Abstract
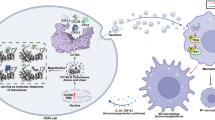
Pancreatic ductal adenocarcinoma (PDAC) remains one of the deadliest malignancies due to its highly immunosuppressive tumor microenvironment (TME), which limits effective therapeutic interventions. Here, we demonstrate that V-domain immunoglobulin suppressor of T cell activation (VISTA) plays a crucial role in orchestrating macrophage polarity within the PDAC TME. Using murine PDAC models, we show that VISTA deficiency markedly impairs tumor growth, leading to prolonged survival. Functionally, VISTA deficiency is linked to a shift in tumor-associated macrophages (TAMs) from an immunosuppressive phenotype marked by secreted phosphoprotein 1 (SPP1), to one enriched for C-X-C motif chemokine ligand 9 (CXCL9), indicative of a pro-inflammatory state. This shift is accompanied by enhanced recruitment of CXCR3⁺ CD8⁺ T cells with sustained cytotoxic potential, among which terminal exhaustion-like CD8+ T cell states are less prevalent. Additionally, VISTA-deficient TAMs exhibit increased antigen cross-presentation, further amplifying CD8+ T cell response against tumors. These findings are corroborated by human PDAC data, which reflect similar immune reprogramming trends. By defining the role of VISTA in controlling Cxcl9:Spp1 ratio and modulating CD8⁺ T cell dynamics, this study positions VISTA inhibition as a promising strategy to reshape the TME and potentiate anti-tumor immunity in PDAC.
Similar content being viewed by others
Data availability
The single-cell RNA sequencing data generated in this study have been deposited in NCBI Gene Expression Omnibus (GEO) under the accession code GSE282101. The publicly available TCGA-PAAD dataset used in this study can be accessed via the GDC Data Portal [https://portal.gdc.cancer.gov/projects/TCGA-PAAD]. Source data are provided with this paper.
Code availability
All custom-written code can be accessed at [https://github.com/gracesshin/VISTA_PDAC].
References
Connor, A. A. & Gallinger, S. Pancreatic cancer evolution and heterogeneity: integrating omics and clinical data. Nat. Rev. Cancer 22, 131–142 (2022).
Alors-Perez, E. et al. Splicing alterations in pancreatic ductal adenocarcinoma: a new molecular landscape with translational potential. J. Exp. Clin. Cancer Res. 42, 282 (2023).
Slodkowski, M. et al. Current approaches for the curative-intent surgical treatment of pancreatic ductal adenocarcinoma. Cancers 15, 2584 (2023).
Koltai, T. et al. Resistance to gemcitabine in pancreatic ductal adenocarcinoma: a physiopathologic and pharmacologic review. Cancers 14, 2486 (2022).
Kleeff, J. et al. Pancreatic cancer. Nat. Rev. Dis. Prim. 2, 16022 (2016).
Truong, L. H. & Pauklin, S. Pancreatic cancer microenvironment and cellular composition: current understandings and therapeutic approaches. Cancers 13, 5028 (2021).
Bear, A. S., Vonderheide, R. H. & O’Hara, M. H. Challenges and opportunities for pancreatic cancer immunotherapy. Cancer Cell 38, 788–802 (2020).
Mucileanu, A., Chira, R. & Mircea, P. A. PD-1/PD-L1 expression in pancreatic cancer and its implication in novel therapies. Med Pharm. Rep. 94, 402–410 (2021).
Ni, R., Hu, Z. & Tao, R. Advances of immune-checkpoint inhibition of CTLA-4 in pancreatic cancer. Biomed. Pharmacother. 179, 117430 (2024).
Miyazawa, M. et al. Advances in immunotherapy for pancreatic ductal adenocarcinoma. J. Hepatobiliary Pancreat. Sci. 28, 419–430 (2021).
Ho, W. J., Jaffee, E. M. & Zheng, L. The tumour microenvironment in pancreatic cancer - clinical challenges and opportunities. Nat. Rev. Clin. Oncol. 17, 527–540 (2020).
Feng, M. et al. PD-1/PD-L1 and immunotherapy for pancreatic cancer. Cancer Lett. 407, 57–65 (2017).
Noelle, R. J. et al. Clinical and research updates on the VISTA immune checkpoint: immuno-oncology themes and highlights. Front. Oncol. 13, 1225081 (2023).
ElTanbouly, M. A., Croteau, W., Noelle, R. J. & Lines, J. L. VISTA: a novel immunotherapy target for normalizing innate and adaptive immunity. Semin. Immunol. 42, 101308 (2019).
Muller, S. et al. V-domain Ig-containing suppressor of T-cell activation (VISTA), a potentially targetable immune checkpoint molecule, is highly expressed in epithelioid malignant pleural mesothelioma. Mod. Pathol. 33, 303–311 (2020).
Huang, X. et al. VISTA: an immune regulatory protein checking tumor and immune cells in cancer immunotherapy. J. Hematol. Oncol. 13, 83 (2020).
ElTanbouly, M. A. et al. VISTA is a checkpoint regulator for naive T cell quiescence and peripheral tolerance. Science 367, eaay0524 (2020).
Zhang, R. J. & Kim, T. K. VISTA-mediated immune evasion in cancer. Exp. Mol. Med. 56, 2348–2356 (2020).
Blando, J. et al. Comparison of immune infiltrates in melanoma and pancreatic cancer highlights VISTA as a potential target in pancreatic cancer. Proc. Natl. Acad. Sci. USA 116, 1692–1697 (2019).
Hou, Z. et al. Prognostic significance and therapeutic potential of the immune checkpoint VISTA in pancreatic cancer. J. Cancer Res. Clin. Oncol. 147, 517–531 (2021).
Park, J. G. et al. Kidney residency of VISTA-positive macrophages accelerates repair from ischemic injury. Kidney Int. 97, 980–994 (2020).
Wherry, E. J. T cell exhaustion. Nat. Immunol. 12, 492–499 (2011).
Bengsch, B. et al. Bioenergetic insufficiencies due to metabolic alterations regulated by the inhibitory receptor PD-1 are an early driver of CD8(+) T cell exhaustion. Immunity 45, 358–373 (2016).
Tegowski, M. & Baldwin, A. Noncanonical NF-kappaB in cancer. Biomedicines 6, 66 (2018).
Dai, L. et al. SOCS3 acts as an onco-immunological biomarker with value in assessing the tumor microenvironment, pathological staging, histological subtypes, therapeutic effect, and prognoses of several types of cancer. Front. Oncol. 12, 881801 (2022).
Pascual-Garcia, M. et al. LIF regulates CXCL9 in tumor-associated macrophages and prevents CD8(+) T cell tumor-infiltration impairing anti-PD1 therapy. Nat. Commun. 10, 2416 (2019).
Sathe, A. et al. Colorectal cancer metastases in the liver establish immunosuppressive spatial networking between tumor-associated SPP1+ macrophages and fibroblasts. Clin. Cancer Res 29, 244–260 (2023).
Bill, R. et al. CXCL9:SPP1 macrophage polarity identifies a network of cellular programs that control human cancers. Science 381, 515–524 (2023).
Su, X., Liang, C., Chen, R. & Duan, S. Deciphering tumor microenvironment: CXCL9 and SPP1 as crucial determinants of tumor-associated macrophage polarity and prognostic indicators. Mol. Cancer 23, 13 (2024).
Weulersse, M. et al. Eomes-dependent loss of the co-activating receptor CD226 restrains CD8(+) T $$$erapy. Immunity 53, 824–839 e810 (2020).
Franco, F., Jaccard, A., Romero, P., Yu, Y. R. & Ho, P. C. Metabolic and epigenetic regulation of T-cell exhaustion. Nat. Metab. 2, 1001–1012 (2020).
Richter, F. C., Saliutina, M., Hegazy, A. N. & Bergthaler, A. Take my breath away-mitochondrial dysfunction drives CD8(+) T cell exhaustion. Genes Immun. 25, 4–6 (2024).
Jin, S. et al. Inference and analysis of cell-cell communication using CellChat. Nat. Commun. 12, 1088 (2021).
Pan, M. et al. Targeting CXCL9/10/11-CXCR3 axis: an important component of tumor-promoting and antitumor immunity. Clin. Transl. Oncol. 25, 2306–2320 (2023).
Chia, T. Y. et al. The CXCL16-CXCR6 axis in glioblastoma modulates T-cell activity in a spatiotemporal context. Front. Immunol. 14, 1331287 (2023).
Yang, H. et al. Single-cell RNA sequencing reveals recruitment of the M2-like CCL8(high) macrophages in Lewis lung carcinoma-bearing mice following hypofractionated radiotherapy. J. Transl. Med. 22, 306 (2024).
Quemener, C. et al. Dual roles for CXCL4 chemokines and CXCR3 in angiogenesis and invasion of pancreatic cancer. Cancer Res 76, 6507–6519 (2016).
Harrison, O. J. et al. Epithelial-derived IL-18 regulates Th17 cell differentiation and Foxp3(+) Treg cell function in the intestine. Mucosal Immunol. 8, 1226–1236 (2015).
Yang, R. et al. Galectin-9 interacts with PD-1 and TIM-3 to regulate T cell death and is a target for cancer immunotherapy. Nat. Commun. 12, 832 (2021).
Ring, A. M. et al. Mechanistic and structural insight into the functional dichotomy between IL-2 and IL-15. Nat. Immunol. 13, 1187–1195 (2012).
Cassetta, L. & Pollard, J. W. Targeting macrophages: therapeutic approaches in cancer. Nat. Rev. Drug Discov. 17, 887–904 (2018).
Xiong, C. et al. Tumor-associated macrophages promote pancreatic ductal adenocarcinoma progression by inducing epithelial-to-mesenchymal transition. Aging (Albany NY) 13, 3386–3404 (2021).
Qian, B. Z. & Pollard, J. W. Macrophage diversity enhances tumor progression and metastasis. Cell 141, 39–51 (2010).
Ruffell, B. & Coussens, L. M. Macrophages and therapeutic resistance in cancer. Cancer Cell 27, 462–472 (2015).
Biswas, S. K. & Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat. Immunol. 11, 889–896 (2010).
Murray, P. J. et al. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41, 14–20 (2014).
Lines, J. L. et al. VISTA is an immune checkpoint molecule for human T cells. Cancer Res 74, 1924–1932 (2014).
Wang, L. et al. VISTA, a novel mouse Ig superfamily ligand that negatively regulates T cell responses. J. Exp. Med. 208, 577–592 (2011).
Lim, R. J. et al. CXCL9/10-engineered dendritic cells promote T cell activation and enhance immune checkpoint blockade for lung cancer. Cell Rep. Med. 5, 101479 (2024).
Hu, J. K., Kagari, T., Clingan, J. M. & Matloubian, M. Expression of chemokine receptor CXCR3 on T cells affects the balance between effector and memory CD8 T-cell generation. Proc. Natl. Acad. Sci. USA 108, E118–E127 (2011).
Rittling, S. R. & Singh, R. Osteopontin in immune-mediated diseases. J. Dent. Res. 94, 1638–1645 (2015).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
Siddiqui, I. et al. Intratumoral Tcf1(+)PD-1(+)CD8(+) T cells with stem-like properties promote tumor control in response to vaccination and checkpoint blockade immunotherapy. Immunity 50, 195–211 e110 (2019).
Bangs, D. J. et al. CXCR3 regulates stem and proliferative CD8+ T cells during chronic infection by promoting interactions with DCs in splenic bridging channels. Cell Rep. 38, 110266 (2022).
Spranger, S. & Gajewski, T. F. Impact of oncogenic pathways on evasion of antitumour immune responses. Nat. Rev. Cancer 18, 139–147 (2018).
Peranzoni, E., Ingangi, V., Masetto, E., Pinton, L. & Marigo, I. Myeloid cells as clinical biomarkers for immune checkpoint blockade. Front. Immunol. 11, 1590 (2020).
Marcovecchio, P. M., Thomas, G. & Salek-Ardakani, S. CXCL9-expressing tumor-associated macrophages: new players in the fight against cancer. J. Immunother. Cancer 9, e002045 (2021).
Nowak, E. C. et al. Immunoregulatory functions of VISTA. Immunol. Rev. 276, 66–79 (2017).
Johnston, R. J. et al. VISTA is an acidic pH-selective ligand for PSGL-1. Nature 574, 565–570 (2019).
Ta, H. M. et al. LRIG1 engages ligand VISTA and impairs tumor-specific CD8(+) T cell responses. Sci. Immunol. 9, eadi7418 (2024).
Rogers, B. M. et al. VISTA is an activating receptor in human monocytes. J. Exp. Med. 218, e20201601 (2021).
Shekari, N. et al. VISTA and its ligands: the next generation of promising therapeutic targets in immunotherapy. Cancer Cell Int. 23, 265 (2023).
Kim, M. G. et al. Kidney VISTA prevents IFN-gamma/IL-9 axis-mediated tubulointerstitial fibrosis after acute glomerular injury. J. Clin. Invest. 132, e151189 (2022).
Sharma, N. S. et al. Targeting tumor-intrinsic hexosamine biosynthesis sensitizes pancreatic cancer to anti-PD1 therapy. J. Clin. Invest. 130, 451–465 (2020).
Kim, D. K. et al. PD-L1-directed PlGF/VEGF blockade synergizes with chemotherapy by targeting CD141(+) cancer-associated fibroblasts in pancreatic cancer. Nat. Commun. 13, 6292 (2022).
Cao, J. et al. The single-cell transcriptional landscape of mammalian organogenesis. Nature 566, 496–502 (2019).
Rozenblatt-Rosen, O. et al. The human tumor atlas network: charting tumor transitions across space and time at single-cell resolution. Cell 181, 236–249 (2020).
Wickham, H. ggplot2. Wiley Interdiscip. Rev.: Comput. Stat. 3, 180–185 (2011).
Acknowledgements
Figures 7a and 7c were created with BioRender.com. This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (No. RS-2024-00406325 to D.-S.L.). This research was also supported by the National Research Foundation of Korea (NRF) grant, funded by the Korea government (MSIT) (No. RS-2024-00345658 and RS-2025-25403101 to D.-S.L.) and by the Education and Research Encouragement Fund of Seoul National University Hospital. Suk-Kyung Shin was supported by BK21 Four Biomedical Science Program, Seoul National University College of Medicine.
Author information
Authors and Affiliations
Contributions
S.-K.S. conceived and designed the study, performed experiments, analyzed murine single-cell data, and wrote the manuscript. G.K. performed and led the analysis of murine and human single-cell data and contributed to manuscript revision. G.H.K. guided human tissue staining and performed human IHC scoring. K.J. provided key reagents and methodological support. S.M.P. assisted with experiments. E.-B.S., S.-K.Y., and H.M.S. contributed to data discussion and manuscript revision. H.-R.K. and D.-S.L. supervised the study and edited the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Casey Ager, Lei Zheng, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shin, SK., Kim, G., Park, S.M. et al. VISTA drives pancreatic tumor progression through modulation of the tumor-associated macrophage polarity. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70215-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70215-7