–1019 (2013).
Zhang, S. et al. Catalysis on singly dispersed bimetallic sites. Nat. Commun. 6, 7938 (2015).
Wang, C. et al. Product selectivity controlled by nanoporous environments in zeolite crystals enveloping rhodium nanoparticle catalysts for co2 hydrogenation. J. Am. Chem. Soc. 141, 8482–8488 (2019).
Zhang, S. et al. Dual-active-sites design of co@c catalysts for ultrahigh selective hydrogenation of N-heteroarenes. Chem 6, 2994–3006 (2020).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Henkelman, G. & Jónsson, H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J. Chem. Phys. 113, 9978–9985 (2000).
Henkelman, G. & Jónsson, H. A dimer method for finding saddle points on high dimensional potential surfaces using only first derivatives. J. Chem. Phys. 111, 7010–7022 (1999).
Acknowledgements
This work was supported by the National Key R&D Program of China (2022YFA1504500, 2021YFA1502802), the National Natural Science Foundation of China (U21B2092, 22202213, 22072162, 22203012, and 22402210), and the International Partnership Program of the Chinese Academy of Sciences (172GJHZ2022028MI). The XAS experiments were conducted in the Beijing Synchrotron Radiation Facility (BSRF) and the Shanghai Synchrotron Radiation Facility (SSRF).
Author information
Authors and Affiliations
Contributions
H.L. and D.M. conceived the research. J.C. conducted material synthesis and performed catalytic performance tests. J.C. conducted the X-ray absorption fine structure spectroscopic measurements and analyzed the data. Q.C. and G.S. performed the DFT calculations. X.C. contributed to the aberration-corrected high-angle annular dark-field scanning transmission electron microscopy. M.P., J.D., B.S., and Y.W. performed some of the experiments. The manuscript was primarily written by J.C., H.L., and D.M. J.C., H.C., X.C., Y.W., G.S., D.M., and H.L. contributed to discussions and manuscript review.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, J., Chen, H., Cai, X. et al. Dual-atom Rh-Co catalysts for synergistically boosting nitrile hydrogenation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69778-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69778-2