Abstract
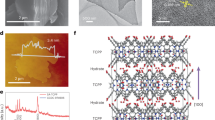
Photosynthesis of H2O2 under sunlight is a sustainable method; however, most developed photocatalysts utilize limited near-infrared light, which accounts for 52% of the solar spectrum. In typical near-infrared photocatalysts, excited electrons fall into low-energy sub-gap states, reducing the driving force for H2O2 generation. Here, a polydopamine-loaded porphyrin supramolecular photocatalyst efficiently utilizes near-infrared light for H2O2 production from H2O and O2, achieving an apparent quantum yield of 2.8% at 1020 nm. This substantial near-infrared utilization significantly boosts activity under full-spectrum irradiation, with an H2O2 generation rate of 3.37 mM/h and solar-to-chemical conversion efficiency of 2.2%. Persistent semiquinone radicals in polydopamine are demonstrated to enable ultrafast sub-gap electron transfer (ca. 79 fs) from porphyrin to polydopamine and facilitate near-infrared-driven •OOH radical generation, thereby accelerating H2O2 production. This study sheds light on the potential of near-infrared-responsive photocatalysts and offers insights into optimizing their performance for sustainable H2O2 synthesis.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the main text and its Supplementary Information. Atomic coordinates of the optimized computational models of PDA/SA-TCPP are available in Supplementary Data 1. Source data are provided in this paper.
References
Choi, J. et al. Photocatalytic Hydrogen peroxide production through functionalized semiconductive metal-organic frameworks. J. Am. Chem. Soc. 146, 11319–11327 (2024).
Hou, H., Zeng, X. & Zhang, X. Production of hydrogen peroxide by photocatalytic processes. Angew. Chem. Int. Ed. 59, 17356–17376 (2020).
Global hydrogen peroxide market forecast to experience a CAGR of 5.1% to 2030. Focus on Catalysts 2020, 3 (2020).
Campos-Martin, J., Blanco-Brieva, G. & Fierro, J. Hydrogen peroxide synthesis: an outlook beyond the anthraquinone process. Angew. Chem. Int. Ed. 45, 6962–6984 (2006).
Teng, Z. et al. Atomically dispersed antimony on carbon nitride for the artificial photosynthesis of hydrogen peroxide. Nat. Catal. 4, 374–384 (2021).
Kou, M. et al. Molecularly engineered covalent organic frameworks for hydrogen peroxide photosynthesis. Angew. Chem. Int. Ed. 61, e202200413 (2022).
Yoshikawa, A. et al. Near-infrared light-driven H2O2 generation via metalloporphyrin-based covalent organic frameworks and nanodisks. ACS Appl. Energy Mater. 7, 9084–9088 (2024).
Ou, H. et al. Hydroxyl-bonded Co single atom site on boroncarbonitride surface realizes nonsacrificial H2O2 synthesis in the near-infrared region. Adv. Mater. 36, e2404851 (2024).
Zhang, Y. et al. H2O2 generation from O2 and H2O on a near-infrared absorbing porphyrin supramolecular photocatalyst. Nat. Energy 8, 361–371 (2023).
Qi, F. et al. Interfacial reaction-induced defect engineering: enhanced visible and near-infrared absorption of wide band gap metal oxides with abundant oxygen vacancies. ACS Appl. Mater. Interfaces 12, 55417–55425 (2020).
Zhang, T. et al. Constructing oxygen-related defects in carbon nanodots with Janus optical properties: noninvasive NIR fluorescent imaging and effective photocatalytic therapy. Adv. Mater. 35, e2302705 (2023).
Zhang, H. et al. Hydrogen radical mediated concerted electron−proton transfer in 1D sulfone-based covalent organic framework for boosting photosynthesis of H2O2. Angew. Chem. Int. Ed. 64, e2025166572025 (2025).
Shiraishi, Y. et al. Resorcinol-formaldehyde resins as metal-free semiconductor photocatalysts for solar-to-hydrogen peroxide energy conversion. Nat. Mater. 18, 985–993 (2019).
Rao, P. & Hayon, E. Experimental determination of the redox potential of the superoxide radical •O2−. Biochem. Biophys. Res. Commun. 51, 468–473 (1973).
Petlicki, J. & van de Ven, T. The equilibrium between the oxidation of hydrogen peroxide by oxygen and the dismutation of peroxyl or superoxide radicals in aqueous solutions in contact with oxygen. J. Chem. Soc. Faraday. Trans. 94, 2763–2767 (1998).
Andrés, C. et al. Superoxide anion chemistry—its role at the core of the innate immunity. Int. J. Mol. Sci. 24, 1841 (2023).
Liu, Y. et al. Dopamine-melanin colloidal nanospheres: an efficient near-infrared photothermal therapeutic agent for in vivo cancer therapy. Adv. Mater. 25, 1353–1359 (2013).
Zangmeister, R., Morris, T. & Tarlov, M. Characterization of polydopamine thin films deposited at short times by autoxidation of dopamine. Langmuir. 29, 8619–8628 (2013).
Deng, Y. et al. Reduction of superoxide radical intermediate by polydopamine for efficient hydrogen peroxide photosynthesis. Angew. Chem. Int. Ed. 63, e202319216 (2024).
Fei, B. et al. Coating carbon nanotubes by spontaneous oxidative polymerization of dopamine. Carbon 46, 1795–1797 (2008).
Panzella, L. et al. Atypical structural and pi-electron features of a melanin polymer that lead to superior free-radical-scavenging properties. Angew. Chem. Int. Ed. 52, 12684–12687 (2013).
Bailey, C. et al. Polydopamine as a visible-light photosensitiser for photoinitiated polymerisation. Angew. Chem. Int. Ed. 62, e202301678 (2023).
Song, Y. & Buettner, G. Thermodynamic and kinetic considerations for the reaction of semiquinone radicals to form superoxide and hydrogen peroxide. Free Radical Biol. Med. 49, 919–962 (2010).
Guo, Y. et al. Hydrogen atom transfer from HOO• to ortho-quinones explains the antioxidant activity of polydopamine. Angew. Chem. Int. Ed. 60, 15220–15224 (2021).
Liu, H. et al. Role of polydopamine’s redox-activity on its pro-oxidant, radical-scavenging, and antimicrobial activities. Acta Biomater. 88, 181–196 (2019).
Lee, D. et al. Sustainable hydrogen peroxide production based on dopamine through Janus-like mechanism transition from chemical to photocatalytic reactions. J. Catal. 411, 235–244 (2022).
Yaman, H., Baig, M. & Kayan, A. Synthesis and characterization of tetrasubstituted porphyrin Tin(IV) complexes and their adsorption properties over tetracycline antibiotics. Reactions 6, 12 (2025).
Kurochkin, I. et al. Vibrational spectra of 5,10,15,20-Tetraphenylporphyrin (H2TPP) and Platinum(II) 5,10,15,20-Tetra(phenyl/pentafluorophenyl)porphyrins (PtTPP and PtTF5PP). Macroheterocycles 14, 334–341 (2021).
Takei, K., Takahashi, R. & Noguchi, T. Correlation between the hydrogen-bond structures and the C═O stretching frequencies of carboxylic acids as studied by density functional theory calculations: theoretical basis for interpretation of infrared bands of carboxylic groups in proteins. J. Phys. Chem. B. 112, 6725–6731 (2008).
Vasilopoulou, M. et al. Porphyrin oriented self-assembled nanostructures for efficient exciton dissociation in high-performing organic photovoltaics. J. Mater. Chem. A. 2, 182–192 (2014).
Kasha, M., Rawls, H. & El-Bayoumi, M. The exciton model in molecular spectroscopy. Pure. Appl. Chem. 11, 371–392 (1965).
Wei, Z. et al. Artificial Photosynthesis of H2O2 through reversible photoredox transformation between catechol and o-benzoquinone on polydopamine-coated CdS. ACS Catal. 12, 11436–11443 (2022).
Jing, J. et al. Supramolecular Zinc porphyrin photocatalyst with strong reduction ability and robust built-in electric field for highly efficient hydrogen production. Adv. Energy Mater. 11, 2101392 (2021).
Pang, H. et al. Capacitance methodology for investigating defect states in energy gap of organic semiconductor. Org. Electron. 65, 275–299 (2019).
Lian, Z. et al. Near infrared light induced plasmonic hot hole transfer at a nano-heterointerface. Nat. Commun. 9, 2314 (2018).
Zheng, Y. et al. Heavily doped carbon nitride nanocrystal promotes visible-near-infrared photosynthesis of hydrogen peroxide with near-unit photon utilization. ACS Nano. 18, 14583–14594 (2024).
Xiao, Y. et al. Long-lived internal charge-separated state in two-dimensional metal-organic frameworks improving photocatalytic performance. ACS Energy Lett. 7, 2323–2330 (2022).
Liu, H. et al. Delocalized orbitals over metal clusters and organic linkers enable boosted charge transfer in metal-organic framework for overall CO2 photoreduction. Angew. Chem. Int. Ed. 63, e202411508 (2024).
Wolff, C. et al. All-in-one visible-light-driven water splitting by combining nanoparticulate and molecular co-catalysts on CdS nanorods. Nat. Energy 3, 862–869 (2018).
Miao, T. & Tang, J. Characterization of charge carrier behavior in photocatalysis using transient absorption spectroscopy. J. Chem. Phys. 152, 194201 (2020).
Sun, H. et al. Efficient photothermal persulfate activation for rapid removal of antibiotics by heat-promoted Co3+ to Co2+ conversion on Bi12CoO20 photocatalyst. J. Catal. 428, 115172 (2023).
Zhang, L. et al. Visible-light-driven non-oxidative dehydrogenation of alkanes at ambient conditions. Nat. Energy 7, 1042–1051 (2022).
Zhao, L. et al. H2O2 self-supplied CuFeOx nanosystem as fenton-like reaction agents for endogenous/exogenous responsive synergetic antibacterial therapy. Chem. Eng. J. 492, 152265 (2024).
Fan, Y., Liu, J. & Han, X. Important contributions of in-situ produced H2O2 during photocatalytic sterilization of air by self-doped Bi2.15WO6. Sep. Purif. Technol. 363, 132052 (2025).
H2O2, price, Sigma-Aldrich, https://www.sigmaaldrich.com/US/en/product/sial/18304, (accessed Jan 06, 2026).
Ai, K. et al. Sp2 C-dominant N-doped carbon sub-micrometer spheres with a tunable size: a versatile platform for highly efficient oxygen-reduction catalysts. Adv. Mater. 25, 998–1003 (2013).
Dou, S. et al. Boosting photocatalytic oxygen reduction to hydrogen peroxide via chemisorbed oxygen activation on polydopamine-coated zinc oxide. J. Colloid Interface Sci. 691, 137370 (2025).
Pan, C. & Zhu, Y. New type of BiPO4 oxy-acid salt photocatalyst with high photocatalytic activity on degradation of dye. Environ. Sci. Technol. 44, 5570–5574 (2010).
Pan, C. et al. Efficient and stable H2O2 production from H2O and O2 on BiPO4 photocatalyst. Appl. Catal. B Environ. 316, 121675 (2022).
Sellers, R. Spectrophotometric determination of hydrogen peroxide using potassium titanium(IV) oxalate. Analyst 105, 950–954 (1990).
Liu, X. et al. Facile synthesis of biocompatible cysteine-coated CuS nanoparticles with high photothermal conversion efficiency for cancer therapy. Dalton. Trans. 43, 11709–11715 (2014).
Barros, M. C. F. et al. Diffusion of levodopa in aqueous solutions of hydrochloric acid at 25 °C. Chem. Thermodyn. 72, 44–47 (2014).
Wilke, C. & Chang, P. Correlation of diffusion coefficients in dilute solutions. AIChE Journal 1, 264 (1955).
Hines, A. & Maddo, R. Mass Transfer: Fundamentals and Applications (Prentice-Hall, 1985).
Johnny, L. Development of a model to determine mass transfer coefficient and oxygen solubility in bioreactors. Heliyon. 3, e00248 (2017).
Hasan, M. et al. The influence of gas–liquid interfacial transport theory on numerical modelling of plasma activation of water. Plasma. Chem. Plasma. Process. 41, 1363 (2021).
Riet, K. Review of measuring methods and results in nonviscous gas-liquid mass transfer in stirred Vessels. Ind. Eng. Chem. Process Des. Dev. 18, 3 (1979).
Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 113, 7756–7764 (2000).
Acknowledgements
C. Pan acknowledged the National Natural Science Foundation of China (22476071, 22172065), the Natural Science Foundation of Hebei Province (E2024210028), and the Natural Science Foundation of Jiangsu Province (BK20201345).
Author information
Authors and Affiliations
Contributions
S.D. conducted the experiments and performance evaluation. Y.N.Z. performed DFT calculations and analysis of the TAS data. J.X. and Y.Z. performed data analysis. J.Z., Y.D., and Y.L. participated in the paper discussions and interpretation of the results. Y.F.Z. supervised the project. C.P. wrote the manuscript with critical revision from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interest.
Peer review
Peer review information
Nature Communications thanks Minsu Gu, Qingrui Zhang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dou, S., Zhang, Y., Xu, J. et al. Persistent semiquinone radicals enable efficient near-infrared-driven H2O2 photosynthesis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70130-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70130-x