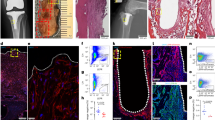
Abstract
Globally, approximately 1.3 million total joint implants are placed annually, and around 80,000 patients develop aseptic loosening, a leading cause of implant failure driven by peri-implant fibrosis. Here, we show that the BMP-antagonist Gremlin-1 (GREM1), expressed by leptin receptor–expressing skeletal (LEPR⁺) cells, is a key regulator of this process. GREM1 is highly expressed by LEPR⁺ cells in peri-implant fibrotic tissue in mice and humans. Conditional deletion of Grem1 in LEPR⁺ cells attenuate peri-implant fibrosis and enhances peri-implant osteogenesis. Transcriptomic and functional analyses show that loss of Grem1 in LEPR⁺ cells upregulate the bone morphogenetic protein (BMP) and WNT pathways, increasing in vivo osteogenesis and reducing fibrous tissue formation. As proof-of-concept, intra-articular administration of a neutralizing antibody against GREM1 (anti-GREM1) in mice prevents and reverses peri-implant fibrous tissue while promoting peri-implant bone formation. Inhibition of GREM1 in LEPR⁺ cells therefore represent a promising strategy to prevent and treat aseptic loosening.
Similar content being viewed by others
Data availability
The main data supporting the results in this study are available in the paper and supplementary information. The raw and analyzed data generated during the study are available for research purposes from the corresponding author within reasonable request. Transcriptomic data from bulk RNA-seq has been deposited at the Gene Expression Omnibus (GEO) under accession numbers GSE313854 and GSE227869. Source data are provided with this paper.
References
Ferguson, R. J. et al. Hip replacement. Lancet 392, 1662–1671 (2018).
Dossett, H. G. Machine learning: The future of total knee replacement. Fed. Pract. 39, 62–63 (2022).
Tomasi, C. & Derks, J. Etiology, occurrence, and consequences of implant loss. Periodontol 2000 88, 13–35 (2022).
Upfill-Brown, A. et al. Epidemiology of revision total knee arthroplasty in the United States, 2012 to 2019. Arthroplast Today 15, 188–195.e6 (2022).
Teeny, S. M., York, S. C., Mesko, J. W. & Rea, R. E. Long-term follow-up care recommendations after total hip and knee arthroplasty: Results of the American Association of Hip and Knee Surgeons’ member survey. J. Arthroplast. 18, 954–962 (2003).
Apostu, D. L. O., Berce, C., Lucaciu, D. & Cosma, D. Current methods of preventing aseptic loosening and improving osseointegration of titanium implants in cementless total hip arthroplasty: a review. J. Int. Med. Res. 46, 2104–2119 (2018).
Drees, P., Eckardt, A., Gay, R. E., Gay, S. & Huber, L. C. Mechanisms of disease: Molecular insights into aseptic loosening of orthopedic implants. Nat. Clin. Pr. Rheumatol. 3, 165–171 (2007).
Wooley, P. H. & Schwarz, E. M. Aseptic loosening. Gene Ther. 11, 402–407 (2004).
Gareb, B., Vissink, A., Terheyden, H., Meijer, H. J. A. & Raghoebar, G. M. Outcomes of implants placed in sites of previously failed implants: A systematic review and meta-analysis. Int J. Oral. Maxillofac. Surg. https://doi.org/10.1016/j.ijom.2024.10.006 (2024).
Yao, J. J. et al. Long-term mortality after revision THA. Clin. Orthop. Relat. Res. 476, 420–426 (2018).
Kienzle, A. et al. High rates of aseptic loosening after revision total knee arthroplasty for periprosthetic joint infection. JB JS Open Access;5 https://doi.org/10.2106/JBJS.OA.20.00026 (2020).
Wik, T. S., Klaksvik, J., Husby, O. S., Rasch, A. & Winther, S. B. Patient-reported outcome after primary and aseptic revision hip arthroplasty: 1-year follow-up of 3,559 primary and 406 revision THAs in an institutional registry. Acta Orthop. 93, 132–137 (2022).
Suhardi, V. J. et al. Prevention and treatment of peri-implant fibrosis by functionally inhibiting skeletal cells expressing the leptin receptor. Nat. Biomed. Eng. 8, 1285–1307 (2024).
Zhou, B. Y. R. et al. Leptin-receptor-expressing mesenchymal stromal cells represent the main source of bone formed by adult bone marrow. Cell 15, 154–168 (2014).
Aoki, K. et al. Identification of CXCL12-abundant reticular cells in human adult bone marrow. Br. J. Haematol. 193, 659–668 (2021).
Omatsu, Y., Seike, M., Sugiyama, T., Kume, T. & Nagasawa, T. Foxc1 is a critical regulator of haematopoietic stem/progenitor cell niche formation. Nature 508, 536–540 (2014).
Chan, C. K. et al. Identification and specification of the mouse skeletal stem cell. Cell 160, 285–298 (2015).
Greenbaum, A. et al. CXCL12 in early mesenchymal progenitors is required for haematopoietic stem-cell maintenance. Nature 495, 227–230 (2013).
Ortinau, L. C. et al. Identification of functionally distinct Mx1+αSMA+ periosteal skeletal stem cells. Cell Stem Cell. 25, 784–796.e5 (2019).
Baccin, C. et al. Combined single-cell and spatial transcriptomics reveal the molecular, cellular and spatial bone marrow niche organization. Nat. Cell Biol. 22, 38–48 (2020).
Jeffery, E. C., Mann, T. L. A., Pool, J. A., Zhao, Z. & Morrison, S. J. Bone marrow and periosteal skeletal stem/progenitor cells make distinct contributions to bone maintenance and repair. Cell Stem Cell. 29, 1547–1561.e6 (2022).
Zhong, L. et al. Single cell transcriptomics identifies a unique adipose lineage cell population that regulates bone marrow environment. Elife. 9. https://doi.org/10.7554/eLife.54695 (2020).
Hsu, D. R., Economides, A. N., Wang, X., Eimon, P. M. & Harland, R. M. The Xenopus dorsalizing factor Gremlin identifies a novel family of secreted proteins that antagonize BMP activities. Mol. Cell 1, 673–683 (1998).
Teng, F. Y., Chen, W. C., Wang, Y. L., Hung, C. C. & Tseng, C. C. Effects of osseointegration by bone morphogenetic protein-2 on titanium implants in vitro and in vivo. Bioinorg. Chem. Appl 2016, 3837679 (2016).
Pederson, L., Ruan, M., Westendorf, J. J., Khosla, S. & Oursler, M. J. Regulation of bone formation by osteoclasts involves Wnt/BMP signaling and the chemokine sphingosine-1-phosphate. Proc. Natl. Acad. Sci. USA. 105, 20764–20769 (2008).
Ye, Q., Taleb, S. J., Zhao, J. & Zhao, Y. Emerging role of BMPs/BMPR2 signaling pathway in treatment for pulmonary fibrosis. Biomed. Pharmacother. 178, 117178 (2024).
Yang, X. R. B. et al. Intermitent parathyroid hormone enchances cancellous osseointegration of a novel murine tibial implant. J. Bone Jt. Surg. Am. 97, 1074–1083 (2015).
Shi, Y. et al. Gli1 identifies osteogenic progenitors for bone formation and fracture repair. Nat. Commun. 8, 2043 (2017).
Debnath, S. et al. Discovery of a periosteal stem cell mediating intramembranous bone formation. Nature 562, 133–139 (2018).
Sarkaria, S. M. et al. Systematic dissection of coordinated stromal remodeling identifies Sox10(+) glial cells as a therapeutic target in myelofibrosis. Cell Stem Cell. 30, 832–850 e6 (2023).
Zhu, S., Chen, W., Masson, A. & Li, Y. P. Cell signaling and transcriptional regulation of osteoblast lineage commitment, differentiation, bone formation, and homeostasis. Cell Discov. 10, 71 (2024).
Liu, Q. et al. Recent Advances of Osterix Transcription Factor in Osteoblast Differentiation and Bone Formation. Front Cell Dev. Biol. 8, 601224 (2020).
Chen, S. et al. Grem1 accelerates nucleus pulposus cell apoptosis and intervertebral disc degeneration by inhibiting TGF-beta-mediated Smad2/3 phosphorylation. Exp. Mol. Med. 54, 518–530 (2022).
Gazzerro, E. et al. Conditional deletion of gremlin causes a transient increase in bone formation and bone mass. J. Biol. Chem. 282, 31549–31557 (2007).
He, L. et al. Parenchymal and stromal tissue regeneration of tooth organ by pivotal signals reinstated in decellularized matrix. Nat. Mater. 18, 627–637 (2019).
Matsumoto, T. et al. BMP-2 Induced Expression of Alx3 That Is a Positive Regulator of Osteoblast Differentiation. PLoS One 8, e68774 (2013).
Lavery, K. et al. New insights into BMP-7 mediated osteoblastic differentiation of primary human mesenchymal stem cells. Bone 45, 27–41 (2009).
Wright, V. et al. BMP4-expressing muscle-derived stem cells differentiate into osteogenic lineage and improve bone healing in immunocompetent mice. Mol. Ther. 6, 169–178 (2002).
Bialek, P. et al. A myostatin and activin decoy receptor enhances bone formation in mice. Bone 60, 162–171 (2014).
DiGirolamo, D. J., Singhal, V., Chang, X., Lee, S. J. & Germain-Lee, E. L. Administration of soluble activin receptor 2B increases bone and muscle mass in a mouse model of osteogenesis imperfecta. Bone Res. 3, 14042 (2015).
Agarwal, S. et al. BMP signaling mediated by constitutively active Activin type 1 receptor (ACVR1) results in ectopic bone formation localized to distal extremity joints. Dev. Biol. 400, 202–209 (2015).
Chen, G. et al. PiRNA-63049 inhibits bone formation through Wnt/beta-catenin signaling pathway. Int J. Biol. Sci. 17, 4409–4425 (2021).
Cawthorn, W. P. et al. Wnt6, Wnt10a and Wnt10b inhibit adipogenesis and stimulate osteoblastogenesis through a beta-catenin-dependent mechanism. Bone 50, 477–489 (2012).
Yu, B. et al. Wnt4 signaling prevents skeletal aging and inflammation by inhibiting nuclear factor-kappaB. Nat. Med. 20, 1009–1017 (2014).
Xu, Z. et al. Triple-gene deletion for osteocalcin significantly impairs the alignment of hydroxyapatite crystals and collagen in mice. Front Physiol. 14, 1136561 (2023).
Ehlermann, J., Weber, S., Pfisterer, P. & Schorle, H. Cloning, expression and characterization of the murine Efemp1, a gene mutated in Doyne-Honeycomb retinal dystrophy. Gene Expr. Patterns 3, 441–447 (2003).
Zhang, D. et al. EFEMP1 binds to STEAP1 to promote osteosarcoma proliferation and invasion via the Wnt/beta-catenin and TGF-beta/Smad2/3 signal pathways. J. Bone Oncol. 37, 100458 (2022).
Tan, T. W. et al. CCN3 increases BMP-4 expression and bone mineralization in osteoblasts. J. Cell Physiol. 227, 2531–2541 (2012).
von Heyking, K. et al. The posterior HOXD locus: Its contribution to phenotype and malignancy of Ewing sarcoma. Oncotarget 7, 41767–41780 (2016).
Guan, R. et al. Bone morphogenetic protein 4 inhibits pulmonary fibrosis by modulating cellular senescence and mitophagy in lung fibroblasts. Eur. Respir J. 60. https://doi.org/10.1183/13993003.02307-2021 (2022).
Li, R. X., Yiu, W. H. & Tang, S. C. Role of bone morphogenetic protein-7 in renal fibrosis. Front Physiol. 6, 114 (2015).
Yuan, Y., Han, Q., Li, S., Tian, Z. & Zhang, J. Wnt2b attenuates HSCs activation and liver fibrosis through negative regulating TLR4 signaling. Sci. Rep. 7, 3952 (2017).
Liu, J. et al. Wnt4 negatively regulates the TGF-beta1-induced human dermal fibroblast-to-myofibroblast transition via targeting Smad3 and ERK. Cell Tissue Res. 379, 537–548 (2020).
Beaton, H. et al. Wnt6 regulates epithelial cell differentiation and is dysregulated in renal fibrosis. Am. J. Physiol. Ren. Physiol. 311, F35–F45 (2016).
Gu, X. et al. The loss of Kruppel-like factor 15 in Foxd1(+) stromal cells exacerbates kidney fibrosis. Kidney Int. 92, 1178–1193 (2017).
Basu, M. & Roy, S. S. Wnt/beta-catenin pathway is regulated by PITX2 homeodomain protein and thus contributes to the proliferation of human ovarian adenocarcinoma cell, SKOV-3. J. Biol. Chem. 288, 4355–4367 (2013).
Mohr, M. E. et al. Cardiomyocyte-fibroblast interaction regulates ferroptosis and fibrosis after myocardial injury. iScience 27, 109219 (2024).
Yang, T. et al. EFEMP1 is repressed by estrogen and inhibits the epithelial-mesenchymal transition via Wnt/beta-catenin signaling in endometrial carcinoma. Oncotarget 7, 25712–25725 (2016).
Liu, R. et al. FGFR4 promotes stroma-induced epithelial-to-mesenchymal transition in colorectal cancer. Cancer Res. 73, 5926–5935 (2013).
Yu, C. et al. Increased carbon tetrachloride-induced liver injury and fibrosis in FGFR4-deficient mice. Am. J. Pathol. 161, 2003–2010 (2002).
Baron, R. & Rawadi, G. Targeting the Wnt/beta-catenin pathway to regulate bone formation in the adult skeleton. Endocrinology 148, 2635–2643 (2007).
Zhang, S., van de Peppel, J., Koedam, M., van Leeuwen, J. & van der Eerden, B. C. J. Tensin-3 is involved in osteogenic versus adipogenic fate of human bone marrow stromal cells. Cell Mol. Life Sci. 80, 277 (2023).
He, G. et al. NSD2-mediated H3K36me2 exacerbates osteoporosis via activation of hoxa2 in bone marrow mesenchymal stem cells. Cell Signal 121, 111294 (2024).
Ishikawa, M., Iwamoto, T., Fukumoto, S. & Yamada, Y. Pannexin 3 inhibits proliferation of osteoprogenitor cells by regulating Wnt and p21 signaling. J. Biol. Chem. 289, 2839–2851 (2014).
Leddy, H. A., McNulty, A. L., Guilak, F. & Liedtke, W. Unraveling the mechanism by which TRPV4 mutations cause skeletal dysplasias. Rare Dis. 2, e962971 (2014).
Qi, H. et al. FGFR3 induces degradation of BMP type I receptor to regulate skeletal development. Biochim Biophys. Acta 1843, 1237–1247 (2014).
Hioki, T. et al. Oncostatin M suppresses bone morphogenetic protein-4-induced osteoprotegerin synthesis in MC3T3-E1 osteoblast-like cells: p70 S6 kinase attenuation. Cell Biochem Funct. 42, e4068 (2024).
Cui, Y., Huang, R., Wang, Y., Zhu, L. & Zhang, X. Down-regulation of LGR6 promotes bone fracture recovery using bone marrow stromal cells. Biomed. Pharmacother. 99, 629–637 (2018).
Li, X. et al. Role of intercellular adhesion molecule-2 in osteoclastogenesis. Genes Cells 17, 568–575 (2012).
Kondo, T. et al. Insulin-like growth factor 2 promotes osteoclastogenesis increasing inflammatory cytokine levels under hypoxia. J. Pharm. Sci. 149, 93–99 (2022).
Xu, P. et al. Oncostatin M/Oncostatin M Receptor Signal Induces Radiation-Induced Heart Fibrosis by Regulating SMAD4 in Fibroblast. Int J. Radiat. Oncol. Biol. Phys. 118, 203–217 (2024).
Tampe, B. et al. Tet3-mediated hydroxymethylation of epigenetically silenced genes contributes to bone morphogenic protein 7-induced reversal of kidney fibrosis. J. Am. Soc. Nephrol. 25, 905–912 (2014).
Chen, X. et al. The hedgehog and Wnt/beta-catenin system machinery mediate myofibroblast differentiation of LR-MSCs in pulmonary fibrogenesis. Cell Death Dis. 9, 639 (2018).
Cortesi, EE. et al. Increased LGR6 expression sustains long-term Wnt activation and acquisition of senescence in epithelial progenitors in chronic lung diseases. Cells 10. https://doi.org/10.3390/cells10123437 (2021).
ETS1 is a key transcription factor that drives RANKL-expressing, tissue-destructive fibroblasts. Nat. Immunol 23:1303-1304. https://doi.org/10.1038/s41590-022-01298-9 (2022).
Zieba, J. et al. TGFbeta and BMP dependent cell fate changes due to loss of filamin B produces disc degeneration and progressive vertebral fusions. PLoS Genet. 12, e1005936 (2016).
Zhang, N., Guo, F. & Song, Y. HOXC8/TGF-beta1 positive feedback loop promotes liver fibrosis and hepatic stellate cell activation via activating Smad2/Smad3 signaling. Biochem Biophys. Res. Commun. 662, 39–46 (2023).
Bhattacharyya, S., Feferman, L. & Tobacman, J. K. Regulation of chondroitin-4-sulfotransferase (CHST11) expression by opposing effects of arylsulfatase B on BMP4 and Wnt9A. Biochim Biophys. Acta 1849, 342–352 (2015).
Li, C. H., Chan, M. H., Chang, Y. C. & Hsiao, M. The CHST11 gene is linked to lung cancer and pulmonary fibrosis. J. Gene Med. 24, e3451 (2022).
Zhan, L. & Li, J. The role of TRPV4 in fibrosis. Gene 642, 1–8 (2018).
Pereira, B. A. et al. Temporally resolved proteomics identifies nidogen-2 as a cotarget in pancreatic cancer that modulates fibrosis and therapy response. Sci. Adv. 10, eadl1197 (2024).
Waldrep KM, Rodgers JI, Garrett SM, Wolf BJ, Feghali-Bostwick CA. The role of SOX9 in IGF-II-mediated pulmonary fibrosis. Int. J. Mol. Sci. 24. https://doi.org/10.3390/ijms241411234 (2023).
Zhang, F. et al. Podocan unraveled: Understanding its role in tumor proliferation and smooth muscle regulation. Biomed. Pharmacother. 179, 117416 (2024).
Wei, T. et al. Wnt4 signaling is associated with the decrease of proliferation and increase of apoptosis during age-related thymic involution. Mol. Med. Rep. 12, 7568–7576 (2015).
Du, R. et al. Activin receptors in human cancer: Functions, mechanisms, and potential clinical applications. Biochem Pharm. 222, 116061 (2024).
Cai, P., Kovacs, L., Dong, S., Wu, G. & Su, Y. BMP4 inhibits PDGF-induced proliferation and collagen synthesis via PKA-mediated inhibition of calpain-2 in pulmonary artery smooth muscle cells. Am. J. Physiol. Lung Cell Mol. Physiol. 312, L638–L648 (2017).
Maliakal, J. C., Asahina, I., Hauschka, P. V. & Sampath, T. K. Osteogenic protein-1 (BMP-7) inhibits cell proliferation and stimulates the expression of markers characteristic of osteoblast phenotype in rat osteosarcoma (17/2.8) cells. Growth Factors 11, 227–234 (1994).
Jianfeng, W., Yutao, W. & Jianbin, B. Indolethylamine-N-methyltransferase inhibits proliferation and promotes apoptosis of human prostate cancer cells: A mechanistic exploration. Front Cell Dev. Biol. 10, 805402 (2022).
Du, H. F. et al. Expression of hepaCAM inhibits bladder cancer cell proliferation via a Wnt/beta-catenin-dependent pathway in vitro and in vivo. Cancer Biol. Ther. 16, 1502–1513 (2015).
Cui, H. et al. ENO3 inhibits growth and metastasis of hepatocellular carcinoma via Wnt/beta-catenin signaling pathway. Front Cell Dev. Biol. 9, 797102 (2021).
Ervinna, N. et al. Anagliptin, a DPP-4 inhibitor, suppresses proliferation of vascular smooth muscles and monocyte inflammatory reaction and attenuates atherosclerosis in male apo E-deficient mice. Endocrinology 154, 1260–1270 (2013).
Feng, Q., Li, S., Ma, H. M., Yang, W. T. & Zheng, P. S. LGR6 activates the Wnt/beta-catenin signaling pathway and forms a beta-catenin/TCF7L2/LGR6 feedback loop in LGR6(high) cervical cancer stem cells. Oncogene 40, 6103–6114 (2021).
Li, Z. et al. EGFL7 promotes hepatocellular carcinoma cell proliferation and inhibits cell apoptosis through increasing CKS2 expression by activating Wnt/beta-catenin signaling. J. Cell Biochem. 119, 10327–10337 (2018).
Mu, F. et al. The Wnt/β-Catenin/LEF1 pathway promotes cell proliferation at least in part through direct upregulation of miR-17-92 cluster. Front Genet. 10, 525 (2019).
Zhang, P. et al. Pannexin-3 deficiency delays skin wound healing in mice due to defects in channel functionality. J. Invest. Dermatol 139, 909–918 (2019).
Bernier-Latmani, J. et al. Apelin-driven endothelial cell migration sustains intestinal progenitor cells and tumor growth. Nat. Cardiovasc Res. 1, 476–490 (2022).
Wu, J. et al. Co-regulation of the Notch and Wnt signaling pathways promotes supporting cell proliferation and hair cell regeneration in mouse utricles. Sci. Rep. 6, 29418 (2016).
Zhang, J. et al. PIEZO1 functions as a potential oncogene by promoting cell proliferation and migration in gastric carcinogenesis. Mol. Carcinog. 57, 1144–1155 (2018).
Choi, J. et al. TERT promotes epithelial proliferation through transcriptional control of a Myc- and Wnt-related developmental program. PLoS Genet. 4, e10 (2008).
Xiong, Y. & Wang, Q. STC1 regulates glioblastoma migration and invasion via the TGF‑beta/SMAD4 signaling pathway. Mol. Med. Rep. 20, 3055–3064 (2019).
Sun, L. et al. Delta-like noncanonical notch ligand 2 regulates the proliferation and differentiation of sheep myoblasts through the Wnt/beta-catenin signaling pathway. J. Cell Physiol. 239, e31385 (2024).
Chang, S. H. et al. Excessive mechanical loading promotes osteoarthritis through the gremlin-1-NF-kappaB pathway. Nat. Commun. 10, 1442 (2019).
de Steiger, R. N., Lewis, P. L., Harris, I., Lorimer, M. F. & Graves, S. E. What is the outcome of the first revision procedure of primary THA for Osteoarthritis? A study From the Australian orthopaedic association national joint replacement registry. Clin. Orthop. Relat. Res. 480, 1952–1970 (2022).
Church, R. H. et al. Gremlin1 preferentially binds to bone morphogenetic protein-2 (BMP-2) and BMP-4 over BMP-7. Biochem J. 466, 55–68 (2015).
Rivera, I. R. et al. Abstract 5977: A first-in-human, open-label, multi-center phase 1 study of TST003, a GREM 1 inhibitor, in subjects with locally advanced or metastatic solid tumors. Cancer Res. 84, 5977–5977 (2024).
Sarker, D. et al. Abstract CT094: Phase I dose-escalation study evaluating the safety and tolerability of ginisortamab (UCB6114), a first-in-class anti-gremlin-1 monoclonal antibody (mAb), as monotherapy in advanced solid tumors. Cancer Res. 83, CT094–CT094 (2023).
Parvizi, J. et al. New definition for periprosthetic joint infection: From the Workgroup of the Musculoskeletal Infection Society. Clin. Orthop. Relat. Res. 469, 2992–2994 (2011).
Yang, X. et al. Intermittent parathyroid hormone enhances cancellous osseointegration of a novel murine tibial implant. J. Bone Jt. Surg. Am. 97, 1074–1083 (2015).
Bankhead, P. et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 7, 16878 (2017).
Bouxsein, M. L. et al. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J. Bone Min. Res. 25, 1468–1486 (2010).
Hildebrand, T. & Ruegsegger, P. A new method for the model-independent assessment of thickness in three-dimensional images. J. Microsc. 185, 67–75 (1997).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, i884–i890 (2018).
Dobin, A. et al. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Acknowledgements
We thank Ellena Monica for the assistance on manuscript figure editing and formatting. We thank Emile Kuyl for the assistance on patient sample procurement. We thank the Flow Cytometry Core, Genomics Resources Core, Optical Microscopy Core, and Citigroup Biomedical Imaging Core at Weill Cornell Medicine for their technical support. This project was funded by the OREF under awards 994088 and 892405, a Hospital for Special Surgery Surgeon-in-Chief Grant, and a Complex Joint Reconstruction Center grant given to VJS. XY is supported by grant UL1 TR000457 from the Clinical and Translational Science Center at Weill Cornell Medicine, the Feldstein Medical Foundation, and grant W81XWH-21-1-0900 from the Department of Defense. MBG is supported by a Pershing Square Sohn Cancer Research Alliance Award and R01AR075585.
Author information
Authors and Affiliations
Contributions
V.J.S. and A.O. designed, conducted, and analyzed the majority of experiments. X.Y., V.J.S., and M.P.G.B. conceived the project. X.Y. and M.P.G.B. supervised the project. X.Y. performed all mouse surgeries. V.J.S. performed μCT scans and analysis, and RNA-seq data analysis. V.J.S. and A.O. maintained and genotyped all mice. J.M. supervised or conducted flow cytometry. V.J.S., A.O., Y.N., J.A., A.T., and N.S. assisted with surgeries and performed tissue collection, immunofluorescence, and histology analysis. M.P.G.B. provided access to human samples and supervised human studies. U.A., M.B.G., and L.B.I. helped with the study design and data interpretation. V.J.S., A.O., N.S., and X.Y. prepared the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Jill Helms, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Suhardi, V.J., Oktarina, A., Niu, Y. et al. GREM1 acts in leptin receptor-expressing skeletal cells to mediate peri-implant fibrosis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70111-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70111-0