Abstract
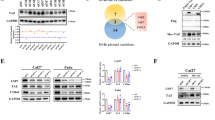
Human FAN1 is a structure-specific endonuclease implicated in the repair of DNA interstrand crosslinks (ICLs) and the excision of extrahelical CAG repeats–whose pathological expansion underlies Huntington’s disease (HD), a progressive and currently incurable neurodegenerative disorder. However, mechanisms of post-translational regulation of FAN1 are still largely unknown. Here, we identify the ubiquitin-specific protease 7 (USP7) as an interactor of FAN1. USP7 stabilizes FAN1 protein levels in a deubiquitination-dependent manner, preventing FAN1 from proteasomal degradation. Consequently, we demonstrate that USP7 depletion leads to reduced chromatin association of FAN1 and increased cellular hypersensitivity following ICL damage. Moreover, loss of USP7 accelerates CAG repeat expansion in an RPE-1 cell model stably expressing mutant huntingtin (mHTT) exon 1 containing 129 CAG repeats (RPE-1HTT-CAG129). Collectively, our findings uncover a link between USP7 and FAN1 in mechanisms that preserve genome stability and influence repeat instability.
Similar content being viewed by others
Data availability
All data are available within the Article, Supplementary Information, or Source data file. Raw data used to generate all graphs and derived statistics are provided in the Source data file. Original, uncropped blots can also be found in the Source data file. All original microscopy images will be made available upon request. The mass spectrometry data generated for this study have been deposited in the PRIDE database via PXD identifier PXD06510175. Source data are provided with this paper.
References
Kratz, K. et al. Deficiency of FANCD2-Associated Nuclease KIAA1018/FAN1 Sensitizes Cells to Interstrand Crosslinking Agents. Cell 142, 77–88 (2010).
MacKay, C. et al. Identification of KIAA1018/FAN1, a DNA Repair Nuclease Recruited to DNA Damage by Monoubiquitinated FANCD2. Cell 142, 65–76 (2010).
Smogorzewska, A. et al. A Genetic Screen Identifies FAN1, a Fanconi Anemia-Associated Nuclease Necessary for DNA Interstrand Crosslink Repair. Mol. Cell 39, 36–47 (2010).
Liu, T., Ghosal, G., Yuan, J., Chen, J. & Huang, J. FAN1 acts with FANCI-FANCD2 to promote DNA interstrand cross-link repair. Science 329, 693–696 (2010).
Zhao, Q., Xue, X., Longerich, S., Sung, P. & Xiong, Y. Structural insights into 5’-flap DNA unwinding and incision by the human FAN1 dimer. Nat. Commun. 5, 5726 (2014).
Pizzolato, J., Mukherjee, S., Schärer, O. D. & Jiricny, J. FANCD2-associated Nuclease 1, but Not Exonuclease 1 or Flap Endonuclease 1, Is Able to Unhook DNA Interstrand Cross-links in Vitro. J. Biol. Chem. 290, 22602–22611 (2015).
Wang, R. et al. Mechanism of DNA interstrand cross-link processing by repair nuclease FAN1. Science 346, 1127–1130 (2014).
Cannavo, E., Gerrits, B., Marra, G., Schlapbach, R. & Jiricny, J. Characterization of the interactome of the human MutL homologues MLH1, PMS1, and PMS2. J. Biol. Chem. 282, 2976–2986 (2007).
Porro, A. et al. FAN1-MLH1 interaction affects repair of DNA interstrand cross-links and slipped-CAG/CTG repeats. Sci. Adv.7, eabf7906 (2021).
Goold, R. et al. FAN1 controls mismatch repair complex assembly via MLH1 retention to stabilize CAG repeat expansion in Huntington’s disease. Cell Rep. 36, 109649 (2021).
Deshmukh, A. L. et al. FAN1, a DNA Repair Nuclease, as a Modifier of Repeat Expansion Disorders. J. Huntington’s Dis. 10, 95–122 (2021).
Lee, J. M. et al. CAG Repeat Not Polyglutamine Length Determines Timing of Huntington’s Disease Onset. Cell 178, 887–900.e14 (2019).
Genetic Modifiers of Huntington’s Disease (GeM-HD) Consortium Identification of Genetic Factors that Modify Clinical Onset of Huntington’s Disease. Cell 162, 516–526 (2015).
McAllister, B. et al. Exome sequencing of individuals with Huntington’s disease implicates FAN1 nuclease activity in slowing CAG expansion and disease onset. Nat. Neurosci. 25, 446–457 (2022).
Phadte, A. S. et al. FAN1 removes triplet repeat extrusions via a PCNA- and RFC-dependent mechanism. Proc. Natl. Acad. Sci. USA 120, e2302103120 (2023).
Mätlik, K. et al. Cell-type-specific CAG repeat expansions and toxicity of mutant Huntingtin in human striatum and cerebellum. Nat. Genet 56, 383–394 (2024).
Goold, R. et al. FAN1 modifies Huntington’s disease progression by stabilizing the expanded HTT CAG repeat. Hum. Mol. Genet 28, 650–661 (2019).
Jackson, S. P. & Durocher, D. Regulation of DNA Damage Responses by Ubiquitin and SUMO. Mol. Cell 49, 795–807 (2013).
Lai, F. et al. Human KIAA1018/FAN1 nuclease is a new mitotic substrate of APC/CCdh1. Chin. J. Cancer 31, 440–448 (2012).
Mevissen, T. E. T. & Komander, D. Mechanisms of Deubiquitinase Specificity and Regulation. Annu Rev. Biochem 86, 159–192 (2017).
Dewson, G., Eichhorn, P. J. A. & Komander, D. Deubiquitinases in cancer. Nat. Rev. Cancer 23, 842–862 (2023).
Ghodgaonkar, M. M. et al. Phenotypic characterization of missense polymerase-δ mutations using an inducible protein-replacement system. Nat. Commun. 5, 4990 (2014).
Porro, A. et al. FAN1 interaction with ubiquitylated PCNA alleviates replication stress and preserves genomic integrity independently of BRCA2. Nat. Commun. 8, 1073 (2017).
Velimezi, G. et al. Map of synthetic rescue interactions for the Fanconi anemia DNA repair pathway identifies USP48. Nat. Commun. 9, 2280 (2018).
Lu, Q., Zhang, F.-L., Lu, D.-Y., Shao, Z.-M. & Li, D.-Q. USP9X stabilizes BRCA1 and confers resistance to DNA-damaging agents in human cancer cells. Cancer Med 8, 6730–6740 (2019).
O’Dea, R. & Santocanale, C. Non-canonical regulation of homologous recombination DNA repair by the USP9X deubiquitylase. J. Cell Sci. 133, jcs233437 (2020).
Zhao, Y. et al. Noncanonical regulation of alkylation damage resistance by the OTUD4 deubiquitinase. EMBO J. 34, 1687–1703 (2015).
Yu, M. et al. USP11 Is a Negative Regulator to γH2AX Ubiquitylation by RNF8/RNF168. J. Biol. Chem. 291, 959–967 (2016).
Perry, M. et al. USP11 mediates repair of DNA-protein cross-links by deubiquitinating SPRTN metalloprotease. J. Biol. Chem. 296, 100396 (2021).
Hyer, M. L. et al. A small-molecule inhibitor of the ubiquitin activating enzyme for cancer treatment. Nat. Med. 24, 186–193 (2018).
An, L. et al. Dual-utility NLS drives RNF169-dependent DNA damage responses. Proc. Natl. Acad. Sci. 114, E2872–E2881 (2017).
Saridakis, V. et al. Structure of the p53 Binding Domain of HAUSP/USP7 Bound to Epstein-Barr Nuclear Antigen 1. Mol. Cell 18, 25–36 (2005).
Cheng, J. et al. Molecular mechanism for USP7-mediated DNMT1 stabilization by acetylation. Nat. Commun. 6, 7023 (2015).
Faesen, A. C. et al. Mechanism of USP7/HAUSP Activation by Its C-Terminal Ubiquitin-like Domain and Allosteric Regulation by GMP-Synthetase. Mol. Cell 44, 147–159 (2011).
Ashton, N. W., Valles, G. J., Jaiswal, N., Bezsonova, I. & Woodgate, R. DNA Polymerase ι Interacts with Both the TRAF-like and UBL1-2 Domains of USP7. J. Mol. Biol. 433, 166733 (2021).
Miyashita, R. et al. The termination of UHRF1-dependent PAF15 ubiquitin signaling is regulated by USP7 and ATAD5. Elife 12, e79013 (2023).
Park, H.-B. & Baek, K.-H. Current and future directions of USP7 interactome in cancer study. Biochimica et. Biophysica Acta (BBA) - Rev. Cancer 1878, 188992 (2023).
Sheng, Y. et al. Molecular recognition of p53 and MDM2 by USP7/HAUSP. Nat. Struct. Mol. Biol. 13, 285–291 (2006).
Georges, A., Marcon, E., Greenblatt, J. & Frappier, L. Identification and Characterization of USP7 Targets in Cancer Cells. Sci. Rep. 8, 15833 (2018).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Cheng, J. et al. Molecular mechanism for the substrate recognition of USP7. Protein Cell 6, 849–852 (2015).
Kumar, M. et al. ELM—the Eukaryotic Linear Motif resource—2024 update. Nucleic Acids Res 52, D442–D455 (2024).
Saha, G. & Ghosh, M. K. The key vulnerabilities and therapeutic opportunities in the USP7-p53/MDM2 axis in cancer. Biochim Biophys. Acta Mol. Cell Res 1872, 119908 (2025).
Turnbull, A. P. et al. Molecular basis of USP7 inhibition by selective small-molecule inhibitors. Nature 550, 481–486 (2017).
Kategaya, L. et al. USP7 small-molecule inhibitors interfere with ubiquitin binding. Nature 550, 534–538 (2017).
Thongthip, S. et al. Fan1 deficiency results in DNA interstrand cross-link repair defects, enhanced tissue karyomegaly, and organ dysfunction. Genes Dev. 30, 645–659 (2016).
Liu, J. et al. De-ubiquitination of SAMHD1 by USP7 promotes DNA damage repair to overcome oncogenic stress and affect chemotherapy sensitivity. Oncogene 42, 1843–1856 (2023).
Zlatanou, A. et al. USP7 is essential for maintaining Rad18 stability and DNA damage tolerance. Oncogene 35, 965–976 (2016).
Valles, G. J., Bezsonova, I., Woodgate, R. & Ashton, N. W. USP7 Is a Master Regulator of Genome Stability. Front. Cell Dev. Biol. 8, 717 (2020).
Agathanggelou, A. et al. USP7 inhibition alters homologous recombination repair and targets CLL cells independently of ATM/p53 functional status. Blood 130, 156–166 (2017).
Zhu, Q., Sharma, N., He, J., Wani, G. & Wani, A. A. USP7 deubiquitinase promotes ubiquitin-dependent DNA damage signaling by stabilizing RNF168*. Cell Cycle 14, 1413–1425 (2015).
Iyer, R. R. & Pluciennik, A. DNA Mismatch Repair and its Role in Huntington’s Disease. J. Huntingt. Dis. 10, 75–94 (2021).
Wang, N. et al. Distinct mismatch-repair complex genes set neuronal CAG-repeat expansion rate to drive selective pathogenesis in HD mice. Cell 188, 1524–1544.e22 (2025).
McLean, Z. L. et al. Splice modulators target PMS1 to reduce somatic expansion of the Huntington’s disease-associated CAG repeat. Nat. Commun. 15, 3182 (2024).
Bunting, E. L. et al. Antisense oligonucleotide–mediated MSH3 suppression reduces somatic CAG repeat expansion in Huntington’s disease iPSC–derived striatal neurons. Sci. Transl. Med. 17, eadn4600 (2025).
Mouro Pinto, R. et al. In vivo CRISPR–Cas9 genome editing in mice identifies genetic modifiers of somatic CAG repeat instability in Huntington’s disease. Nat. Genet 57, 314–322 (2025).
Chaudhury, I., Stroik, D. R. & Sobeck, A. FANCD2-Controlled Chromatin Access of the Fanconi-Associated Nuclease FAN1 Is Crucial for the Recovery of Stalled Replication Forks. Mol. Cell Biol.34, 3939–3954 (2014).
Lachaud, C. et al. Ubiquitinated Fancd2 recruits Fan1 to stalled replication forks to prevent genome instability. Science351, 846–849 (2016).
Zhao, X. N. & Usdin, K. FAN1 protects against repeat expansions in a Fragile X mouse model. DNA Repair (Amst) 69, 1–5 (2018).
Uhlén, M. et al. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Tang, K. et al. USP7 deubiquitinates epigenetic reader ZMYND8 to promote breast cancer cell migration and invasion. J. Biol. Chem. 300, 107672 (2024).
Huang, B. et al. USP7 deubiquitinates KRAS and promotes non-small cell lung cancer. Cell Rep. 43, 114917 (2024).
Kim, K. H. et al. Genetic and Functional Analyses Point to FAN1 as the Source of Multiple Huntington Disease Modifier Effects. Am. J. Hum. Genet. 107, 96–110 (2020).
Loupe, J. M. et al. Promotion of somatic CAG repeat expansion by Fan1 knock-out in Huntington’s disease knock-in mice is blocked by Mlh1 knock-out. Hum. Mol. Genet 29, 3044–3053 (2020).
Pluciennik, A. et al. Deubiquitinase USP7 contributes to the pathogenicity of spinal and bulbar muscular atrophy. J. Clin. Investig. 131, e134565 (2021).
Fountain, M. D. et al. Pathogenic variants in USP7 cause a neurodevelopmental disorder with speech delays, altered behavior, and neurologic anomalies. Genet. Med. 21, 1797–1807 (2019).
Ji, J., Sundquist, K. & Sundquist, J. Cancer incidence in patients with polyglutamine diseases: a population-based study in Sweden. Lancet Oncol. 13, 642–648 (2012).
Coarelli, G. et al. Low cancer prevalence in polyglutamine expansion diseases. Neurology 88, 1114–1119 (2017).
Rycenga, H. B. & Long, D. T. The evolving role of DNA inter-strand crosslinks in chemotherapy. Curr. Opin. Pharm. 41, 20–26 (2018).
Cheng, Y.-C. et al. Increased FAN1 expression by mRNA-LNP attenuates CAG repeat expansion in Huntington patients’ iPSC-derived astrocytes. bioRxiv 2023.11.24.568451 https://doi.org/10.1101/2023.11.24.568451 (2023).
Liu, J. et al. USP7-Based Deubiquitinase-Targeting Chimeras Stabilize AMPK. J. Am. Chem. Soc. 146, 11507–11514 (2024).
Conant, D. et al. Inference of CRISPR Edits from Sanger Trace. Data. CRISPR J. 5, 123–130 (2022).
Wolski, W. E. et al. prolfqua: A Comprehensive R-Package for Proteomics Differential Expression Analysis. J. Proteome Res 22, 1092–1104 (2023).
Gatti, M., Imhof, R., Huang, Q., Baudis, M. & Altmeyer, M. The Ubiquitin Ligase TRIP12 Limits PARP1 Trapping and Constrains PARP Inhibitor Efficiency. Cell Rep. 32, 107985 (2020).
Perez-Riverol, Y. et al. The PRIDE database resources in 2022: a hub for mass spectrometry-based proteomics evidences. Nucleic Acids Res 50, D543–D552 (2022).
Acknowledgements
We thank Lorenza Penengo for helpful discussions and providing plasmids for tagged-ubiquitin expression. We thank Sarah Tabrizi and Rob Goold for providing GFP-FAN1 expression constructs. We gratefully acknowledge Mike Flower, Lucy Coupland, and the Tabrizi Lab for generously providing the CAG expansion tools and their invaluable expertise in somatic instability analyses. We additionally thank Denny Yang Tze Te and Kangning He for their assistance with the ICE analysis used to verify the editing outcome in the MSH3 knockdown line. We gratefully acknowledge the Functional Genomics Center Zurich (FGCZ) of University of Zurich and ETH Zurich, for the support on proteomics analyses. This work was supported by research grants from the Swiss National Science Foundation (31003A_176161 and 310030_208143 to A.A.S.) and the Worldwide Cancer Research (grant reference number: 23-0355 to A.P. and A.A.S.). Work in the Balmus laboratory is supported by the UK Dementia Research Institute through UK DRI Ltd, principally funded by the UK Medical Research Council as well as CHDI Foundation, the Romanian Ministry of Research, Innovation, and Digitization (grant #PNRR-III-C9-2022-I8-66, contract 760114) and the Hereditary Disease Foundation.
Author information
Authors and Affiliations
Contributions
G.C. performed most cloning, interaction studies, western blot experiments, cell cycle analysis, and colony survival assays under the supervision of A.P. and A.A.S. E.R. and S.B. prepared mass spectrometry samples and analysed the proteomics data. F.V. performed in situ-PLA experiments. D.G.V generated HeLa FAN1 KO cells and prepared recombinant human FAN1. K.M.F. prepared recombinant GST-FAN1 fragments. M.G. performed QIBC analysis and in vitro deubiquitination experiments. V.v.A. and C.v.A. contributed to colony formations assays. I.U. and K.U. generated the RPE-1CAG129 cell lines and performed repeat expansion measurements under the supervision of G.B. A.H. prepared recombinant USP7 fragments and performed fluorescence polarization assays under the supervision of R.J.H. R.J.H generated AlphaFold3-based structural models and performed model curation and confidence assessment. R.G. performed multiple sequence alignments and conservation analysis of USP7 binding sites in FAN1. A.A.S. and G.C. designed the project with crucial contributions from M.G. and G.B. G.C., G.B., and A.A.S. wrote the manuscript with inputs from all authors.
Corresponding authors
Ethics declarations
Competing interests
G.B. is the founder and chief executive officer (part-time) of Function RX Ltd. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Vincenzo D’Angiolella, Grant S. Stewart, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Collotta, G., Gatti, M., Ungureanu, IM. et al. USP7 deubiquitinase stabilizes FAN1 to support DNA crosslink repair and suppress CAG repeat expansion. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70051-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70051-9