Abstract
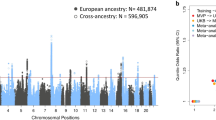
Age-related hearing loss is a prevalent and growing public health issue among the elderly. Here, we perform a multi-ancestry genome-wide association study comprising 456,613 cases and 1,053,834 controls, identifying 140 independent loci associated with age-related hearing loss, including 44 novel signals. We further fine-map 9 likely causal missense variants for age-related hearing loss and provide evidence of purifying selection for age-related hearing loss-associated variants. Notably, genetic risk for age-related hearing loss is strongly correlated with behavior traits such as neuroticism score and irritability. Integration of molecular phenotypes identifies 22 genes and 85 DNA methylation sites significantly associated with age-related hearing loss. Moreover, analyses incorporating spatial and single-cell transcriptomic identify the inner ear as a crucial site of age-related hearing loss, emphasizing the importance of hair cells, supporting cells, basal and root cells of the stria vascularis to its pathogenesis. Our study provides genetic and cellular insights into age-related hearing loss and advance our understanding of its genetics architecture.
Similar content being viewed by others
Data availability
All GWAS summary statistics used for ARHL meta-analysis are available as below: the ARHL GWAS of East Asian from BBJ is publicly available at https://pheweb.jp/pheno/Hearing_Loss; the ARHL GWASs of East Asian, European, African and Admixed American from MVP are available via the dbGap study accession number phs002453; the ARHL GWAS of European from Trpchevska et al. is available at https://zenodo.org/records/5769707#.Ybm6v33MKhx. The summary statistic of the cross ancestry meta-analysis from this study is available at https://zenodo.org/records/17141085. The summary-level xQTL data used for SMR are available as follow: eQTL data from eQTLGen project are available at https://www.eqtlgen.org/cis-eqtls.html and the meta-analysis data of mQTL from LBC and BSGS are available at https://cnsgenomics.com/software/smr/#Download. The Roadmap Epigenomics Mapping Consortium epigenomic annotations data are available for download at http://compbio.mit.edu/roadmap. The 1000 Genome project of European reference data (phase 3) are available at https://ftp.1000genomes.ebi.ac.uk/vol1/ftp/phase3/. The spatial transcriptomics data of mouse embryos at E16.5 used for gsMap is available at https://db.cngb.org/stomics/mosta/download. The single-cell RNA-seq data of mouse cochlea for Jean et al., Iyer et al., Eshel et al., and Sun et al. is available from the dataset access via the gEAR portal (https://umgear.org/p?s=7fd80bf5, https://umgear.org/p?s=728a05e2, https://umgear.org/p?s=bb49463b, and https://umgear.org/p?s=653896d7). Source data are provided in this paper.
Code availability
Code used in GWAS meta-analysis and results visualization are available at Github (https://github.com/Crazzy-Rabbit/project_hearing_loss/), which has been archived on Zenodo and assigned a DOI: (https://doi.org/10.5281/zenodo.17614049).
References
Haile, L. M. et al. Hearing loss prevalence and years lived with disability, 1990–2019: findings from the Global Burden of Disease Study 2019. Lancet 397, 996–1009 (2021).
Vollset, S. E. et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: a forecasting analysis for the Global Burden of Disease Study. Lancet 396, 1285–1306 (2020).
Deafness and hearing loss. https://www.who.int/news-room/fact-sheets/detail/deafness-and-hearing-loss (2025).
Kramer, S. E., Kapteyn, T. S., Kuik, D. J. & Deeg, D. J. H. The association of hearing impairment and chronic diseases with psychosocial health status in older age. J. Aging Health 14, 122–137 (2002).
Monzani, D., Galeazzi, G. M., Genovese, E., Marrara, A. & Martini, A. Psychological profile and social behaviour of working adults with mild or moderate hearing loss. Acta Otorhinolaryngol. Ital. 28, 61–66 (2008).
Momi, S. K., Wolber, L. E., Fabiane, S. M., MacGregor, A. J. & Williams, F. M. K. Genetic and Environmental Factors in Age-Related Hearing Impairment. Twin Res. Hum. Genet. 18, 383–392 (2015).
Christensen, K., Frederiksen, H. & Hoffman, H. J. Genetic and environmental influences on self-reported reduced hearing in the old and oldest old. J. Am. Geriatr. Soc. 49, 1512–1517 (2001).
Karlsson, K. K., Harris, J. R. & Svartengren, M. Description and primary results from an audiometric study of male twins. Ear Hear 18, 114–120 (1997).
Trpchevska, N. et al. Genome-wide association meta-analysis identifies 48 risk variants and highlights the role of the stria vascularis in hearing loss. Am. J. Hum. Genet. 109, 1077–1091 (2022).
Vuckovic, D. et al. Whole-genome sequencing reveals new insights into age-related hearing loss: cumulative effects, pleiotropy and the role of selection. Eur. J. Hum. Genet. 26, 1167–1179 (2018).
Bulik-Sullivan, B. K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Kurki, M. I. et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature 613, 508–518 (2023).
Brown, B. C., Asian Genetic Epidemiology Network Type 2 Diabetes Consortium, Ye, C. J., Price, A. L. & Zaitlen, N. Transethnic Genetic-Correlation Estimates from Summary Statistics. Am. J. Hum. Genet. 99, 76–88 (2016).
Yang, J. et al. Genetic variance estimation with imputed variants finds negligible missing heritability for human height and body mass index. Nat. Genet. 47, 1114–1120 (2015).
Uricchio, L. H., Zaitlen, N. A., Ye, C. J., Witte, J. S. & Hernandez, R. D. Selection and explosive growth alter genetic architecture and hamper the detection of causal rare variants. Genome Res. 26, 863–873 (2016).
Zeng, J. et al. Widespread signatures of natural selection across human complex traits and functional genomic categories. Nat. Commun. 12, 1164 (2021).
Bycroft, C. et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 562, 203–209 (2018).
Yan, D. et al. Biobank-wide association scan identifies risk factors for late-onset Alzheimer’s disease and endophenotypes. Elife 12, RP91360 (2024).
Zhu, Z. et al. Causal associations between risk factors and common diseases inferred from GWAS summary data. Nat. Commun. 9, 224 (2018).
Xue, A. et al. Unravelling the complex causal effects of substance use behaviours on common diseases. Commun. Med. 4, 43 (2024).
Wu, Y. et al. Genome-wide fine-mapping improves identification of causal variants. Eur. Neuropsychopharmacol. 87, 12-13 (2024).
Gazal, S. et al. Linkage disequilibrium–dependent architecture of human complex traits shows action of negative selection. Nat. Genet. 49, 1421–1427 (2017).
Wang, G., Sarkar, A., Carbonetto, P. & Stephens, M. A simple new approach to variable selection in regression, with application to genetic fine mapping. J. R. Stat. Soc. Series B Stat. Methodol. 82, 1273–1300 (2020).
Zou, Y., Carbonetto, P., Wang, G. & Stephens, M. Fine-mapping from summary data with the ‘Sum of Single Effects’ model. PLoS Genet. 18, e1010299 (2022).
Zhu, Z. et al. Integration of summary data from GWAS and eQTL studies predicts complex trait gene targets. Nat. Genet. 48, 481–487 (2016).
Võsa, U. et al. Large-scale cis- and trans-eQTL analyses identify thousands of genetic loci and polygenic scores that regulate blood gene expression. Nat. Genet. 53, 1300–1310 (2021).
McRae, A. F. et al. Identification of 55,000 Replicated DNA Methylation QTL. Sci. Rep. 8, 17605 (2018).
Kundaje, A. et al. Integrative analysis of 111 reference human epigenomes. Nature 518, 317–330 (2015).
Song, L., Chen, W., Hou, J., Guo, M. & Yang, J. Spatially resolved mapping of cells associated with human complex traits. Nature 641, 932–941 (2025).
Chen, A. et al. Spatiotemporal transcriptomic atlas of mouse organogenesis using DNA nanoball-patterned arrays. Cell 185, 1777–1792.e21 (2022).
Jean, P. et al. Single-cell transcriptomic profiling of the mouse cochlea: An atlas for targeted therapies. Proc. Natl. Acad. Sci. USA 120, e2221744120 (2023).
Finucane, H. K. et al. Heritability enrichment of specifically expressed genes identifies disease-relevant tissues and cell types. Nat. Genet. 50, 621–629 (2018).
de Leeuw, C. A., Mooij, J. M., Heskes, T. & Posthuma, D. MAGMA: generalized gene-set analysis of GWAS data. PLoS Comput. Biol. 11, e1004219 (2015).
Zhang, M. J. et al. Polygenic enrichment distinguishes disease associations of individual cells in single-cell RNA-seq data. Nat. Genet. 54, 1572–1580 (2022).
Iyer, A. A. et al. Cellular reprogramming with ATOH1, GFI1, and POU4F3 implicate epigenetic changes and cell-cell signaling as obstacles to hair cell regeneration in mature mammals. Elife 11, e79712 (2022).
Xu, Z. et al. Profiling mouse cochlear cell maturation using 10× Genomics single-cell transcriptomics. Front. Cell Neurosci. 16, 962106 (2022).
Eshel, M., Milon, B., Hertzano, R. & Elkon, R. The cells of the sensory epithelium, and not the stria vascularis, are the main cochlear cells related to the genetic pathogenesis of age-related hearing loss. Am. J. Hum. Genet. 111, 614–617 (2024).
Sun, G. et al. Single-cell transcriptomic atlas of mouse cochlear aging. Protein Cell 14, 180–201 (2023).
Vona, B. et al. A biallelic variant in CLRN2 causes non-syndromic hearing loss in humans. Hum. Genet. 140, 915–931 (2021).
Grillet, N. et al. Mutations in LOXHD1, an evolutionarily conserved stereociliary protein, disrupt hair cell function in mice and cause progressive hearing loss in humans. Am. J. Hum. Genet. 85, 328–337 (2009).
Scott, H. S. et al. Insertion of beta-satellite repeats identifies a transmembrane protease causing both congenital and childhood onset autosomal recessive deafness. Nat. Genet. 27, 59–63 (2001).
Mendia, C. et al. Clarin-2 gene supplementation durably preserves hearing in a model of progressive hearing loss. Mol. Ther. 32, 800–817 (2024).
Najarro, E. H. et al. Dual regulation of planar polarization by secreted Wnts and Vangl2 in the developing mouse cochlea. Development 147, dev191981 (2020).
Landin Malt, A., Hogan, A. K., Smith, C. D., Madani, M. S. & Lu, X. Wnts regulate planar cell polarity via heterotrimeric G protein and PI3K signaling. J. Cell Biol. 219, e201912071 (2020).
Milon, B. et al. A cell-type-specific atlas of the inner ear transcriptional response to acoustic trauma. Cell Rep. 36, 109758 (2021).
Ranum, P. T. et al. Insights into the biology of hearing and deafness revealed by single-cell RNA sequencing. Cell Rep. 26, 3160–3171 (2019).
Han, X. et al. Large-scale multitrait genome-wide association analyses identify hundreds of glaucoma risk loci. Nat. Genet. 55, 1116–1125 (2023).
Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium Electronic address: andrew.mcintosh@ed.ac.uk & Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium. Trans-ancestry genome-wide study of depression identifies 697 associations implicating cell types and pharmacotherapies. Cell 188, 640–652 (2025).
Peterson, R. E. et al. Genome-wide association studies in ancestrally diverse populations: opportunities, methods, pitfalls, and recommendations. Cell 179, 589–603 (2019).
De Angelis, F. et al. Sex differences in the polygenic architecture of hearing problems in adults. Genome Med. 15, 36 (2023).
Ivarsdottir, E. V. et al. The genetic architecture of age-related hearing impairment revealed by genome-wide association analysis. Commun. Biol. 4, 706 (2021).
Praveen, K. et al. Population-scale analysis of common and rare genetic variation associated with hearing loss in adults. Commun. Biol. 5, 540 (2022).
Verma, A. et al. Diversity and scale: genetic architecture of 2068 traits in the VA million veteran program. Science 385, eadj1182 (2024).
Nagai, A. et al. Overview of the bioBank Japan project: study design and profile. J. Epidemiol. 27, S2–S8 (2017).
Cherny, S. S. et al. Self-reported hearing loss questions provide a good measure for genetic studies: a polygenic risk score analysis from UK Biobank. Eur. J. Hum. Genet. 28, 1056–1065 (2020).
Willer, C. J., Li, Y. & Abecasis, G. R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010).
McVean, G. A. et al. An integrated map of genetic variation from 1092 human genomes. Nature 491, 56–65 (2012).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Kalra, G. et al. Biological insights from multi-omic analysis of 31 genomic risk loci for adult hearing difficulty. PLoS Genet. 16, e1009025 (2020).
Burgess, S., Butterworth, A. & Thompson, S. G. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet. Epidemiol. 37, 658–665 (2013).
Verbanck, M., Chen, C.-Y., Neale, B. & Do, R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat. Genet. 50, 693–698 (2018).
Bowden, J., Davey Smith, G., Haycock, P. C. & Burgess, S. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet. Epidemiol. 40, 304–314 (2016).
Zhao, Q., Wang, J., Hemani, G., Bowden, J. & Small, D. S. Statistical inference in two-sample summary-data Mendelian randomization using robust adjusted profile score. Ann. Statist. 48, 1742–1769 (2020).
Zheng, Z. et al. Leveraging functional genomic annotations and genome coverage to improve polygenic prediction of complex traits within and between ancestries. Nat. Genet. 56, 767–777 (2024).
Wu, Y. et al. Integrative analysis of omics summary data reveals putative mechanisms underlying complex traits. Nat. Commun. 9, 918 (2018).
Orvis, J. et al. gEAR: Gene expression analysis resource portal for community-driven, multi-omic data exploration. Nat. Methods 18, 843–844 (2021).
Durinck, S., Spellman, P. T., Birney, E. & Huber, W. Mapping identifiers for the integration of genomic datasets with the R/Bioconductor package biomaRt. Nat. Protoc. 4, 1184–1191 (2009).
Durinck, S. et al. BioMart and Bioconductor: a powerful link between biological databases and microarray data analysis. Bioinformatics 21, 3439–3440 (2005).
Acknowledgements
This research was supported by the Fundamental Research Funds for the Central Universities (Y.W.), the 1·3·5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYYC24006 to Y.W., ZYJC20002 to H.Y.), National Key Research and Development Program of China grant 82171836 (H.Y.). The research was supported by the National Supercomputing Center in Chengdu. The numerical calculations in this paper have been done on the Hefei Advanced Computing Center. This study makes use of data from the UK Biobank (project ID: 151441). We acknowledge the use of Grammarly for assistance with grammar correction.
Author information
Authors and Affiliations
Contributions
Y.W. conceived and designed the experiment. Y.W. supervised the study. L.S. conducted all analyses with the assistance or guidance from Y.W., H.Y., Y.Z., J.L., K.G., W.L., and H.H. L.S. and Y.W. wrote the manuscript with the participation of all authors. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Christopher Cederroth, Yuanhao Yang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shi, L., He, H., Li, J. et al. Multi-ancestry GWAS of age-related hearing loss identifies 140 loci and key cellular mechanisms. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69894-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69894-z