Abstract
N-bridged [3.2.1]octanes (tropanes) and their related bridged bicyclic systems constitute highly sought-after scaffolds in drug discovery and development. Notably, the enantioselective synthesis of chiral 3-aryltropanes which are compounds widely distributed across bioactive pharmaceutical agents remains underdeveloped. Tropinone is a readily available and cost-effective starting material. By initiating the synthesis from tropinone, it is possible to substantially lower the synthesis costs. Here we describe an enantioselective Pd/Ming-Phos-catalyzed β-H elimination reaction of Tropinone-derived N-arylsulfonylhydrazones and aryl bromides to give chiral tropanes and oxatropanes. Strikingly, this study achieves enantioselective β-H elimination which needs to simultaneously control over both diastereoselectivity during the migratory insertion and enantioselectivity during the β-H elimination. This approach shows broad functional group tolerance, good enantiocontrol as well as easy scale-up. Moreover, the synthetic value is further demonstrated by the enantioselective catalytic total synthesis of drugs for treating Alzheimer’s disease and monoamine transporter ligands. Additionally, both the facile elaborations and the preliminary biological activities of the products demonstrate the application potential.
Similar content being viewed by others
Introduction
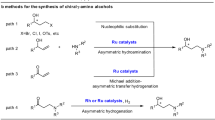
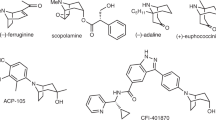
N-bridged [3.2.1]octane (tropane) scaffolds, as a distinguished subclass of the bicyclo[3.2.1] framework, have maintained enduring interest in synthetic and medicinal chemistry, primarily owing to their prevalent occurrence in diverse families of biologically active natural products1,2,3,4. Notably, 3-aryltropane derivatives exhibit considerable therapeutic potential for the treatment of neurological and psychiatric disorders, and also function as potent ligands for monoamine transporters—key molecular targets in neuropharmacology5,6,7,8,9 (Fig. 1a). However, only limited protocols toward asymmetric construction of chiral 3-aryltropanes have been disclosed so far10. In 2013, a three-step protocol was reported for the asymmetric synthesis of 3-aryltropanes, encompassing deprotonation of tropanone with a stoichiometric amount of chiral lithium amide base11, treatment with pyNTf₂ to form the corresponding enol triflate, and subsequent Fe-catalyzed Kumada coupling with benzylmagnesium bromide (Fig. 1b). Following deprotection, reaction with (S)-binoPCl in the presence of NEt₃ afforded a phosphoramidite as an inseparable mixture of two diastereomers in a 79:21 ratio10. This result confirms that chirality is not preserved during the Fe-catalyzed Kumada coupling, precluding the attainment of chiral 3-aryltropanes with high enantioselectivity. To date, no straightforward methodology exists for the synthesis of chiral 3-aryltropanes through asymmetric catalysis, despite their significance in expanding the structural diversity of tropane derivatives and facilitating the discovery of small-molecule therapeutic agents.
a Selected bioactive molecules possessing tropane cores. b Methods for synthesizing chiral tropanes. c The reaction modes for the asymmetric β-H elimination reaction from carbene coupling. This work: Pd/Ming-Phos-catalyzed asymmetric synthesis of tropanes.
The β-H elimination is a fundamental chemical process that appears in many transition-metal-catalyzed transformation12,13,14,15,16,17,18,19,20,21. However, there have been very limited efforts to investigate the stereoselectivity, as this elimination step converts two sp³ carbons into an alkene without forming a common stereocenter. The development of methods to achieve enantioselective β-H elimination represents a highly appealing research topic in asymmetric transition-metal catalysis. Meanwhile, as an important class of carbene precursors, sulfonylhydrazones are being increasingly applied in the field of metal-catalyzed carbene transformation reactions22,23,24,25,26,27,28,29,30,31,32,33,34. Nevertheless, reports on enantioselective β-H elimination reactions involving sulfonylhydrazones are still relatively scarce30,31,32,33,34. As shown in Fig. 1c, there have been only a few examples of constructing axially chiral30,33,34 and inherently chiral compounds31,32, while the construction of bridged ring skeletons remains unexplored. Moreover, previous strategies relied on substrate control to ensure that only one β-H elimination site was available for Pd elimination, thereby simplifying enantioselective control. In recent years, desymmetrization strategies have become one of the important approaches for constructing chiral compounds35,36,37,38,39. Recently, Zi’s group reported an elegant desymmetrization β-H elimination strategy that enabled the control of remote stereogenicity in six-membered rings39. However, the reaction requires the starting material to be in the trans configuration. Consequently, the trans relationship between the leaving group and the 4-substituent also restricts the elimination to only two of the four theoretically possible β-H atoms, thereby confining chiral induction to merely two β-H elimination pathways and simplifying enantiocontrol.
However, in our reaction, the presence of two distinct β-H elimination sites can lead to four possible elimination pathways during the migratory insertion and β-H elimination. This necessitates simultaneous control over both diastereoselectivity during the migratory insertion and enantioselectivity during the β-H elimination. To the best of our knowledge, no literature has reported the successful achievement of enantioselective β-H elimination in the presence of two distinct β-H sites.
In this work, we report the meticulous design of a Pd/Ming-Phos catalyst that enables enantioselective β-H elimination (Fig. 1b, c). This catalytic strategy, which initiates from cost-effective tropinone, achieves the synthesis of chiral tropanes and oxatropane scaffolds with good enantiocontrol. Preliminary biological activity evaluations reveal that compound 3l exhibits promising potential for further development as an anticancer drug. Notably, we introduce a method for the preparation of pharmaceuticals used in the treatment of Alzheimer’s disease. Unlike previous synthetic routes that relied on chiral resolution and harsh reaction conditions, our approach offers a more efficient pathway.
Results and discussion
To test our working hypothesis, we initially reacted N-arylsulfonylhydrazone 1a with phenyl bromide 2a in the presence of 10% mol Pd(OAc)2, 20 mol% of ligand, and tBuOLi in 1,4-Dioxane at 80 °C for 20 h. Initially, an exhaustive screening of various types of commercially available ligands failed to yield satisfactory results (See Supplementary information Table S1).
Then, we turned attention to our developed ligands (Sadphos), which have emerged as powerful ligands in various asymmetric catalysis40,41,42,43. The examination of the Sadphos ligand kit indicated that only N-H ligands could deliver good ee, in which M1 was the optimal choice, affording the tropane product 3a in 54% yield with 42% ee. Additionally, the use of N-Me ligands obviously shut down the enantioselectivity (See SI Table S1).
We next proposed a model for the interaction between Ming- Phos and 1a (Fig. 2a). Inspired by the easy synthesis and modification of Ming-Phos, we proceeded to screen a series of Ming-Phos (Fig. 2b). Subsequently, the tuning of the C4-substituted group (R) of Ming-Phos (M2-M8) was first carried out. The OMe-substitution (M2) conferred a slight improvement in ee values without yield erosion. By contrast, ligands M3-M8 failed to work satisfactorily. We then systematically evaluated the OMe group at different positions of the benzene ring, aiming to elucidate aromatic substitution effects. Satisfyingly, when the OMe group was positioned at the ortho-site (M10), a significant enhancement in yield (80%) and ee (74%) was observed. However, when the OMe group was positioned at the meta- or para-position, irrespective of using mono- or disubstituted ligands, the ee and yield of product remained low. These results indicated that the ortho position is closer to the reactive site to control the chiral pocket. Therefore, a series of ortho-OMe-modified ligands were evaluated. Fortunately, M14 afforded an improved 81% ee of 3a. Next, in view of the matching of the rate between oxidative addition and metal carbene generation, the variation of the aryl group (Ar2) on the P atom was investigated (M16–M21). Unfortunately, those ligands did not give better results. Finally, on the basis of the ligand model presented in Fig. 2a, we hypothesized that the electronic properties of R” might influence the interaction between the ligand and the β-H atoms. This prompted us to modify the R” and, with an electron-donating OMe group, M26 gave the best ee (83%).
a Proposed interaction model for β-H elimination. Preliminary screening results indicated that the formation of hydrogen bonds between the substrate and the ligand is crucial for enantioselectivity. b Detailed modification and rational design of ligands. Unless otherwise noted, all reactions were GC yield determined with 1,3-Dimethoxybenzene as an internal standard.
With the best ligand M26, further studies of the influence of the solvent, palladium, and temperature were conducted (Fig. 3). We systematically evaluated different polar and nonpolar solvents (Fig. 3, entries 1–7) and found that the polar solvent DMA performed better (82% yield, 86% ee). Subsequently, we focused on the optimization of metal salt and base (entries 8-17). When the metal salt and base were changed to Pd2dba3·CHCl3 and Cs2CO3, respectively, the desired product 3a was obtained in 84% yield with 89% ee (entry 17). At the same time, we found that lowering the temperature could improve the ee to 91% but resulted in a sharply lower yield (entry 18). The unsatisfactory yield of 3a may be ascribed to the mismatched rates for carbene generation and benzylpalladium intermediate formation. To this end, carbene precursors with distinctive substituents on the sulfonylphenyls were investigated. It is satisfactory to discover that easy-decomposed (2,4,6-triisopropylphenyl) sulfonylhydrazone 1c outperformed the other carbene precursors screened, affording 3a in 88% yield with 91% ee.
Reaction conditions: aUnless otherwise noted, all reactions were carried out with 0.1 mmol of 1a with M26 as the chiral ligand in 1.0 mL solvent at 80 °C for 20 h. bGC yield determined with 1,3-Dimethoxybenzene as an internal standard. cEnantioselectivity was determined by HPLC. d50 °C. eThe reaction was performed at 50 °C for 36 h.
Having established the optimized conditions, the scope of this reaction was examined by using various aryl bromides (Fig. 4). The results showed that aryl bromides bearing para-electron-donating groups or electron-withdrawing groups proceeded well to deliver 3a–3p. In regard to the substrates with para-strong electron-withdrawing substituents, the products 3o–3p were exclusively obtained in 76-77% yields with 93-94% ees when changing M26 to M27. Some functional groups, such as ester (3p), trimethylsilyl (3q), and vinyl (3r), were incorporated, allowing the products to be amenable to further transformations. Generally, good functional group tolerance was also achieved for meta-substituents of aryl bromides (3s–3y). Furthermore, ortho-substituted substrates (3z–3ba) achieved good ees, although the yields were moderate. This outcome may be attributed to the steric hindrance posed by the bulky substituents. Notably, this strategy could also be extended to disubstituted aryl bromides, generating the products 3bb–3bi in satisfactory yields and ees. We also found that our method was applicable to benzannulated ring systems, regardless of bearing electron-withdrawing or electron-donating substituents (3bj–3bo). To explore the scope of our reaction further, we incorporated heterocyclic rings into the aryl bromides, successfully synthesizing products 3bp–3bv with 60-87% yields and 87-94% ees. Notably, the transformation was effective even with the more challenging pyridine substrate (3bv). Furthermore, the absolute configuration of the product was unambiguously confirmed by X-ray diffraction analysis of compound 3bd.
Reaction conditions: Unless otherwise noted, all reactions were performed with 1 (0.3 mmol), 2 (0.36 mmol), Cs2CO3 (1.2 mmol), 5 mol% Pd2dba3•CHCl3, and 20 mol% M26 in 3.0 mL DMA at 50 °C for 36 h. Isolated yields were given. The ee values were determined by chiral HPLC analysis. aReactions were performed with 20 mol% M27. bReactions were performed with 20 mol% M27 and in 3.0 mL PhMe.
Next, the effect of substituents on the tropane scaffolds was investigated under the standard reaction conditions (Fig. 4). Satisfactorily, this asymmetric β-H elimination was also applicable to the tropanes bearing different N-protecting groups, delivering the products 4a–4k with high yields (71–96%) and ees (87–95%). Furthermore, changing N-substituents to O-substituents on the tropanes proceeded smoothly, affording the corresponding products 4k with satisfactory results.
To ascertain the scope of this method, several aryl bromides derived from the core structures of natural products were further investigated (Fig. 5a). All these structurally complex substrates (3ca–3ce) underwent the β-H elimination to afford the corresponding products in high yields and diastereoselectivities. To illustrate the practicality of this β-H elimination reaction, a larger-scale synthesis of 3c and 4b was carried out (Fig. 5b, c). Under the standard conditions, the products 3c and 4b were isolated in 86% yield with 92% ee and 71% yield with 91% ee, respectively. Subsequently, the Boc group could be substituted by different other groups, thus leading to 5-7 in 71-81% yields and 89-91% ee. Moreover, upon oxidation by NaBO3·4H2O, the 3c could undergo hydroboration-oxidation reaction to furnish 8 and further proceed Dess-Martin oxidation reaction to give unsaturated 9 in an overall 60% yield with 89% ee. Meanwhile, with the existence of m-CPBA, the epoxidation of 3c could deliver 10 in 83% yield and 91% ee. In addition, 3c was able to afford dihydroxylation product 11 in good yield and ee. Finally, enantioenriched compound 12 that bears an intrinsically synthetic challenge could be easily accessed via three-step transformations of 3c.
All yields in the figure refer to the isolated yields after flash column chromatography. a Late-stage modification of drug molecules. b The functionalization of the product 3c. c The functionalization of the product 4b.
It was found that the chiral N-bridged [3.2.1]octane 15 and 19 were present in pharmaceuticals treating Alzheimer’s disease (Fig. 6a)44. Our method provided a straightforward approach to 15 in 58% overall yield and 94% ee. In contrast to previous routes, which required chiral resolution and harsh reaction conditions involving high-pressure, high-temperature, and toxic CO gas, this reaction produced 15 in high ee under mild conditions, significantly reducing the synthesis cost. Furthermore, 4c could also undergo hydroboration-oxidation and be transformed into 18, which was a key intermediate in the total synthesis of 19 (Fig. 6b). To our delight, removal of the Boc group of 4g and 4c afforded 21 and 22, which were recently shown to have affinity and high selectivity for monoamine transporters (Fig. 6c)45. Therefore, our reaction provided an efficient strategy to prepare analogs by starting with different aryl bromides. It was suited for finding new compounds that display a broad spectrum of monoamine transporter selectivity. In view of the broad bioactive properties of tropanes family, we were intrigued by the potential biological activities of these chiral N-bridged [3.2.1]octane scaffolds. Accordingly, the cytotoxic effects of these compounds against cancer cells were evaluated by Cell Counting Kit-8 (CCK-8) assay. Our preliminary studies demonstrated that many of them exhibited significant cytotoxic effects on cancer cells at the concentration of 20 μM (see Table S2 in Supplementary information for details). Next, we further determined the cytotoxic IC50 Values of some selected active compounds (Fig. 6d). Notably, the IC50 value of 3l was in the range of 10-20 μM, which indicated that 3l showed potential for further development into an anticancer drug.
All yields in the figure refer to the isolated yields after flash column chromatography. a Enantioselective synthesis of 15, which can treat Alzheimer’s disease. b Enantioselective synthesis of 19, which can treat Alzheimer’s disease. c Enantioselective synthesis of monoamine transporter ligands 21 and 22. d The preliminary biological investigations. Concentration-dependent inhibitory curves of compounds 3l, 3bc, and 3ca in A549 cells. The data are presented as the means ± SEM (n = 3).
In order to gain insights into this asymmetric β-H elimination reaction, mechanistic investigations were carried out (Fig. 7). First, nonlinear effect studies indicated that there was a significant first-order dependence on catalyst (Fig. 7a). To identify the rate-determining step of the reaction, we next investigated the Hammett plots of various para-substituted aryl bromides (Fig. 7b). These results indicated that electronic variation of the aryl bromides had no impact on the rate. Therefore, oxidative addition of aryl bromides might not be the rate-determining step. Additionally, if 0 < ρ < 1 (ρ is the slope of the Hammett plot), it indicates that the rate-determining step involved only minor charge separation and the mechanism or rate-determining step might be concerted46. Moreover, we performed experiments with N-sulfonylhydrazones 1 and [D]−1 to measure the initial reaction rate, respectively. The side-by-side experiments provided a KH/KD value of 1.08 (Fig. 7c). The intermolecular competition reaction of 1 and [D]−1c in one pot showed a KH/KD value of 1.2 calculated from the generation of 3a and [D]−3a (Fig. 7d). These results suggested that the β-H elimination process might not be the rate-determining step. Next, the kinetic analysis experiments were conducted employing 1c and 2a (Fig. 7e). Based on the kinetic experiment’s data, we plotted the logarithm of the reaction rate (-Ln(K)) against the logarithm of the concentration of Pd, sulfonylhydrazones 1c, and PhBr 2a, respectively. Thus, we obtained the initial reaction rate: Rateinitial = Kobs[1c]1.01[2a]0.07[PdL*]0.58 (Supplementary information Fig. S1). Therefore, we determined that the reaction exhibits approximately first-order kinetics with substrate 1c and fractional order for the catalyst, and approximately zero-order kinetics with PhBr 2a. Firstly, the kinetic results indicated that the reaction was zero-order in aryl bromide 2a, which suggested that changes of aryl bromide (bond cleavage or 1,2-migratory insertion) were not involved in the rate-determining intermediate. Secondly, the first-order dependence for 1c and the fractional order for palladium catalyst disclosed that the rate-determining step involved interactions (bond formation or cleavage) between the Pd and 1c. In our reaction, carbene formation involved bond formation between Pd and 1c, and β-H elimination involved bond cleavage between Pd and 1c. Since KIE experiments excluded β-H elimination as the rate-determining step, we inferred that carbene generation might be the rate-determining step and proceeded via a concerted process, which was consistent with the result of the Hammett plot. According to the previous reports34,35, the decomposition rates of Ts-, Mes-, and Tris-groups increased with the steric bulk of the substituent. Finally, we determined the initial reaction rates for 1a (Ts), 1b (Mes), 1c (Tris) at 50 °C, the experimental results showed that the initial reaction rates follow the order 1c (Tris) > 1b (Mes) > 1a (Ts)47,48, which further supports that carbene formation might be the rate-determining step in this reaction (see SI Fig. S9). Additionally, some control experiments were then conducted. We conducted radical inhibition experiments under standard conditions (Supplementary information Fig. S10). The reaction was not inhibited regardless of whether TEMPO or BHT was added. Therefore, a single-electron transfer mechanism had been ruled out. Then, after the reaction proceeded under standard conditions for 36 h, the product 3a was obtained with an 88% yield and 91% ee. Then we added additional starting material 1c, bromobenzene 2a, and Cs2CO3 to the mixture, and the product 3a was still obtained with an 85% yield and 90% ee (See SI Fig. S11). Therefore, we inferred that no significant catalyst deactivation occurred in this reaction.
a Nonlinear experiment. b Hammett study for the formation of 3 versus the corresponding σ value (k = reaction rate). c Deuterium kinetic isotope effect (KIE): side-by-side reaction. d Deuterium kinetic isotope effect (KIE): intermolecular competition. e Kinetic studies of catalyst, substrate 1, and aryl bromides. f Proposed mechanism.
Based on the experimental results and previous reports29,49, we propose a mechanism as illustrated in Fig. 7f. The reaction is initiated with the oxidative addition of phenyl bromide 2 to the Pd(0) catalyst. The Pd(II) intermediate A is generated, followed by the reaction with diazo compound 1′, delivering the Pd carbene intermediates C. As an alternative, the Pd carbene intermediate B is generated by the coordination of Pd(0) with 1’. Then intermediate B undergoes oxidation addition with 2 to furnish the Pd carbene intermediate C. Path a is favored over Path b in this reaction49. Subsequently, the carbene migratory insertion and ligand exchange occur to form benzylpalladium species D and E. Finally, E undergoes β-H elimination to afford the final product 3 or 4 as well as regenerate the palladium catalyst with the aid of the base. Alternatively, the ligand can also be exchanged first to generate intermediate F, and then carbene migratory insertion to form E. The irreversibility of the β-H elimination process is critical for maintaining the ee value of product 3 or 4. Moreover, based on experiment results and our proposed model, the hydrogen bond between ligand and the substrate is crucial for the reaction to proceed, which enables the substrate to bind with Pd and promote carbene formation (Fig. 7f and SI Fig. S13).
In summary, we have developed a newly identified Ming-Phos ligand, allowing highly enantioselective β-H elimination for the synthesis of multifunctional N-bridged [3.2.1]octane scaffolds, which requires modulating the reactivity of the strained carbene in an enantioselective and stereospecific manner. The value of these N-bridged [3.2.1]octane scaffolds is demonstrated by the synthesis of drugs for treating Alzheimer’s disease and monoamine transporter ligands. Moreover, these tropane compounds exhibit diversified biological activities. Additionally, mechanistic studies indicate that β-H elimination might not be the rate-determining step in this process. We anticipate that this methodology will be of broad interest to researchers in asymmetric β-H elimination reactions and medicinal sciences.
Methods
Representative procedure A for asymmetric arylation of N-bridged [3.2.1]octane derivatives (3 or 4)
To a 10 mL oven-dried sealed tube was added substrate 1 (0.30 mmol, 1.0 equiv.), Pd2(dba)3•CHCl3 (15.5 mg, 0.015 mmol, 5 mol%), M26 (38.3 mg, 0.06 mmol, 20 mol%), Cs2CO3 (391.2 mg, 1.2 mmol, 4.0 equiv.). The flask was evacuated and refilled with argon. Then, substrate 2 (0.36 mmol, 1.2 equiv.), DMA (3 mL) was added to the tube and stirred at room temperature for 1 h. Then the mixture was stirred at 50 °C for 36–60 h. After the reaction was complete (monitored by TLC), the solvent was removed under reduced pressure. The crude product was then purified by flash column chromatography on silica gel to afford the desired product 3.
Representative procedure B for asymmetric arylation of N-bridged [3.2.1]octane derivatives (3 or 4)
To a 10 mL oven-dried sealed tube was added substrate 1 (0.30 mmol, 1.0 equiv.), Pd2(dba)3•CHCl3 (15.5 mg, 0.015 mmol, 5 mol%), M27 (39.3 mg, 0.06 mmol, 20 mol%), Cs2CO3 (391.2 mg, 1.2 mmol, 4.0 equiv.). The flask was evacuated and refilled with argon. Then, substrate 2 (0.36 mmol, 1.2 equiv.), DMA (3 mL) was added to the tube and stirred at room temperature for 1 h. Then the mixture was stirred at 50 °C for 36–60 h. After the reaction was complete (monitored by TLC), the solvent was removed under reduced pressure. The crude product was then purified by flash column chromatography on silica gel to afford the desired product 3 or 4.
Representative procedure C for asymmetric arylation of N-bridged [3.2.1]octane derivatives (3 or 4)
To a 10 mL oven-dried sealed tube was added substrate 1 (0.30 mmol, 1.0 equiv.), Pd2(dba)3•CHCl3 (15.5 mg, 0.015 mmol, 5 mol%), M27 (39.3 mg, 0.06 mmol, 20 mol%), Cs2CO3 (391.2 mg, 1.2 mmol, 4.0 equiv.). The flask was evacuated and refilled with argon. Then, substrate 2 (0.36 mmol, 1.2 equiv.), PhMe (3 mL) was added to the tube and stirred at room temperature for 1 h. Then the mixture was stirred at 50 °C for 36–60 h. After the reaction was complete (monitored by TLC), the solvent was removed under reduced pressure. The crude product was then purified by flash column chromatography on silica gel to afford the desired product 3 or 4.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information. And all data are available from the corresponding author upon request. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2451377 (3bd). Copies of the data can be obtained free of charge via www.ccdc.cam.ac.uk/data_request/cif.
References
Huang, J.-P. et al. Tropane alkaloid biosynthesis: a centennial review. Nat. Prod. Rep. 38, 1634–1658 (2021).
Shim, K. H., Kang, M. J., Sharma, N. & An, S. S. A. Beauty of the beast: anticholinergic tropane alkaloids in therapeutics. Nat. Prod. Bioprospect. 12, 33–47 (2022).
Fodor, G. & Dharanipragada, R. Tropane alkaloids. Nat. Prod. Rep. 11, 443–450 (1994).
Kohnen-Johannsen, K. L. & Kayser, O. Tropane alkaloids: chemistry, pharmacology, biosynthesis and production. Molecules 24, 796–818 (2019).
Grynkiewicz, G. & Gadzikowska, M. Tropane alkaloids as medicinally useful natural products and their synthetic derivatives as new drugs. Pharmacol. Rep. 60, 439–462 (2008).
Afewerki, S., Wang, J.-X., Liao, W.-W. & Córdova, A. The chemical synthesis and applications of tropane alkaloids. Alkaloids Chem. Biol. 81, 151–233 (2019).
Monn, J. A., Thurkauf, A., Mattson, M. V., Jacobson, A. E. & Rice, K. C. Synthesis and structure-activity relationship of C5-substituted analogs of (.+-.)−10,11-dihydro-5H-dibenzo[a,d]cyclohepten-5, 10-imine [(.+-.)-desmethyl-MK801]: ligands for the NMDA receptor-coupled phen-cyclidine binding site. J. Med. Chem. 33, 1069–1076 (1990).
Jin, C., Navarro, H. A. & Carroll, F. I. Development of 3-phenyltropane analogues with transporters and low affinity for the norepinephrine transporter. J. Med. Chem. 51, 8048–8056 (2008).
Zayene, M. et al. Site-selective palladium(II)-catalyzed methylene C(sp3)-H diaraylation of a tropane scaffold. J. Org. Chem. 87, 16399–16409 (2022).
Vlahovic, S., Schädel, N., Tussetschläger, S. & Laschat, S. Tropanes as scaffolds for phosphorus–olefin ligands and their application in asymmetric catalysis. Eur. J. Org. Chem. 2013, 1580–1590 (2013).
Majewski, M. & Lazny, R. Synthesis of tropane alkaloids via enantioselective deprotonation of tropinone. J. Org. Chem. 60, 5825–5830 (1995).
Wilkinson, G. The long search for stable transition metal alkyls. Science 185, 109–112 (1974).
Han, X.-L., Liu, G.-X. & Lu, X.-Y. Hydride elimination in palladium catalyzed reactions. Chin. J. Org. Chem. 25, 1182–1197 (2005).
Bras, J. L. & Muzart, J. Intermolecular dehydrogenative Heck reactions. Chem. Rev. 111, 1170–1214 (2011).
Li, Y. & Yin, G. Nickel chain-walking catalysis: a journey tomigratory. carboboration of alkenes. Acc. Chem. Res. 56, 3246–3259 (2023).
Turlik, A., Chen, Y. & Newhouse, T. R. Dehydrogenation adjacent to carbonyls using palladium-allyl intermediates. Synlett 27, 331–336 (2016).
Bogdos, M. K., Stepanović, O., Bismuto, A., Luraschi, M. G. & Morandi, B. Mechanistically informed selection rules for competing β-hydride and β-heteroatom eliminations. Nat. Synth. 1, 787–793 (2022).
Lu, X. Control of the β-hydride elimination making palladium catalyzed coupling reactions more diversified. Top. Catal. 35, 73–86 (2005).
Yip, S. Y. Y. & Aïssa, C. Isomerization of olefins triggered by rhodium-catalyzed C-H bond activation: control of endocyclic β-hydrogen elimination. Angew.Chem. Int. Ed. 54, 6870–6873 (2015).
Yang, M.-H. & Altman, R. A. -Elimination rules for Pd. Nat. Synth. 1, 753–754 (2022).
Yin, G., Mu, X. & Liu, G. Palladium(II)-catalyzed oxidative difunctionalization of alkenes: bond forming at a high-valent palladium center. Acc. Chem. Res. 49, 2413–2423 (2016).
Xia, Y. & Wang, J. N-Tosylhydrazones: versatile synthons in the construction of cyclic compounds. Chem. Soc. Rev. 46, 2306–2362 (2017).
Xia, Y., Qiu, D. & Wang, J. Transition-metal-catalyzed cross-couplings through carbene migratory insertion. Chem. Rev. 117, 13810–13889 (2017).
Xia, Y. & Wang, J. Transition-metal-catalyzed cross-coupling with ketones or aldehydes via N-tosylhydrazones. J. Am. Chem. Soc. 142, 10592–10605 (2020).
Sivaguru, P., Pan, Y., Wang, N. & Bi, X. Who is who in the carbene chemistry of N-sulfonyl hydrazones. Chin. J. Chem. 42, 2071–2108 (2024).
Zhang, X. et al. The carbene chemistry of N-sulfonyl hydrazones: the past, present, and future. Chem. Rev. 125, 1049–1190 (2025).
Barluenga, J., Moriel, P., Valdés, C. & Aznar, F. N-tosylhydrazones as reagents for cross-coupling reactions: a route to polysubstituted olefins. Angew. Chem. Int. Ed. 46, 5587–5590 (2007).
Zhao, G., Wu, Y., Wu, H., Yang, J. & Zhang, J. Pd/GF-Phos-catalyzed asymmetric three-component coupling reaction to access chiral diarylmethyl alkynes. J. Am. Chem. Soc. 143, 17983–17988 (2021).
Yang, B., Cao, K., Zhao, G., Yang, J. & Zhang, J. Pd/Ming-Phos-catalyzed asymmetric three-component arylsilylation of N-sulfonylhydrazones: enantioselective synthesis of gem-diarylmethine silanes. J. Am. Chem. Soc. 144, 15468–15474 (2022).
Feng, J., Li, B., He, Y. & Gu, Z. Enantioselective synthesis of atropisomeric vinyl arene compounds by palladium catalysis: a carbene strategy. Angew. Chem. Int. Ed. 55, 2186–2190 (2016).
Wang, X. et al. Enantioselective synthesis of inherently chiral 9-benzylidene-9H-tribenzo[a,c,e][7]annulene and its application as a ligand platform. Chem Catal. 4, 100904–100916 (2024).
Zhang, H. et al. Palladium-catalyzed asymmetric carbene coupling enroute to inherently chiral heptagon-containing polyarenes. Nat. Commun. 15, 3353–3361 (2024).
Ning, X., Chen, Y., Hu, F. & Xia, Y. Palladium-catalyzed carbene coupling reactions of cyclobutanone N-sulfonylhydrazones. Org. Lett. 23, 8348–8352 (2021).
Ning, X. et al. Enantioselective synthesis of axially chiral alkylidenecyclobutanes via palladium-catalyzed N-tosylhydrazone-based carbene coupling. J. Am. Chem. Soc. 147, 16773–16780 (2025).
Zeng, X.-P., Cao, Z.-Y., Wang, Y.-H., Zhou, F. & Zhou, J. Catalytic enantioselective desymmetrization reactions to all-carbon quaternary stereocenters. Chem. Rev. 116, 7330–7396 (2016).
Xu, Y., Zhai, T.-Y., Xu, Z. & Ye, L.-W. Recent advances towards organocatalytic enantioselective desymmetrizing reactions. Trends. Chem. 4, 191–205 (2022).
Nájera, C., Foubelo, F., Sansano, J. M. & Yus, M. Enantioselective desymmetrization reactions in asymmetric catalysis. Tetrahedron 106-107, 132629 (2022).
Zhao, S. et al. Enantioselective synthesis of P-chiral phosphine oxides bearing an all-carbon quaternary stereogenic center via palladium-catalyzed Domino Heck−Suzuki Reaction. ACS Catal. 14, 14762–14768 (2024).
Sun, S., Sun, S. & Zim, W. Palladium-catalyzed enantioselective β-hydride elimination for the construction of remote stereocenters. Nat. Commun. 16, 2227–2236 (2025).
Xu, B. et al. Palladium/Xu-Phos-catalyzed enantioselective cascade Heck/remote C(sp2)–H alkylation reaction. Chem 8, 836–849 (2022).
Tu, Y. et al. Palladium/TY-Phos-catalyzed asymmetric Heck/Tsuji–Trost reaction of o-bromophenols with 1,3-dienes. J. Am. Chem. Soc. 145, 4378–4383 (2023).
Fang, C., Wang, Q.-P., Xu, B., Zhang, Z.-M. & Zhang, J. Palladium/Xu-Phos-catalyzed enantioselective cascade Heck/intermolecular C(sp2)–H alkylation reaction. Chem. Sci. 15, 5573–5580 (2024).
Wang, J., Xu, B., Wang, Y., Zhang, Z.-M. & Zhang, J. Pd-catalyzed enantioselective three-component carboamination of 1,3-cyclohexadiene. J. Am. Chem. Soc. 146, 21231–21238 (2024).
Nieman, J. A., Fang, L. & Barbara, J. Methods of treating or preventing Alzheimer’s disease using 4-aryl-3-aralkoxypiperidines and aza-bicyclooctanes. World Intellect. Prop. Organ. WO2002076440. A2. 2002-10-03 (2022).
Kaur, H. et al. Synthesis and monoamine transporter affinity of 3α-arylmethoxy-3β-arylnortropanes. Bioorg. Med. Chem. Lett. 19, 6865–6868 (2009).
Hammett, L. P. The effect of structure upon the reactions of organic compounds. Benzene derivatives. J. Am. Chem. Soc. 59, 96–103 (1937).
Lipták, A., Oláh, V. A. & Kerékgyártó, J. A convenient synthesis of carbohydrate methylene acetals. Synthesis 5, 419–421 (1982).
Liu, Z., Babu, K. R., Wang, F., Yang, Y. & Bi, X. Influence of sulfonyl substituents on the decomposition of N-sulfonylhydrazones at room temperature. Org. Chem. Front. 6, 121–124 (2019).
Xiao, B., Sun, T.-Y., Zhang, J. & Wu, Y.-D. Theoretical insight into the activity and selectivity in palladium/ Ming-Phos-catalyzed three-component asymmetric synthesis of gem-diarylmethine silanes. Sci. China Chem. 66, 2817–2827 (2023).
Acknowledgements
The authors gratefully acknowledge the funding support of the National Key R&D Program of China (No. 2021YFF0701600), NSFC (No. 22031004, 22501294), the Shanghai Municipal Education Commission (No. 20212308), and STCSM (No. 23ZR1445600). The authors also particularly thank Dr. Chengbin Yang for his contributions to the evaluation of biological activity.
Author information
Authors and Affiliations
Contributions
C.F. carried out the experimental and data-analysis work. J.A. and Q.W. carried out the experimental work. B.X. carried out data-analysis work and wrote the paper. Z.-M.Z. and J.Z. designed the reaction, directed the project, and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Lingchao Cai, Ying Xia, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fang, C., Ai, J., Wang, Q. et al. Asymmetric synthesis of Heteroatom-bridged [3.2.1]Octane scaffolds via enantioselective β-H elimination reaction. Nat Commun 17, 3315 (2026). https://doi.org/10.1038/s41467-026-69960-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69960-6