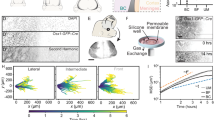
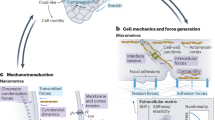
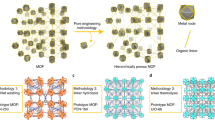
Abstract
Cellular mechanotransduction, mediated by specialized structures such as microvilli, regulates processes ranging from tissue homeostasis to disease progression. Existing tools for microvilli-specific biomechanical intervention suffer from limited spatiotemporal precision and non-physiological constraints, restricting mechanistic studies and targeted therapies. Here, we develop a magnetically driven gear-like metal-organic framework microrobot (MOFbot) for programmable mechanical manipulation of single-cell microvilli. MOFbots are fabricated through epitaxial growth of heterogeneous MOF structures followed by deposition of Ni/Au nanofilms. Under a rotating magnetic field, they perform rolling and obstacle negotiation. Their rotating gear structure entangles microvilli, exerting quantified pulling forces via Förster resonance energy transfer and traction force microscopy. This mechanical stimulation triggers intracellular calcium influx and enhanced focal adhesion kinase phosphorylation, indicating mechanotransduction pathway activation. Consequently, rotating MOFbots increase membrane permeability, enabling on-demand transmembrane delivery of therapeutics into targeted single cells. This work establishes a targeted cellular mechanomodulation strategy and informs future micro/nanorobotic biomedical designs.
Similar content being viewed by others
Data availability
The data generated or analysed during this study are included in this published article/Supplementary Information/Source Data file. Source data is available for Figs.1d–g, 2c, 2d, 2f, 2i, 2k, 3e–g, 4c, 4d, 4h, 4i, 5c–f, 5h, 6d, 6f–h and Supplementary Figs. 3, 4, 6, 8, 10b, 12b, 12c, 13b, 13c, 15b, 19c, 20b, 21, 22a, 22b, 24 in the associated source data file. The raw data generated in this study have also been deposited in the Zenodo database under accession code [10.5281/zenodo.18495219]. Source data are provided with this paper.
Code availability
Code associated with our paper is available at https://github.com/alfredyang93/Demo-datasets.
References
Zhang, Y. & Habibovic, P. Delivering mechanical stimulation to cells: state of the art in materials and devices design. Adv. Mater. 34, e2110267 (2022).
Humphrey, J. D., Dufresne, E. R. & Schwartz, M. A. Mechanotransduction and extracellular matrix homeostasis. Nat. Rev. Mol. Cell Biol. 15, 802–812 (2014).
Lin, Y. C. et al. Force-induced conformational changes in PIEZO1. Nature 573, 230 (2019).
Savadipour, A. et al. Membrane stretch as the mechanism of activation of PIEZO1 ion channels in chondrocytes. Proc. Natl. Acad. Sci. Usa. 120, e2221958120 (2023).
Andreu, I. et al. The force loading rate drives cell mechanosensing through both reinforcement and cytoskeletal softening. Nat. Commun. 12, 4229 (2021).
Dasgupta, I. & McCollum, D. Control of cellular responses to mechanical cues through YAP/TAZ regulation. J. Biol. Chem. 294, 17693–17706 (2019).
Elosegui-Artola, A. et al. Force triggers YAP nuclear entry by regulating transport across nuclear pores. Cell 171, 1397 (2017).
Korkmazhan, E. & Dunn, A. R. The membrane-actin linker ezrin acts as a sliding anchor. Sci. Adv. 8, eabo2779 (2022).
Du, Z. et al. Mechanosensory function of microvilli of the kidney proximal tubule. Proc. Natl. Acad. Sci. Usa. 101, 13068–13073 (2004).
Cai, E. et al. Visualizing dynamic microvillar search and stabilization during ligand detection by T cells. Science 356, eaal3118 (2017).
Weinbaum, S., Guo, P. & You, L. D. A new view of mechanotransduction and strain amplification in cells with microvilli and cell processes. Biorheology 38, 119–142 (2001).
Iyer, S., Gaikwad, R. M., Subba-Rao, V., Woodworth, C. D. & Sokolov, I. Atomic force microscopy detects differences in the surface brush of normal and cancerous cells. Nat. Nanotechnol. 4, 389–393 (2009).
Schneeberger, K. et al. An inducible mouse model for microvillus inclusion disease reveals a role for myosin Vb in apical and basolateral trafficking. Proc. Natl. Acad. Sci. USA. 112, 12408–12413 (2015).
Delon, L. C. et al. A systematic investigation of the effect of the fluid shear stress on Caco-2 cells towards the optimization of epithelial organ-on-chip models. Biomaterials 225, 119521 (2019).
Miura, S., Sato, K., Kato-Negishi, M., Teshima, T. & Takeuchi, S. Fluid shear triggers microvilli formation via mechanosensitive activation of TRPV6. Nat. Commun. 6, 8871 (2015).
Aramesh, M. et al. Nanoconfinement of microvilli alters gene expression and boosts T cell activation. Proc. Natl. Acad. Sci. USA. 118, e2107535118 (2021).
Luo, M., Feng, Y., Wang, T. & Guan, J. Micro-/nanorobots at work in active drug delivery. Adv. Funct. Mater. 28, 1706100 (2018).
Li, J. X., De Avila, B. E. F., Gao, W., Zhang, L. F. & Wang, J. Micro/nanorobots for biomedicine: delivery, surgery, sensing, and detoxification. Sci. Robot. 2, eaam6431 (2017).
Gultepe, E. et al. Biopsy with thermally-responsive untethered microtools. Adv. Mater. 25, 514–519 (2013).
Wu, Z. et al. A swarm of slippery micropropellers penetrates the vitreous body of the eye. Sci. Adv. 4, eaat4388 (2018).
Gao, Y. et al. Magnetically manipulated optoelectronic hybrid microrobots for optically targeted non-genetic neuromodulation. Adv. Mater. 36, 2305632 (2024).
Lin, Z. et al. Magnetically actuated peanut colloid motors for cell manipulation and patterning. ACS Nano 12, 2539–2545 (2018).
Chen, W. J. et al. Recent progress of micro/nanorobots for cell delivery and manipulation. Adv. Funct. Mater. 32, 2110625 (2022).
Gao, Y. X. et al. Template-guided silicon micromotor assembly for enhanced cell manipulation. Angew. Chem. 63, e202405895 (2024).
Zhou, H. J., Mayorga-Martinez, C. C., Pané, S., Zhang, L. & Pumera, M. Magnetically driven micro and nanorobots. Chem. Rev. 121, 4999–5041 (2021).
Cao, Y. et al. Magnetic continuum robot with modular axial magnetization: design, modeling, optimization, and control. IEEE Trans. Rob. 41, 1513–1532 (2025).
Liu, C. et al. Site-specific growth of MOF-on-MOF heterostructures with controllable nano-architectures: beyond the combination of MOF analogues. Chem. Sci. 11, 3680–3686 (2020).
Bergert, M. et al. Confocal reference free traction force microscopy. Nat. Commun. 7, 12814 (2016).
Guo, J., Wang, Y., Sachs, F. & Meng, F. Actin stress in cell reprogramming. Proc. Natl. Acad. Sci. Usa. 111, E5252–E5261 (2014).
Yang, J. et al. Photo-tunable hydrogels reveal cellular sensing of rapid rigidity changes through the accumulation of mechanical signaling molecules. Cell Stem Cell 32, 121–136 (2025).
Zhang, M. et al. Cell mechanical responses to subcellular perturbations generated by ultrasound and targeted microbubbles. Acta Biomater. 155, 471–481 (2023).
Yuan, F., Yang, C. & Zhong, P. Cell membrane deformation and bioeffects produced by tandem bubble-induced jetting flow. Proc. Natl. Acad. Sci. USA. 112, E7039–E7047 (2015).
Yang, T. et al. Microwheels on microroads: enhanced translation on topographic surfaces. Sci. Robot. 4, eaaw9525 (2019).
Chien, S. Molecular and mechanical bases of focal lipid accumulation in arterial wall. Prog. Biophys. Mol. Biol. 83, 131–151 (2003).
Shi, Z., Graber, Z. T., Baumgart, T., Stone, H. A. & Cohen, A. E. Cell membranes resist flow. Cell 175, 1769–1779 (2018).
Katta, S., Krieg, M. & Goodman, M. B. Feeling force: physical and physiological principles enabling sensory mechanotransduction. Annu. Rev. Cell Dev. Biol. 31, 347–371 (2015).
Acknowledgements
This work was supported by Shenzhen Science and Technology Program (RCJC20231211090000001 to X.M.), Shenzhen Medical Research Fund (A2403068 to D.J.), Shenzhen Science and Technology Program (GXWD20231129101105001 to X.M., KJZD20231023100302006 to D.J.), National Natural Science Foundation of China (52472280 to D.J., 82402730 to Y.W.), Macau Foundation for Development of Sicence and Technology (0008/2024/RIA1 to X.M.), China Postdoctoral Science Foundation (2024M763882 to X.L.).
Author information
Authors and Affiliations
Contributions
X.L. synthesized and characterized the MOFbots, analyzed the interaction between MOFbots and cellular microvilli, and wrote the paper. Y.W. was responsible for video recording of the MOFbots and data analysis. L.L. simulated the flow field around the MOFbots. N.L. simulated the interaction between the gear-like MOFbot and cellular microvilli. Z.Y. analyzed the distribution of gear-like MOFbots within cellular microvilli using the Unet + + network model. P.W. analyzed the cell substrate deformation induced by the MOFbot. X.Y. and J.G. revised the manuscript. Y.W., D.J. and X.M. conceived the initial idea, supervised the project, and edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Pierre Bongrand and Jianguo Guan for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, X., Wang, Y., Lin, L. et al. Gear-like MOF microrobots for single cell mechanotransduction of microvilli. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70052-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70052-8