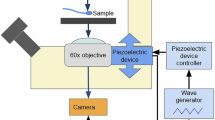
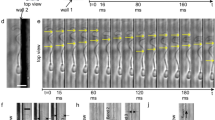
Abstract
During migration through the female reproductive tract, sperm undergo physiological changes known as capacitation, including a motility transition termed hyperactivation. Hyperactivation is essential for various aspects of fertilization, particularly effective migration within the tract. However, how hyperactivation facilitates this migration remains elusive. Here, we profiled bull sperm hyperactivation and swimming in Newtonian and complex fluids, using microfluidic surfaces to mimic confinement of the tract. We identified three swim gaits: wandering (persistent random walks), circling, and an intriguing circling-and-wandering mode marked by stochastic transitions between the two. All gaits exhibit diffusive behavior over long time scales, with wandering showing a tenfold higher diffusivity than circling, and the effective diffusivity of circling-and-wandering falling in between. We found that while wandering sperm scatter from convex and concave surfaces, circling sperm become trapped around pillars, highlighting a distinctive feature of each phase. Additionally, stochastic simulations of active transport in porous media showed that as the geometrical complexity of the environment increases, circling-and-wandering outperforms either motility alone in spreading through the media. Our findings suggest that wandering may broaden the search landscape, while circling could help maintain local focus. Therefore, the combined circling-and-wandering swimming behavior might provide a flexible mechanism for modulating motility and facilitating migration in complex environments. Our results may have implications for understanding the physical aspects of sperm migration in the female reproductive tract.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are available in the article, its Supplementary Information, and the Source Data. Source data are provided with this paper.
Code availability
The codes used in this study are publicly available at https://doi.org/10.5281/zenodo.18495070.
References
Miki, K. & Clapham, D. E. Rheotaxis guides mammalian sperm. Curr. Biol. 23, 443–452 (2013).
Kantsler, V., Dunkel, J., Blayney, M. & Goldstein, R. E. Rheotaxis facilitates upstream navigation of mammalian sperm cells. Elife 3, e02403 (2014).
Tung, C.-K. et al. Microgrooves and fluid flows provide preferential passageways for sperm over pathogen tritrichomonas foetus. Proc. Natl. Acad. Sci. USA 112, 5431–5436 (2015).
Denissenko, P., Kantsler, V., Smith, D. J. & Kirkman-Brown, J. Human spermatozoa migration in microchannels reveals boundary-following navigation. Proc. Natl. Acad. Sci. USA 109, 8007–8010 (2012).
Raveshi, M. R. et al. Curvature in the reproductive tract alters sperm–surface interactions. Nat. Commun. 12, 3446 (2021).
Zaferani, M., Palermo, G. D. & Abbaspourrad, A. Strictures of a microchannel impose fierce competition to select for highly motile sperm. Sci. Adv. 5, eaav2111 (2019).
Nosrati, R., Driouchi, A., Yip, C. M. & Sinton, D. Two-dimensional slither swimming of sperm within a micrometre of a surface. Nat. Commun. 6, 8703 (2015).
Friedrich, B. M., Riedel-Kruse, I. H., Howard, J. & Jülicher, F. High-precision tracking of sperm swimming fine structure provides strong test of resistive force theory. J. Exp. Biol. 213, 1226–1234 (2010).
Chen, Z. et al. In situ cryo-electron tomography reveals the asymmetric architecture of mammalian sperm axonemes. Nat. Struct. Mol. Biol. 30, 360–369 (2023).
Gadadhar, S. et al. Tubulin glycylation controls axonemal dynein activity, flagellar beat, and male fertility. Science 371, eabd4914 (2021).
Keller, J. & Rubinow, S. Swimming of flagellated microorganisms. Biophys. J. 16, 151–170 (1976).
Ishijima, S., Hamaguchi, M. S., Naruse, M., Ishijima, S. A. & Hamaguchi, Y. Rotational movement of a spermatozoon around its long axis. J. Exp. Biol. 163, 15–31 (1992).
Elgeti, J., Kaupp, U. B. & Gompper, G. Hydrodynamics of sperm cells near surfaces. Biophys. J. 99, 1018–1026 (2010).
Woolley, D. Motility of spermatozoa at surfaces. Reproduction 126, 259–270 (2003).
Zaferani, M., Javi, F., Mokhtare, A., Li, P. & Abbaspourrad, A. Rolling controls sperm navigation in response to the dynamic rheological properties of the environment. Elife 10, e68693 (2021).
Babcock, D. F., Wandernoth, P. M. & Wennemuth, G. Episodic rolling and transient attachments create diversity in sperm swimming behavior. BMC Biol. 12, 1–12 (2014).
Publicover, S., Harper, C. V. & Barratt, C. [ca2+] i signalling in sperm-making the most of what you’ve got. Nat. Cell Biol. 9, 235–242 (2007).
Eisenbach, M. & Giojalas, L. C. Sperm guidance in mammals-an unpaved road to the egg. Nat. Rev. Mol. Cell Biol. 7, 276–285 (2006).
Saggiorato, G. et al. Human sperm steer with second harmonics of the flagellar beat. Nat. Commun. 8, 1415 (2017).
Carlson, A. E. et al. Catsper1 required for evoked ca2+ entry and control of flagellar function in sperm. Proc. Natl. Acad. Sci. USA 100, 14864–14868 (2003).
Gagnon, C. & Lamirande, E. D. Controls of Sperm Motility 108–133 (Cambridge University Press, 2006).
Ho, H.-C. & Suarez, S. S. Hyperactivation of mammalian spermatozoa: function and regulation. Reproduction 122, 519–526 (2001).
Suarez, S. S. Control of hyperactivation in sperm. Hum. Reprod. Update 14, 647–657 (2008).
Kirichok, Y., Navarro, B. & Clapham, D. E. Whole-cell patch-clamp measurements of spermatozoa reveal an alkaline-activated ca2+ channel. Nature 439, 737–740 (2006).
Ren, D. et al. A sperm ion channel required for sperm motility and male fertility. Nature 413, 603–609 (2001).
Chung, J.-J. et al. Structurally distinct ca2+ signaling domains of sperm flagella orchestrate tyrosine phosphorylation and motility. Cell 157, 808–822 (2014).
Lin, S., Ke, M., Zhang, Y., Yan, Z. & Wu, J. Structure of a mammalian sperm cation channel complex. Nature 595, 746–750 (2021).
Zhao, Y. et al. 3d structure and in situ arrangements of catsper channel in the sperm flagellum. Nat. Commun. 13, 3439 (2022).
Wang, H., McGoldrick, L. L. & Chung, J.-J. Sperm ion channels and transporters in male fertility and infertility. Nat. Rev. Urol. 18, 46–66 (2021).
Cai, X., Wang, X. & Clapham, D. E. Early evolution of the eukaryotic ca2+ signaling machinery: conservation of the catsper channel complex. Mol. Biol. Evol. 31, 2735–2740 (2014).
Qi, H. et al. All four catsper ion channel proteins are required for male fertility and sperm cell hyperactivated motility. Proc. Natl. Acad. Sci. USA 104, 1219–1223 (2007).
Quill, T. A. et al. Hyperactivated sperm motility driven by catsper2 is required for fertilization. Proc. Natl. Acad. Sci. USA 100, 14869–14874 (2003).
Chang, H. & Suarez, S. S. Rethinking the relationship between hyperactivation and chemotaxis in mammalian sperm. Biol. Reprod. 83, 507–513 (2010).
Armon, L. & Eisenbach, M. Behavioral mechanism during human sperm chemotaxis: involvement of hyperactivation. PLoS ONE 6, e28359 (2011).
Bahat, A. et al. Thermotaxis of mammalian sperm cells: a potential navigation mechanism in the female genital tract. Nat. Med. 9, 149–150 (2003).
Bahat, A. & Eisenbach, M. Sperm thermotaxis. Mol. Cell. Endocrinol. 252, 115–119 (2006).
Suarez, S. S. & Pacey, A. Sperm transport in the female reproductive tract. Hum. Reprod. Update 12, 23–37 (2006).
Clapham, D. E. & Hulse, R. E. Sperm catsper ion channel swims into sharper focus. (2021).
Demott, R. P. & Suarez, S. S. Hyperactivated sperm progress in the mouse oviduct. Biol. Reprod. 46, 779–785 (1992).
Suarez, S. S. & Dai, X. Hyperactivation enhances mouse sperm capacity for penetrating viscoelastic media. Biol. Reprod. 46, 686–691 (1992).
Zaferani, M., Suarez, S. S. & Abbaspourrad, A. Mammalian sperm hyperactivation regulates navigation via physical boundaries and promotes pseudo-chemotaxis. Proc. Natl. Acad. Sci. USA 118, e2107500118 (2021).
Ho, K., Wolff, C. A. & Suarez, S. S. Catsper-null mutant spermatozoa are unable to ascend beyond the oviductal reservoir. Reprod. Fertil. Dev. 21, 345–350 (2009).
Ardon, F. et al. Dynamics of bovine sperm interaction with epithelium differ between oviductal isthmus and ampulla. Biol. Reprod. 95, 90–1 (2016).
Friedrich, B. M. & Jülicher, F. Chemotaxis of sperm cells. Proc. Natl. Acad. Sci. USA 104, 13256–13261 (2007).
Friedrich, B. M. & Jülicher, F. Steering chiral swimmers along noisy helical paths. Phys. Rev. Lett. 103, 068102 (2009).
Alvarez, L. et al. The rate of change in ca2+ concentration controls sperm chemotaxis. J. Cell Biol. 196, 653–663 (2012).
Jikeli, J. F. et al. Sperm navigation along helical paths in 3d chemoattractant landscapes. Nat. Commun. 6, 7985 (2015).
Suarez, S. S. Regulation of sperm storage and movement in the mammalian oviduct. Int. J. Dev. Biol. 52, 455–462 (2008).
Barakat, I. A., Danfour, M. A., Galewan, F. A. & Dkhil, M. A. Effect of various concentrations of caffeine, pentoxifylline, and kallikrein on hyperactivation of frozen bovine semen. Biomed. Res. Int. 2015, 948575 (2015).
Colás, C., Cebrián-Pérez, J. & Muiño-Blanco, T. Caffeine induces ram sperm hyperactivation independent of camp-dependent protein kinase. Int. J. Androl. 33, e187–e197 (2010).
Tung, C.-K. et al. Fluid viscoelasticity promotes collective swimming of sperm. Sci. Rep. 7, 3152 (2017).
Ren, X. & Bloomfield-Gadêlha, H. Swimming by spinning: spinning-top type rotations regularize sperm swimming into persistently progressive paths in 3d. Adv. Sci. 12, 2406143 (2024).
Zaferani, M. & Abbaspourrad, A. Biphasic chemokinesis of mammalian sperm. Phys. Rev. Lett. 130, 248401 (2023).
van Teeffelen, S. & Löwen, H. Dynamics of a brownian circle swimmer. Phys. Rev. E 78, 020101 (2008).
Kurzthaler, C. & Franosch, T. Intermediate scattering function of an anisotropic brownian circle swimmer. Soft Matter 13, 6396–6406 (2017).
Kurzthaler, C. et al. Characterization and control of the run-and-tumble dynamics of Escherichia Coli. Phys. Rev. Lett. 132, 038302 (2024).
Zhao, Y. et al. Quantitative characterization of run-and-tumble statistics in bulk bacterial suspensions. Phys. Rev. E 109, 014612 (2024).
Solon, A. P. et al. Pressure is not a state function for generic active fluids. Nat. Phys. 11, 673–678 (2015).
Granek, O. et al. Colloquium: Inclusions, boundaries, and disorder in scalar active matter. Rev. Mod. Phys. 96, 031003 (2024).
Torquato, S. et al. Random Heterogeneous Materials: Microstructure and Macroscopic Properties, Vol. 16 (Springer, 2002).
Bertrand, T., Zhao, Y., Bénichou, O., Tailleur, J. & Voituriez, R. Optimized diffusion of run-and-tumble particles in crowded environments. Phys. Rev. Lett. 120, 198103 (2018).
Kurzthaler, C. et al. A geometric criterion for the optimal spreading of active polymers in porous media. Nat. Commun. 12, 7088 (2021).
Bénichou, O., Loverdo, C., Moreau, M. & Voituriez, R. Intermittent search strategies. Rev. Mod. Phys. 83, 81–129 (2011).
Perez Ipiña, E., Otte, S., Pontier-Bres, R., Czerucka, D. & Peruani, F. Bacteria display optimal transport near surfaces. Nat. Phys. 15, 610–615 (2019).
Kurzthaler, C. et al. Probing the spatiotemporal dynamics of catalytic janus particles with single-particle tracking and differential dynamic microscopy. Phys. Rev. Lett. 121, 078001 (2018).
Lauga, E., DiLuzio, W. R., Whitesides, G. M. & Stone, H. A. Swimming in circles: motion of bacteria near solid boundaries. Biophys. J. 90, 400–412 (2006).
Takagi, D. Dispersion of self-propelled rods undergoing fluctuation-driven flips. Phys. Rev. Lett. 110, 038301 (2013).
Kümmel, F. et al. Circular motion of asymmetric self-propelling particles. Phys. Rev. Lett. 110, 198302 (2013).
Narinder, N., Bechinger, C. & Gomez-Solano, J. R. Memory-induced transition from a persistent random walk to circular motion for achiral microswimmers. Phys. Rev. Lett. 121, 078003 (2018).
Takagi, D., Palacci, J., Braunschweig, A. B., Shelley, M. J. & Zhang, J. Hydrodynamic capture of microswimmers into sphere-bound orbits. Soft Matter 10, 1784 (2014).
Spagnolie, S. E., Moreno-Flores, G. R., Bartolo, D. & Lauga, E. Geometric capture and escape of a microswimmer colliding with an obstacle. Soft Matter 11, 3396–3411 (2015).
Berg, H. C. & Brown, D. A. Chemotaxis in Escherichia coli analysed by three-dimensional tracking. Nature 239, 500–504 (1972).
Gaffney, E. A., Ishimoto, K. & Walker, B. J. Modelling motility: the mathematics of spermatozoa. Front. Cell Dev Biol. 9, 710825 (2021).
Swain, D. K., Vergara, C., Castro-Arnau, J. & Lishko, P. V. The essential calcium channel of sperm catsper is temperature-gated. Nat. Commun. 16, 3657 (2025).
Leung, E. T. et al. Simulating nature in sperm selection for assisted reproduction. Nat. Rev. Urol. 19, 16–36 (2022).
Zaferani, M., Cheong, S. H. & Abbaspourrad, A. Rheotaxis-based separation of sperm with progressive motility using a microfluidic corral system. Proc. Natl. Acad. Sci. USA 115, 8272–8277 (2018).
Nosrati, R. Lab on a chip devices for fertility: from proof-of-concept to clinical impact. Lab Chip 22, 1680–1689 (2022).
Cates, M. E. & Tailleur, J. When are active brownian particles and run-and-tumble particles equivalent? Consequences for motility-induced phase separation. Europhys. Lett. 101, 20010 (2013).
Feller, W. An Introduction to Probability Theory and its Applications: Volume II (John Wiley & Sons, 1971).
Angelani, L. Averaged run-and-tumble walks. Europhys. Lett. 102, 20004 (2013).
Zeitz, M., Wolff, K. & Stark, H. Active brownian particles moving in a random lorentz gas. Eur. Phys. J. E 40, 23 (2017).
Martínez, L. Celllistmap.jl: efficient and customizable cell list implementation for calculation of pairwise particle properties within a cutoff. Comput. Phys. Commun. 279, 108452 (2022).
Acknowledgements
M.Z. thanks Ned S. Wingreen for insightful discussions on sperm circling and wandering dynamics. C.K. acknowledges helpful discussions with S. Mandal and P. Das. M.Z. acknowledges support from the Omenn–Darling Postdoctoral Fellowship, which supported this study. We also acknowledge support from NSF Grant No. DMS/NIGMS 2245850 (to H.A.S. and S.P.).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
M.Z., C.K., and H.A.S. conceived the project. M.Z., Y.L.A., and S.H.C. performed a portion of the experiments, and M.Z. and S.P. carried out the remaining experiments. Y.B., A.P., and C.K. performed the analytical calculations and simulations. M.Z., C.K., and H.A.S. wrote the manuscript, and all authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Anton Bukatin, Daisuke Takagi, and the other, anonymous, reviewer for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zaferani, M., Baouche, Y., Lago-Alvarez, Y. et al. Sperm hyperactivation drives a circling-and-wandering swimming behavior. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70143-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70143-6