Abstract
The effects of transcranial focused ultrasound (tFUS) on the human brain are poorly understood. Currently, the field is at odds with whether tFUS is subthreshold modulating the brain’s excitability towards other stimuli, producing suprathreshold neural stimulation on its own, or if it even has a spatially specific non-auditory induced effect. Herein, we investigated the ability of tFUS, transcranial direct current stimulation (tDCS), and a combination of the two (transcranial electro-acoustic stimulation; tEAS) to evoke cortical target-location specific activity in 27 resting state humans with whole-brain electroencephalography recordings. In none of the exogenous event-related potentials did tFUS or tDCS result in location-specific activations. However, the co-modulatory combination of tEAS provided evidence that tFUS has a location-specific subthreshold modulatory effect. We propose a minimally modified Hodgkin-Huxley model that explains our results and provides a unifying framework for the field-wide observed effects (or lack thereof) of tFUS.
Similar content being viewed by others
Introduction
Therapeutic intervention of the human nervous system dates back centuries. Medical texts from c. 1st century CE Rome1 explicitly prescribe uses of electric fish for pain management, and surviving records from c. 1550 BCE Egypt2 imply the practice may date even earlier. A modern, philosophical descendant of these methods, transcranial direct current stimulation (tDCS), applies subthreshold (meaning it will modulate spontaneous firing, but will not induce stimulation time-locked spikes on its own) current across brain tissue using non-invasive scalp electrodes3,4,5,6. The subthreshold current depolarizes (or hyperpolarizes, in the case of inhibitory current) the neurons towards (or away from) their action potential threshold, which will make the neurons more (or less) sensitive to additional stimuli. One of the biggest limitations with this form of neuromodulation has to do with the spatial precision at which it can be applied due to the volume conduction effect7. Non-invasive electrical stimulation has spatial precision on the order of centimeters8, which can contain tens of millions of neurons9, and unintentionally modulate off-target brain regions as well3,10.
A recently introduced technique, transcranial focused ultrasound (tFUS), promises to overcome that limitation with a spatial precision on the order of millimeters11,12. tFUS’ mechanism of action continues to be under investigation, but one prevalent theory is that the ultrasound pressure waves open mechanosensitive ion channels13,14. This theory would be consistent with behavioral outcomes dependent on the specific brain region targeted with tFUS, which have been reported in a range of human studies11,15,16,17,18,19,20.
However, there is a critical lack of understanding when it comes to tFUS’ effect on the human brain. The bulk of human tFUS studies pair it with a secondary stimulation (such as tactile vibrations11,15, transcranial magnetic stimulation (TMS) pulses21, or visual stimuli18), which point toward, but do not confirm, a subthreshold modulation-based mechanism of action. Other studies directly looked into tFUS’ ability to elicit percepts as the sole stimulus, but their results pointed towards, at best, an inconsistent ability to do so—a study investigating S1 activation only elicited percepts in 54% of trials16, and a study investigating V1 stimulation reported visual percepts in only 11 of 19 participants17. Beyond even the question of suprathreshold stimulation vs. subthreshold modulation, some studies report that the effects of tFUS may not actually be due to the pressure wave, but rather auditory conduction. tFUS, typically, is not applied as one continuous wave, but instead pulsed at some lower frequency (pulse repetition frequency; PRF; Fig. 1D). This PRF makes a beeping noise, which is associated with activating auditory pathways22,23, and some studies could not detect an effect beyond auditory activation24,25. In humans, it was reported that actively playing a tone over the duration of tFUS sonication led to no significant differences in EEG between sham and active conditions26.
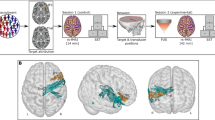
A The experiment consisted of applying neuromodulation to eyes-closed resting-state humans. Transcranial electro-acoustic stimulation (tEAS) involved overlapping transcranial focused ultrasound (tFUS) and transcranial direct current stimulation (tDCS). The tFUS condition consisted of only tFUS. The tDCS control condition also involved a spatial control ultrasound, which did not overlap with the region enclosed by tDCS electrodes, to account for possible auditory confounds. B A diagram of the experimental set up for each condition is provided with tDCS electrodes and ultrasound transducers placed on their relative location to EEG electrodes. C A free water scan of ultrasound is provided to illustrate its 5 mm full-width half-max (−6 dB) spatial specificity. D For the experiment, 500 kHz fundamental frequency (f0) ultrasound was pulsed every two seconds with up to an additional 20% random jitter for 200 trials. The pulse repetition frequencies (PRFs) were either 30 Hz or 3 kHz, as specified. The cycles per pulse (CPP) and pulse number (PN) varied based on PRF to normalize all conditions to 500 ms sonication duration and a 30% duty cycle. For conditions involving tDCS, the electrical stimulation remained on throughout the entirety of the trials. Head model icon created in BioRender. Kosnoff, J. (2026) https://BioRender.com/38kr3b9.
These studies raise critical questions about whether tFUS merely activates auditory pathways or if it evokes location-specific neuronal responses—and, if it does have a location-specific effect, whether that effect is subthreshold or suprathreshold. Resolving these questions requires a comprehensive analysis of whole-brain responses. While functional magnetic resonance imaging (fMRI) studies have investigated these questions and reported activations at the focal site of tFUS sonication27,28, their results may be confounded by multiple mechanisms. fMRI detects changes in blood-oxygen-level-dependent (BOLD) signal as a correlate for neural firing, but blood flow can be changed through a variety of methods not necessarily dictated by neuronal firing, such as vasodilation, which is directly inducible by ultrasound29. Further, unless tFUS is applied strictly parallel to the MRI’s magnetic field, physical interaction between the ultrasound pressure waves and the MRI’s magnetic fields will generate localized electric fields30, potentially creating artifactual neuronal activations absent in non-MRI environments. Consequently, fMRI measurements of tFUS’ neural activation ability are not definitive. Electroencephalography (EEG) based recordings, which directly measure electrical activity and do not require a magnetic resonance environment, may offer more insight into the true effects of tFUS as a sole stimulus.
Herein, we apply excitatory (e) and inhibitory (i) tDCS, tFUS (500 kHz fundamental frequency), and a combination of the modalities (transcranial electro-acoustic stimulation; tEAS) to the cortex of resting-state humans (Fig. 1). In follow-up studies, we broaden our tFUS parameter scope. For all tFUS experiments, we record and analyze simultaneous whole-scalp EEG recordings and image their corresponding cortical sources to characterize targeted-location specific, or lack-thereof, responses. We also present a first-principles verification of our findings using a minimally modified Hodgkin-Huxley model.
Results
Transcranial electro-acoustic stimulation produces a hemisphere-specific exogenous response
Butterfly plots of each condition’s preprocessed EEG are shown in Fig. 2A (dosing information is available in Supplementary Table T1). Statistical differences between EEG signals of the region of interest (ROI; electrodes C1, C3, FC1, and FC3) and their contralateral homologous counterparts (C2, C4, FC2, and FC4) were assessed using spatio-temporal permutation cluster tests within the 0 – 200 ms exogenous (stimulus-evoked) response window. The tests identified significant differences (p < 0.05) in the itEAS (30 Hz PRF tFUS with overlapping cathodal tDCS) and etEAS (3 kHz PRF tFUS with overlapping anodal tDCS) conditions, but none for itFUS (30 Hz PRF), etFUS (3 kHz PRF), itDCS (cathodal tDCS with spatial-control 30 Hz PRF tFUS) or etDCS (anodal tDCS with spatial-control 3 kHz PRF tFUS) conditions (Fig. 2B). Both significant clusters encompassed electrode C3 and the 10–63 ms time window. We used the shared components across both tEAS conditions’ significant clusters as spatial and temporal windows for further analysis.
A Preprocessed EEG butterfly plots of the experiment show exogenous ( <200 ms post stimulation) event-related potentials (ERPs) at 30, 90, and 170 ms for conditions with 3 kHz PRF tFUS. B Spatio-temporal permutation cluster tests (using the two-tailed paired t-test) were performed comparing the ipsilateral ROI electrodes (C1, C3, FC1, and FC3) against their homologous contralateral counterparts (C2, C4, FC2, and FC4) for the 0 to 200 ms window. Significant electrodes are marked with white circles on the left subplot t-statistic topographic map, and significant corresponding time windows are shaded in gray on the right subplot. The temporal traces (mean ± 95% confidence interval) are a grand average of the significant electrodes, or, if there are no significant clusters, of all the electrodes. Only the tEAS conditions yielded significant clusters, indicating the 3 kHz PRF ERPs are symmetrically distributed across the brain. The largest overlapping time window between the significant clusters, 10–63 ms, was selected for further analysis. C The overlapping windows are visualized by taking the mean electrode activity in the 10–63 ms window. Visually, there is a strong ROI response in i/etEAS conditions. Electrode C3 is marked with a white circle. D Electrode C3 was identified in both significant clusters from tEAS permutation cluster tests. Further statistical analysis (two-tailed McNemar’s paired binary data test with FDR multiple comparison correction) was conducted to test for a significant effect on C3 polarity between e/i conditions for all modulation parameters. i/etEAS was found to have a significant effect (padj < 0.001). No significant effects on polarity were detected for tFUS (padj = 0.434) or tDCS (padj = 1.00).
Inhibitory vs. excitatory tEAS produces significant changes to EEG polarity
EEG data were averaged over the permutation cluster test-identified time frame of 10–63 ms. Resultant topographic maps present with visually apparent ROI activations for tEAS conditions and primarily hemisphere-symmetric topographies for the other conditions (Fig. 2C). Effects on electrode C3 (selected based on its presence in permutation cluster test results) polarity for this time average were assessed with McNemar’s test and False Discovery Rate (FDR) multiple comparison correction (Fig. 2D). Inhibitory tEAS led to a significant change (padj < 0.001, χ2 = 12.1) in ROI polarity compared to etEAS. Neither e/i tFUS nor e/i tDCS conditions induced significant polarity changes (padj > 0.05).
tEAS induces significantly localizable responses to the targeted ROI
EEG data were back-projected to the subjects’ cortices using the dynamic statistical parametric mapping (dSPM) source localization method, and the resultant power of source waveforms was averaged over the 10–63 ms window. Source-morphing to a common brain model and averaging the response across subjects showed strong concordance with the targeted ROI, defined by the subspace contained within electrodes C1, C3, FC1, and FC3 orthogonally projected to the cortex (Fig. 3A). Statistical analysis to determine if ipsilateral (IL) ROI power was significantly higher than the homologous contralateral (CL) power was conducted with permutation t-tests with FDR multiple comparison correction. Both tEAS conditions had significantly increased IL ROI source power (etEAS IL = 2.59 ± 1.18 zdSPM, CL = 1.62 ± 0.63 zdSPM, padj < 0.001, t = 4.10, d = 1.03; itEAS IL = 2.40 ± 1.08 zdSPM, CL = 1.22 ± 0.45 zdSPM, padj < 0.001, t = 4.78, d = 1.42). None of the other conditions had significantly increased IL source power (padj > 0.05; Fig. 3B).
A EEG data were projected to the cortex using the dSPM source imaging method. Subject data were averaged over the 10–63 ms post-stimulation window and source morphed to FreeSurfer’s FSAverage common brain model and averaged together for visualization. The cut-off levels of activity for brain sources were defined based on Otsu’s threshold. B e/itEAS (N = 20/21) led to significantly higher ipsilateral ROI source activity than contralateral ROI source activity (one-tailed t-test with FDR multiple comparison correction; ***padj < 0.001). e/itFUS (N = 22/22; padj = 0.998/0.998) and e/itDCS (N = 20/10, padj = 0.912/0.551) did not. Each subplot contains a violin plot highlighting the distribution of the data. A box plot is embedded within the violin plot, marking the data median (center line), 1st and 3rd quartiles (whiskers = 1.5 * quartile ranges) and means (white circle). The individual datapoints are presented in color-matched circles.
Pairwise comparisons using each subject’s condition average were made between IL source power, as well as precision, recall, and F1 score of source localization with respect to the ROI (Supplementary Fig. S1). For all metrics, tEAS conditions were found to have significantly higher outcomes. Cohen’s d analysis yielded that tEAS conditions had at least moderate effect sizes (d > 0.5) for all comparisons with other conditions, and often large ones (d > 0.8). Pairwise FDR-adjusted p-values and Cohen’s d values are provided within the relevant sub-figures.
Transcranial focused ultrasound results in significant auditory activations
Despite the presence of three distinct exogenous time window event-related potentials (ERPs) induced from 3 kHz tFUS in the topographic maps (30, 90, and 170 ms peaks; Fig. 2A), spatio-temporal permutation cluster tests did not return any significant differences for left hemisphere ROI electrodes compared to their right hemisphere counterparts. This points to a hemisphere-symmetric response, despite tFUS only being applied to one side. Topographic map visualizations of each peak (20–40 ms, 80–100 ms, and 160–180 ms averages; Fig. 4A) do not provide any evidence of a target-location specific effect. Source imaging metrics of these peak windows continue to support a lack of ROI response (20–40 ms precision w.r.t. ROI: 0.013 ± 0.014; 80–100 ms: 0.015 ± 0.019; 160–180 ms: 0.022 ± 0.020).
In all three conditions with 3 kHz tFUS (etFUS, etDCS, and etEAS), distinct ERPs at 30, 90, and 170 ms were apparent (Fig. 2A). A For etFUS, EEG and dSPM source activities were averaged for ±10 ms around these ERPs to elucidate their origin. At all three time windows, the topographic maps present with symmetric responses across hemispheres. Despite tFUS being targeted to the left primary motor cortex area, source imaging results of the peaks show the underlying sources are located in the auditory or deep brain. B The electrophysiological response of tFUS applied to resting-state left motor cortex is indistinguishable from tFUS applied to resting-state prefrontal cortex. B i In 21 of the subjects, a 3 kHz spatial control was conducted where tFUS was applied to the right prefrontal cortex (PFC). Even though tFUS was applied contralaterally and to another region, the resultant waveform and ERP spatial distributions are strikingly similar to tFUS applied at M1. B.ii Spatio-temporal cluster testing did not return any significant clusters between tFUS to left hemisphere (LH) M1 and tFUS to right hemisphere (RH) PFC. The plotted values are the grand averages and 95% confidence intervals of all electrodes for both conditions. B.iii The lack of significant differences through traditional hypothesis testing methods does not indicate statistical similarity. However, using Bayes Factor (BF) analysis, the evidence in favor of the null (i.e., that two distributions are statistically the same) can be quantified. A log10 BF < 0 is evidence for the null, whereas log10 BF > 0.5 is considered evidence against the null. To address BF analysis’ sensitivity to its scaling factor (the commonly selected default value is 0.707), we ran BF analysis for RH PFC tFUS vs. LH M1 tFUS ROI electrodes over a wide range of values (0.1–2.0). In the 10–63, 20–40, and 80–100 ms windows, for every scaling factor tested, all four electrodes resulted in negative log10 BFs, providing robust evidence that the ROI response is the same across these two conditions. For C1, C3, and FC3 in the 160–180 ms window, BF analysis was inconclusive for possibly small-medium effect sizes (scaling factor <0.707; 0 <log10 BF < 0.5), though it continued to provide robust evidence in favor of the null for medium-large effect sizes (scaling factor > 0.707). C Granger Causality analysis reveals significant tFUS-induced auditory cortex to deep brain projections. The Net Granger Causality (GCNet) Wald statistic was calculated for each time bin by subtracting the GC of the reverse flow from the GC of the forward flow of information of auditory cortex (A1) to the deep brain and the left M1 ROI to the left deep brain for both etFUS (N = 22) and itFUS (N = 22). The GCNet values were normalized by subtracting the GCNet for the baseline period (−0.5 to −0.1 s) for each subject’s pathways (ΔGCNet). For auditory to deep brain activations, the cortical hemisphere with the maximum net flow was considered in the analysis for each subject. For M1, only the tFUS-targeted left hemisphere ROI to the left deep brain parcellation was considered. The differences between the baseline GCNet and each time bin’s GCNet were analyzed with two-sided permutation t-tests. FDR multiple comparison correction was applied locally. Both etFUS and itFUS induced significant auditory to deep brain Granger causality, while neither induced a significant ROI to deep brain flow of information. The individual p-values are listed in the Source Data file. The box plots mark the data median (center line), and 1st and 3rd quartiles (whiskers = 1.5 * quartile ranges). The individual datapoints are presented in color-matched circles. *padj < 0.05, **padj < 0.01, ***padj < 0.001.
In 21 of the 22 subjects with etFUS, we also tested a 3 kHz PRF right prefrontal cortex (RH PFC) spatial control condition. The resultant waveforms and ERP spatial topographies (Fig. 4B.i) share a striking resemblance to those that arose from applying tFUS to the left motor cortex area (LH M1). Spatio-temporal permutation cluster testing did not yield any significantly different clusters between LH M1 and RH PFC conditions (Fig. 4B.ii). Further, when we directly quantified statistical similarity between the LH ROI electrodes for the two conditions using Bayes Factor (BF) analysis, the evidence that the 10–63, 20–40, and 80–100 ms responses of the two conditions were the same was robust (log10 BF < 0 for all electrodes/scaling factors; Fig. 4B.iii). For the 160–180 ms window, BF analysis was inconclusive for small-medium effect sizes (scaling factors <0.707; 0 <log10 BF < 0.5), though it continued to provide evidence in favor of similarity for medium-large effect sizes (scaling factors > 0.707). For comparison, BF analysis of etEAS compared to etFUS yielded evidence that certain ROI electrode responses were significantly different (log10 BF > 0.5; Supplementary Fig. S2).
Granger causality analysis of both itFUS and etFUS conditions showed significant increases in information flow from the primary auditory cortex to the deep brain, and no changes in net information flow from the targeted ROI to the deep brain (Fig. 4C). In a follow-up study with 11 subjects, it was found that even an inaudible PRF (30 kHz) continued to induce significant auditory to deep brain Granger causality (Supplementary Fig. S3).
tEAS conditions were also found to induce significant increases in auditory to deep brain connectivity and have no effect on the net flow of information from the targeted ROI to deep brain (Supplementary Fig. S4).
itFUS, but not tEAS, induces endogenous dissociations between the ROI and deep brain
Despite not inducing a significant effect on net Granger causality, itFUS was found to have significant dissociation effects on undirected endogenous ( >200 ms) spectral imaginary coherence (ImCoh; Supplementary Fig. S5). None of these dissociations were present in the tEAS conditions.
Increased tFUS parameter searches continued to yield no evidence of ROI suprathreshold stimulation
A follow-up call-back study was conducted in 10 of the 22 original subjects (Supplementary Table T2), in which additional tFUS parameters were tested. These parameters included 1,500 and 40 Hz PRFs with a 30% duty cycle (DC), and, initially, 10% and 60% DCs with both 30 and 3000 Hz PRFs. All four of the first four subjects experienced thermal discomfort with 60% DC, and so we did not test it further. From the fourth subject onward, we tested 0.6% DC instead. Source imaging results continued to point towards deep brain and temporal lobe activations (Fig. 5A) and ROI analysis continued to find no increases in ipsilateral source power for any of the new tested conditions (Fig. 5B). Analysis of the effects of holding the DC or PRF constant and changing the other parameter yielded no significant differences (permutation one-way ANOVA; all p > 0.05) and Bayes Factors provide robust evidence that the responses across conditions are statistically similar (log10 BF < 0; Fig. 5C).
A follow-up study was conducted in 10 subjects (Supplementary Table T2) to test for possible changes in activity from varying the duty cycle or PRF from the original study parameters. A EEG data were projected to the cortex using the dSPM source imaging method. Subject data were averaged over the 10–63 ms post-stimulation window and source morphed to FreeSurfer’s FSAverage common brain model and averaged together for visualization. The cut-off level of activity for brain sources were defined based on Otsu’s threshold. Visualizations continue to show strong deep brain and temporal lobe activations, but nothing concordant with the ROI. B No tFUS-only parameter combination yielded significantly higher ipsilateral than contralateral ROI source activity (one-tailed permutation t-test with FDR multiple comparison correction; all padj > 0.05). Each subplot contains a violin plot highlighting the distribution of the data. A box plot is embedded within the violin plot, marking the data median (center line), 1st and 3rd quartiles (whiskers = 1.5 * quartile ranges) and means (white circle). The individual datapoints are presented in color-matched circles. C Comparisons across the subjects’ tFUS-only ipsilateral ROI responses were compared for constant PRFs and a constant duty cycle. Their 30 Hz and 3 kHz PRF with 30% DC data from the initial session were also included in this analysis. No significant changes were detected with a permutation one-way ANOVA test (all padj > 0.05). Beyond the lack of statistic differences, Bayes Factor analysis indicates the resting-state tFUS responses are statistically similar across the parameter spaces. The p-values for all tests are listed in the Source Data file. The box plots mark the data median (center line), and 1st and 3rd quartiles (whiskers = 1.5 * quartile ranges). The individual datapoints are presented in color-matched circles.
In an additional follow-up study with 11 subjects, the effects of higher pressure were investigated by normalizing the in situ pressure to 300 kPa with Sim4Life simulations (original study in situ mean pressure was 231 kPa). The sonication duration was reduced from 500 ms to 100 ms to compensate for this increase. Even still, 2 of the 11 subjects (18.2%) experienced scalp thermal discomfort and their experiments were aborted. Analysis of the other 9 subjects’ data continued to yield no significant increases in IL compared to CL ROI source power (Supplementary Fig. S6; p > 0.05).
tEAS is more than a linear sum of its parts
We took the summation of the source-space tFUS and tDCS conditions to produce a condition representing the sum of independent electric and acoustic stimulation. The summation did not result in increased concordance with the ROI (Supplementary Fig. S7A), nor to significant increases in ipsilateral over contralateral ROI source power (Supplementary Fig. S7B; permutation t-test p > 0.05). We also modeled the tEAS ROI response as function of tDCS scalp current, cortical current, and tFUS intensity and found none of these components to be meaningfully predictive (Supplementary Fig. S7C–E).
tEAS response is driven by tDCS polarity, not tFUS PRF
Anodal tDCS was combined with 30 and 3000 Hz PRF tFUS and analyzed over the 10–63 ms window (Supplementary Fig. S8). Both conditions led to brain sources concordant with the ROI. Comparisons of the ROI source power yielded no significant differences across conditions (permutation t-test p > 0.05), and Bayes Factor analysis provided evidence that the response powers were statistically similar (log10 BF < 0).
A minimally modified Hodgkin-Huxley model provides support for an electro-acoustic co-modulatory effect
The Hodgkin-Huxley model was modified (Supplementary Methods) to have an additional parallel pathway representing piezo-electric ion channels (Supplementary Fig. 9A). Simulations revealed that subthreshold ultrasound-based opening of piezo-electric channels (Supplementary Fig. S9B) and subthreshold direct current, representing tDCS (Supplementary Fig. S9C), could combine to form a suprathreshold stimulus (Supplementary Fig. S9D-F).
Discussion
While the effect of tFUS is commonly considered to be based on the targeted brain region11,12,15,16,17,18,19,20,31, there is a lack of agreement on the magnitude of that effect. Specifically, there are unanswered questions on whether tFUS is modulating neural excitability or stimulating the neurons on its own. Beyond that, recent discoveries in tFUS research have suggested that auditory pathways, but not the focused ultrasound per se, are responsible for modulating brain signals24,25,26,32. We investigated this phenomenon by applying tFUS, tDCS, and a co-modulatory combination of the two, tEAS, to resting-state humans and characterizing the electrophysiological responses with whole brain EEG and EEG source imaging. In follow-up studies, we broadened our tFUS parameter search to include additional PRFs, duty cycles, and higher in situ pressure. Our results indicated that, while neither resting-state tFUS nor tDCS produced targeted location EEG source localizable brain activities on their own, when they were applied together as tEAS, the multi-modal co-modulatory combination could. Our data provide strong evidence that, while tFUS produces prominent whole brain auditory related activations, it also induces location-specific subthreshold modulatory effects.
When the ipsilateral ROI electrodes’ activities were compared against the contralateral homologous counterparts, only the co-modulatory tEAS conditions yielded significant differences (Fig. 2B). While the clusters from permutation cluster tests are not guaranteed to be significant for their entire extent33, we did consider shared clusters from both itEAS and etEAS to warrant further analysis. Thus, the remainder of our work focused on the shared window of 10–63 ms. We continued our analysis in the cortical domain using dSPM EEG source imaging. The subject average response had strong concordance with the targeted ROI for both itEAS and etEAS, but not for the other conditions (Fig. 3A). The ipsilateral ROI source power was significantly higher than that of the homologous contralateral ROI for only the tEAS conditions (Fig. 3B). In a follow-up study, we expanded our resting-state tFUS parameter search to include additional 40 and 1500 Hz PRFs with 30% DC, as well as 0.6%, 10%, and 60% DCs with both 30 and 3000 Hz PRFs. 60% DC led to unpleasant scalp sensations34 in all initial (4/4) subjects and the experiments were aborted. Analysis of the other parameters continued to show a lack of sonication-location specific suprathreshold responses, providing further support that tFUS’ location-specific effect is subthreshold (Fig. 5).
Directly comparing ipsilateral ROI source power and source localization classification metrics (precision, recall, and F1 score) across conditions indicated tEAS had significantly higher ipsilateral outcomes compared to the others (Supplementary Fig. S1). Summing the separate tDCS and tFUS conditions together did not yield spatially specific information (Supplementary Fig. S7), providing additional evidence for a compounded electro-acoustic effect.
Not only did itEAS and etEAS induce significantly higher source localization metrics and source power than the other tested conditions, but they also led to a significant change in ROI polarity (Fig. 2D). While this polarity change is evidence of differing electrophysiological effects, a follow-up study indicated that this change in response is driven by tDCS polarity and not tFUS PRF (Supplementary Fig. S8). However, further analysis showed no linear relationship between the applied tDCS dose and tEAS response magnitude (Supplementary Fig. S7D–E), nor between tFUS intensity and tEAS response (Supplementary Fig. S7C). This points to a non-linear response from a combined transcranial electro-acoustic effect. Anodal tEAS experiments were repeated with both 30 Hz and 3 kHz PRF tFUS, and the resultant ROI responses were found to be statistically similar through Bayes Factor analysis. This is consistent with another research group’s report that neural response may not be a function of PRF35. Our prior human research has indicated tFUS may act on feature-based attention19, so it may also be possible that PRF-dependent responses would be brought out with non-resting-state task engagement. Understanding the translation from animal models to human models remains a critical challenge in neuroscience work, and additional research into neuron response to tFUS PRF at the human brain scale is warranted.
Despite a lack of source localizable activity and changes to Granger causality net information flow, itFUS was found to have significantly reduced spectral connectivity from the ipsilateral ROI to the deep brain compared to the contralateral ROI to the deep brain (Supplementary Fig. S5). This is consistent with other literature that has reported tFUS to have target-location specific dissociation effects27,36. itFUS significantly modulated different time windows of gamma band connectivity, while etFUS did not, which also provides some evidence of circuit-level PRF-dependencies. However, there were no spectral connectivity changes present in the tEAS conditions. At first, this seems counterintuitive, as one would expect the suprathreshold tEAS condition to have stronger, compounded effects on brain connectivity changes. Instead, our data indicate that itEAS overrides the spectral dissociation itFUS otherwise induces. The timings of these connectivity changes point to why this might be the case. Both of the spectral connectivity changes occur outside of the initial 200 ms window. The first 200 ms of brain activity post-stimulation is often considered exogenous, or stimulus-driven, while activity occurring later than that is considered endogenous, and is reflective of internal cognitive processing37. Given this, these dissociations are likely a result of itFUS inducing some internal change in cognitive brain-state, which can be overridden with suprathreshold stimulation. Additional research is warranted on the nature and function of tFUS’ dissociations.
Excitatory (3 kHz PRF) tFUS targeted to the left motor region, as part of etFUS, etEAS, and etDCS (which included a tFUS spatial control), induced distinct ERPs at approximately 30, 90, and 170 ms post stimulation onset (Figs. 2A, 4A). This is consistent with a previous study that identified V1-targeted tFUS induced EEG peaks, though their reported peak times were labeled as 55 ms, 100 ms, and 150 ms post sonication17. However, when we compared these ERPs to those induced by applying tFUS to an entirely different brain region (right hemisphere prefrontal cortex), the resultant waveforms did not just lack statistical differences from each other—they were found to be statistically similar through Bayes Factor analysis (Fig. 4B). These results provide critical evidence that location-specific effects of tFUS are subthreshold; by itself, tFUS produces the same responses regardless of where it is targeted.
Of course, these shared responses are time-locked event-related potentials. To put it another way: tFUS does induce a suprathreshold response. However, since the topology of these ERPs do not differ across targeting locations, they can be attributed to a whole-brain auditory response. Indeed, the 3 kHz PRF EEG topographic spatial distributions of the 90 and 170 ms ERPs do share visual resemblance to the previously reported auditory-induced N1 and P2 ERPs26. Granger causality investigations, which revealed significantly increased A1 to deep brain information flow for both 30 Hz and 3 kHz tFUS conditions (Fig. 4C), provide additional support that the ERPs are auditory related. Nevertheless, the present results that combining tFUS with tDCS led to spatially specific responses, when neither could do so individually (Fig. 3), as well as itFUS dissociating the spectral connectivity between the ROI and the deep brain (Supplementary Fig. S5), indicate that tFUS does have targeted-location specific effects.
We note that these auditory effects persist within the tEAS conditions (Supplementary Fig. S4). This is unsurprising, as we would have no reason to think the electro-acoustic effect at M1 would cancel out auditory pathways. However, what is surprising is the continued lack of net information flow from the ROI to the deep brain. M1 is connected to deep brain structures38, so on one hand, it would be expected that increased M1 activity would lead to increased deep brain activity, and this would be reflected as an increased net flow of information. However, this was not our finding. One possible explanation for this phenomenon is that M1 neuron connections with the deep brain are dependent on cell type38. Prior work has demonstrated tFUS’ parameter choices can alter cell-type selectivity39. Our tFUS parameters may not have been optimal for eliciting M1 to deep brain connections. Future research on tEAS connectivity changes (or lack thereof) is warranted.
tDCS driving tEAS polarity also raises the question of whether the focality of tEAS’ response could be due to tDCS locally subthreshold modulating tFUS’ induced whole brain auditory response. However, this is already accounted for in our experimental setup. For our tDCS conditions, we implemented a spatial tFUS control (the setup is shown in Fig. 1B). If the source response of tEAS was the result of tDCS interacting with the whole brain auditory response, the tDCS conditions would have resulted in focal activations as well.
A minimally modified Hodgkin-Huxley (HH) model sheds more insight into what might be occurring (Supplementary Fig. S9). We modeled the electro-acoustic effect with an additional piezo-electric ion channel within the traditional HH neuron. It supported that subthreshold current can be combined with subthreshold ultrasound pressure to induce a suprathreshold response. Since the model was created with parameters calculated on different species (the original HH parameters are from a squid40, piezo-electric components are from rodent measurements41,42,43, and the model is currently being used to test findings in humans), particular values of the current or piezo-electric channel open probability needed to produce spikes likely do not hold much meaning to any particular species. However, the model does provide a high-level unifying framework on the previously reported effects of tFUS.
In most of the published literature, tFUS is used explicitly in conjunction with another stimulus. These published paired stimuli include tactile vibration11,15, physical movement20, transcranial magnetic stimulation21, visual stimulation18,19, fMRI (which can introduce artifacts mentioned in the introduction)27,28,30, and now, as part of this study, tDCS. Even in the studies that investigated the effects of tFUS to induce tactile16 or visual17 perceptions, tFUS may have co-interacted with attention-modulated somatosensory or visual circuity, as the participants were instructed to describe the location and type of perceived sensations, which could put their brains into attended states. This is further supported by another ultrasound-to-visual cortex perception study that found even the sham condition led to visual hallucinations44. Indeed, taking all of this into account within the context of our findings, it does follow that tFUS may be a subthreshold co-modulator, and not a suprathreshold stimulator. This may be why some researchers24,25,26, present study included, have not found tFUS to have a non-auditory related effect when not paired with another stimulus.
At the same time, the HH model supports that there is some open probability of piezo-electric channels, and thereby ultrasound pressure level, that will induce neuronal firing. This is in line with rodent cortex studies that have reported tFUS’ ability to be the sole stimulus12 and induce source localizable effects45. To further investigate this in humans, we repeated our experiment on a cohort of 11 subjects, increasing the target pressure to a normalized 300 kPa using Sim4Life simulations (Supplementary Fig. S6). To lower the possibility of the transducer overheating, we reduced the sonication duration from 500 ms to 100 ms. Even still, 2 of the 11 (18.2%) subjects reported uncomfortable scalp sensations, and we aborted those experiments. Analysis of the other nine subjects’ data continued to find no statistical ipsilateral ROI power increases compared to the contralateral hemisphere, though subject average visualizations of the 30 ms ERP (Supplementary Fig. S6C) had a stronger concordance with the ROI than with the (on-average) lower target pressures used in our original study (Fig. 3). The increased concordance provides some support for the argument that higher pressures may be more efficacious as a sole stimulus (as opposed to a co-modulator), though the balance of efficacy and the risk of scalp burns must always be weighed (especially given a report that the pressure levels needed for tFUS to generate direct suprathreshold stimulation in mice sciatic nerves lead to irreversible tissue damage46).
Why tFUS may be capable of stimulating the rodent cortex12,45, yet seems to be limited to subthreshold co-modulating the human cortex cannot be confirmed, but if we are to speculate, based on all that has been discussed thus far, the reason could be that the amount of ultrasound pressure loss through the skull in larger brain models does not reach the levels of cortical pressure needed to open enough piezo-electric channels. As the pressure is increased, so is the amplitude of the PRF beeping noise. Even if there is a small tFUS-induced targeted-location specific effect, it might be masked by the auditory evoked response. Another possibility could be that humans may not have as high a density of mechanosensitive channels for the ultrasound to activate as rodents.
We investigated whether a silent ultrasound waveform could potentially ‘unmask’ an ROI response. We tested a smoothed waveform (pulse duration: 3.25 ms, 1 ms tukey ramping, 250 Hz pulse-repetition frequency)47 in six human subjects (Supplementary Fig. S10, Supplementary Table T3). All six subjects reported that they could not hear the ultrasound. However, the smoothed tFUS by itself continued to lack increased ipsilateral ROI source power, whereas the smoothed tEAS waveform continued to have higher IL power. We also note that the smoothed tEAS waveform appeared overall less focal than the one that resulted from our rectangular pulses, with additional strong activations in the ipsilateral frontal/premotor regions. While the results of this study indicate that ultrasound PRF may not play a substantial role in the outcome of tEAS (Supplementary Fig. S8), it may be that the ultrasound pulse shape does. Future tEAS parameter optimization studies are warranted.
There have been some studies, in humans31,48 and in non-human primates49, that indicate tFUS may have suprathreshold stimulation electrophysiological effects on deep-brain targets. This does, initially, seem contradictory to our results and theory. However, we postulate three reasons as to why these are actually complementary pieces of evidence. First, in rodents, the deep brain has been found to have a much higher concentration of piezo-electric ion channels than the cortex and is therefore more sensitive to ultrasound modulation14. In other words, for our HH model, Gpiezo, deep brain » Gpiezo, cortex, where G is the membrane conductance. Second, in addition to having higher piezo-electric channel conductance, deep brain neurons have also been reported to have much lower activation thresholds than cortical ones50. As a result, what is subthreshold pressure on the cortex may be suprathreshold in the deep brain31. Third, despite targeting the motor cortex, we observed large-scale activation in the auditory and deep brain structures for tFUS-only conditions at the 30, 90, and 170 ms peak windows (Fig. 4A, B). This is not surprising, as the auditory pathways directly project into the deep brain. In fact, when we investigated connectivity from primary auditory cortex to the deep brain in our subjects, we found significantly increased A1 to deep brain Granger causality for both etFUS and itFUS (Fig. 4C). Other research has demonstrated that frequencies beyond which humans can perceive as sound (20 Hz –20 kHz) can still affect auditory pathways51,52, meaning that no ultrasound PRF is inherently exempt from this phenomenon. We corroborated this possibility with an additional follow-up study with an inaudible (30 kHz) PRF and continued to find significantly increased A1 to deep brain Granger causality (Supplementary Fig. S3). As a result, we deem it highly possible that deep brain regions can be “activated” by the auditory aspect of ultrasound, but then co-modulated from the pressure wave itself. In any case, more research into the piezo-electric properties of the human brain is warranted.
It is important to discuss the actual values of the localization scores beyond their statistical significance. In recent years, tFUS has garnered rising attention as a neuromodulation technology with millimeter precision11,53,54. Our ex vivo scans of the ultrasound’s pressure wave profile (Fig. 1C) support this, but the average of our subject precisions of the tEAS reconstructed sources are only about 0.10, calculated by Eq. 1 (Supplementary Fig. S1C). This is not altogether unsurprising, as we applied modulation to resting-state brains. The resting-state brain is not off in a traditional electronics sense, but rather unfocused. Some regions of the brain, known as the default mode network, are actually more active when a person is at rest55. The low precision scores may be partially attributed to heightened off-target activities.
The low scores may also be somewhat attributed to our choice of using Otsu’s method to determine the source threshold. Otsu’s method was first proposed for computer vision purposes to find a threshold value that minimized the intraclass variance (alternatively, it maximizes the interclass variance) between an image’s foreground and background56, and it has become a commonly used technique in EEG source localization to analytically and objectively determine the threshold between a source signal of the brain and noise in the recording57. For individual subject-level data, the thresholds were around 30% (etEAS range: 25–42%, itEAS range: 21–39%). Since brain tissue is conductive, any electrical activations induced by itEAS will propagate outwards until they spatially decay. These peripheral activations are still likely to be detected as sources, given the low threshold values.
Even with persisting off-target activations, the increase in activity levels for the targeted area is apparent. The source-morphed subject averages (Fig. 3A) as well as the grand average ERP (Supplementary Fig. S11) converged to cover the ROI, indicating the off-target activations are inconsistent across subjects compared to the stimulated area. Indeed, when we calculated the Euclidean distance from the grand average brain voxel with the highest activity to the closest point in the ROI, both tEAS conditions had 0 mm localization error (compared to 73, 23, 73, and 26 mm for etFUS, itFUS, etDCS, and itDCS, respectively).
We note the varied levels of scalp current applied to subjects during our experiments. For tDCS experiments, the dose is most commonly normalized for all subjects (either at the scalp- or the cortical-level using simulations). However, the dose can also be varied across subjects58,59,60. We opted for the latter approach in our study. tDCS is generally considered safe, but can induce transient sensations of tingling, itching, and/or burning on the scalp61. The primary goal of our study was to utilize tDCS to draw out possible subthreshold effects of tFUS in a resting state. Skin perceptions are the result of activated sensory neurons62, which can contaminate the resting-state EEG recordings, both from the firing of the scalp (i.e., non-cortical) neurons, but also from the activated pain circuits within the brain itself. We wished to avoid such possible contamination. Beyond this, we also note it is possible for tDCS to induce skin lesions after only a single session63. Since our participants were healthy human subjects, we wanted to avoid this possibility.
We would also like to emphasize that this is not a traditional tDCS study looking at after-effects from prolonged stimulation64,65,66. Instead, this study uses the online effects of subthreshold direct current to investigate possible subthreshold effects of tFUS. Due to the unpredictability of tDCS dose-response relationships5,64,65,66, it is typically recommended to choose dosing levels based on prior research and the intended neurological effect5. However, we did not have such literature to draw upon for investigating simultaneous tDCS and tFUS. Instead, we prioritized a dose that would not activate sensory pathways62 by inducing itching, burning, and tingling sensations61. Such skin perceptions from tDCS would contaminate and undermine our investigations of tFUS.
Since we used the same current density for each condition within a subject, the varied current density across subjects is not a major concern. This is because our cross-over study design looks at the changes in brain response across conditions for subjects. Our results show a significant increase in ROI activity for tEAS conditions, regardless of tDCS dosage, compared to tFUS and tDCS conditions. We did conduct further analysis to determine if scalp or cortical (estimated with SIMNibs67; Supplementary Methods) tDCS dose (Supplementary Table T1) was a meaningful predictor of ROI response. Our results indicate tDCS dose, at the between-subjects level, is not a meaningful predictor of tEAS activation (Supplementary Fig. S7; R2 < 0.05, p > 0.05). This is consistent with other studies that have reported inconsistent outcomes of different tDCS doses5,64,65,66, and further supports that our subject-specific dosage should not be viewed as a study limitation. In a follow-up study, we used a constant 0.7 mA tDCS for all subjects and found a consistent activation pattern across subjects for the tEAS condition (Supplementary Fig. S12C).
We randomized the order of our experiments across subjects, which is also known as counterbalancing. While it is possible that conditions tested within a single session may experience a carry-over or sequence-based effect, counterbalancing, at the group-level, helps ensure that these effects were randomly distributed across conditions and should not confound the main results68. To further validate our main findings, we conducted an additional follow-up study, in which we repeated our etEAS and etFUS experiments with a 7-day washout period (8 participants; split 50/50 for which condition was first; Supplementary Table T4). On the day the condition was tested, we tested it twice: once ‘pre’ 5-minute break and once ‘post’ break. The ROI activity for each condition was statistically similar before and after the break (Supplementary Fig. S12A, B), suggesting that, even without the counterbalancing of the main study, the results may not have suffered from any sequence-based confounds. The source localizability of the experimental conditions was also consistent with the main study, where etEAS yielded strong targeting-location-specific activity, and etFUS did not (Supplementary Fig. S12C).
This study harnesses and demonstrates the electro-acoustic compounding effect in neural stimulation. In doing so, we develop a non-invasive technique that does not just modulate, but rather stimulates, targeted brain regions. Our results provide evidence that tFUS is a subthreshold co-modulator, and we have discussed how this theory is a unifying framework for, up until now, conflicting reports of tFUS neuromodulatory capabilities.
Methods
MRI and MRI segmentation
Subjects underwent a T1-weighted 3 T Magnetic Resonance Imaging (MRI) structural scan of their brain at the CMU-Pitt BRIDGE Center (RRID:SCR_023356). The brain images were then segmented in FreeSurfer (version 7)69,70 using the Desikan-Killiany atlas for post-hoc analysis. One subject was not MRI eligible due to an implanted device, so the FreeSurfer FSAverage brain was used71.
For EEG source imaging cortical parcellation, the primary auditory cortices and deep brain regions were defined by the left and right transversetemporal and medialwall parcellations, respectively. The IL/CL M1 ROI was defined by projecting the four IL/CL ROI electrodes orthogonally to the brain model’s inflated cortex and creating a square slice whose corners were defined by the electrodes.
Free water ultrasound calibration
This experiment used a 500 kHz f0 (fundamental frequency) single-element ultrasound transducer (1.5 inch focal length, 1 inch acoustic aperture diameter, 0.75 inch radius of curvature; Blatek Industries Inc., Boalsburg, PA, USA). To characterize the beam shape and determine the needed input voltage for the desired free water pressure, the transducer was calibrated in a water tank. The water tank was filled with deionized water. Pressure measurements were recorded with an HNR-0500 hydrophone (Onda Corp., Sunnyvale, CA, USA).
In silico transcranial focused ultrasound simulations
In the initial subject cohort, tFUS pressure was normalized with respect to the energy delivered on the scalp (0.98 MPa). To calculate the resulting on-cortex target ultrasound metrics (denoted with.T) across subjects, Sim4Life (Version 7.0, Zurich Med Tech; Zurich, Switzerland)72 computer simulations of the single-element tFUS transducer were conducted. Structural MRI files were converted to pseudo computed tomography (pCT) for use with the software73,74. The pCTs were manually segmented using iSeg (v3.10) into air (1.16 kg/m3 density, 343 m/s speed of sound, 0.000399 MRayl acoustic impedance), bone (1910 kg/m3 density, 3510 m/w speed of sound, 6.71 MRayl acoustic impedance), and soft tissue (1050 kg/m3 density, 1550 m/s speed of sound, 1.61 MRayl acoustic impedance). Sim4Life solved the acoustic simulations with the Linear Acoustic Pressure Wave Equation and the automatic discretization settings (0.45 mm resolution, 0.3 mm maximum step, and a time step of 1.0/(speed of soundmax * \(\sqrt{1/{{{{\rm{dx}}}}}^{2}+1/{{{{\rm{dy}}}}}^{2}+1/{{{{\rm{dz}}}}}^{2}}\))). For the simulations, the transducer was positioned over the subject’s left motor cortex, and the resultant pressure loss from brain tissue and skull bone attenuation was used to calculate spatial-peak temporal-average intensity (ISPTA.T; subject range = 48.6–372 mW/cm2), derated spatial-peak pulse-average intensity (ISPPA.T; 0.16 – 1.24 W/cm2), and Mechanical Index (MI.T; 0.10–0.28). The values for each subject are listed in Supplementary Table T1. In the case of two subjects, a pCT could not be generated (one subject did not have an MRI, and the other’s MRI had quality issues). All metrics were within the Food and Drug Administration’s guidelines for ultrasound (MI < 1.9, ISPPA < 190 W/cm2, and ISPTA < 720 mW/cm2)75.
For later subject cohorts, Sim4Life simulations were used to normalize in situ tFUS pressure to 250 kPa and 300 kPa. The equivalent free water pressures are provided in the relevant Supplementary data.
Neuromodulation-EEG experiment
Once the MRI segmentation was completed, subjects were asked to return for the neuromodulation-EEG portion of the study, which took place in an acoustic and electromagnetically shielded booth (IAC Acoustics; Naperville, Illinois, USA). Subjects were fit with a 64-channel EEG cap (BrainCap TMS, Brain Products, Gilching, Germany). EEG caps were aligned with subject landmarks following standard convention (centering electrode Cz between both the inion and nasion, as well as the right pre-auricular and left pre-auricular). EEG electrode impedances were lowered to be less than 10 kΩ using ABRALYT HiCl electrode gel.
The 4×1 high-definition tDCS (Soterix Medical Inc., Woodbridge, NJ, USA) electrodes were placed in the following locations: 1 peripheral electrode between AFz (ground), Fz, F1, and AF3, 1 peripheral electrode between CP1, CPz, Pz, and P1, 1 peripheral electrode between FC3, C3, C5, and FC5, 1 peripheral electrode between FC2, FC4, C2, and C4, and the center electrode between FCz (reference), Cz, C1, and FC1 (Fig. 1B). The electrical dosage for each subject was based on their own personal threshold for feeling scalp sensations. To determine the dosage, current was slowly ramped up from 0 until the subjects reported sensations. The dosage was set at 0.1 mA below their perception threshold, up to a maximum of 2 mA (Supplementary Table T1). The same amperage was used across all conditions involving tDCS (etEAS, itEAS, etDCS, and itDCS). Excitatory tDCS consisted of center-anode current, while center-cathode current was defined as inhibitory tDCS current. tDCS remained on throughout for the entirety of the 200 trials per condition.
For on-target ultrasound conditions (etEAS, itEAS, etFUS, and itFUS), the transducer was attached between EEG electrodes C1, C3, FC1, and FC3. Using an image-guided neuronavigation system (MagVenture Localite TMS Navigator; Alpharetta, Georgia, USA), the ultrasound target was visually confirmed to align with the subject’s left motor cortex. For off-target ultrasound spatial control conditions (etDCS, itDCS, and an additional 3 kHz tFUS), the ultrasound was targeted to the subject’s right prefrontal cortex area between electrodes AF4, AF8, F4, and F6. Based on our lab’s prior work, excitatory ultrasound was defined by a PRF of 3 kHz, and inhibitory ultrasound by a PRF of 30 Hz39 (Fig. 1B). The duty cycle (30%) and sonication duration (500 ms) were held constant.
For each condition, subjects were asked to assume a resting state for 200 trials (Fig. 1D). Specifically, they were instructed to close their eyes, remain as still as possible, and to relax their thoughts in a meditative-like state. The inter-trial interval was 2 seconds with up to a 20% (0.4 s) random jitter. A five-minute break was taken between each condition to allow the subjects to stretch and let any cumulative effects of neuromodulation dissipate. Experimental condition order was randomized across subjects, though data for e/itDCS control conditions were not collected in the initial subjects (Fig. 2D).
One follow-up study was conducted to investigate additional duty cycles (0.6%, 10%, and 60%) for 30 Hz and 3 kHz PRFs, and additional pulse repetition frequencies (1500 Hz and 40 Hz) with 30% DC. Changing tEAS parameters was also done in this follow-up to determine if the tEAS response differed as a function of PRF, but these conditions were always tested after the resting-state tFUS ones in order to minimize possible long-term contamination effects of tDCS. Another follow-up session was done to investigate the effects of normalizing in situ brain pressure. Other than these parameter changes, the protocol remained the same.
EEG analysis
EEG preprocessing
EEG data were bandpass filtered between 1 and 40 Hz with a zero-phase infinite impulse response filter. Data were epoched from 500 ms before the trial onset time to 1.5 seconds after. Bad channels were detected by averaging all the trials and identifying double median absolute deviation (double MAD; threshold = 3) outlier pre-stimulus variance or total trial peak-to-peak amplitude. On the un-averaged trials, independent component analysis eyeblink and muscle artifacts were removed using MNE Python’s76 automatic artifact detection with a z-threshold of 2.5 (Python version 3.9, MNE Python version 1.8). The data were common average referenced, and the previously identified bad channels were interpolated back into the signal using MNE Python’s interpolate_bads function. The cleaned data were averaged over all the trials to get the evoked waveform and z-scored with respect to the −500 to −100 ms pre-stimulus window. In a few instances, active tDCS created noticeable EEG artifacts. These data were excluded from the analysis (Fig. 2D).
EEG sensor domain analysis
Preprocessed EEG waveforms were averaged across subjects for each condition and used to produce butterfly plots (Fig. 2A). Statistical analysis was conducted using non-parametric spatio-temporal two-tailed cluster tests (5000 permutations, α = 0.05; Fig. 2B). Paired t-tests were conducted comparing ipsilateral ROI electrodes C1, C3, FC1, and FC3 against their homologous contralateral counterparts C2, C4, FC2, and FC4 within the exogenous (0–200 ms37) brain response window. Significantly different adjacent data were clustered together in time and space. Data were randomly shuffled, and the largest significant cluster from each permutation was used to construct a cluster-size histogram. The cluster-forming threshold was set at a significance level of 0.02. Clusters from the original data with sizes greater than the α percentile (corresponding to p < 0.05) were marked as significant.
ROI polarity analysis for the largest overlapping significant cluster components of tEAS was determined by taking the sign of electrode C3’s average over the response window (10–63 ms). McNemar’s test, a statistical procedure for assessing paired binary data, was used to investigate the effect of excitatory and inhibitory modulation parameters on ROI polarity for each condition. False discovery rate (FDR) multiple comparison correction was applied.
EEG source imaging
EEG source imaging (ESI) was solved by minimizing the difference between cortical current density produced by scalp EEG and recorded EEG7 using MNE Python’s implementation of dSPM, which normalizes the minimum norm estimation relative to a noise covariance matrix77. The noise covariance used for dSPM was calculated from each condition’s baseline window (−500 to −100 ms). EEG electrodes were anatomically aligned to the subject’s specific MRI model using the standard montage setup. The dSPM source maps were averaged over the 10–63 ms response window for ROI activity and localization analysis. Subject source maps were morphed from their individual head models to FreeSurfer’s FSAverage71 brain model for group-level visualizations (taking the average of each subject’s max-min scaled data) and connectivity analysis. For grand average ERP analysis (Supplementary Fig. S11), the subjects’ EEG data were averaged prior to EEG source imaging with the FSAverage brain model.
For each subject, the mean activity, precision (Eq. 1), recall (Eq. 2), and F1 score (Eq. 3) were calculated for the source-imaged activity with respect to the ipsilateral ROI. The ROI was defined by all the cortical space within a bounding box formed by projecting electrodes C1, C3, FC1, and FC3 (the electrodes between which tFUS was targeted) orthogonally from the scalp to the cortex. For precision, recall, and F1 score, true or false positives were defined as active brain sources that were inside, or outside, respectively, the region of interest. False negatives were defined as brain voxels within the region of interest that were not estimated as active sources. The threshold for what constituted a brain source, compared to noise, was defined using Otsu’s method56,57. Pairwise statistical comparisons between the mean activity, precision, recall, and F1 score were conducted with linear mixed effect modeling (see Linear Mixed Effect Modeling for more details).
The contralateral ROI was defined by the cortical space contained within the projection of electrodes C2, C4, FC2, and FC4. Comparisons between ipsilateral and contralateral mean activity were conducted with 5000 permutation one-tailed paired t-tests and FDR multiple comparison correction (H1: μIL > μCL). A permutation t-test is a nonparametric alternative to the parametric t-test, which relies on no underlying assumptions about data distributions. Instead, it shuffles the data between conditions an Npermutations number of times (in this case 5000) and calculates the T-statistic for each instance. This generates a histogram of T-statistics, against which the unshuffled data’s T-statistic is compared against to determine its significance. The p-value represents the proportion of permuted T-statistics that are at least as extreme as the unpermuted T-statistic. In some instances, comparisons across the ipsilateral activity for multiple conditions were assessed with permutation ANOVA tests. The permutation ANOVA test follows the same logic as the permutation t-test, except is it based on the F-statistic. Bayes Factor ANOVA testing was performed in R (version 4.3.3) with the BayesFactor package.
Connectivity analysis
Changes in Granger causality were calculated on source waveforms78 for the primary auditory cortex to deep brain and the targeted ROI to deep brain. Granger causality was calculated using the Toda and Yamamoto procedure79. The lag for each pathway was selected using the Akaike Information Criterion with possible choices between 10 and 15 ms based on reported values for transmission delays80. Net Granger causality (GCNet) was calculated by subtracting the reverse Y → X Granger causality Wald statistic from the forward X → Y Granger causality Wald statistic. GCNet was calculated for each 200 ms time bin from stimulus onset to one second after, as well as for the −0.5 to −0.1 s pre-stimulus baseline period. Permutation paired two-tailed t-tests were used to compare each time bin’s GCNet to the pre-stimulus period. FDR multiple comparison correction was applied locally for each series.
Spectral connectivity was assessed with imaginary coherence (Eq. 4), a metric used for quantifying non-zero time-lagged undirected connectivity81. The power spectral density (PSD) for each series was calculated using Welch’s method with a 0.2 s window in 0.2 s bins over the 0 to 1 second post-stimulus period and mean-normalized against the −0.5 to −0.1 s baseline period. Imaginary coherence was calculated for alpha (10–13 Hz), beta (13–30 Hz), and gamma (30–40 Hz) frequency bands. Comparison between the ipsilateral and contralateral changes in ImCoh for each time bin was assessed with 5000 permutation t-tests. FDR multiple comparison correction was applied locally for each condition/frequency band level.
Linear mixed effect modeling
The partially-paired data (Fig. 2D) made linear mixed-effect models excellent candidates for analyzing statistical differences across conditions. Linear mixed effect models account for both fixed effects (experimental conditions) and random effects (subject-to-subject differences) and estimate the true mean of the predictors on the outcome variables. For all our analyses, we modeled the relationship as Eq. 5. The outcome variable name is a stand-in for mean ROI activity, precision, recall, or F1 score. The outcomej,k variable for condition j and subject k is a function of some fixed offset for all subjects and all conditions, \({\beta }_{0}\), the estimated true mean effect of experimental condition j, \({\beta }_{1,j}\), and a subject-specific random effect for subject k, \({b}_{k}\). The difference between the model’s prediction and the observed outcome is the error term \({\epsilon }_{j,k}\). Since our independent variables are categorical, \({{{{\mathbf{\beta }}}}}_{{{{\bf{1}}}}}\) is a (1 x J) vector of the estimated true means of the effects, and Condition is a (J x 1) one-hot vector. Modeling was done in R (version 4.3.3) with the lme4 and multcomp packages.
Pairwise comparisons were made using R’s glht function, which calculates the mean difference of the estimated true effect across conditions and their pooled standard error. These values are used in z-tests to statistically compare condition means with FDR multiple comparison correction. The pooled standard error, used in calculating Cohen’s d (Eq. 6), was found by multiplying the pooled standard error by the square root of the subject number.
The primary assumptions of a linear mixed effect model are: (1) the response variable is linearly related to the predictor variable(s), (2) the model errors are independent, (3) the error variances are homogeneous, and (4) the errors are normally distributed. Since the independent variables are categorical (i.e., experimental condition), the model is calculating group means and not fitting a line, so assumption 1 is automatically satisfied. Non-violation of assumption 2 was tested by plotting the autocorrelation of the model residuals and identifying no significant systematic patterns. Assumptions 3 and 4 were assessed with Levene and Shapiro-Wilks tests, respectively. To meet the requirements of assumptions 3 and 4, ROI activity (strictly positive) was natural log transformed, and classification metric data (precision, recall, and F1; inclusively bound between 0 and 1) were square-root transformed.
Ethics
27 healthy human subjects volunteered to participate in the study. Subjects took part in a pre-experiment session where they were safety-screened for magnetic resonance imaging (MRI) and tFUS eligibility. Each participant gave informed written consent. Every experiment involving human participants has been carried out following a protocol approved by an ethical commission (Advarra Institutional Review Board). Subject recruitment and experiments were conducted in cohorts of 22 (subjects 101–122; 14F/8M; mean ± std age: 24.5 ± 4.0 years old), 11 (subjects 201-211; 7F/4M; mean ages = 23.22 ± 1.10 years old), 11 (subjects 301-311; 5F/6 M; mean age = 24.2 ± 2.3 years old), 6 (subjects 401–406; 4F/2M; mean ages = 23.6 ± 2.6 years old), and 8 (subjects 501–508; 5F/3M; mean ages = 23.6 ± 2.3 years old) participants. Some subjects participated in multiple cohorts of experimentation. Subjects were compensated at a rate of $20 per hour. We did not obtain consent to publish subject-identifying information.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings of this study are available within the article and its supplementary files. Any additional requests for information can be directed to and will be fulfilled by the corresponding authors. Source data are provided with this paper. Raw EEG data and defaced MRI files82 have been deposited in a publicly available Figshare repository [https://doi.org/10.6084/m9.figshare.31029691]83. Source data are provided with this paper.
Code availability
Python and R scripts generated and used for this study are publicly available, without restriction, on GitHub [https://github.com/bfinl/tFUS-ESI]84.
References
Cambiaghi, M. & Sconocchia, S. Scribonius Largus (probably before 1CE–after 48CE). J. Neurol. 265, 2466–2468 (2018).
Popko, L. & Sinclair, A. Papyrus Ebers. https://sae.saw-leipzig.de/en/documents/papyrus-ebers?version=445 (2024).
Nitsche, M. A. et al. Transcranial direct current stimulation: State of the art 2008. Brain Stimul. 1, 206–223 (2008).
Kricheldorff, J. et al. Evidence of Neuroplastic Changes after Transcranial Magnetic, Electric, and Deep Brain Stimulation. Brain Sci. 12, 929 (2022).
Antal, A. et al. Low intensity transcranial electric stimulation: Safety, ethical, legal regulatory and application guidelines (2017–2025: An update) – endorsed by the European Society for Brain Stimulation and by the International Federation for Clinical Neurophysiology. Clin. Neurophysio. 2111436 https://doi.org/10.1016/j.clinph.2025.2111436 (2025).
San-Juan, D. Cathodal transcranial direct current stimulation in refractory epilepsy: a noninvasive neuromodulation therapy. J. Clin. Neurophysiol. 38, 503 (2021).
He, B., Sohrabpour, A., Brown, E. & Liu, Z. Electrophysiological source imaging: a noninvasive window to brain dynamics. Annu Rev. Biomed. Eng. 20, 171–196 (2018).
Edwards, D. et al. Physiological and modeling evidence for focal transcranial electrical brain stimulation in humans: A basis for high-definition tDCS. Neuroimage 74, 266–275 (2013).
Collins, C. E. et al. Cortical cell and neuron density estimates in one chimpanzee hemisphere. Proc. Natl. Acad. Sci. USA 113, 740–745 (2016).
Huo, L. et al. Transcranial direct current stimulation enhances episodic memory in healthy older adults by modulating retrieval-specific activation. Neural Plast. 2020, e8883046 (2020).
Legon, W. et al. Transcranial focused ultrasound modulates the activity of primary somatosensory cortex in humans. Nat. Neurosci. 17, 322–329 (2014).
Tufail, Y. et al. Transcranial pulsed ultrasound stimulates intact brain circuits. Neuron 66, 681–694 (2010).
Yoo, S., Mittelstein, D. R., Hurt, R. C., Lacroix, J. & Shapiro, M. G. Focused ultrasound excites cortical neurons via mechanosensitive calcium accumulation and ion channel amplification. Nat. Commun. 13, 493 (2022).
Zhu, J. et al. The mechanosensitive ion channel Piezo1 contributes to ultrasound neuromodulation. Proc. Natl. Acad. Sci. 120, e2300291120 (2023).
Liu, C., Yu, K., Niu, X. & He, B. Transcranial focused ultrasound enhances sensory discrimination capability through somatosensory cortical excitation. Ultrasound Med. Biol. 47, 1356–1366 (2021).
Lee, W. et al. Image-guided transcranial focused ultrasound stimulates human primary Somatosensory Cortex. Sci. Rep. 5, 8743 (2015).
Lee, W. et al. Transcranial focused ultrasound stimulation of human primary visual cortex. Sci. Rep. 6, 34026 (2016).
Butler, C. R. et al. Transcranial ultrasound stimulation to human middle temporal complex improves visual motion detection and modulates electrophysiological responses. Brain Stimul. 15, 1236–1245 (2022).
Kosnoff, J., Yu, K., Liu, C. & He, B. Transcranial focused ultrasound to V5 enhances human visual motion brain-computer interface by modulating feature-based attention. Nat. Commun. 15, 4382 (2024).
Yu, K., Liu, C., Niu, X. & He, B. Transcranial Focused Ultrasound Neuromodulation of Voluntary Movement-related Cortical Activity in Humans. IEEE Trans. Biomed. Eng. 68, 1923–1931 (2021).
Fomenko, A. et al. Systematic examination of low-intensity ultrasound parameters on human motor cortex excitability and behavior. eLife 9, e54497 (2020).
Sanguinetti, J. L. et al. Transcranial focused ultrasound to the right prefrontal cortex improves mood and alters functional connectivity in humans. Front. Hum. Neurosci. 14, 52 (2020).
Niu, X., Yu, K. & He, B. On the neuromodulatory pathways of the in vivo brain by means of transcranial focused ultrasound. Curr. Opin. Biomed. Eng. 8, 61–69 (2018).
Guo, H. et al. Ultrasound produces extensive brain activation via a cochlear pathway. Neuron 98, 1020–1030.e4 (2018).
Sato, T., Shapiro, M. G. & Tsao, D. Y. Ultrasonic neuromodulation causes widespread cortical activation via an indirect auditory mechanism. Neuron 98, 1031–1041.e5 (2018).
Braun, V., Blackmore, J., Cleveland, R. O. & Butler, C. R. Transcranial ultrasound stimulation in humans is associated with an auditory confound that can be effectively masked. Brain Stimul. Basic Transl. Clin. Res. Neuromodul. 13, 1527–1534 (2020).
Folloni, D. et al. Manipulation of subcortical and deep cortical activity in the primate brain using transcranial focused ultrasound stimulation. Neuron 101, 1109–1116.e5 (2019).
Yang, P.-F. et al. Bidirectional and state-dependent modulation of brain activity by transcranial focused ultrasound in non-human primates. Brain Stimul. 14, 261–272 (2021).
Iida, K. et al. Noninvasive low-frequency ultrasound energy causes vasodilation in humans. J. Am. Coll. Cardiol. 48, 532–537 (2006).
Webb, T., Cheeniyil, R., Wilson, M. & Kubanek, J. Remote targeted electrical stimulation. J. Neural Eng. 20, 036030 (2023).
Darmani, G. et al. Individualized non-invasive deep brain stimulation of the basal ganglia using transcranial ultrasound stimulation. Nat. Commun. 16, 2693 (2025).
Kop, B. R. et al. Auditory confounds can drive online effects of transcranial ultrasonic stimulation in humans. eLife 12, RP88762 (2024).
Sassenhagen, J. & Draschkow, D. Cluster-based permutation tests of MEG/EEG data do not establish significance of effect latency or location. Psychophysiology 56, e13335 (2019).
Legon, W. et al. A retrospective qualitative report of symptoms and safety from transcranial focused ultrasound for neuromodulation in humans. Sci. Rep. 10, 5573 (2020).
Plaksin, M., Kimmel, E. & Shoham, S. Cell-Type-Selective Effects of Intramembrane Cavitation as a Unifying Theoretical Framework for Ultrasonic Neuromodulation. eNeuro 3, ENEURO.0136-15.2016 (2016).
Verhagen, L. et al. Offline impact of transcranial focused ultrasound on cortical activation in primates. eLife 8, e40541 (2019).
Pratt, H. Sensory ERP Components. in The Oxford Handbook of Event-Related Potential Components (eds Kappenman, E. S. & Luck, S. J.) 0 (Oxford University Press, 2011). https://doi.org/10.1093/oxfordhb/9780195374148.013.0050.
McColgan, P., Joubert, J., Tabrizi, S. J. & Rees, G. The human motor cortex microcircuit: insights for neurodegenerative disease. Nat. Rev. Neurosci. 21, 401–415 (2020).
Yu, K., Niu, X., Krook-Magnuson, E. & He, B. Intrinsic functional neuron-type selectivity of transcranial focused ultrasound neuromodulation. Nat. Commun. 12, 2519 (2021).
Hodgkin, A. L. & Huxley, A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. 117, 500–544 (1952).
Gottlieb, P. A. Chapter One - A Tour de Force: The Discovery, Properties, and Function of Piezo Channels. In Current Topics in Membranes (ed. Gottlieb, P. A.) vol. 79 1–36 (Academic Press, 2017).
Wu, J., Lewis, A. H. & Grandl, J. Touch, Tension, and Transduction – The Function and Regulation of Piezo Ion Channels. Trends Biochem. Sci. 42, 57–71 (2017).
Lewis, A. H. & Grandl, J. Piezo1 ion channels inherently function as independent mechanotransducers. eLife 10, e70988 (2021).
Schimek, N. et al. Repeated application of transcranial diagnostic ultrasound towards the visual cortex induced illusory visual percepts in healthy participants. Front. Hum. Neurosci. 14, 66 (2020).
Yu, K., Sohrabpour, A. & He, B. Electrophysiological source imaging of brain networks perturbed by low-intensity transcranial focused ultrasound. IEEE Trans. Biomed. Eng. 63, 1787–1794 (2016).
Downs, M. E. et al. Non-invasive peripheral nerve stimulation via focused ultrasound in vivo. Phys. Med. Biol. 63, 035011 (2018).
Johnstone, A. et al. A range of pulses commonly used for human transcranial ultrasound stimulation are clearly audible. Brain Stimul. 14, 1353–1355 (2021).
Beisteiner, R. et al. Transcranial pulse stimulation with ultrasound in alzheimer’s disease—a new navigated focal brain therapy. Adv. Sci. 7, 1902583 (2020).
Yu, K. & He, B. Transcranial focused ultrasound modulates visual thalamus in a nonhuman primate model. IEEE Trans. Biomed. Eng. 1–10 https://doi.org/10.1109/TBME.2025.3554935 (2025).
Stephani, C. & Koubeissi, M. Differences of intracranial electrical stimulation thresholds in the human brain. Brain Stimul. 8, 724–729 (2015).
Hensel, J., Scholz, G., Hurttig, U., Mrowinski, D. & Janssen, T. Impact of infrasound on the human cochlea. Hear. Res. 233, 67–76 (2007).
Oohashi, T. et al. Inaudible high-frequency sounds affect brain activity: hypersonic effect. J. Neurophysiol. 83, 3548–3558 (2000).
Zhang, T., Pan, N., Wang, Y., Liu, C. & Hu, S. Transcranial focused ultrasound neuromodulation: a review of the excitatory and inhibitory effects on brain activity in human and animals. Front. Hum. Neurosci. 15, 749162 (2021).
Blackmore, J., Shrivastava, S., Sallet, J., Butler, C. R. & Cleveland, R. O. Ultrasound neuromodulation: a review of results, mechanisms and safety. Ultrasound Med. Biol. 45, 1509–1536 (2019).
Raichle, M. E. The brain’s default mode network. Annu. Rev. Neurosci. 38, 433–447 (2015).
Otsu, N. A Threshold Selection Method from Gray-Level Histograms. IEEE Trans. Syst. Man, Cybern. 9, 62–66 (1979).
Sun, R., Sohrabpour, A., Worrell, G. A. & He, B. Deep neural networks constrained by neural mass models improve electrophysiological source imaging of spatiotemporal brain dynamics. Proc. Natl. Acad. Sci. 119, e2201128119 (2022).
Pilloni, G. et al. Tolerability and feasibility of at-home remotely supervised transcranial direct current stimulation (RS-tDCS): Single-center evidence from 6,779 sessions. Brain Stimul.: Basic, Transl., Clin. Res. Neuromodul. 15, 707–716 (2022).
Simpson, E. A. et al. Remotely monitored transcranial direct current stimulation in pediatric cerebral palsy: open label trial protocol. BMC Pediatr. 22, 566 (2022).
Lafon, B. et al. Low frequency transcranial electrical stimulation does not entrain sleep rhythms measured by human intracranial recordings. Nat. Commun. 8, 1199 (2017).
Poreisz, C., Boros, K., Antal, A. & Paulus, W. Safety aspects of transcranial direct current stimulation concerning healthy subjects and patients. Brain Res. Bull. 72, 208–214 (2007).
van Boekholdt, L., Kerstens, S., Khatoun, A., Asamoah, B. & Mc Laughlin, M. tDCS peripheral nerve stimulation: a neglected mode of action?. Mol. Psychiatry 26, 456–461 (2021).
Wang, J., Wei, Y., Wen, J. & Li, X. Skin Burn After Single Session of Transcranial Direct Current Stimulation (tDCS). Brain Stimul. 8, 165–166 (2015).
Kidgell, D. J. et al. Different current intensities of anodal transcranial direct current stimulation do not differentially modulate motor cortex plasticity. Neural Plast. 2013, 603502 (2013).
Agboada, D., Mosayebi Samani, M., Jamil, A., Kuo, M.-F. & Nitsche, M. A. Expanding the parameter space of anodal transcranial direct current stimulation of the primary motor cortex. Sci. Rep. 9, 18185 (2019).
Jamil, A. et al. Systematic evaluation of the impact of stimulation intensity on neuroplastic after-effects induced by transcranial direct current stimulation. J. Physiol. 595, 1273–1288 (2017).
Thielscher, A., Antunes, A. & Saturnino, G. B. Field modeling for transcranial magnetic stimulation: A useful tool to understand the physiological effects of TMS? in 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 222–225 https://doi.org/10.1109/EMBC.2015.7318340 (2015).
Corriero, E. F. Counterbalancing. In The SAGE Encyclopedia of Communication Research Methods, 278–281 (SAGE Publications, Inc, 2017). https://doi.org/10.4135/9781483381411.
Dale, A. M., Fischl, B. & Sereno, M. I. Cortical surface-based analysis: i. segmentation and surface reconstruction. NeuroImage 9, 179–194 (1999).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage 31, 968–980 (2006).
Fischl, B., Sereno, M. I., Tootell, R. B. H. & Dale, A. M. High-resolution intersubject averaging and a coordinate system for the cortical surface. Hum. Brain Mapp. 8, 272–284 (1999).
Sim4Life by www.zmt.swiss. Zurich Med Tech (version 7.0).
Yaakub, S. N. et al. Pseudo-CTs from T1-weighted MRI for planning of low-intensity transcranial focused ultrasound neuromodulation: An open-source tool. Brain Stimul. Basic Transl. Clin. Res. Neuromodulation 16, 75–78 (2023).
Yaakub, S. N. et al. Transcranial focused ultrasound-mediated neurochemical and functional connectivity changes in deep cortical regions in humans. Nat. Commun. 14, 5318 (2023).
FDA Center for Devices and Radiological Health. Marketing Clearance of Diagnostic Ultrasound Systems and Transducers - Guidance for Industry and Food and Drug Administration Staff. (2023).
Gramfort, A. et al. MNE software for processing MEG and EEG data. NeuroImage 86, 446–460 (2014).
Dale, A. M. et al. Dynamic statistical parametric mapping: combining fmri and meg for high-resolution imaging of cortical activity. Neuron 26, 55–67 (2000).
Babiloni, F. et al. Estimation of the cortical functional connectivity with the multimodal integration of high-resolution EEG and fMRI data by directed transfer function. NeuroImage 24, 118–131 (2005).
Toda, H. Y. & Yamamoto, T. Statistical inference in vector autoregressions with possibly integrated processes. J. Econ. 66, 225–250 (1995).
Tovée, M. J. Neuronal Processing: How fast is the speed of thought?. Curr. Biol. 4, 1125–1127 (1994).
Nolte, G. et al. Identifying true brain interaction from EEG data using the imaginary part of coherency. Clin. Neurophysiol. 115, 2292–2307 (2004).
Bischoff-Grethe, A. et al. A technique for the deidentification of structural brain MR images. Hum. Brain Mapp. 28, 892–903 (2007).
Kosnoff, J. et al. tFUS-ESI EEG Data Set for ‘Transcranial focused ultrasound induces source localizable cortical activation in resting state humans when applied concurrently with transcranial electric stimulation. figshare https://doi.org/10.6084/m9.figshare.31029691.v1 (2026).
Kosnoff, J. tFUS-ESI-Analysis. Zenodo https://doi.org/10.5281/zenodo.18202975 (2026).
Acknowledgements
This work was supported in part by NIH grants R01NS124564-01 (PI: B.H.), RF1NS131069 (PI: B.H.), R01NS127849 (PI: B.H.), R01NS096761 (PI: B.H.), RF1NS124564-02 (PI: B.H.), and T32EB029365 (J.K. and C.G.; PI: B.H.). J.K.’s work was supported in part by the National Science Foundation Graduate Research Fellowship Program under Grant No. DGE2140739. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Institutes of Health and the National Science Foundation. The authors would also like to thank Kings Jiang and Zach Johnson for their assistance in EEG capping, Elena Bondi, Jesse Rong, Chih-Yu Yeh, and Annabel Frake for discussions on EEG analysis and preprocessing, Yunruo Ni and Chih-Yu Yeh for assistance in 3D printing tFUS transducer holders, and Zherui Li for assistance in converting subject MRIs into pseudo-CT images. In silico tFUS simulations were conducted using Sim4Life by ZMT, www.zmt.swiss.
Author information
Authors and Affiliations
Contributions
Conceptualization: J.K., K.Y., and B.H. Methodology: J.K., C.G., K.Y., and B.H. Investigation: J.K., C.G., K.Y., and B.H. Data curation: J.K., J.Z., Y.D., and Y.Z. Formal analysis: J.K., C.G. Visualization: J.K. Writing—original draft: J.K. Writing—reviewing and editing: J.K., C.G., K.Y., and B.H. Supervision: B.H.
Corresponding author
Ethics declarations
Competing interests
B.H. and K.Y. are co-inventors of a US patent for EEG source imaging-informed tFUS neuromodulation technology (Patent Number: 12,515,079). B.H., K.Y., and J.K. are co-inventors of a pending US patent application for tFUS application (Application Number: PCT/US2025/041121). B.H. and J.K. are co-inventors of a pending provisional patent application for the tEAS technique (Application number: 63/915,778). The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Thomas Tarnaud and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kosnoff, J., Gonsisko, C., Yu, K. et al. Transcranial focused ultrasound induces source localizable cortical activation in resting state humans when applied concurrently with transcranial electric stimulation. Nat Commun 17, 2023 (2026). https://doi.org/10.1038/s41467-026-69853-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69853-8