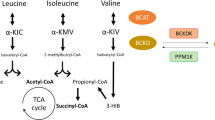
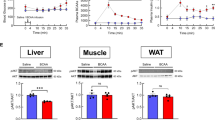
Abstract
Dysmetabolism of branched-chain amino acid (BCAA) causes insulin resistance in type 2 diabetes, yet its effect on insulin-producing β-cells remains unclear. Here, we demonstrate that branched-chain α-ketoacids (BCKAs), derived from BCAAs, inhibited glucose-stimulated insulin secretion (GSIS) and glucose fluxes across human islets, mouse islets, and mouse β-cells. In diabetic humans, elevated circulating BCKAs negatively correlated with insulin secretory ability. Treatment with BCKA or its impaired catabolism suppressed GSIS in human islets and male mice, while reducing BCKA improved glucose tolerance and GSIS in male and female diabetic mice. Mechanistically, BCKA redirected glucose metabolism from the TCA cycle to the “β-cell disallowed” lactate dehydrogenase A (LDHA)-lactate axis. BCKA directly bound to LDHA, promoting its dimerization and enhancing enzymatic activity. β-cell-specific LDHA ablation restored GSIS and glucose tolerance in BCKA-fed male mice. Our findings demonstrate that BCKA disrupts insulin secretion through LDHA reactivation, linking aberrant BCAA metabolism to β-cell dysfunction in diabetes.
Similar content being viewed by others
Data availability
All experimental data generated in this study are provided in the Supplementary Information/Source Data file. A PDF file with uncropped scans of western blots and source data used to generate the graphs in the paper are provided. The processed transcriptome and proteome datasets for BCAA catabolic enzyme expression were downloaded from Kolic et al. (PMID: 38959864) and are publicly available at www.humanislets.com and were reanalyzed. Additional information can be obtained from the lead contact upon reasonable request. Source data are provided in this paper.
References
Straub, S. G. & Sharp, G. W. Glucose-stimulated signaling pathways in biphasic insulin secretion. Diab. Metab. Res. Rev. 18, 451–463 (2002).
Campbell, J. E. & Newgard, C. B. Mechanisms controlling pancreatic islet cell function in insulin secretion. Nat. Rev. Mol. Cell Biol. 22, 142–158 (2021).
Wang, B. et al. The adaptor protein APPL2 controls glucose-stimulated insulin secretion via F-actin remodeling in pancreatic beta-cells. Proc. Natl. Acad. Sci. USA 117, 28307–28315 (2020).
Hasan, N. M. et al. Impaired anaplerosis and insulin secretion in insulinoma cells caused by small interfering RNA-mediated suppression of pyruvate carboxylase. J. Biol. Chem. 283, 28048–28059 (2008).
Haythorne, E. et al. Altered glycolysis triggers impaired mitochondrial metabolism and mTORC1 activation in diabetic beta-cells. Nat. Commun. 13, 6754 (2022).
Lu, D. et al. 13 C NMR isotopomer analysis reveals a connection between pyruvate cycling and glucose-stimulated insulin secretion (GSIS). Proc. Natl. Acad. Sci. USA 99, 2708–2713 (2002).
Zhang, G. F. et al. Reductive TCA cycle metabolism fuels glutamine- and glucose-stimulated insulin secretion. Cell Metab. 33, 804–817 (2021).
Ristow, M., Vorgerd, M., Mohlig, M., Schatz, H. & Pfeiffer, A. Deficiency of phosphofructo-1-kinase/muscle subtype in humans impairs insulin secretion and causes insulin resistance. J. Clin. Invest. 100, 2833–2841 (1997).
Ainscow, E. K., Zhao, C. & Rutter, G. A. Acute overexpression of lactate dehydrogenase-A perturbs beta-cell mitochondrial metabolism and insulin secretion. Diabetes 49, 1149–1155 (2000).
Zhou, M. et al. Targeting BCAA catabolism to treat obesity-associated insulin resistance. Diabetes 68, 1730–1746 (2019).
White, P. J. et al. The BCKDH kinase and phosphatase integrate BCAA and lipid metabolism via regulation of ATP-citrate lyase. Cell Metab. 27, 1281–1293.e1287 (2018).
Mansoori, S., Ho, M. Y., Ng, K. K. & Cheng, K. K. Branched-chain amino acid metabolism: Pathophysiological mechanism and therapeutic intervention in metabolic diseases. Obes. Rev. 26, e13856 (2025).
Sivanand, S. & Vander Heiden, M. G. Emerging roles for branched-chain amino acid metabolism in cancer. Cancer Cell 37, 147–156 (2020).
Lian, K. et al. Impaired adiponectin signaling contributes to disturbed catabolism of branched-chain amino acids in diabetic mice. Diabetes 64, 49–59 (2015).
Liu, S. et al. Elevated branched-chain α-keto acids exacerbate macrophage oxidative stress and chronic inflammatory damage in type 2 diabetes mellitus. Free Radic. Biol. Med. 175, 141–154 (2021).
Yue, S. J. et al. Berberine alleviates insulin resistance by reducing peripheral branched-chain amino acids. Am. J. Physiol. Endocrinol. Metab. 316, E73–e85 (2019).
Vanweert, F. et al. A randomized placebo-controlled clinical trial for pharmacological activation of BCAA catabolism in patients with type 2 diabetes. Nat. Commun. 13, 3508 (2022).
Neinast, M. D. et al. Quantitative analysis of the whole-body metabolic fate of branched-chain amino acids. Cell Metab. 29, 417–429 (2019).
Panten, U., Christians, J., von Kriegstein, E., Poser, W. & Hasselblatt, A. Studies on the mechanism of L-leucine-and alpha-ketoisocaproic acid-induced insulin release from perifused isolated pancreatic islets. Diabetologia 10, 149–154 (1974).
Branstrom, R., Efendic, S., Berggren, P. O. & Larsson, O. Direct inhibition of the pancreatic beta-cell ATP-regulated potassium channel by alpha-ketoisocaproate. J. Biol. Chem. 273, 14113–14118 (1998).
Yang, J. et al. Leucine regulation of glucokinase and ATP synthase sensitizes glucose-induced insulin secretion in pancreatic β-cells. Diabetes 55, 193–201 (2006).
Lotta, L. A. et al. Genetic predisposition to an impaired metabolism of the branched-chain amino acids and risk of type 2 diabetes: a mendelian randomisation analysis. PLoS Med. 13, e1002179 (2016).
Sekine, N. et al. Low lactate dehydrogenase and high mitochondrial glycerol phosphate dehydrogenase in pancreatic beta-cells Potential role nutrient Sens. J. Biol. Chem. 269, 4895–4902 (1994).
Kolic, J. et al. Proteomic predictors of individualized nutrient-specific insulin secretion in health and disease. Cell Metab. 36, 1619–1633.e1615 (2024).
Taneera, J. et al. A systems genetics approach identifies genes and pathways for type 2 diabetes in human islets. Cell Metab. 16, 122–134 (2012).
Vanweert, F., Schrauwen, P. & Phielix, E. Role of branched-chain amino acid metabolism in the pathogenesis of obesity and type 2 diabetes-related metabolic disturbances BCAA metabolism in type 2 diabetes. Nutr. Diab. 12, 35 (2022).
Lynch, C. J. & Adams, S. H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 10, 723–736 (2014).
Ratheiser, K. et al. Quantitative and qualitative differences in basal and glucose- and arginine-stimulated insulin secretion in healthy subjects and different stages of NIDDM. Acta Diabetol. Lat. 27, 197–213 (1990).
Shen, Y. et al. Association between 1,5-anhydroglucitol and acute C peptide response to arginine among patients with type 2 diabetes. J. Diab. Res. 2020, 4243053 (2020).
Tso, S. C. et al. Benzothiophene carboxylate derivatives as novel allosteric inhibitors of branched-chain alpha-ketoacid dehydrogenase kinase. J. Biol. Chem. 289, 20583–20593 (2014).
Zhou, Y., Jetton, T. L., Goshorn, S., Lynch, C. J. & She, P. Transamination is required for alpha-ketoisocaproate but not leucine to stimulate insulin secretion. J. Biol. Chem. 285, 33718–33726 (2010).
Gheibi, S. et al. Reduced expression level of protein phosphatase PPM1E serves to maintain insulin secretion in type 2. Diab. Diab. 72, 455–466 (2023).
Li, X. et al. The MDM2-p53-pyruvate carboxylase signalling axis couples mitochondrial metabolism to glucose-stimulated insulin secretion in pancreatic β-cells. Nat. Commun. 7, 11740 (2016).
Yang, Y. et al. The mitochondrial enzyme pyruvate carboxylase restricts pancreatic β-cell senescence by blocking p53 activation. Proc. Natl. Acad. Sci. USA 121, e2401218121 (2024).
Lee, H. et al. Beta cell dedifferentiation induced by IRE1α deletion prevents type 1 diabetes. Cell Metab. 31, 822–836 (2020).
Lee, H. et al. Stress-induced β cell early senescence confers protection against type 1 diabetes. Cell Metab. 35, 2200–2215 (2023).
Pullen, T. J. et al. Overexpression of monocarboxylate transporter-1 (SLC16A1) in mouse pancreatic β-cells leads to relative hyperinsulinism during exercise. Diabetes 61, 1719–1725 (2012).
Sanchez, P. K. M. et al. LDHA is enriched in human islet alpha cells and upregulated in type 2 diabetes. Biochem. Biophys. Res. Commun. 568, 158–166 (2021).
Schuit, F. et al. β-cell-specific gene repression: a mechanism to protect against inappropriate or maladjusted insulin secretion? Diabetes 61, 969–975 (2012).
Rutter, G. A., Georgiadou, E., Martinez-Sanchez, A. & Pullen, T. J. Metabolic and functional specialisations of the pancreatic beta cell: gene disallowance, mitochondrial metabolism and intercellular connectivity. Diabetologia 63, 1990–1998 (2020).
Montemurro, C. et al. IAPP toxicity activates HIF1α/PFKFB3 signaling delaying β-cell loss at the expense of β-cell function. Nat. Commun. 10, 2679 (2019).
Cornell, D. et al. Pseudoislet aggregation of pancreatic β-cells improves glucose-stimulated insulin secretion by altering glucose metabolism and increasing ATP production. Cells 11, https://doi.org/10.3390/cells11152330 (2022).
Hals, I. et al. Time dynamics of elevated glucose and beta-hydroxybutyrate on beta cell mitochondrial metabolism. Islets 17, 2503515 (2025).
Sentjens, K., Pillai, R. & Joseph, J. W. The effects of free fatty acid-free bovine serum albumin and palmitate on pancreatic β-cell function. Islets 17, 2479911 (2025).
Xiong, X. et al. Sirtuin 6 regulates glucose-stimulated insulin secretion in mouse pancreatic beta cells. Diabetologia 59, 151–160 (2016).
Mookerjee, S. A., Goncalves, R. L. S., Gerencser, A. A., Nicholls, D. G. & Brand, M. D. The contributions of respiration and glycolysis to extracellular acid production. Biochim. Biophys. Acta 1847, 171–181 (2015).
Tsonkova, V. G. et al. The EndoC-βH1 cell line is a valid model of human beta cells and applicable for screenings to identify novel drug target candidates. Mol. Metab. 8, 144–157 (2018).
Davis, J. C. et al. Glucose response by stem cell-derived β cells in vitro is inhibited by a bottleneck in glycolysis. Cell Rep. 31, 107623 (2020).
Rabaglia, M. E. et al. Alpha-Ketoisocaproate-induced hypersecretion of insulin by islets from diabetes-susceptible mice. Am. J. Physiol. Endocrinol. Metab. 289, E218–E224 (2005).
Ishihara, H., Maechler, P., Gjinovci, A., Herrera, P. L. & Wollheim, C. B. Islet beta-cell secretion determines glucagon release from neighbouring alpha-cells. Nat. Cell Biol. 5, 330–335 (2003).
Stabenow, L.K. et al. Oxidative glucose metabolism promotes senescence in vascular endothelial cells. Cells 11, 2213 (2022).
Li, Y. et al. Acetate supplementation restores chromatin accessibility and promotes tumor cell differentiation under hypoxia. Cell Death Dis. 11, 102 (2020).
Srinivasan, M. et al. ß-Cell-specific pyruvate dehydrogenase deficiency impairs glucose-stimulated insulin secretion. Am. J. Physiol. Endocrinol. Metab. 299, E910–E917 (2010).
Wang, X. et al. Conditional knockout of pyruvate dehydrogenase in mouse pancreatic β‑cells causes morphological and functional changes. Mol. Med. Rep. 21, 1717–1726 (2020).
Zhao, C. & Rutter, G. A. Overexpression of lactate dehydrogenase A attenuates glucose-induced insulin secretion in stable MIN-6 beta-cell lines. FEBS Lett. 430, 213–216 (1998).
Hicks, K. G. et al. Protein-metabolite interactomics of carbohydrate metabolism reveal regulation of lactate dehydrogenase. Science 379, 996–1003 (2023).
Yang, Y. et al. αKG-driven RNA polymerase II transcription of cyclin D1 licenses malic enzyme 2 to promote cell-cycle progression. Cell Rep. 42, 112770 (2023).
Jafary, F., Ganjalikhany, M. R., Moradi, A., Hemati, M. & Jafari, S. Novel peptide inhibitors for lactate dehydrogenase a (LDHA): a survey to inhibit LDHA activity via disruption of protein-protein interaction. Sci. Rep. 9, 4686 (2019).
Yamamoto, S. & Storey, K. B. Dissociation-association of lactate dehydrogenase isozymes: influences on the formation of tetramers versus dimers of M4-LDH and H4-LDH. Int. J. Biochem. 20, 1261–1265 (1988).
Liu, Y. et al. Nuclear lactate dehydrogenase A senses ROS to produce α-hydroxybutyrate for HPV-induced cervical tumor growth. Nat. Commun. 9, 4429 (2018).
Girg, R., Rudolph, R. & Jaenicke, R. The dimeric intermediate on the pathway of reconstitution of lactate dehydrogenase is enzymatically active. FEBS Lett. 163, 132–135 (1983).
Girg, R., Jaenicke, R. & Rudolph, R. Dimers of porcine skeletal muscle lactate dehydrogenase produced by limited proteolysis during reassociation are enzymatically active in the presence of stabilizing salt. Biochem. Int. 7, 433–441 (1983).
Newgard, C. B. et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 9, 311–326 (2009).
Jang, C. et al. A branched-chain amino acid metabolite drives vascular fatty acid transport and causes insulin resistance. Nat. Med. 22, 421–426 (2016).
Neinast, M., Murashige, D. & Arany, Z. Branched chain amino acids. Annu. Rev. Physiol. 81, 139–164 (2019).
Prentki, M., Matschinsky, F. M. & Madiraju, S. R. Metabolic signaling in fuel-induced insulin secretion. Cell Metab. 18, 162–185 (2013).
Zhao, C., Wilson, M. C., Schuit, F., Halestrap, A. P. & Rutter, G. A. Expression and distribution of lactate/monocarboxylate transporter isoforms in pancreatic islets and the exocrine pancreas. Diabetes 50, 361–366 (2001).
Xu, J., Han, J., Long, Y. S., Epstein, P. N. & Liu, Y. Q. The role of pyruvate carboxylase in insulin secretion and proliferation in rat pancreatic beta cells. Diabetologia 51, 2022–2030 (2008).
Li, T. et al. Defective branched-chain amino acid catabolism disrupts glucose metabolism and sensitizes the heart to ischemia-reperfusion injury. Cell Metab. 25, 374–385 (2017).
Haythorne, E. et al. Diabetes causes marked inhibition of mitochondrial metabolism in pancreatic β-cells. Nat. Commun. 10, 2474 (2019).
Aguayo-Mazzucato, C. et al. Acceleration of beta cell aging determines diabetes and senolysis improves disease outcomes. Cell Metab. 30, 129–142 (2019).
Chen, Y. et al. LDHA-mediated metabolic reprogramming promoted cardiomyocyte proliferation by alleviating ROS and inducing M2 macrophage polarization. Redox Biol. 56, 102446 (2022).
Heemskerk, M. M., van Harmelen, V. J., van Dijk, K. W. & van Klinken, J. B. Reanalysis of mGWAS results and in vitro validation show that lactate dehydrogenase interacts with branched-chain amino acid metabolism. Eur. J. Hum. Genet. 24, 142–145 (2016).
Yoneshiro, T. et al. BCAA catabolism in brown fat controls energy homeostasis through SLC25A44. Nature 572, 614–619 (2019).
Walejko, J. M. et al. Branched-chain α-ketoacids are preferentially reaminated and activate protein synthesis in the heart. Nat. Commun. 12, 1680 (2021).
Silva, L. S. et al. Branched-chain ketoacids secreted by glioblastoma cells via MCT1 modulate macrophage phenotype. EMBO Rep. 18, 2172–2185 (2017).
Chen, L. et al. MCT1-mediated Lactate shuttle to mitochondria governs macrophage polarization and modulates glucose homeostasis by affecting β cells. Adv. Sci. 12, e14760 (2025).
Malmgren, S. et al. Tight coupling between glucose and mitochondrial metabolism in clonal beta-cells is required for robust insulin secretion. J. Biol. Chem. 284, 32395–32404 (2009).
Marselli, L. et al. Gene expression profiles of Beta-cell enriched tissue obtained by laser capture microdissection from subjects with type 2 diabetes. PLoS ONE 5, e11499 (2010).
Nishi, K. et al. Branched-chain keto acids inhibit mitochondrial pyruvate carrier and suppress gluconeogenesis in hepatocytes. Cell Rep. 42, 112641 (2023).
Xiao, W. et al. Branched-chain α-ketoacids aerobically activate HIF1α signalling in vascular cells. Nat. Metab. 6, 2138–2156 (2024).
Wang, T. J. et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 17, 448–453 (2011).
Biswas, D. et al. Branched-chain ketoacid overload inhibits insulin action in the muscle. J. Biol. Chem. 295, 15597–15621 (2020).
Blair, M. C. et al. Branched-chain amino acid catabolism in muscle affects systemic BCAA levels but not insulin resistance. Nat. Metab. 5, 589–606 (2023).
Karusheva, Y. et al. Branched-Chain Amino Acids Associate Negatively With Postprandial Insulin Secretion in Recent-Onset Diabetes. J. Endocr. Soc. 5, bvab067 (2021).
Oresic, M. et al. Dysregulation of lipid and amino acid metabolism precedes islet autoimmunity in children who later progress to type 1 diabetes. J. Exp. Med. 205, 2975–2984 (2008).
Funchal, C., Gottfried, C., De Almeida, L. M., Wajner, M. & Pessoa-Pureur, R. Evidence that the branched-chain alpha-keto acids accumulating in maple syrup urine disease induce morphological alterations and death in cultured astrocytes from rat cerebral cortex. Glia 48, 230–240 (2004).
Guo, X. et al. BCKA down-regulates mTORC2-Akt signal and enhances apoptosis susceptibility in cardiomyocytes. Biochem. Biophys. Res. Commun. 480, 106–113 (2016).
Lu, G. et al. A novel mitochondrial matrix serine/threonine protein phosphatase regulates the mitochondria permeability transition pore and is essential for cellular survival and development. Genes Dev. 21, 784–796 (2007).
D’Addio, F. et al. The IGFBP3/TMEM219 pathway regulates beta cell homeostasis. Nat. Commun. 13, 684 (2022).
D’Addio, F. et al. TMEM219 regulates the transcription factor expression and proliferation of beta cells. Front. Endocrinol. 15, 1306127 (2024).
Karusheva, Y. et al. Short-term dietary reduction of branched-chain amino acids reduces meal-induced insulin secretion and modifies microbiome composition in type 2 diabetes: a randomized controlled crossover trial. Am. J. Clin. Nutr. 110, 1098–1107 (2019).
American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2019. Diabetes Care 42, S13-s28 (2019).
Supriya, R. et al. Adipokine profiling in adult women with central obesity and hypertension. Front. Physiol. 9, 294 (2018).
Cheong, L. Y. et al. Fibroblastic reticular cells in lymph node potentiate white adipose tissue beiging through neuro-immune crosstalk in male mice. Nat. Commun. 14, 1213 (2023).
Lu, G. et al. Functional characterization of a mitochondrial Ser/Thr protein phosphatase in cell death regulation. Methods Enzymol. 457, 255–273 (2009).
Olson, K. C., Chen, G. & Lynch, C. J. Quantification of branched-chain keto acids in tissue by ultra fast liquid chromatography–mass spectrometry. Anal. Biochem. 439, 116–122 (2013).
Walvekar, A., Rashida, Z., Maddali, H. & Laxman, S. A versatile LC-MS/MS approach for comprehensive, quantitative analysis of central metabolic pathways [version 1; referees: 2 approved]. Wellcome Open Res. 3, 122–122 (2018).
Yan, M. et al. Metabolomics profiling of metformin-mediated metabolic reprogramming bypassing AMPKα. Metabolism 91, 18–29 (2019).
Lomenick, B. et al. Target identification using drug affinity responsive target stability (DARTS). Proc. Natl. Acad. Sci. USA 106, 21984–21989 (2009).
Acknowledgements
We would like to thank Dr. Xi Chen for her kind help in collecting serum and tissue samples in BCKA-fed mice. We thank the Department of University Life Science at The Hong Kong Polytechnic University for providing instruments and instructions. This project was supported by National Natural Science Foundation of China (81970675 and 92357305), Hong Kong Research Grant Council (RGC) Collaborative Research Fund (C5044-23G) and General Research Fund (15101221), Shenzhen Municipal Science and Technology Innovation Commission Basic Research General Program: 20210324130202006 and PolyU internal funding (P0040979, P0036848 and P0059102) to Dr. Kenneth King-yip Cheng, AoE/M-707/18/RGC Area of Excellence to Dr. Aimin Xu and Department of Defense (W81XWH2010592) to Dr. Chen Cao.
Author information
Authors and Affiliations
Contributions
H.L. and M.Y.H. conducted most of the experiments and drafted and revised the manuscript. Y.Y. and B.W. collected human samples and conducted data analysis. S.M. and A.M.M. performed some immunohistochemical experiments. W.W., P.S., and H.P. helped with LC-MS/MS measurement for BCAA/BCKA and TCA cycle intermediates. A.X. provided LDHA floxed mice and revised the manuscript. S.Y. and J.C. supervised and conducted BCKA-LDHA docking site analysis. C.G. provided PPM1K global KO mice, provided supervision on the animal study and reviewed the manuscript. P.M.S. collected human serum samples and clinical data in Hong Kong. X.L. provided pancreatic slides, islets, serum and clinical data from T2D humans. K.K.C. acquired the funding, provided resources, supervised the project and wrote the manuscript. K.K.C. and X.L. jointly supervised this work. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Gang Bai, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lin, H., Ho, M.Y.M., Wang, B. et al. Branched-chain α-keto acids impair glucose-stimulated insulin secretion in pancreatic β-cells under diabetes by reactivating the LDHA-lactate axis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70004-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70004-2