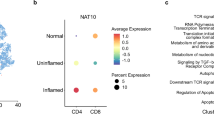
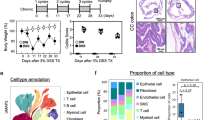
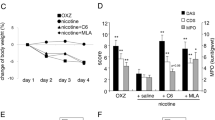
Abstract
Intestinal aging characterized by imbalance between cell senescence and mucosal self-renewal, increases susceptibility to the elderly-onset ulcerative colitis (UC), while the underlying mechanisms remain elusive. Here, we identify mRNA N4-acetylcytidine (ac4C) modification and its specific writer, N-acetyltransferase 10 (NAT10), as critical regulators of human colonic epithelial cell senescence. Knockdown of NAT10 significantly alleviates human colonic epithelial cell senescence in vitro and colonoid and intestinal aging in vivo in aged mice. Using ac4C-modified transcriptome sequencing, we reveal that NAT10 stabilizes DYRK1A mRNA through ac4C modification, thereby driving colon epithelial senescence. Moreover, NAT10 and DYRK1A are markedly upregulated in ulcerative colitis tissues from elderly patients and positively correlate with disease severity. Knockdown of NAT10, treatment with Nat10 or Dyrk1a inhibitor, alleviates colitis in aged mice. Collectively, these findings suggest that modulating NAT10-mediated RNA ac4C modification could rejuvenate intestinal aging and provide a novel therapeutic strategy for elderly-onset colitis.
Similar content being viewed by others
Data availability
The RNA-seq data generated in this study have been deposited in the SRA database under accession code PRJNA1394239 (https://www.ncbi.nlm.nih.gov/sra/?term=PRJNA1394239). The acRIP-seq data generated in this study have been deposited in the SRA database under accession code PRJNA1347399 (https://www.ncbi.nlm.nih.gov/sra/?term=PRJNA1347399). Other data from the findings of this study are available from the corresponding author upon request. Source data are provided in this paper.
References
Kennedy, B. K. et al. Geroscience: linking aging to chronic disease. Cell 159, 709–713 (2014).
Campisi, J. et al. From discoveries in ageing research to therapeutics for healthy ageing. Nature 571, 183–192 (2019).
Hohman, L. S. & Osborne, L. C. A gut-centric view of aging: Do intestinal epithelial cells contribute to age-associated microbiota changes, inflammaging, and immunosenescence?. Aging Cell 21, e13700 (2022).
Cai, Y. et al. The landscape of aging.Sci. China Life Sci. 65, 2354–2454 (2022).
Wang, Q. et al. The aged intestine: performance and rejuvenation. Aging Dis. 12, 1693–1712 (2021).
Liu, A. et al. Aging increases the severity of colitis and the related changes to the gut barrier and gut microbiota in humans and mice. J. Gerontol. A Biol. Sci. Med. Sci. 75, 1284–1292 (2020).
Sun, T. Y. et al. MiR-1-3p and MiR-124-3p Synergistically damage the intestinal barrier in the ageing colon. J. Crohns Colitis 16, 656–667 (2022).
Hu, X. et al. Intestinal epithelial cell-specific deletion of cytokine-inducible SH2-containing protein alleviates experimental colitis in ageing mice. J. Crohns Colitis 17, 1278–1290 (2023).
Huang, L. et al. Elevated miR-124-3p in the aging colon disrupts mucus barrier and increases susceptibility to colitis by targeting T-synthase. Aging Cell 19, e13252 (2020).
Sienkiewicz, M., Sroka, K., Binienda, A., Jurk, D. & Fichna, J. A new face of old cells: An overview about the role of senescence and telomeres in inflammatory bowel diseases. Ageing Res. Rev. 91, 102083 (2023).
Ruel, J., Ruane, D., Mehandru, S., Gower-Rousseau, C. & Colombel, J. F. IBD across the age spectrum: is it the same disease?. Nat. Rev. Gastroenterol. Hepatol. 11, 88–98 (2014).
Agrawal, M. et al. The rising burden of inflammatory bowel disease in Denmark over two decades: a ntionwide cohort study. Gastroenterology 163, 1547–1554 (2022).
Ha, C. Y. & Katz, S. Clinical implications of ageing for the management of IBD. Nat. Rev. Gastroenterol. Hepatol. 11, 128–138 (2014).
Sousa, P., Bertani, L. & Rodrigues, C. Management of inflammatory bowel disease in the elderly: A review. Dig. Liver Dis. 55, 1001–1009 (2023).
Rozich, J. J., Dulai, P. S., Fumery, M., Sandborn, W. J. & Singh, S. Progression of elderly onset inflammatory bowel diseases: a systematic review and meta-analysis of population-based cohort studies. Clin. Gastroenterol. Hepatol. 18, 2437–2447 (2020).
Bollegala, N., Jackson, T. D. & Nguyen, G. C. Increased postoperative mortality and complications among elderly patients with inflammatory bowel diseases: an analysis of the national surgical quality improvement program cohort. Clin. Gastroenterol. Hepatol. 14, 1274–1281 (2016).
Meng, G., Monaghan, T.M., Duggal, N.A., Tighe, P. & Peerani, F. Microbial-immune crosstalk in elderly-onset inflammatory bowel disease: unchartered territory. J. Crohns Colitis 17, 1309–1325 (2023).
Clevers, H. & Batlle, E. SnapShot: the intestinal crypt. Cell 152, 1198–1198 (2013).
Di Micco, R., Krizhanovsky, V., Baker, D. & d’Adda di Fagagna, F. 1-Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 22, 75–95 (2021).
Lopez-Otin, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: An expanding universe. Cell 186, 243–278 (2023).
Wang, K. et al. Epigenetic regulation of aging: implications for interventions of aging and diseases. Signal Transduct. Target Ther. 7, 374 (2022).
Sun, H., Li, K., Liu, C. & Yi, C. Regulation and functions of non-m(6)A mRNA modifications. Nat. Rev. Mol. Cell Biol. 24, 714–731 (2023).
Arango, D. et al. Acetylation of Cytidine in mRNA Promotes Translation Efficiency. Cell 175, 1872–1886 (2018).
Dalhat, M. H., Narayan, S., Serio, H. & Arango, D. Dissecting the oncogenic properties of essential RNA-modifying enzymes: a focus on NAT10. Oncogene 43, 1077–1086 (2024).
Liu, R. et al. NAT10-mediated N4-acetylcytidine mRNA modification regulates self-renewal in human embryonic stem cells. Nucleic Acids Res. 51, 8514–8531 (2023).
Chen, L. et al. NAT10-mediated N4-acetylcytidine modification is required for meiosis entry and progression in male germ cells. Nucleic Acids Res. 50, 10896–10913 (2022).
Huang, W., Hickson, L. J., Eirin, A., Kirkland, J. L. & Lerman, L. O. Cellular senescence: the good, the bad and the unknown. Nat. Rev. Nephrol. 18, 611–627 (2022).
Liu, H. et al. Targeting N4-acetylcytidine suppresses hepatocellular carcinoma progression by repressing eEF2-mediated HMGB2 mRNA translation. Cancer Commun. 44, 1018–1041 (2024).
Hunt, D. P. J. & Hofer, M. J. Unweaving type I interferons in age-related neuroinflammation. Trends Neurosci. 47, 751–752 (2024).
Chen, X. et al. The role of oxidative stress in intervertebral disc degeneration: Mechanisms and therapeutic implications. Ageing Res. Rev. 98, 102323 (2024).
Liu, D. et al. NAT10 promotes synovial aggression by increasing the stability and translation of N4-acetylated PTX3 mRNA in rheumatoid arthritis. Ann. Rheum. Dis. 83, 1118–1131 (2024).
Qi, Y. et al. Heat-inactivated Bifidobacterium adolescentis ameliorates colon senescence through Paneth-like-cell-mediated stem cell activation. Nat. Commun. 14, 6121 (2023).
He, D. et al. Gut stem cell aging is driven by mTORC1 via a p38 MAPK-p53 pathway. Nat. Commun. 11, 37 (2020).
Omrani, O. et al. IFNgamma-Stat1 axis drives aging-associated loss of intestinal tissue homeostasis and regeneration. Nat. Commun. 14, 6109 (2023).
Wang, Z. et al. FUT2-dependent fucosylation of HYOU1 protects intestinal stem cells against inflammatory injury by regulating unfolded protein response. Redox Biol. 60, 102618 (2023).
Zheng, X. et al. N-acetyltransferase 10 promotes colon cancer progression by inhibiting ferroptosis through N4-acetylation and stabilization of ferroptosis suppressor protein 1 (FSP1) mRNA. Cancer Commun. 42, 1347–1366 (2022).
Yang, W. et al. ac4C acetylation of RUNX2 catalyzed by NAT10 spurs osteogenesis of BMSCs and prevents ovariectomy-induced bone loss. Mol. Ther. Nucleic Acids 26, 135–147 (2021).
Xie, R. et al. NAT10 Drives cisplatin chemoresistance by enhancing ac4C-associated DNA repair in bladder cancer. Cancer Res. 83, 1666–1683 (2023).
Guo, G. et al. Epitranscriptomic N4-Acetylcytidine Profiling in CD4(+) T Cells of Systemic Lupus Erythematosus. Front. Cell Dev. Biol. 8, 842 (2020).
Balmus, G. et al. Targeting of NAT10 enhances healthspan in a mouse model of human accelerated aging syndrome. Nat. Commun. 9, 1700 (2018).
Rammohan, M. et al. The chromosome 21 kinase DYRK1A: emerging roles in cancer biology and potential as a therapeutic target. Oncogene 41, 2003–2011 (2022).
Litovchick, L., Florens, L. A., Swanson, S. K., Washburn, M. P. & DeCaprio, J. A. DYRK1A protein kinase promotes quiescence and senescence through DREAM complex assembly. Genes Dev. 25, 801–813 (2011).
Park, J. et al. Dyrk1A phosphorylates p53 and inhibits proliferation of embryonic neuronal cells. J. Biol. Chem. 285, 31895–31906 (2010).
Chen, J. Y., Lin, J. R., Tsai, F. C. & Meyer, T. Dosage of Dyrk1a shifts cells within a p21-cyclin D1 signaling map to control the decision to enter the cell cycle. Mol. Cell 52, 87–100 (2013).
Soppa, U. et al. The Down syndrome-related protein kinase DYRK1A phosphorylates p27(Kip1) and Cyclin D1 and induces cell cycle exit and neuronal differentiation. Cell Cycle 13, 2084–2100 (2014).
Hu, K. et al. MicroRNA-221-3p inhibits the inflammatory response of keratinocytes by regulating the DYRK1A/STAT3 signaling pathway to promote wound healing in diabetes. Commun. Biol. 7, 300 (2024).
Bhansali, R. S. et al. DYRK1A regulates B cell acute lymphoblastic leukemia through phosphorylation of FOXO1 and STAT3. J. Clin. Invest. 131, https://doi.org/10.1172/jci135937 (2021).
Murray, A. et al. Dose imbalance of DYRK1A kinase causes systemic progeroid status in Down syndrome by increasing the un-repaired DNA damage and reducing LaminB1 levels. EBioMedicine 94, 104692 (2023).
Kim, M. Y. et al. A repressor complex, AP4 transcription factor and geminin, negatively regulates expression of target genes in nonneuronal cells. Proc. Natl. Acad. Sci. USA 103, 13074–13079 (2006).
Wang, P., Wang, L., Chen, L. & Sun, X. Dual-specificity tyrosine-phosphorylation regulated kinase 1A Gene Transcription is regulated by Myocyte Enhancer Factor 2D. Sci. Rep. 7, 7240 (2017).
Saccon, T. D. et al. Senolytic combination of dasatinib and quercetin alleviates intestinal senescence and inflammation and modulates the gut microbiome in aged mice. J. Gerontol. A Biol. Sci. Med. Sci. 76, 1895–1905 (2021).
Zhou, M., Liu, Z. L., Liu, J. Y. & Wang, X. B. Tedizolid phosphate alleviates DSS-induced ulcerative colitis by inhibiting senescence of cell and colon tissue through activating AMPK signaling pathway. Int. Immunopharmacol. 135, 112286 (2024).
Wellman, A. S. et al. Intestinal epithelial sirtuin 1 regulates intestinal inflammation during aging in mice by altering the intestinal microbiota. Gastroenterology 153, 772–786 (2017).
Zhao, Y., Simon, M., Seluanov, A. & Gorbunova, V. DNA damage and repair in age-related inflammation. Nat. Rev. Immunol. 23, 75–89 (2023).
Bao, W. et al. Inhibiting sorting nexin 10 promotes mucosal healing through SREBP2-mediated stemness restoration of intestinal stem cells. Sci. Adv. 9, eadh5016 (2023).
Gonzalez, E. A. et al. TET3-mediated DNA oxidation is essential for intestinal epithelial cell response to stressors. Proc. Natl. Acad. Sci. USA 120, e2221405120 (2023).
Ren, X. et al. DHX9 maintains epithelial homeostasis by restraining R-loop-mediated genomic instability in intestinal stem cells. Nat. Commun. 15, 3080 (2024).
Choi, Y. W. et al. Senescent tumor cells build a cytokine shield in colorectal cancer. Adv. Sci. 8, 2002497 (2021).
Tollefsbol, T.O. Biological Aging: Methods and Protocols. (2013).
Acknowledgements
We thank Xiaoyu Li (Zhejiang University School of Medicine) and Liang Lu (Zhejiang University) for their helpful discussions and technical support in the detection and analysis of RNA modifications. We thank Chunchun Li, Xiaoye Li, Liya Lin and Jiaying Li (Clinical Research Center, The Second Affiliated Hospital of Zhejiang University School of Medicine) for the laboratory management and technical support. We thank Yunlu Chen, Hui Xie and Zhenlin Jiang (Animal Experiment Center, The Second Affiliated Hospital of Zhejiang University School of Medicine) for their technical support in small animal endoscopy techniques. This project was financially supported by the National Foundation of Natural Science of China (82525010 to S.C.) and the Provincial Key R&D Program of Zhejiang (2023C03163 to L.W.).
Author information
Authors and Affiliations
Contributions
S.C., L.W., and M.X. designed and supervised the study. J.C. performed most experiments with the assistance of S.M., A.F., W.C., Y.S., S.X., Q.G., J.L., and Q.Y.; J.C. and M.X. analyzed and interpreted the data. J.C. drafted the manuscript. S.C. and L.W. critically revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, J., Xue, M., Mi, S. et al. Targeting NAT10 alleviates colonic senescence and elderly-onset colitis by disrupting N4-acetylation of DYRK1A. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70220-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70220-w