Abstract
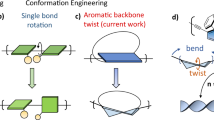
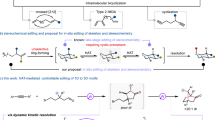
Topology plays an important role in polymeric materials. Herein, we present an iterative, modular approach for creating tetra-arylsubstituted alkene (TAA)-based dynamic conjugated oligomers with diverse topologies, using boronate-protected Suzuki-Miyaura coupling chemistry. The TAA building blocks involving spontaneous alkene isomerization are found to induce conformational dynamics in the conjugated backbones, exhibiting steric-controlled transitions. These transitions occur from a twisted backbone rich in cis-alkenes in the linear PL9 oligomer, to a stretched backbone with a trans-alkene center and multiple cis-alkene end in the three-armed planar PY12 oligomer and the four-armed 3D PX16 oligomer. Consequently, these topological oligomers exhibit distinct photoluminescence and photochemical properties depending on their physical state. Experimental characterization and molecular dynamics simulations (MD) reveal a topology-dependent adaptive self-assembly of helices: linear PL9 forms long flexible helical fibers with a pitch of 28 nm; planar Y-type PY12 oligomers often occur in neural-like networks, connected by nanofibers and cell-like central aggregates; and stereo X-type PX16 adopts short helical rod-like morphology with a mesoscopic pitch of 86 nm in crystalline phases. This work may inspire concepts and the practical construction of helical and neural-like fiber materials by altering unit topology in dynamic conjugated oligomers.
Similar content being viewed by others
Data availability
The data supplementary the findings of this study are available in the article. All data are available from the corresponding author upon request. Source Data are provided with this manuscript. The data in this manuscript do not include clinical data or third-party data; all data originate from the authors’ experimental work. The experimental characterization and computational data generated in this paper are provided in the supplementary information. Source data are provided with this paper.
References
Hemminger, J. et al. From quanta to the continuum: opportunities for mesoscale science. U.S. Department of Energy, 3-11 (Argonne National Laboratory, United States, (2012).
Hughes, J., Tran, C. & Rappporteurs, J. A. Mesoscale chemistry: a workshop summary. National Academies Press, 2–8 (Washington, DC, 2015).
Tu, Y., Zhao, Z., Lam, J. W. Y. & Tang, B. Z. Aggregate science: much to explore in the meso world. Matter 4, 338–349 (2021).
Zhang, B., Watuthanthrige, N. D. A., Wanasinghe, S. V., Averick, S. & Konkolewicz, D. Complementary dynamic chemistries for multifunctional polymeric materials. Adv. Funct. Mater. 32, 2108431 (2022).
Ni, Z. et al. Mesopolymer synthesis by ligand-modulated direct arylation polycondensation towards n-type and ambipolar conjugated systems. Nat. Chem. 11, 271–277 (2019).
Genabeek, B. V. et al. Properties and applications of precision oligomer materials; where organic and polymer chemistry join forces. J. Polym. Sci. 59, 373–403 (2021).
Shi, Q., Deng, Z., Hou, M., Hu, X. & Liu, S. Engineering precise sequence-defined polymers for advanced functions. Prog. Polym. Sci. 141, 101677 (2023).
Murphy, E. A., Zhang, C., Bates, C. M. & Hawker, C. J. Chromatographic separation: a versatile strategy to prepare discrete and well-defined polymer libraries. Acc. Chem. Res. 57, 1202–1213 (2024).
Pasgrimaud, C. et al. Synthesis of electron-deficient BisAzaCoroneneDiimide-conjugated polymers by light-locking dynamic covalent bonds. J. Am. Chem. Soc. 147, 12218–12227 (2025).
Anderson, P. W. More is different. Science 177, 393–396 (1972).
Barua, H., Svärd, M., Rasmuson, ÅC., Hudson, S. P. & Cookman, J. Mesoscale clusters in the crystallisation of organic molecules. Angew. Chem. Int. Ed. 63, e202312100 (2024).
Zhang, Y., Xie, S., Zeng, Z. & Tang, B. Z. Functional scaffolds from AIE building blocks. Matter 3, 1862–1892 (2020).
Xu, G. et al. -Cyano triaryl[3]radialene: unsymmetrical stereo-configuration, clustering-enhanced excimer emission, and radical-involved multimodal information switching. Angew. Chem. Int. Ed. 62, e202305011 (2023).
Wang, X. et al. Photoelectromagnetic responsive adaptive porous frameworks through dynamic covalent chemistry of tetraarylethylene-backboned aryldicyanomethyl radicals. Angew. Chem. Int. Ed. 62, e202301719 (2023).
Liu, H. et al. A photoelectromagnetic 3D metal-organic framework from flexible tetraarylethylene-backboned ligand and dynamic copper-based coordination chemistry. Small 20, 2306956 (2024).
Meng, S. et al. Anisotropic flexibility and rigidification in a TPE-based Zr-MOFs with scutopology. Nat. Commun. 14, 5347 (2023).
Wang, S., Zhang, Q., Wang, Z., Guan, S. & Zhang, X. Tetraphenylethylene-based hydrogen-bonded organic frameworks (HOFs) with brilliant fluorescence. Angew. Chem. Int. Ed. 62, e202315382 (2023).
Li, C., Zhao, W., He, J. & Zhang, Y. Topology controlled all- (meth) acrylic thermoplastic elastomers by multi-functional Lewis pairs-mediated polymerization. Angew. Chem. Int. Ed. 63, e202401265 (2024).
Irie, M., Fukaminato, T., Matsuda, K. & Kobatake, S. Photochromism of diarylethene molecules and crystals: memories, switches, and actuators. Chem. Rev. 114, 12174–12277 (2014).
Zhou, Z. et al. Spiro-functionalized diphenylethenes: suppression of a reversible photocyclization contributes to the aggregation-induced emission effect. J. Am. Chem. Soc. 141, 9803–9807 (2019).
Xie, Y. & Li, Z. Recent advances in the Z/E isomers of tetraphenylethene derivatives: stereoselective synthesis, AIE mechanism, photophysical properties, and application as chemical probes. Chem. Asian J. 14, 2524–2541 (2019).
Huang, Y. et al. Tuning proapoptotic activity of a phosphoric-acid-tethered tetraphenylethene by visible-light-triggered isomerization and switchable protein interactions for cancer therapy. Angew. Chem. Int. Ed. 61, e202208378 (2022).
Huang, Y., Zhang, G., Zhao, R. & Zhang, D. Tetraphenylethene-based cis/trans isomers for targeted fluorescence sensing and biomedical applications. Chem. Eur. J. 29, e202300539 (2023).
Liu, X., Li, Y., Li, X. & Hahn, F. E. Photoinduced E to Z isomerization of tetraphenylethylene derivatives within organometallic supramolecular assemblies. Sci. China Chem. 64, 1709–1715 (2021).
Yu, X. Trans/cis-stereoisomers of triterpenoid-substituted tetraphenylethene: aggregation-induced emission, aggregate morphology, and mechano-chromism. Nanoscale 13, 15257–15266 (2021).
Nie, X. et al. Kinetic and thermodynamic control of tetraphenylethene aggregation-induced emission behaviors. Aggregate 3, e165 (2022).
Bian, Q. et al. Iterative synthesis of dynamic conjugated oligomers displaying length-dependent configuration metastability, state-dependent properties, and helix emergence. Angew. Chem. Int. Ed. 64, e202505703 (2025).
Xia, Q. et al. Direct visualization of chiral ampli fication of chiral aggregation induced emission molecules in nematic liquid crystals. ACS Nano 15, 4956–4966 (2021).
Li, Z. et al. Visual β-sheet-to-β-turn transition in luminescent polymeric vesicles for color-reporting drug delivery. Angew. Chem. Int. Ed. 64, e202503875 (2025).
Wu, Z. et al. Dynamic cross-linked topological network reconciles the longstanding contradictory properties of polymers. Sci. Adv. 11, e0825 (2025).
Webber, M. J. & Tibbitt, M. W. Dynamic and reconfigurable materials from reversible network interactions. Nat. Rev. Mater. 7, 541–556 (2022).
Fang, Z. et al. 3D printing of dynamic covalent polymer network with on-demand geometric and mechanical reprogrammability. Nat. Commun. 14, 1313 (2023).
Reith, M. A. et al. Sequence-dezined mikto-arm star-shaped macromolecules. J. Am. Chem. Soc. 144, 7236–7244 (2022).
Reilly, L. T., Kottage, D., Cavallo, L., Falivene, L. & Chen, E. Y. Entropy-regulated selective synthesis of cyclic polymers and polycatenanes by Lewis pair polymerization. J. Am. Chem. Soc. 147, 31689–31703 (2025).
Weil, T. & Chen, C. Cyclic polymers: synthesis, characteristics, and emerging applications. Nanoscale Horiz. 7, 1121–1135 (2022).
Xu, C. et al. Regio- and sequence-controlled conjugated topological oligomers and polymers via boronate-tag assisted solution-phase strategy. Nat. Commun. 12, 5853 (2021).
Yin, J., Choi, S., Pyle, D., Guest, J. R. & Dong, G. Backbone engineering of monodisperse conjugated polymers via integrated iterative binomial synthesis. J. Am. Chem. Soc. 145, 19120–19128 (2023).
Mills, H. A. et al. Sequence-defined conjugated oligomers in donor-acceptor dyads. J. Am. Chem. Soc. 145, 23519–23526 (2023).
Li, Z. & Tang, Y. Stair-like narrow N-doped nanographene with unusual diradical character at the topological interface. Chem 9, 1–14 (2023).
Kim, M. P. et al. Iterative SuFEx approach for sequence-regulated oligosulfates and its extension to periodic copolymers. Nat. Commun. 15, 3381 (2024).
Lai, Y. et al. Modulating ligand-exchange dynamics on metal-organic polyhedra for reversible sorting and hybridization of miktoarm star polymers. Angew. Chem. Int. Ed. 62, e202311954 (2023).
Gkolfi, E. & Harmandaris, V. Soft character of star-like polymer melts: from linear-like chains to impenetrable nanoparticles. Nano Lett. 23, 1608–1614 (2023).
Hu, R., Leung, N. L. C. & Tang, B. Z. AIE macromolecules: syntheses, structures and functionalities. Chem. Soc. Rev. 43, 4494–4562 (2014).
Aida, T., Meijer, E. W. & Stupp, S. I. Functional supramolecular polymers. Science 335, 813–817 (2012).
Zhang, G., Cheng, X., Wang, Y. & Zhang, W. Supramolecular chiral polymeric aggregates: construction and applications. Aggregate 4, e262 (2023).
Wang, W. et al. Rapid automated iterative small-molecule synthesis. Nat. Synth. 3, 1031–1038 (2024).
Xu, C., Dong, J., He, C., Yun, J. & Pan, X. Precise control of conjugated polymer synthesis from step-growth polymerization to iterative synthesis. Giant 14, 100154 (2023).
Blair, D. J. et al. Automated iterative Csp3-C bond formation. Nature 604, 92–97 (2022).
Lehmann, J. W., Blair, D. J. & Burke, M. D. Towards the generalized iterative. Nat. Rev. Chem. 2, 0115 (2018).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (52525306 and 22375059), Shenzhen Science and Technology Innovation Program (JCYJ20220530160403008), and Natural Science Foundation of Hunan Province (2023JJ50002 and 2024RC1027). Thanks to the Analytical Instrumentation Center of Hunan University, and the AIE institute (www.aietech.org.cn) for providing technical assistance.
Author information
Authors and Affiliations
Contributions
S.X., Z.Z., and B.Z.T. designed the research. Q.B., Ying Zhao, C.Z., Y.Z. ‘affiliation 1’, and Y.Z. ‘affiliation 3’ performed the research. Q.B. and S.X. conducted the analyses. S.X., Z.Z., and B.Z.T. supervised the research. Q.B. and S.X. wrote the paper. All authors substantially revised and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Anna Mielańczyk and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bian, Q., Zhao, Y., Zhang, C. et al. Topology-controlled dynamic conjugated oligomers from tetra-arylsubstituted alkene building blocks. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70106-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70106-x