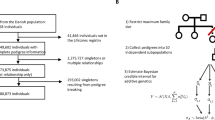
Abstract
Heritability estimates are essential for understanding genetic and environmental contributions to disease, yet large-scale studies remain scarce. In this study, we leverage the Danish national health registers, including medical records for more than 10 million individuals, to estimate heritability for more than 1000 health outcomes. We estimate heritability using both twins and siblings born in Denmark between 1955-2021, providing insight into the influence of shared sibling environment with estimates that show strong concordance with published twin studies and meta-analyses. We consider the impact of left-truncation by conducting analyses in both the full cohort and in individuals born after 1977. In a nested genotype case-cohort sample, we contrasted twin- and sibling-based heritabilities for psychiatric and neurological disorders with single-nucleotide polymorphism (SNP)-heritability, revealing disorder-specific “missing heritability” gaps. Together, these results map disease heritability in a single population, providing comprehensive insights for future genetic studies and preventive strategies using population health registers.
Similar content being viewed by others
Data availability
Data are not publicly available due to Danish data protection regulations. The study used individual-level data from the Danish Civil Registration System, the Danish National Patient Register, and the iPSYCH case-cohort sample, which are protected under national legislation. Data can only be accessed through secure servers and international researchers need a collaboration with a Danish research institution.
Maps to translate diagnosis codes can be found at ref. 31 (ICD8 to ICD10) and https://phewascatalog.org/phewas/#phe12 (ICD10 to phecodes).
Code availability
Code used to map ICD10 diagnosis codes to phecodes and to generate results can be found at https://github.com/janneah/heritability, https://doi.org/10.5281/zenodo.18480233.
References
Chatterjee, N., Shi, J. & Garcia-Closas, M. Developing and evaluating polygenic risk prediction models for stratified disease prevention. Nat. Rev. Genet. 17, 392–406 (2016).
Lakhani, C. M. et al. Repurposing large health insurance claims data to estimate genetic and environmental contributions in 560 phenotypes. Nat. Genet. 51, 327–334 (2019).
Polderman, T. J. et al. Meta-analysis of the heritability of human traits based on fifty years of twin studies. Nat. Genet. 47, 702–709 (2015).
Falconer, D. S. & Mackay, T. F. C. Introduction to Quantitative Genetics, (Benjamin-Cummings Pub Co, 1996).
Boomsma, D., Busjahn, A. & Peltonen, L. Classical twin studies and beyond. Nat. Rev. Genet. 3, 872–882 (2002).
Lykken, D. T., McGue, M. & Tellegen, A. Recruitment bias in twin research: the rule of two-thirds reconsidered. Behav. Genet. 17, 343–362 (1987).
Tambs, K. et al. The Norwegian Institute of Public Health twin study of mental health: examining recruitment and attrition bias. Twin Res. Hum. Genet. 12, 158–168 (2009).
Schoeler, T. et al. Participation bias in the UK Biobank distorts genetic associations and downstream analyses. Nat. Hum. Behav. 7, 1216–1227 (2023).
Skytthe, A., Kyvik, K. O., Holm, N. V. & Christensen, K. The Danish Twin Registry. Scand. J. Public Health 39, 75–78 (2011).
Pedersen, D. A. et al. The Danish Twin Registry: an updated overview. Twin Res. Hum. Genet. 22, 499–507 (2019).
Barry, C. J. S. et al. How to estimate heritability: a guide for genetic epidemiologists. Int. J. Epidemiol. 52, 624–632 (2023).
Manolio, T. A. et al. Finding the missing heritability of complex diseases. Nature 461, 747–753 (2009).
McGuire, D. et al. Dissecting heritability, environmental risk, and air pollution causal effects using > 50 million individuals in MarketScan. Nat. Commun. 15, 5357 (2024).
Polubriaginof, F. C. G. et al. Disease heritability inferred from familial relationships reported in medical records. Cell 173, 1692–1704 e11 (2018).
Wang, K., Gaitsch, H., Poon, H., Cox, N. J. & Rzhetsky, A. Classification of common human diseases derived from shared genetic and environmental determinants. Nat. Genet. 49, 1319–1325 (2017).
Westergaard, D. et al. Uncovering the heritable components of multimorbidities and disease trajectories: a nationwide cohort study. Nat. Commun. 15, 7457 (2024).
Cain, K. C. et al. Bias Due to left truncation and left censoring in longitudinal studies of developmental and disease processes. Am. J. Epidemiol. 173, 1078–1084 (2011).
Sondhi, A. Estimating survival parameters under conditionally independent left truncation. Pharm. Stat. 21, 895–906 (2022).
Hazelbag, C. M., Klungel, O. H., van Staa, T. P., de Boer, A. & Groenwold, R. H. H. Left truncation results in substantial bias of the relation between time-dependent exposures and adverse events. Ann. Epidemiol. 25, 590–596 (2015).
Tenesa, A. & Haley, C. S. The heritability of human disease: estimation, uses and abuses. Nat. Rev. Genet. 14, 139–149 (2013).
Owen, M. J. & Williams, N. M. Explaining the missing heritability of psychiatric disorders. World Psychiatry 20, 294–295 (2021).
International League Against Epilepsy Consortium on Complex, E. GWAS meta-analysis of over 29,000 people with epilepsy identifies 26 risk loci and subtype-specific genetic architecture. Nat. Genet. 55, 1471–1482 (2023).
Berkovic, S. F. & Scheffer, I. E. Febrile seizures: genetics and relationship to other epilepsy syndromes. Curr. Opin. Neurol. 11, 129–134 (1998).
Epi, C. Exome sequencing of 20,979 individuals with epilepsy reveals shared and distinct ultra-rare genetic risk across disorder subtypes. Nat. Neurosci. 27, 1864–1879 (2024).
Athanasiadis, G. et al. A comprehensive map of genetic relationships among diagnostic categories based on 48.6 million relative pairs from the Danish genealogy. Proc. Natl. Acad. Sci. USA. 119, e2118688119. (2022).
Pedersen, C. B. The Danish Civil Registration System. Scand. J. Public Health 39, 22–25 (2011).
Lynge, E., Sandegaard, J. L. & Rebolj, M. The Danish National Patient Register. Scand. J. Public Health 39, 30–33 (2011).
Plana-Ripoll, O. et al. Analysis of mortality metrics associated with a comprehensive range of disorders in Denmark, 2000 to 2018: A population-based cohort study. PloS Med. 19, e1004023 (2022).
Denny, J. C. et al. Systematic comparison of phenome-wide association study of electronic medical record data and genome-wide association study data. Nat. Biotechnol. 31, 1102–1110 (2013).
Bastarache, L. Using Phecodes for Research with the Electronic Health Record: From PheWAS to PheRS. Annu. Rev. Biomed. Data Sci. 4, 1–19 (2021).
Pedersen, M. K. et al. A unidirectional mapping of ICD-8 to ICD-10 codes, for harmonized longitudinal analysis of diseases. Eur. J. Epidemiol. 38, 1043–1052 (2023).
Christensen, J., Vestergaard, M., Olsen, J. & Sidenius, P. Validation of epilepsy diagnoses in the Danish National Hospital Register. Epilepsy Res. 75, 162–170 (2007).
Pedersen, C. B. et al. A Comprehensive nationwide study of the incidence rate and lifetime risk for treated mental disorders. JAMA Psychiatry 71, 573–581 (2014).
Weinberg, W. Beiträge zur Physiologie und Pathologie der Mehrlingsgeburten Beim Menschen. Pflüger., Arch. 88, 638 (1901).
Davison, A. C. & Hinkley, D. V. Bootstrap Methods and Their Applications (Cambridge University Press, Cambridge, 1997).
Canty, A. & Ripley, B. D. boot: Bootstrap R (S-Plus) Functions. (R package version 1.3-31, 2024).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. 57, 289–300 (1995).
Pedersen, C. B. et al. The iPSYCH2012 case-cohort sample: new directions for unravelling genetic and environmental architectures of severe mental disorders. Mol. Psychiatry 23, 6–14 (2018).
Bybjerg-Grauholm, J. et al. The iPSYCH2015 Case-Cohort sample: updated directions for unravelling genetic and environmental architectures of severe mental disorders. Preprint at: https://doi.org/10.1101/2020.11.30.20237768 (2020).
Lam, M. et al. RICOPILI: rapid imputation for COnsortias PIpeLIne. Bioinformatics 36, 930–933 (2020).
McCarthy, S. et al. A reference panel of 64,976 haplotypes for genotype imputation. Nat. Genet. 48, 1279–1283 (2016).
Manichaikul, A. et al. Robust relationship inference in genome-wide association studies. Bioinformatics 26, 2867–2873 (2010).
Prive, F., Luu, K., Blum, M. G. B., McGrath, J. J. & Vilhjalmsson, B. J. Efficient toolkit implementing best practices for principal component analysis of population genetic data. Bioinformatics 36, 4449–4457 (2020).
Prive, F., Arbel, J. & Vilhjalmsson, B. J. LDpred2: better, faster, stronger. Bioinformatics 36, 5424–5431 (2021).
Privé, F., Albiñana, C., Arbel, J., Pasaniuc, B. & Vilhjálmsson, B. J. Inferring disease architecture and predictive ability with LDpred2-auto. Am. J. Hum. Genet. 110, 2042–2055 (2023).
Acknowledgements
J.A. was supported by The Central Denmark Region, the Novo Nordisk Foundation (NNF16OC0019126 and NNF22OC0075033), and a Lundbeck Foundation Fellowship (R335-2019-2339). J.C. and B.B.T. were supported by Novo Nordisk Foundation (NNF16OC0019126 and NNF22OC0075033), the Lundbeck Foundation (R402-2022−1485), the Central Denmark Region, and the Danish Epilepsy Association. J.W.D. was supported by The Independent Research Fund Denmark (4253-00007B, 3166-00134B, 4308-00142B). B.J.W. was supported by Independent Research Fund (2034-00241B), Lundbeck Fellow Grant (R335-2019-2339), and Danish National Research Foundation (P4).
Author information
Authors and Affiliations
Contributions
J.A. performed the data processing and analyses and wrote the manuscript. B.B.T. reviewed the code. J.W.D., J.C., and B.J.V. supervised the study. All authors contributed with revisions to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
J.C. has received honoraria from serving on the scientific advisory board of UCB Nordic and Eisai AB, received honoraria for giving lectures from UCB Nordic and Eisai AB, and received funding for a trip funded by UCB Nordic. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Wouter van Rheenen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Auning, J., Trabjerg, B.B., Dreier, J.W. et al. Mapping the heritability of disease: a nationwide study. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69991-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69991-z