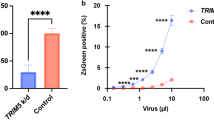
Abstract
Current animal models of HIV-1 infection are either immunocompromised or rely on proxy viruses instead of HIV-1. Here, we establish an immunocompetent animal model for CXCR4-tropic HIV-1 in owl monkeys (Aotus nancymaae). Through analysis of 191 owl monkeys, genetic characterization and functional testing demonstrate that CD4 and Tetherin in this species support HIV-1 replication. Although owl monkeys do carry restrictive TRIMCyp and APOBEC3G alleles, small changes to the HIV-1 genome allow the virus to overcome these barriers. The resulting virus remains 93% wildtype HIV-1 in sequence. Fully immunocompetent owl monkeys can be infected with this virus, recapitulating key aspects of HIV-1 infection in humans: an initial surge of virus replication, subsequent establishment of a durable set point viremia, seroconversion, and the formation of a viral reservoir. The owl monkey model broadens the experimental options for HIV-1 research, and future studies will explore its utility for CCR5-tropic virus strains.
Similar content being viewed by others
Data availability
All data are available within this manuscript or its supporting files. Sequencing data generated in this study have been deposited in the GenBank database under accession numbers PX876883-PX877642, PX877926-PX878685, PX911634-PX912013, PX925169-PX925171. Source data are provided with this paper.
References
Bibollet-Ruche, F. et al. CD4 receptor diversity in chimpanzees protects against SIV infection. Proc. Natl. Acad. Sci. USA. 116, 3229–3238 (2019).
Warren, C. J. et al. A glycan shield on chimpanzee CD4 protects against infection by primate lentiviruses (HIV/SIV). Proc. Natl. Acad. Sci. Usa. 116, 11460–11469 (2019).
Hatziioannou, T. & Evans, D. T. Animal models for HIV/AIDS research. Nat. Rev. Microbiol. 10, 852–867 (2012).
Agarwal, Y. et al. Moving beyond the mousetrap: current and emerging humanized mouse and rat models for investigating prevention and cure strategies against HIV infection and associated pathologies. Retrovirology 17, 8 (2020).
Sharma, M., Nag, M. & DelPrete, G. Q. Minimally modified HIV-1 infection of macaques: Development, utility, and limitations of current models. Viruses 16, 1618 (2024).
Hatziioannou, T. et al. HIV-1-induced AIDS in monkeys. Science 344, 1401–1405 (2014).
Jasinska, A. J., Apetrei, C. & Pandrea, I. Walk on the wild side: SIV infection in African non-human primate hosts—from the field to the laboratory. Front. Immunol. 13, 1060985 (2023).
Bauer, A. M. & Bar, K. J. Advances in simian - human immunodeficiency viruses for nonhuman primate studies of HIV prevention and cure. Curr. Opin. HIV AIDS 15, 275–281 (2020).
Kaur, A. & Vaccari, M. Exploring HIV vaccine progress in the pre-clinical and clinical setting: From history to future prospects. Viruses 16, 368 (2024).
Ambrose, Z., KewalRamani, V. N., Bieniasz, P. D. & Hatziioannou, T. HIV/AIDS: in search of an animal model. Trends Biotechnol. 25, 333–337 (2007).
Warren, C. J. & Sawyer, S. L. Identifying animal viruses in humans. Science 379, 982–983 (2023).
Sharp, P. M. & Hahn, B. H. Origins of HIV and the AIDS pandemic. CSH Perspect. Med 1, a006841 (2011).
Warren, C. J. & Sawyer, S. L. How host genetics dictates successful viral zoonosis. PLoS Biol. 17, e3000217 (2019).
Sauter, D. & Kirchhoff, F. Key viral adaptations preceding the AIDS pandemic. Cell Host Microbe 25, 27–38 (2019).
Dalgleish, A. G. et al. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature 312, 763–767 (1984).
Stremlau, M. et al. The cytoplasmic body component TRIM5α restricts HIV-1 infection in Old World monkeys. Nature 427, 848–853 (2004).
Sheehy, A. M., Gaddis, N. C., Choi, J. D. & Malim, M. H. Isolation of a human gene that inhibits HIV-1 infection and is suppressed by the viral Vif protein. Nature 418, 646–650 (2002).
Neil, S. J. D., Zang, T. & Bieniasz, P. D. Tetherin inhibits retrovirus release and is antagonized by HIV-1 Vpu. Nature 451, 425–430 (2008).
VanDamme, N. et al. The interferon-induced protein BST-2 restricts HIV-1 release and is downregulated from the cell surface by the viral Vpu protein. Cell Host Microbe 3, 245–252 (2008).
Sawyer, S. L., Wu, L. I., Emerman, M. & Malik, H. S. Positive selection of primate TRIM5alpha identifies a critical species-specific retroviral restriction domain. Proc. Natl. Acad. Sci. USA. 102, 2832–2837 (2005).
Sawyer, S. L., Emerman, M. & Malik, H. S. Ancient adaptive evolution of the primate antiviral DNA-editing enzyme APOBEC3G. PLoS Biol. 2, e275 (2004).
McNatt, M. W. et al. Species-specific activity of HIV-1 Vpu and positive selection of Tetherin transmembrane domain variants. PLoS Pathog. 5, e1000300 (2009).
Gupta, R. K. et al. Mutation of a single residue renders human Tetherin resistant to HIV-1 Vpu-mediated depletion. PLoS Pathog. 5, e1000443 (2009).
Sawyer, S. L., Wu, L. I., Akey, J. M., Emerman, M. & Malik, H. S. High-frequency persistence of an impaired allele of the retroviral defense gene TRIM5α in humans. Curr. Biol. 16, 95–100 (2006).
Warren, C. J. et al. Adaptation of CD4 in gorillas and chimpanzees conveyed resistance to simian immunodeficiency viruses. eLife 13, RP93316 (2025).
Warren, C. J. et al. Selective use of primate CD4 receptors by HIV-1. PLoS Biol. 17, e3000304 (2019).
Russell, R. M. et al. CD4 receptor diversity represents an ancient protection mechanism against primate lentiviruses. Proc. Natl. Acad. Sci. USA. 118, e2025914118 (2021).
Meyerson, N. R. & Sawyer, S. L. Two-stepping through time: mammals and viruses. Trends Microbiol 19, 286–294 (2011).
Johnson, W. E. & Sawyer, S. L. Molecular evolution of the antiretroviral TRIM5 gene. Immunogenetics 61, 163–176 (2009).
Ohainle, M. & Malik, H. S. A balancing act between primate lentiviruses and their receptor. Proc. Natl. Acad. Sci. Usa. 118, e2104741118 (2021).
Nisole, S., Lynch, C., Stoye, J. P. & Yap, M. W. A Trim5-cyclophilin A fusion protein found in owl monkey kidney cells can restrict HIV-1. Proc. Natl. Acad. Sci. Usa. 101, 13324–13328 (2004).
Sayah, D. M., Sokolskaja, E., Berthoux, L. & Luban, J. Cyclophilin A retrotransposition into TRIM5 explains owl monkey resistance to HIV-1. Nature 430, 569–573 (2004).
Choe, H. et al. The beta-chemokine receptors CCR3 and CCR5 facilitate infection by primary HIV-1 isolates. Cell 85, 1135–1148 (1996).
Feng, Y., Broder, C. C., Kennedy, P. E. & Berger, E. A. HIV-1 entry cofactor: Functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science 272, 872–877 (1996).
Meyerson, N. R. et al. Positive selection of primate genes that promote HIV-1 replication. Virology 291–298 https://doi.org/10.1016/j.virol.2014.02.029 (2014).
Zhang, Y. W., Ryder, O. A. & Zhang, Y. P. Sequence evolution of the CCR5 chemokine receptor gene in primates. Mol. Biol. evolution 16, 1145–1154 (1999).
Meyerson, N. R., Sharma, A., Wilkerson, G. K., Overbaugh, J. & Sawyer, S. L. Identification of owl monkey CD4 receptors broadly compatible with early-stage HIV-1 isolates. J. Virol. 89, 8611–8622 (2015).
Humes, D., Emery, S., Laws, E. & Overbaugh, J. A species-specific amino acid difference in the macaque CD4 receptor restricts replication by global circulating HIV-1 variants representing viruses from recent infection. J. Virol. 86, 12472–12483 (2012).
Wickenhagen, Tol, A., van, S. & Munster, V. Molecular determinants of cross-species transmission in emerging viral infections. Microbiol. Mol. Biol. Rev. 88, e00001–23 (2024).
Parrish, C. R. & Kawaoka, Y. The origins of new pandemic viruses: The acquisition of new host ranges by canine parvovirus and influenza A viruses. Annu Rev. Microbiol 59, 553–586 (2005).
Warren, C. J. et al. Primate hemorrhagic fever-causing arteriviruses are poised for spillover to humans. Cell 185, 3980–3991.e18 (2022).
Wong, S. K. et al. A New World primate deficient in Tetherin-mediated restriction of human immunodeficiency virus type 1. J. Virol. 83, 8771–8780 (2009).
Nahabedian, J. et al. Owl monkey CCR5 reveals synergism between CD4 and CCR5 in HIV-1 entry. Virology 512, 180–186 (2017).
Adachi, A. et al. Production of acquired immunodeficiency syndrome-associated retrovirus in human and nonhuman cells transfected with an infectious molecular clone. J. Virol. 59, 284–291 (1986).
Barden, A. et al. Antiemetic doses of dexamethasone and their effects on immune cell populations and plasma mediators of inflammation resolution in healthy volunteers. Prost. Leukot. Essent. Fat. Acids 139, 31–39 (2018).
Robb, M. L. et al. Prospective study of acute HIV-1 infection in adults in East Africa and Thailand. N. Engl. J. Med 374, 2120–2130 (2016).
Bayón-Gil, Á, Martinez-Picado, J. & Puertas, M. C. Viremic non-progression in HIV/SIV infection: A tied game between virus and host. Cell Rep. Med. 6, 101921 (2025).
Guadalupe, M. et al. Severe CD4+ T-cell depletion in gut lymphoid tissue during primary human immunodeficiency virus type 1 infection and substantial delay in restoration following highly active antiretroviral therapy. J. Virol. 77, 11708–11717 (2003).
Mattapallil, J. J. et al. Massive infection and loss of memory CD4+ T cells in multiple tissues during acute SIV infection. Nature 434, 1093–1097 (2005).
Veazey, R. S. et al. Gastrointestinal tract as a major site of CD4+ T cell depletion and viral replication in SIV infection. Science 280, 427–431 (1998).
Vanhamel, J., Bruggemans, A. & Debyser, Z. Establishment of latent HIV-1 reservoirs: what do we really know?. J. Virus Erad. 5, 3–9 (2019).
Pantaleo, G. et al. Studies in subjects with long-term nonprogressive human immunodeficiency virus infection. N. Engl. J. Med. 332, 209–216 (1995).
Boritz, E. A. et al. Multiple origins of virus persistence during natural control of HIV infection. Cell 166, 1004–1015 (2016).
Baiyegunhi, O. O. et al. CD8 lymphocytes mitigate HIV-1 persistence in lymph node follicular helper T cells during hyperacute-treated infection. Nat. Commun. 13, 4041 (2022).
Lichterfeld, M., Gao, C. & Yu, X. G. An ordeal that does not heal: understanding barriers to a cure for HIV-1 infection. Trends Immunol. 43, 608–616 (2022).
Churchill, M. J., Deeks, S. G., Margolis, D. M., Siliciano, R. F. & Swanstrom, R. HIV reservoirs: What, where and how to target them. Nat. Rev. Microbiol. 14, 55–60 (2016).
Feinberg, M. B. & Moore, J. P. AIDS vaccine models: challenging challenge viruses. Nat. Med. 8, 207–210 (2002).
Shepherd, J. et al. Emergence and persistence of CXCR4-tropic HIV-1 in a population of men from the multicenter AIDS cohort study. J. Infect. Dis. 198, 1104–1112 (2008).
Apetrei, C., Pandrea, I. & Mellors, J. W. Nonhuman primate models for HIV cure research. PLoS Pathog. 8, e1002892 (2012).
Srinivasan, P. et al. A malaria vaccine protects aotus monkeys against virulent plasmodium falciparum infection. Vaccines 2, 14 (2017).
Fernandez-Duque, E. et al. Owl Monkeys: Biology, Adaptive Radiation, and Behavioral Ecology of the Only Nocturnal Primate in the Americas. (Springer, 2023).
Montoya, G. E., Vernot, J. & Patarroyo, M. E. Comparative analysis of CD45 proteins in primate context: owl monkeys vs humans. Tissue Antigens 64, 165–172 (2004).
Hatziioannou, T. et al. A macaque model of HIV-1 infection. Proc. Natl. Acad. Sci. Usa. 106, 4425–4429 (2009).
Schmidt, F. et al. Derivation of simian tropic HIV-1 infectious clone reveals virus adaptation to a new host. Proc. Natl. Acad. Sci. Usa. 116, 10504–10509 (2019).
Duquenne, C. et al. The two human CXCR4 isoforms display different HIV receptor activities: consequences for the emergence of X4 strains. J. Immunol. 193, 4188–4194 (2014).
Connell, B. J. et al. Immune activation correlates with and predicts CXCR4 co-receptor tropism switch in HIV-1 infection. Sci. Rep. 10, 15866 (2020).
Swanstrom, R., Graham, W. D. & Zhou, S. Sequencing the biology of entry: the retroviral env gene. Curr. Top. Microbiol Immunol. 407, 65–82 (2017).
Marx, P. A. & Chen, Z. The function of simian chemokine receptors in the replication of SIV. Semin. Immunol. 10, 215–23 (1998).
Sina, S. T., Ren, W. & Cheng-Mayer, C. Coreceptor use in nonhuman primate models of HIV infection. J. Transl. Med. 9, S7 (2011).
Dudek, A. M. et al. A simultaneous knockout knockin genome editing strategy in HSPCs potently inhibits CCR5- and CXCR4-tropic HIV-1 infection. Cell Stem Cell 31, 499–518.e6 (2024).
Li, H. et al. Envelope residue 375 substitutions in simian–human immunodeficiency viruses enhance CD4 binding and replication in rhesus macaques. Proc. Natl. Acad. Sci. Usa. 113, E3413–E3422 (2016).
DelPrete, G. Q. et al. A single gp120 residue can affect HIV-1 tropism in macaques. PLoS Pathog. 13, e1006572 (2017).
Humes, D. & Overbaugh, J. Adaptation of subtype a human immunodeficiency virus type 1 envelope to pig-tailed macaque cells. J. Virol. 85, 4409–4420 (2011).
Pollom, E. et al. Comparison of SIV and HIV-1 genomic RNA structures reveals impact of sequence evolution on conserved and non-conserved structural motifs. PLoS Pathog. 9, e1003294 (2013).
Wilson, N. A. et al. Vaccine-Induced cellular immune responses reduce plasma viral concentrations after repeated low-dose challenge with pathogenic simian immunodeficiency virus SIVmac239. J. Virol. 80, 5875–5885 (2006).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. Available online at: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (2010).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Li, H. et al. Aligning sequences reads, clone sequences and assembly contigs with BWA-MEM. arXiv https://doi.org/10.48550/arXiv.1303.3997 (2013).
Danecek, P. et al. Twelve years of SAMtools and BCFtools. GigaScience 10, giab008 (2021).
der Auwera, G. A. V. & O’Connor, B. D. Genomics in the Cloud: Using Docker, GATK, and WDL in Terra. (O’Reilly Media, 2020).
Librado, P. & Rozas, J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25, 1451–1452 (2009).
Yamashita, M. & Emerman, M. Capsid is a dominant determinant of retrovirus infectivity in nondividing cells. J. Virol. 78, 5670–5678 (2004).
Dull, T. et al. A third-generation lentivirus vector with a conditional packaging system. J. Virol. 72, 8463–8471 (1998).
Meyerson, N. R., Warren, C. J., Vieira, D. A. S. A., Diaz-Griffero, F. & Sawyer, S. L. Species-specific vulnerability of RanBP2 shaped the evolution of SIV as it transmitted in African apes. PLoS Pathog. 14, e1006906 (2018).
Poss, M. & Overbaugh, J. Variants from the diverse virus population identified at seroconversion of a clade A human immunodeficiency virus type 1-infected woman have distinct biological properties. J. Virol. 73, 5255–5264 (1999).
Wei, X. et al. Emergence of resistant human immunodeficiency virus type 1 in patients receiving fusion inhibitor (T-20) monotherapy. Antimicrob. Agents. Chemother. 6, 1896–905 (2002).
Platt, E. J., Wehrly, K., Kuhmann, S. E., Chesebro, B. & Kabat, D. Effects of CCR5 and CD4 cell surface concentrations on infections by macrophage-tropic isolates of human immunodeficiency virus type 1. J. Virol. 72, 2855–2864 (1998).
Kimpton, J. & Emerman, M. Detection of replication-competent and pseudotyped human immunodeficiency virus with a sensitive cell line on the basis of activation of an integrated beta-galactosidase gene. J. Virol. 66, 2232–2239 (1992).
Vermeire, J. et al. Quantification of reverse transcriptase activity by real-time PCR as a fast and accurate method for titration of HIV, lenti- and retroviral vectors. PLoS One 7, e50859 (2012).
Wong, J. K. & Yukl, S. A. Tissue reservoirs of HIV. Curr. Opin. HIV AIDS 11, 362–370 (2016).
Li, K., Liu, B., Ma, R. & Zhang, Q. HIV tissue reservoirs: current advances in research. AIDS Patient Care STDs 37, 284–296 (2023).
Spach, D. H. & Kalapila, A. G. Occupational postexposure prophylaxis. National HIV Curriculum https://www.hiv.uw.edu/go/prevention/occupational-postexposure-prophylaxis/core-concept/all (2025).
Leigh, J. W. & Bryant, D. Popart: Full-feature software for haplotype network construction. Methods Ecol. Evol. 6, 1110–1116 (2015).
Sultana, T. et al. Novel mutant human immunodeficiency virus type 1 strains with high degree of resistance to cynomolgus macaque TRIMCyp generated by random mutagenesis. J. Gen. Virol. 97, 963–976 (2016).
Kroon, E. et al. Paradoxically greater persistence of HIV RNA-positive cells in lymphoid tissue when ART is initiated in the earliest stage of infection. J. Infect. Dis. 225, 2167–2175 (2022).
Acknowledgements
We thank Greg Del Prete for valuable conversations, Jiro Wada for help with data visualization, and Anya Crane for critically editing the manuscript. Hut78 cells stably expressing human CCR5 were a gift from Vineet KewalRamani. This work was funded by grants from the National Institutes of Health (DP1-AI-175471, DP1-DA-046108, R01-AI-137011, R01-OD-034046 to SLS); by NIAID through two contracts, Simian Vaccine Evaluation Unit (SVEU) (75N93020D00007) to Tulane National Biomedical Research Center and the Virology Core Laboratory (75N93025C00002) to Duke University; by the University of Colorado Flow Cytometry Shared Facility in JSCBB (RRID:SCR_019309), supported by NIH grant S10OD021601. This work was supported in part through the prime contract of Laulima Government Solutions, LLC, with NIAID under contract no. HHSN272201800013C. J.H.K. performed this work as an employee of Tunnell Government Services (TGS), a subcontractor of Laulima Government Solutions, LLC, under contract no. HHSN272201800013C. The views and conclusions contained in this document are those of the authors and should not be interpreted as necessarily representing the official policies or positions, either expressed or implied, of the US Department of Health and Human Services, or any of the institutions and companies affiliated with the authors.
Author information
Authors and Affiliations
Contributions
N.R.M., V.L.B., C.J.W., A.B.-G., M.J.W., P.N.N., K.A.S., P.P.A., R.S.V., S.M.K., G.K.W., and S.L.S. conceived and designed the experiments. N.R.M., V.L.B., W.T.F., C.J.W., A.B.-G., M.J.W., O.Dirasantha, B.L.B., A.S.A., J.L.T., E.R.F., R.T.F., P.G., A.F., K.A.S., A.G., G.W.T., P.J.K., M.D.S., O.Davila, E.I.L., S.E.C., M.L.G., C.T.D., S.R.S., T.N.D., S.M.K., and G.K.W. performed the experiments. N.R.M., V.L.B., W.T.F., C.J.W., A.B.-G., M.J.W., O.Dirasantha, B.L.B., A.S.A., J.L.T., E.R.F., R.T.F., P.G., A.F., A.G., O.Davila, R.S.V., G.K.W., and S.L.S. analyzed the data. P.N.N. contributed materials and analysis tools. N.R.M., V.L.B., J.H.K., G.K.W., and S.L.S. wrote the manuscript. All authors edited and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
NRM, WTF, and SLS are employees of, or hold financial interests in, Darwin Biosciences in Boulder, CO, USA. The remaining authors declare no competing interests. A patent application is related to this work; PCT Application No. PCT/US2023/082939 for “Novel HIV-1 Variants and their Methods of Use in an Animal Challenge Model.”
Peer review
Peer review information
Nature Communications thanks Roger Le Grand and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Meyerson, N.R., Bauer, V.L., Fattor, W.T. et al. CXCR4-tropic HIV-1 infection in an immunocompetent monkey model. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70209-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70209-5