Abstract
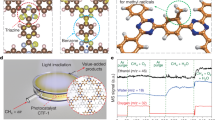
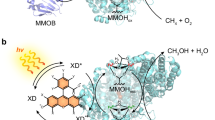
Direct catalytic conversion of methane to methanol offers a pathway for transforming a potent greenhouse gas into a portable clean liquid fuel, thereby mitigating carbon emissions and supporting sustainable energy. However, this process faces challenges from thermodynamically favorable methanol overoxidation. Here, we show that spatiotemporal regulation of photogenerated charge carriers on engineered catalytic sites enables a bio-inspired ordered two-step photocatalytic process that imitates methane monooxygenase. In a platinum-loaded cadmium sulfide photocatalyst, unsaturated sulfur sites modulate hole migration while platinum sites modulate electron migration, ensuring their concurrent surface arrival within picoseconds and prolonged localization. This dynamics temporarily anchors methane at hole-enriched sulfur sites while hydroxyl radical generation occurs at electron-rich platinum sites, decoupling hydroxyl radical formation from methane dehydrogenation to suppress overoxidation. The approach achieves methane-to-methanol conversion with selectivity of 83.5%, offering a bio-inspired solar-driven strategy for C1 valorization.
Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information files. Source data are provided as a source data file with this paper and are available from the corresponding authors upon request. Source data are provided with this paper.
References
Schwach, P. et al. Direct conversion of methane to value-added chemicals over heterogeneous catalysts: challenges and prospects. Chem. Rev. 117, 8497–8520 (2017).
Han, C. et al. Selective cleavage of chemical bonds in targeted intermediates for highly selective photooxidation of methane to methanol. J. Am. Chem. Soc. 145, 8609–8620 (2023).
Upham, D. C. et al. Catalytic molten metals for the direct conversion of methane to hydrogen and separable carbon. Science 358, 917–921 (2017).
Cao, Y. et al. Methane photooxidation with nearly 100 % selectivity towards oxygenates: proton rebound ensures the regeneration of methanol. Angew. Chem. Int. Ed. 62, e202302196 (2023).
Jiménez, J. D. et al. From methane to methanol: Pd-iC-CeO2 catalysts engineered for high selectivity via mechanochemical synthesis. J. Am. Chem. Soc. 146, 25986–25999 (2024).
Xie, J. et al. Methane oxidation to ethanol by a molecular junction photocatalyst. Nature 639, 368–374 (2025).
Song, X. et al. Methane bubbled through seawater can be converted to methanol with high efficiency. Adv. Sci. 12, 2412246 (2025).
Fan, Y. et al. Selective photocatalytic oxidation of methane by quantum-sized bismuth vanadate. Nat. Sustain. 4, 509–515 (2021).
Mao, J. et al. Direct conversion of methane with O2 at room temperature over edge-rich MoS2. Nat. Catal. 6, 1052–1061 (2023).
Snyder, B. E. R. et al. The active site of low-temperature methane hydroxylation in iron-containing zeolites. Nature 536, 317–321 (2016).
Grundner, S. et al. Single-site trinuclear copper oxygen clusters in mordenite for selective conversion of methane to methanol. Nat. Commun. 6, 7546 (2015).
Ravi, M. et al. Misconceptions and challenges in methane-to-methanol over transition-metal-exchanged zeolites. Nat. Catal. 2, 485–494 (2019).
Groothaert, M. H. et al. Selective oxidation of methane by the bis(μ-oxo)dicopper core stabilized on ZSM-5 and mordenite zeolites. J. Am. Chem. Soc. 127, 1394–1395 (2005).
Sushkevich, V. L. et al. Selective anaerobic oxidation of methane enables direct synthesis of methanol. Science 356, 523–527 (2017).
Tomkins, P. et al. Isothermal cyclic conversion of methane into methanol over copper-exchanged zeolite at low temperature. Angew. Chem. Int. Ed. 55, 5467–5471 (2016).
Feng, C. et al. Optimizing the reaction pathway of methane photo-oxidation over single copper sites. Nat. Commun. 15, 9088 (2024).
Jiang, Y. et al. Enabling specific photocatalytic methane oxidation by controlling free radical type. J. Am. Chem. Soc. 145, 2698–2707 (2023).
Zhou, Q. et al. Selective photocatalytic oxidation of methane to methanol by constructing a rapid O2 conversion pathway over Au-Pd/ZnO. ACS Catal. 14, 955–964 (2024).
Zhang, C. et al. Design principles for maximizing hole utilization of semiconductor quantum wires toward efficient photocatalysis. Angew. Chem. Int. Ed. 62, e202305571 (2023).
Li, Y. et al. Revealing the importance of hole transfer: boosting photocatalytic hydrogen evolution by delicate modulation of photogenerated holes. ACS Catal. 13, 8281–8292 (2023).
Liu, Y. et al. Inorganic ligands-mediated hole attraction and surface structural reorganization in InP/ZnS QD photocatalysts studied via ultrafast visible and midinfrared spectroscopies. Sci. China Mater. 65, 2529–2539 (2022).
Dan, M. et al. Balancing the charge separation and surface reaction dynamics in twin-interface photocatalysts for solar-to-hydrogen production. Adv. Mater. 37, 2415138 (2024).
Tang, Y. et al. Surface unsaturated sulfur modulates Pt sub-nanoparticles on tandem homojunction CdS for efficient electron extraction. Adv. Energy Mater. 13, 2203827 (2023).
Zhai, G. et al. Direct photocatalytic oxidation of methane to formic acid with high selectivity via a concerted proton-electron transfer process. J. Am. Chem. Soc. 147, 2444–2454 (2025).
Vrubel, H. et al. Hydrogen evolution catalyzed by MoS3 and MoS2 particles. Energy Environ. Sci. 5, 6136–6144 (2012).
Liu, P. et al. Electronic interactions on platinum/(metal-oxide)-based photocatalysts boost selective photoreduction of CO2 to CH4. Angew. Chem. Int. Ed. 62, e202309443 (2023).
Naskar, S. et al. Synthesis of ternary and quaternary Au and Pt decorated CdSe/CdS heteronanoplatelets with controllable morphology. Adv. Funct. Mater. 27, 1604685 (2017).
Zhu, Z. et al. Oriented crystal polarization tuning bulk charge and single-site chemical state for exceptional hydrogen photo-production. Adv. Mater. 36, 2411339 (2024).
You, Y. et al. Distinct selectivity control in solar-driven bio-based α-hydroxyl acid conversion: A comparison of Pt nanoparticles and atomically dispersed Pt on CdS. Angew. Chem. Int. Ed. 62, e202306452 (2023).
Qiao, B. et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 3, 634–641 (2011).
Zhang, Z. et al. Thermally stable single atom Pt/m-Al2O3 for selective hydrogenation and CO oxidation. Nat. Commun. 8, 16100 (2017).
Zhang, X. et al. Crystal-phase-mediated restructuring of Pt on TiO2 with tunable reactivity: redispersion versus reshaping. ACS Catal. 12, 3634–3643 (2022).
Guo, C. et al. The zinc vacancy induced CdS/ZnS Z-scheme structure as a highly stable photocatalyst for hydrogen production. J. Alloy. Compd. 888, 161620 (2021).
Torres-Castanedo, C. G. et al. Optical properties of CdS nanocrystalline thin films in the abrupt phase transition from zinc blende to wurtzite. J. Mater. Sci. Mater. Electron. 31, 16561–16568 (2020).
Vamvasakis, I. et al. Mesoporous dual-semiconductor ZnS/CdS nanocomposites as efficient visible light photocatalysts for hydrogen generation. Nanomaterials 13, 2426 (2023).
Wu, K. et al. Ultrafast charge separation and long-lived charge separated state in photocatalytic CdS-Pt nanorod heterostructures. J. Am. Chem. Soc. 134, 10337–10340 (2012).
Wang, J. et al. Charge transfer from n-doped nanocrystals: mimicking intermediate events in multielectron photocatalysis. J. Am. Chem. Soc. 140, 7791–7794 (2018).
van Stokkum, I. H. M. et al. Global and target analysis of time-resolved spectra. Biochim. Biophys. Acta - Bioenerg. 1657, 82–104 (2004).
Ghosh, S. et al. Introducing Ag dopants into CdSe nanoplatelets (NPLs) leads to effective charge separation for better photodetector performance. Chem. Asian J. 19, e202400528 (2024).
Xu, Y. et al. Efficient methane oxidation to formaldehyde via photon-phonon cascade catalysis. Nat. Sustain. 7, 1171–1181 (2024).
Qureshi, M. & Takanabe, K. Insights on measuring and reporting heterogeneous photocatalysis: efficiency definitions and setup examples. Chem. Mater. 29, 158–167 (2017).
Jiang, Y. et al. Best practices for experiments and reports in photocatalytic methane conversion. Angew. Chem. Int. Ed. 63, e202404658 (2024).
Cao, S. & Piao, L. Considerations for a more accurate evaluation method for photocatalytic water splitting. Angew. Chem. Int. Ed. 59, 18312–18320 (2020).
Melchionna, M. & Fornasiero, P. Updates on the roadmap for photocatalysis. ACS Catal. 10, 5493–5501 (2020).
Chen, X. et al. Photocatalytic oxidation of methane over silver decorated zinc oxide nanocatalysts. Nat. Commun. 7, 12273 (2016).
Xu, Y. et al. Regulating Au coverage for the direct oxidation of methane to methanol. Nat. Commun. 15, 564 (2024).
Socrates, G., Infrared and Raman characteristic group frequencies: tables and charts. 2004.
Bahou, M. & Lee, Y.-P. Diminished cage effect in solid p-H2: Infrared absorption of CH3S observed from photolysis in situ of CH3SH, CH3SCH3, or CH3SSCH3 isolated in p-H2 matrices. J. Chem. Phys. 133, 164316 (2010).
Chen, Y.-Q. et al. An efficient Au/ZnO catalyst for the photocatalytic conversion of methane to formaldehyde. Chem. Eng. J. 498, 155792 (2024).
Chen, T. et al. Mechanistic studies of photocatalytic reaction of methanol for hydrogen production on Pt/TiO2 by in situ fourier transform IR and time-resolved IR spectroscopy. J. Phys. Chem. C. 111, 8005–8014 (2007).
Cao, Y. et al. Modulating electron density of vacancy site by single Au atom for effective CO2 photoreduction. Nat. Commun. 12, 1675 (2021).
Jiang, W. et al. Pd-modified ZnO-Au enabling alkoxy intermediates formation and dehydrogenation for photocatalytic conversion of methane to ethylene. J. Am. Chem. Soc. 143, 269–278 (2021).
Muoz, M. et al. Continuous Cauchy wavelet transform of XAFS spectra. Phys. Scr. 2005, 221 (2005).
Muñoz, M. et al. Continuous Cauchy wavelet transform analyses of EXAFS spectra: A qualitative approach. Am. Mineral. 88, 694–700 (2003).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron. Radiat. 12, 537–541 (2005).
Song, H. et al. Direct and selective photocatalytic oxidation of CH4 to oxygenates with O2 on cocatalysts/ZnO at room temperature in water. J. Am. Chem. Soc. 141, 20507–20515 (2019).
Acknowledgements
This work was supported by the National Science Fund for Distinguished Young Scholars (Grant No. 52325401, Y.Z.), the National Natural Science Foundation of China (Grant No. 22209135, Y.C.), the National Key R&D Project of China (Grant No. 2020YFA0710000, Y.Z.) and Key Research and Development Program of Sichuan Province (No. 2023ZDZX0005, Y.C.). We thank staff at the BL13SSW beamline of the Shanghai Synchrotron Radiation Facilities (SSRF) for assistance with the XAFS. This work was also supported by the User Experiment Assist System of SSRF. The group of Nano-Catalysis Group at Sum Yat-sen University and Shanghai Synchrotron Radiation Facility was thanked for sharing XAFS of standard Pt foil and PtO2. We would like to thank Analysis and Testing Center, Southwest Petroleum University for the support of EPR test.
Author information
Authors and Affiliations
Contributions
Y.Z. conceived and designed the study. Y.L. and C.H. conducted photocatalytic CH4 oxidation evaluation experiments. Y.L. and Y.C. guided most of characterizations and data analysis. Y.L., K.Z. and J.M. carried out the excited-state dynamics measurements and finished the data analysis. S.C carried out in-situ XPS characterizations and Y.L. finished the data analysis. Y.L. and Y.C. organized the manuscripts and co-wrote the paper. Y.Z. supervised the project and co-wrote the paper. The manuscript has been reviewed by all authors and the results are presented after the discussions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Lingyu Piao, Yong Xu, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Cao, Y., Han, C. et al. Spatiotemporal photon distribution control on active sites enables bio-inspired methane-to-methanol conversion. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70134-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70134-7