Abstract
The differences in response times between vegetation physiology and vegetation structure to water stress at the global scale remain unclear. Here, we integrate solar-induced chlorophyll fluorescence satellite observations, optical remote sensing indices, and hydrometeorological data to globally disentangle the sequence and related factors of vegetation physiological and structural responses to drought. We isolated fluorescence efficiency by normalizing solar-induced chlorophyll fluorescence by absorbed photosynthetically active radiation; We show that fluorescence efficiency is a robust proxy for ecosystem-level vegetation physiology and responds to drought within ~3 days, while structural changes emerge after ~12 days. The contrast in timing is clearest in humid regions, owing to the sustained soil moisture availability during the initial stage of drought. Physiological responses are more temporally aligned with changes in vapor pressure deficit, whereas structural changes coincide more with soil moisture dynamics, reflecting differing patterns of association under increasing drought stress. These findings advance mechanistic understanding of vegetation drought responses across the continuum from physiological to structural processes.
Similar content being viewed by others
Introduction
Drought is one of the most prevalent extreme climate events globally, with its frequency, intensity, and duration increasing in a warmer world1. Because of prolonged water deficit, drought impairs photosynthetic efficiency2, inhibits carbon fixation in plants3, and may lead to plant hydraulic failure4,5. This can lead to individual plant mortality6 and potentially ecosystem-level dieback7. The response time of vegetation to drought stress reveals the time required for plants to exhibit detectable physiological and structural changes after sensing water scarcity. Quantifying these time scales not only enhances our understanding of ecosystem drought response but also enables more accurate prediction of drought impacts on land surface carbon and water balances.
Reflectance-based optical remote sensing to measure vegetation greenness and structure has been well established. Vegetation indices (VIs) such as the enhanced vegetation index (EVI) or the near-infrared reflectance of vegetation (NIRv) are effective in capturing key physical properties like density and coverage8,9. These indices are therefore widely used as proxies in ecosystem-scale studies of vegetation responses to drought10,11,12,13. Since the dynamics of these indices primarily reflect slow changes in plant tissues (e.g., above-ground biomass)9,14, their response to drought is generally lagged at the ecosystem scale14,15. This could introduce delays in representing drought-induced vegetation stress in global carbon cycle modeling15,16,17, assessments of ecosystem services18,19, and wildfire forecasting20,21.
Vegetation physiology typically exhibits a more rapid response to stress than vegetation greenness and structure22. Specifically, plants respond to increased transpiration demand and soil water deficit by adjusting several physiological processes, such as reducing stomatal conductance23. This reduction subsequently diminishes both transpiration and photosynthesis rates, which in turn leads to a decline in carbon fixation and biomass accumulation24, and triggers a series of ecosystem–atmosphere feedbacks25,26. Hence, physiological adjustments later manifest in greenness and structure. Vegetation physiological parameters, including maximum carboxylation efficiency, electron transport rate, and stomatal conductance, have been shown to exhibit greater sensitivity and faster response to stress than greenness and structure at both leaf and ecosystem levels27,28. However, ecosystem-level physiological drought responses remain understudied as vegetation physiological parameters have so far remained difficult to isolate unambiguously from space-based remote sensing.
Recent advances in solar-induced fluorescence (SIF) satellite observations provide new opportunities to study global physiological responses to drought. SIF is the release of energy in the form of radiation when excited chlorophyll molecules return to their ground state after absorbing sunlight. Closely associated with key physiological processes related to plant photosynthesis, SIF serves as a comprehensive proxy for vegetation physiology by capturing the dynamics of energy partitioning within the photosynthetic apparatus29. Under non-stress conditions, high SIF emission generally indicates active photosynthesis and high light use, whereas under stress (e.g., drought), changes in SIF can indicate shifts toward enhanced non-photochemical energy dissipation and reduced photosynthetic efficiency30. However, factors such as solar radiation and canopy properties govern the SIF signal to a great extent, and the fluorescence quantum yield (SIFyield), the fraction of absorbed light re-emitted as fluorescence and leaving the canopy, represents only a small portion of the variability in SIF. This is, however, the component that is closely tied to vegetation physiology and regulated by photochemical and non-photochemical quenching. Consequently, isolating the physiological signal from these structural and radiative contributions is essential to accurately extract information on vegetation function and stress responses. Recent studies have also shown the potential of isolating SIFyield to diagnose gross primary productivity, in specific vegetation types or under regional drought events25,31,32,33,34,35,36,37,38,39.
Here, we explore the response times of global vegetation physiology, greenness, and structure to drought stress at a daily scale. We consider satellite SIF observations as influenced by three factors: incoming radiation, vegetation canopy properties, and SIFyield31,40,41. To quantify these factors, we leverage satellite observations of PAR and the fraction of absorbed PAR (i.e., fPAR) to compute the absorbed PAR (i.e., APAR; see “Methods”: Isolating vegetation physiology). Because SIF primarily responds to light regardless of stress conditions, we normalize it to account for variations in incoming radiation (i.e., SIF/PAR). Specifically, we consider SIF/PAR as a mixed proxy of vegetation canopy properties and SIFyield, while SIF normalized by absorbed photosynthetically active radiation (SIF/APAR) isolates the SIFyield itself, thus serving as a refined proxy for vegetation physiological functioning. Meanwhile, EVI, NIRv, fPAR and leaf area index (LAI) are considered as indicators of vegetation greenness and/or structure.
While the influence of soil moisture (SM) deficits during drought on vegetation dynamics has been well studied42,43,44, the impact of high vapor pressure deficit (VPD) during drought on plant physiology is less well understood45,46,47. As drought persists, SM is slowly depleted at time scales that match the gradual structural impacts observed in vegetation. This temporal alignment may lead to a potentially misleading conclusion that SM is the dominant driver of vegetation decay during droughts. In contrast, VPD rapidly responds to factors such as air temperature, evaporation, radiation, and wind speed45,48,49,50, and it directly influences the plant economics at the leaf level, triggering faster physiological changes47,51. In response to increased VPD during drought, immediate physiological adjustments at the leaf level often precede significant declines in SM47,51. From this perspective, plant physiology may show an earlier response to atmospheric dryness (high VPD) than to SM deficits during droughts. Here, we simultaneously explore the effects of soil and atmospheric drought and leverage vegetation physiological indicators like SIFyield for detecting earlier vegetation responses that are not reflected by traditional VIs. The evaporative stress index (ESI) is used to identify the most severe drought stress event between 2018 and 2022 for every terrestrial location on Earth. For these drought events, we then explore the covariance of the anomalies in VPD or SM with the anomalies in the SIF-derived and other VIs, and calculate the time lags of the cross-correlations to reveal mean vegetation response times.
Results and discussion
Impact of drought on vegetation
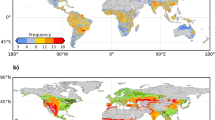
About 74% of global vegetated areas experienced at least one drought event persisting for over 30 days during the 5-year study period (Fig. 1). These drought events correspond well with major reported droughts (Fig. 1a), such as the 2019 European52,53, the 2019 eastern China54, or the 2018–2019 Australian55 events. Across both the Northern and Southern Hemispheres, drought occurrences predominantly cluster during summer months (Fig. 1b).
a Year of the most severe drought onset during the 2018–2022 study period. b Month of drought onset. The source data can be found in Supplementary Data 1.
Drought stress is well correlated with vegetation decline globally (Fig. 2). We observe significant correlations (p < 0.01, two-tailed t-test) between the anomalies in SM or VPD and the anomalies in vegetation state across ~83% of the drought-affected land area for at least one VI, with ~93% of the significant correlations found to be positive. Regionally, the VI anomalies in southern North America, southeastern South America, southeastern Africa, Indonesia, and northeastern Australia are most strongly correlated with SM anomalies and VPD anomalies, consistent with previous studies56. Nearly identical patterns emerge when SM is taken from an alternative database (ERA5-Land instead of GLEAM, Supplementary Fig. 1), and they remain independent of the selection of vegetation indices (Supplementary Fig. 2), soil depth (Supplementary Fig. 3), spatial scale (Supplementary Fig. 4), and whether non-linear rather than linear relationships between variables are considered (Supplementary Fig. 5). Negative correlations are primarily concentrated in energy-limited high latitudes in the Northern Hemisphere, where increased radiation during drought may promote vegetation activity by warming temperatures toward, but not beyond, the optimal range for plant growth57. We also find some negative correlations in the Amazon region, in line with the well-documented findings of canopy greening during drought due to enhanced radiation and access to deep water reserves12,58,59,60.
a Violin plots of Pearson’s correlations between the VI anomalies and the soil moisture (SM) or vapor pressure deficit (VPD) anomalies. The correlations refer to the correlation values at the peak position in the cross-correlation results (See “Methods”: Response time to drought). The horizontal dashed lines are the lower quartile (Q1), median (Q2) and upper quartile (Q3). Maps of correlations between the VIs anomalies and the SM anomalies (b, d, f, h, j) and between the VIs anomalies and the VPD anomalies (c, e, g, i, k). The VIs include SIF/APAR (b, c), SIF/PAR (d, e), SIF (f, g), EVI (h, i) and LAI (j, k). The correlations for all colored pixels in b–k are significant (p < 0.01, two-tailed t-test). * Indicates the correlation sign has been inverted. The source data can be found in Supplementary Data 1.
The strong correlations between anomalies of EVI or LAI and SM anomalies indicate that SM is the primary factor associated with vegetation canopy changes during drought (Fig. 2), or that at least their temporal scale of variability matches, as previously reported by other studies42,56. However, the impact of SM on vegetation physiology (SIF/APAR, as a proxy for SIFyield) is less pronounced (Fig. 2a), reflecting how vegetation physiology changes are faster than those of SM during drought, and may be governed by other factors such as VPD61.
Response time of vegetation to drought
The kernel density distribution curves of the global vegetation drought response time based on all available pixels are plotted in Fig. 3a, b. The curves indicate a significant global pattern in the response time of vegetation characteristics to drought, from physiology to greenness, and finally to structure. Specifically, among all VIs examined, SIF/APAR shows the most rapid response to drought (~3 days to VPD and ~5 days to SM), whereas LAI shows the most delayed response (~13 days to VPD and ~12 days to SM). This confirms the physiological character of SIF/APAR, which captures plant biochemical processes at the ecosystem scale. Furthermore, the enhanced sensitivity of SIF/APAR to VPD confirms that signal decomposition transforms SIF from a biophysical metric primarily sensitive to SM to a biochemical metric primarily controlled by VPD. These findings remain independent of the choice of SM dataset (Supplementary Fig. 6), specific vegetation index products (Supplementary Fig. 7), soil depth (Supplementary Fig. 8), spatial scale (Supplementary Fig. 9), and whether linear or non-linear relationships between variables are considered (Supplementary Fig. 10). The drought response times of SIF/PAR and SIF resemble those of EVI (Fig. 3a, b), indicating that their behavior is more influenced by variations in vegetation canopy structure and greenness. This is likely because SIF/PAR accounts for changes in incoming radiation but still retains the impact of fPAR, which predominantly reflects canopy structure. In contrast, SIF/APAR removes this structural component, isolating physiological changes.
a, b Kernel density distribution curves of response times of the VIs to soil moisture (SM) (a) and vapor pressure deficit (VPD) (b). The vertical dashed lines in the curves indicate the mode (i.e., the most repeated value). Maps of response times of the VIs to SM (c, e, g, i, k) and to VPD (d, f, h, j, l). The VIs include SIF/APAR (c, d), SIF/PAR (e, f), SIF (g, h), EVI (i, j) and LAI (k, l). The source data can be found in Supplementary Data 1.
Spatially, the response time of vegetation physiology to drought shows clear latitudinal variability (Fig. 3c, d). The shortest response time of SIF/APAR to drought occurs in intertropical regions, in line with other studies signaling the high sensitivity of vegetation to water deficits in these areas62,63,64, mostly driven by limitations in rainfall57,65 and temperature increases over the photosynthetic optimum57,66,67,68,69. A similar, albeit less distinct, spatial pattern is observed in vegetation greenness metrics (Fig. 3i, j), which may be attributed to the intrinsic coupling between leaf pigment dynamics and plant physiological mechanisms70.
Vegetation response to drought per biome and hydroclimatic regime
The correlations of the VI anomalies to VPD and SM anomalies across diverse biomes and hydroclimatic regimes remain consistent (Fig. 4a–h), but the response times vary (Fig. 4i–p). The response time of vegetation physiology (i.e. SIF/APAR) exhibits a gradual decrease along the aridity gradient from dry to wet, attributable to the enhanced drought-tolerance mechanisms evolved by vegetation in water-limited regions71,72,73. In humid environments, vegetation physiology is more responsive to VPD (Fig. 4l, p), consistent with site-level observations27,74. In the early stages of drought, the higher soil water availability in humid regions is insufficient to trigger plant stress responses in response to the declining SM, yet short-term VPD changes can rapidly regulate vegetation physiology at the leaf scale27,46,72,74.
We summarize correlations and response times in four background hydrological environments, including (Hyper) Arid (a, e, i, m), Semi-Arid (b, f, j, n), Dry-sub humid (c, g, k, o) and Humid (d, h, l, p). a–h, Correlations. i–p Response times. Results correspond to soil moisture (SM) (a–d, i–l) and vapor pressure deficit (VPD) (e–h, m–p). Values indicate the mode of the kernel density distribution curve for each group. TRO-BF: tropical broadleaf forests, TEM-BF: temperate broadleaf forests, NF: needleleaf forests, MF: mixed forests, SAV: savannas, GRA: grasslands, CRO: croplands. Evergreen broadleaf forests and deciduous broadleaf forests in the MCD12C1 are merged into one category. Then, the broadleaf forests are classified into TRO-BF and TEM-BF according to Köppen-Geiger climate classification78. Evergreen needleleaf forests and deciduous needleleaf forests in the MCD12C1 are also merged, the same as for savannas (i.e., open shrublands, closed shrublands, savannas and woody savannas). Hydroclimatic conditions are based on aridity index, defined as AI = Precipitation/Potential evaporation79: (Hyper) Arid: AI < 0.2; Semi-Arid: AI = 0.2–0.5; Dry sub-humid: AI = 0.5–0.65; Humid: AI > 0.65. The vertex labels of the unlabeled triangles and heptagons are the same as those of the first triangle and the first heptagon, respectively. The source data can be found in Supplementary Data 1.
Humid tropical broadleaf forest regions exhibit an almost immediate response to drought stress (Fig. 4l, p), likely due to high hydraulic vulnerability and stomatal sensitivity75,76. Croplands show slower drought responses relative to savannas and grasslands in dry sub-humid regions (Fig. 4o), presumably due to irrigation occasionally offsetting the rise in VPD and the declines in SM46,77. In general, the biome-specific analysis confirms the global observational patterns (Fig. 3), with physiological processes (SIF/APAR anomalies) showing faster response to atmospheric drought while morphological changes appear more in phase with soil water depletion.
Overall, our global analysis reveals distinct temporal sequences in vegetation responses to drought stress (Fig. 5), with physiological adjustments preceding changes in greenness and structural modifications. By decomposing satellite-derived fluorescence signals, we have demonstrated that vegetation physiology, measured through SIF/APAR, responds to drought immediately, particularly to VPD increases, while morphological changes manifest after weeks. This sequence suggests a mechanistic cascade in which VPD-induced physiological regulations function as early indicators of drought stress before changes in leaf pigments or canopy structure can be observed by traditional optical remote sensing proxies. The response patterns show marked spatial heterogeneity, with intertropical regions exhibiting heightened sensitivity to water stress. Notably, drought impacts exhibit a transition from patterns associated with VPD-linked physiological changes in the early stages to those aligned with SM-related morphological changes as drought stress persists. Factors such as drought frequency and intensity, legacy effects, and canopy overshooting may also influence vegetation responses to drought80,81; these factors are not explicitly accounted for, being a limitation of this study. In addition, compound events such as wildfires, plant diseases and insect pests, or other extreme disturbances occurring concurrently with drought, may affect the strength and timing of vegetation signals, and their potential impacts on vegetation responses warrant further investigation. Nevertheless, our results provide satellite-based evidence for the decoupling of physiological and morphological responses to water stress at the ecosystem scale, providing insights into global vegetation–drought interactions (Fig. 5). This refined mechanistic understanding of how vegetation responds to water stress across temporal and spatial scales at the physiological level has implications for climate prediction and the understanding of carbon and water balances26.
At drought onset, plant leaves initially respond to soil moisture (SM) declines and vapor pressure deficit (VPD) increases by closing stomata as a rapid physiological response to minimize water loss, while still maintaining their morphology. As drought persists, plants mitigate oxidative stress and photooxidation by degrading chlorophyll, which manifests as leaf chlorosis. Under prolonged drought conditions, severe cellular dehydration triggers plasmolysis and subsequent leaf abscission, leading to substantial alterations in canopy structure. The numbers are the days after drought onset and are the most repeated values globally for SIF/APAR (~3 days), enhanced vegetation index (~8 days) and leaf area index (~13 days) in response to VPD. Created in BioRender.
Methods
Vegetation data
A diverse array of satellite-based vegetation data was utilized to conduct a global study on vegetation physiological and morphological responses to drought, including SIF, EVI, NIRv, LAI, and fPAR.
The TROPOspheric Monitoring Instrument (TROPOMI) onboard Sentinel-5 Precursor provides global daily SIF observations with high spatial resolution82,83,84. Daily TROPOMI SIF data, retrieved at 743 nm, were gridded to the spatial resolution of 0.25° for the period from 1 May 2018 to 31 December 2022. SIF varies with changes in incoming radiation throughout the day at the leaf scale, even in the absence of drought stress. Since instantaneous SIF is an inadequate representation of the average state of vegetation photosynthetic potential over the course of a day, a day-length corrected SIF product was used82,84,85.
LAI was used as a proxy to measure vegetation structural characteristics. The LAI dataset, sourced from the Moderate Resolution Imaging Spectroradiometer (MODIS) MCD15A3H product (4 July 2002—31 December 2022, 500 m, 4-day), was spatially resampled to 0.25° using Google Earth Engine. A temporal linear interpolation was applied to obtain the daily LAI. The fPAR in MCD15A3H was processed in the same way as LAI and used to examine the effect of different vegetation metrics on the results.
EVI and NIRv were used to diagnose vegetation greenness, since they reflect changes in both canopy structure and pigment concentration. Daily EVI and daily NIRv were calculated from the MODIS MCD43C4 products (24 February 2000—31 December 2022, 0.05°, daily) according to Eqs. (1, 2), and then resampled to 0.25°.
where NIR is the near-infrared band, R is the red band, B is the blue band. 0.08 was used to eliminate the influence of soil background8. Only EVI and NIRv values in the range of 0 to 1 were retained.
Hydrometeorological data
ESI, the ratio of actual to potential evaporation, reflects transpiration and soil evaporation decrease under stress conditions86. ESI was employed to identify the most severe droughts during the study period (See Section: Data pre-processing). Daily ESI data were obtained from the Global Land Evaporation Amsterdam Model (GLEAM) v3.7b87,88 at the spatial resolution of 0.25° (1 January 2003—31 December 2022). Using the ESI for drought event identification allows us to examine whether there are differences in vegetation response to soil drought and atmospheric drought within the same drought event. SM and VPD were used as indicators of soil drought and atmospheric drought, respectively. Surface SM daily data with the same resolution and period as the ESI were also sourced from GLEAM v3.7b. Root zone SM in GLEAM v3.7b was used to test the influence of soil depth. To avoid dependence effects between GLEAM surface SM data and GLEAM ESI data, ERA-5 Land SM data were additionally used.
Global daily 0.25° air temperature (AT) and relative humidity (RH) were obtained by downscaling observations from the Atmospheric Infrared Sounder (AIRS). Global daily 0.25° VPD was then calculated according to Eq. (3)89,90.
where VPD (Pa) represents the difference between saturated and actual vapor pressure, AT is near-surface air temperature (°C), and RH is relative humidity (%).
PAR data were sourced from the Clouds and the Earth’s Radiant Energy System (CERES) SYN1deg Ed4.1 product (1 May 2018 – 31 December 2022, 1°, daily). The all-sky surface PAR direct flux and the all-sky surface PAR diffuse flux were summed to represent surface total incoming radiation. The surface total incoming radiation data were resampled to 0.25° using a spatial linear interpolation method.
Data pre-processing
All data were resampled to the spatial resolution of 0.25° and daily temporal resolution. Masks were applied using snow water equivalent (SWE) data from GlobSnow and the land cover mask band in the TROPOMI SIF product to mitigate the effects of snow cover and include only terrestrial ecosystems. Data points with SWE > 0 were excluded, and pixels identified as water bodies or bare land were filtered out. Data with quality control band values below 0.582,84 and cloud fraction greater than 50% were excluded to ensure SIF accuracy. The choice of cloud fraction is motivated by the trade-off between accuracy and amount of data (Supplementary Fig. 11). SIF was further decomposed into SIF/APAR to compute SIFyield as a proxy of vegetation physiology (see Section: Isolating vegetation physiology).
Our focus is on the interaction between vegetation and water stress during anomalous decreases in ESI. First, long-term trends were extracted using a locally weighted smoothing function91, with the overlap period set to 40% of the observation period; the influence of this parameter is limited (Supplementary Fig. 12). Subsequently, the anomalies were derived by subtracting the long-term trend and the daily average seasonal cycle from the original daily observations, with the period from 1 May 2018 to 31 December 2022 used as reference. A spatiotemporal smoothing using a 15-day and 3 × 3-pixel moving window was applied to reduce random errors. The 15-day window strikes a balance between capturing short-term dynamics and maintaining data continuity (Supplementary Fig. 13).
To identify the most severe drought event locally between 1 May 2018 and 31 December 2022, fluctuations for which the ESI anomalies remain below zero during at least 30 consecutive days were first detected (Supplementary Fig. 14a). Events separated by less than 30 days were merged into a single episode if no precipitation occurred in the interim. The droughts between 1 July 2018 and 31 October 2022 were further screened out (Supplementary Fig. 14b) to ensure the accuracy of the response time results (see Section: Response time to drought). The drought event with the most negative ESI anomaly was selected as the most severe drought event (Supplementary Fig. 14c). The first date on which the ESI anomaly dropped below 0 during the selected drought event was taken as the drought onset. The entire drought period was defined as extending from 30 days prior to the drought onset until the end of the drought.
The MODIS MCD12C1 land cover product was used to diagnose drought response times across different vegetation types. To minimize the influence of land cover change and non-vegetated surface, only pixels with consistent vegetation type over the five-year period (2018–2022) and a vegetation cover exceeding 50% were included in the analysis. The vegetation cover was defined as the proportion of vegetated pixels within each 0.25° grid in the raw land cover data. The dominant vegetation type corresponds to the one covering >50% of the pixel; mixed pixels without a dominant vegetation type were not further analysed. Ecosystem types in focus are forests, savannas, grasslands and croplands (Supplementary Fig. 15). All data processing was implemented in Python 3.10.4.
Isolating vegetation physiology
The satellite-observed SIF signal is conceptualized in a way that accounts for incoming solar radiation, canopy structure, and SIFyield40,41, thereby enabling the isolation of plant physiological signals92:
where PAR is photosynthetically active radiation, fPAR is the fraction of absorbed photosynthetically active radiation, SIFyield is the fluorescence quantum yield, known as the SIF photons re-emitted from the canopy, and it was computed as SIF/APAR in this study. APAR is absorbed photosynthetically active radiation. Then SIF/PAR can be computed as:
SIF/PAR reflects the product of fPAR and SIFyield, and was obtained here by dividing SIF by PAR. SIF/APAR was calculated as follows, with values falling below the 1st percentile or exceeding the 99th percentile being excluded from further analysis.
The ratio SIF/APAR used here can be regarded as a combined metric that implicitly incorporates both the intrinsic SIFyield and the fluorescence escape probability (fesc), since the fesc is not explicitly separated in our formulation. This treatment is consistent with the definition of apparent SIFyield by Wang et al.93
Response time to drought
Response times were determined by calculating cross-correlations between the VIs and drought indicators during drought. The drought index anomaly time series was shifted sequentially within a lag window of −5 to +31 days, in 1-day steps. For each shift, Pearson’s correlation and its corresponding p-value between the vegetation index anomalies and the drought index anomalies were calculated. Vegetation activity is theoretically expected to decay after the onset of drought; however, considering measurement errors, the correlation peak (trough for VPD) closest to zero with p-value (two-tailed, t-test) <0.01 within the lag window range of −5 to +31 days was searched. The correlation and lag of the first local peak (or trough) closest to the 0-point was defined as the covariation and response time between vegetation and drought. In the ideal case, the distribution of values in the correlation array is unimodal (Supplementary Fig. 16a) with a single peak value. However, in practice, a more complex correlation pattern was occasionally observed, with multiple peaks (Supplementary Fig. 16b). To capture the first response of vegetation to drought, we used the peak index closest to zero (zero in the lag range) as the response time within the lag range of −5 to +31 days (the gray area in Supplementary Fig. 16), provided that the p-value (two-tailed, t-test) <0.01. Pixels where no p-value was significant (i.e., no p-value < 0.01) were removed from further analysis. This rule was used for all vegetation physiological and structural indices in this study. A 3 × 3-pixel mean filter was applied to spatially smooth the results. Spearman’s correlation was additionally used to examine the influence of potential non-linear relationships between vegetation and drought stress on the results (Supplementary Fig. 5, Supplementary Fig. 10).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
TROPOMI SIF can be accessed from https://data-portal.s5p-pal.com/products/troposif.html. MCD43C4 EVI and MCD43C4 NIRv are available at https://lpdaac.usgs.gov/products/mcd43c4v061. MCD15A3H LAI and MCD15A3H fPAR are available at https://lpdaac.usgs.gov/products/mcd15a3hv061. MCD12C1 land cover data are from https://lpdaac.usgs.gov/products/mcd12c1v061. GLEAM ESI, GLEAM surface SM and GLEAM root zone SM can be downloaded from https://www.gleam.eu. ERA-5 Land SM and precipitation are available at https://cds.climate.copernicus.eu/datasets/reanalysis-era5-land?tab=overview. CERES SYN1deg Ed4.1 PAR can be accessed from https://ceres-tool.larc.nasa.gov/ord-tool/jsp/SYN1degEd41Selection.jsp. AIRS AT and AIRS RH are from https://airs.jpl.nasa.gov/data/get-data/standard-data. SWE data are from https://www.globsnow.info/swe. Köppen-Geiger climate classification maps can be downloaded from https://www.gloh2o.org/koppen. Global-AI_PET_v3 aridity index dataset is from https://doi.org/10.6084/m9.figshare.7504448.v5. The source data for Figs. 1−4 and Supplementary Figs. 1−16 can be found in Supplementary Data 1.
Code availability
The data and codes required to reproduce the main figures of the paper are available at [https://doi.org/10.5281/zenodo.18327559].
References
Gebrechorkos, S. H. et al. Warming accelerates global drought severity. Nature 642, 628–635 (2025).
Pinheiro, C. & Chaves, M. M. Photosynthesis and drought: can we make metabolic connections from available data? J. Exp. Bot. 62, 869–882 (2011).
Peters, W. et al. Increased water-use efficiency and reduced CO2 uptake by plants during droughts at a continental scale. Nat. Geosci. 11, 744–748 (2018).
Anderegg, W. R. L. et al. The roles of hydraulic and carbon stress in a widespread climate-induced forest die-off. Proc. Natl. Acad. Sci. USA 109, 233–237 (2012).
Choat, B. et al. Triggers of tree mortality under drought. Nature 558, 531–539 (2018).
McDowell, N. G. et al. Mechanisms of woody-plant mortality under rising drought, CO2 and vapour pressure deficit. Nat. Rev. Earth Environ. 3, 294–308 (2022).
McDowell, N. G. & Allen, C. D. Darcy’s law predicts widespread forest mortality under climate warming. Nat. Clim. Change 5, 669–672 (2015).
Badgley, G., Field, C. B. & Berry, J. A. Canopy near-infrared reflectance and terrestrial photosynthesis. Sci. Adv. 3, e1602244 (2017).
Huete, A. et al. Overview of the radiometric and biophysical performance of the MODIS vegetation indices. Remote Sens. Environ. 83, 195–213 (2002).
Liu, X. F. et al. Compound droughts slow down the greening of the Earth. Glob. Change Biol. 29, 3072–3084 (2023).
Li, X. Y. et al. Global variations in critical drought thresholds that impact vegetation. Natl. Sci. Rev. 10, nwad049 (2023).
Chen, S. L. et al. Amazon forest biogeography predicts resilience and vulnerability to drought. Nature 631, 111–117 (2024).
Huang, M. T. & Zhai, P. M. Protracted vegetation recovery after compound drought and hot extreme compared to general drought. Environ. Res. Lett. 20, 024001 (2025).
Wu, D. et al. Time-lag effects of global vegetation responses to climate change. Glob. Change Biol. 21, 3520–3531 (2015).
Morton, D. C. et al. Amazon forests maintain a consistent canopy structure and greenness during the dry season. Nature 506, 221–224 (2014).
Stocker, B. D. et al. Drought impacts on terrestrial primary production are underestimated by satellite monitoring. Nat. Geosci. 12, 264–270 (2019).
Hu, Z. et al. Decoupling of greenness and gross primary productivity as aridity decreases. Remote Sens. Environ. 279, 113120 (2022).
Zhou, G. Y. et al. Resistance of ecosystem services to global change weakened by the increasing number of environmental stressors. Nat. Geosci. 17, 882–888 (2024).
Millar, C. I. & Stephenson, N. L. Temperate forest health in an era of emerging megadisturbance. Science 349, 823–826 (2015).
Lai, G. K. et al. Earlier peak photosynthesis timing potentially escalates global wildfires. Natl. Sci. Rev. 11, nwae292 (2024).
Syphard, A. D., Velazco, S. J. E., Rose, M. B., Franklin, J. & Regan, H. M. The importance of geography in forecasting future fire patterns under climate change. Proc. Natl. Acad. Sci. 121, e2310076121 (2024).
Li, W. et al. Widespread and complex drought effects on vegetation physiology inferred from space. Nat. Commun. 14, 4640 (2023).
Lawlor, D. W. & Cornic, G. Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ. 25, 275–294 (2002).
Wong, S. C., Cowan, I. R. & Farquhar, G. D. Stomatal conductance correlates with photosynthetic capacity. Nature 282, 424–426 (1979).
Li, W. T. et al. Disentangling Effects of Vegetation Structure and Physiology on Land-Atmosphere Coupling. Glob. Change Biol. 31, e70035 (2025).
Miralles, D. G., de Arellano, J. V. G., Mcvicar, T. R. & Mahecha, M. D. Vegetation-climate feedbacks across scales. Ann. Ny. Acad. Sci. 1544, 27–41 (2025).
Fu, Z. et al. Atmospheric dryness reduces photosynthesis along a large range of soil water deficits. Nat. Commun. 13, 989 (2022).
Johnson, D. M. et al. Co-occurring woody species have diverse hydraulic strategies and mortality rates during an extreme drought. Plant Cell Environ. 41, 576–588 (2018).
Mohammed, G. H. et al. Remote sensing of solar-induced chlorophyll fluorescence (SIF) in vegetation: 50 years of progress. Remote Sens. Environ. 231, 111177 (2019).
Xu, S. et al. Structural and photosynthetic dynamics mediate the response of SIF to water stress in a potato crop. Remote Sens. Environ. 263, 112555 (2021).
Song, L. et al. Satellite sun-induced chlorophyll fluorescence detects early response of winter wheat to heat stress in the Indian Indo-Gangetic Plains. Glob. Change Biol. 24, 4023–4037 (2018).
Magney, T. S. et al. Mechanistic evidence for tracking the seasonality of photosynthesis with solar-induced fluorescence. Proc. Natl. Acad. Sci. USA 116, 11640–11645 (2019).
Yang, X. et al. Solar-induced chlorophyll fluorescence that correlates with canopy photosynthesis on diurnal and seasonal scales in a temperate deciduous forest. Geophys. Res. Lett. 42, 2977–2987 (2015).
Liu, X. J. et al. Modelling the influence of incident radiation on the SIF-based GPP estimation for maize. Agric. For. Meteorol. 307, 108522 (2021).
Wieneke, S. et al. Linking photosynthesis and sun-induced fluorescence at sub-daily to seasonal scales. Remote Sens. Environ. 219, 247–258 (2018).
Dechant, B. et al. Canopy structure explains the relationship between photosynthesis and sun-induced chlorophyll fluorescence in crops. Remote Sens. Environ. 241, 111733 (2020).
Zhang, Y. et al. Satellite solar-induced chlorophyll fluorescence tracks physiological drought stress development during the 2020 Southwest US drought. Glob. Change Biol. 29, 3395–3408 (2023).
Zeng, Y. et al. Combining near-infrared radiance of vegetation and fluorescence spectroscopy to detect effects of abiotic changes and stresses. Remote Sens. Environ. 270, 112856 (2022).
Yoshida, Y. et al. The 2010 Russian drought impact on satellite measurements of solar-induced chlorophyll fluorescence: insights from modeling and comparisons with parameters derived from satellite reflectances. Remote Sens. Environ. 166, 163–177 (2015).
Lu, H. B. et al. Large influence of atmospheric vapor pressure deficit on ecosystem production efficiency. Nat. Commun. 13, 1653 (2022).
Zhang, Y. G. et al. Estimation of vegetation photosynthetic capacity from space-based measurements of chlorophyll fluorescence for terrestrial biosphere models. Glob. Change Biol. 20, 3727–3742 (2014).
Yao, Y., Liu, Y. X., Zhou, S., Song, J. X. & Fu, B. J. Soil moisture determines the recovery time of ecosystems from drought. Glob. Change Biol. 29, 3562–3574 (2023).
Li, X. & Xiao, J. F. Global climatic controls on interannual variability of ecosystem productivity: Similarities and differences inferred from solar-induced chlorophyll fluorescence and enhanced vegetation index. Agric. For. Meteorol. 288, 108018 (2020).
Liu, L. B. et al. Soil moisture dominates dryness stress on ecosystem production globally. Nat. Commun. 11, 4892 (2020).
Grossiord, C. et al. Plant responses to rising vapor pressure deficit. N. Phytol. 226, 1550–1566 (2020).
Novick, K. A. et al. The impacts of rising vapour pressure deficit in natural and managed ecosystems. Plant Cell Environ. 47, 3561–3589 (2024).
Vicente-Serrano, S. M., McVicar, T. R., Miralles, D. G., Yang, Y. T. & Tomas-Burguera, M. Unraveling the influence of atmospheric evaporative demand on drought and its response to climate change. Wires Clim. Change 11, e632 (2020).
Vicente-Serrano, S. M. et al. Evidence of increasing drought severity caused by temperature rise in southern Europe. Environ. Res. Lett. 9, 44001 (2014).
Donohue, R. J., McVicar, T. R. & Roderick, M. L. Assessing the ability of potential evaporation formulations to capture the dynamics in evaporative demand within a changing climate. J. Hydrol. 386, 186–197 (2010).
McVicar, T. R. et al. Global review and synthesis of trends in observed terrestrial near-surface wind speeds: implications for evaporation. J. Hydrol. 416, 182–205 (2012).
Miralles, D. G., Gentine, P., Seneviratne, S. I. & Teuling, A. J. Land-atmospheric feedbacks during droughts and heatwaves: state of the science and current challenges. Ann. Ny. Acad. Sci. 1436, 19–35 (2019).
Rakovec, O. et al. The 2018-2020 multi-year drought sets a new benchmark in Europe. Earths Future 10, e2021EF002394 (2022).
Hari, V., Rakovec, O., Markonis, Y., Hanel, M. & Kumar, R. Increased future occurrences of the exceptional 2018–2019 Central European drought under global warming. Sci. Rep. 10, 12207 (2020).
Yan, X. et al. GRACE and land surface models reveal severe drought in eastern China in 2019. J. Hydrol. 601, 126640 (2021).
Dunne, A. & Kuleshov, Y. Drought risk assessment and mapping for the Murray-Darling Basin, Australia. Nat. Hazards 115, 839–863 (2023).
Li, W. T. et al. Widespread increase in vegetation sensitivity to soil moisture. Nat. Commun. 13, 3959 (2022).
Huang, M. T. et al. Air temperature optima of vegetation productivity across global biomes. Nat. Ecol. Evol. 3, 772–779 (2019).
Huete, A. R. et al. Amazon rainforests green up with sunlight in the dry season. Geophys. Res. Lett. 33, L06405 (2006).
She, X. J. et al. Varied responses of Amazon forests to the 2005, 2010, and 2015/2016 droughts inferred from multi-source satellite data. Agric. For. Meteorol. 353, 110051 (2024).
Saleska, S. R., Didan, K., Huete, A. R. & da Rocha, H. R. Amazon forests green-up during 2005 drought. Science 318, 612–612 (2007).
De Cannière, S., Baur, M. J., Chaparro, D., Jagdhuber, T. & Jonard, F. Water availability and atmospheric dryness controls on spaceborne sun-induced chlorophyll fluorescence yield. Remote Sens. Environ. 301, 113922 (2024).
González, R. et al. Diverging functional strategies but high sensitivity to an extreme drought in tropical dry forests. Ecol. Lett. 24, 451–463 (2021).
Zhou, L. M. et al. Widespread decline of Congo rainforest greenness in the past decade. Nature 509, 86–90 (2014).
Bennett, A. C. et al. Sensitivity of South American tropical forests to an extreme climate anomaly. Nat. Clim. Change 13, 967–974 (2023).
Guan, K. Y. et al. Photosynthetic seasonality of global tropical forests constrained by hydroclimate. Nat. Geosci. 8, 284–289 (2015).
Doughty, C. E. et al. Tropical forests are approaching critical temperature thresholds. Nature 621, 105–111 (2023).
Wang, X. H. et al. A two-fold increase of carbon cycle sensitivity to tropical temperature variations. Nature 506, 212–215 (2014).
Kullberg, A. T., Coombs, L., Ahuanari, R. D. S., Fortier, R. P. & Feeley, K. J. Leaf thermal safety margins decline at hotter temperatures in a natural warming ‘experiment’ in the Amazon. N. Phytol. 241, 1447–1463 (2024).
Piao, S. L. et al. Characteristics, drivers and feedback of global greening. Nat. Rev. Earth Environ. 1, 14–27 (2020).
Chaves, M. M., Maroco, J. P. & Pereira, J. S. Understanding plant responses to drought - from genes to the whole plant. Funct. Plant Biol. 30, 239–264 (2003).
Frank, D. et al. Effects of climate extremes on the terrestrial carbon cycle: concepts, processes and potential future impacts. Glob. Change Biol. 21, 2861–2880 (2015).
Vicente-Serrano, S. M. et al. Response of vegetation to drought time-scales across global land biomes. Proc. Natl. Acad. Sci. USA 110, 52–57 (2013).
Gupta, A., Rico-Medina, A. & Caño-Delgado, A. I. The physiology of plant responses to drought. Science 368, 266–269 (2020).
Novick, K. A. et al. The increasing importance of atmospheric demand for ecosystem water and carbon fluxes. Nat. Clim. Change 6, 1023–1027 (2016).
Martínez-Vilalta, J., Poyatos, R., Aguadé, D., Retana, J. & Mencuccini, M. A new look at water transport regulation in plants. N. Phytol. 204, 105–115 (2014).
Liu, L. Y. et al. Tropical tall forests are more sensitive and vulnerable to drought than short forests. Glob. Change Biol. 28, 1583–1595 (2022).
Zhang, J. W. et al. Sustainable irrigation based on co-regulation of soil water supply and atmospheric evaporative demand. Nat. Commun. 12, 5549 (2021).
Beck, H. E. et al. High-resolution (1 km) Köppen-Geiger maps for 1901-2099 based on constrained CMIP6 projections. Sci. Data 10, 724 (2023).
Zomer, R. J., Xu, J. C. & Trabucco, A. Version 3 of the global aridity index and potential evapotranspiration database. Sci. Data 9, 409 (2022).
Zhang, Y., Keenan, T. F. & Zhou, S. Exacerbated drought impacts on global ecosystems due to structural overshoot. Nat. Ecol. Evol. 5, 1490–1498 (2021).
Liu, Y. et al. Drought legacies delay spring green-up in northern ecosystems. Nat. Clim. Change 15, 444–451 (2025).
Köhler, P. et al. Global retrievals of solar-induced chlorophyll fluorescence with TROPOMI: first results and intersensor comparison to OCO-2. Geophys. Res. Lett. 45, 10456–10463 (2018).
Guanter, L. et al. Potential of the TROPOspheric Monitoring Instrument (TROPOMI) onboard the Sentinel-5 Precursor for the monitoring of terrestrial chlorophyll fluorescence. Atmos. Meas. Tech. 8, 1337–1352 (2015).
Guanter, L. et al. The TROPOSIF global sun-induced fluorescence dataset from the Sentinel-5P TROPOMI mission. Earth Syst. Sci. Data 13, 5423–5440 (2021).
Frankenberg, C. et al. New global observations of the terrestrial carbon cycle from GOSAT: Patterns of plant fluorescence with gross primary productivity. Geophys. Res. Lett. 38, L17706 (2011).
Anderson, M. C. et al. Evaluation of drought indices based on thermal remote sensing of evapotranspiration over the continental United States. J. Clim. 24, 2025–2044 (2011).
Miralles, D. G. et al. Global land-surface evaporation estimated from satellite-based observations. Hydrol. Earth Syst. Sc. 15, 453–469 (2011).
Martens, B. et al. GLEAM v3: satellite-based land evaporation and root-zone soil moisture. Geosci. Model Dev. 10, 1903–1925 (2017).
Murray, F. W. On the computation of saturation vapor pressure. J. Appl. Meteorol. Climatol. 6, 203–204 (1967).
Valiantzas, J. D. Simplified forms for the standardized FAO-56 Penman-Monteith reference evapotranspiration using limited weather data. J. Hydrol. 505, 13–23 (2013).
Cleveland, W. S. Robust locally weighted regression and smoothing scatterplots. J. Am. Stat. Assoc. 74, 829–836 (1979).
Maes, W. H. et al. Sun-induced fluorescence is closely linked to ecosystem transpiration as evidenced by satellite data and radiative transfer models. Remote Sens. Environ. 249, 112030 (2020).
Wang, C. et al. Satellite footprint data from OCO-2 and TROPOMI reveal significant spatio-temporal and inter-vegetation type variabilities of solar-induced fluorescence yield in the U.S. Midwest. Remote Sens. Environ. 241, 111728 (2020).
Acknowledgments
The authors thank Oscar Baez Villanueva at the Ghent University for providing snow water equivalent, relative humidity and air temperature data, and Xiaosheng Xia at the Sun Yat-sen University for help with code writing. D.G.M. acknowledges support from the European Research Council (ERC) through the HEAT Consolidator Grant (101088405). Z.T. acknowledges support from the China Scholarship Council (202306140085). The computing resources and services used were provided by the VSC (Flemish Supercomputer Center), funded by the Research Foundation – Flanders (FWO) and the Flemish government.
Author information
Authors and Affiliations
Contributions
Z.T. conducted data analysis, visualized results, and wrote the manuscript with the suggestions from D.G.M., W.H.M., and Z.Y.G.; D.G.M. and W.H.M. conceived the study concept, developed the methods and designed the experiment; Z.T. revised the manuscript based on critical comments from D.G.M. and W.H.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tang, Z., Miralles, D.G., Guo, Z. et al. Fast response of satellite fluorescence-derived plant physiology to drought stress. Nat Commun 17, 2886 (2026). https://doi.org/10.1038/s41467-026-70076-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-70076-0