Abstract
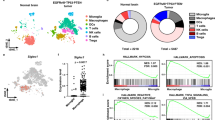
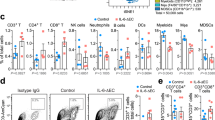
Macrophage-mediated phagocytosis of tumor cells elicits potent antitumor immunity. Nonetheless, sole-blockade of the anti-phagocytosis molecule CD47 has yielded insufficient therapeutic outcomes. Here, we report that glioblastoma (GBM) cells expressed abundant levels of phagocytosis checkpoint CD24. We further show that dual blockade of CD24 and CD47 synergistically enhances the pro-phagocytic activity of macrophages, thereby improving tumor antigen cross-presentation and activating the cyclic GMP-AMP synthase–stimulator of interferon genes (cGAS-STING) pathway. This innate immune activation facilitates T cell infiltration into tumors and sensitizes tumors to anti-PD1 therapy, improving survival outcomes in murine GBM models, including immunosuppressive tumors reflecting human GBM-like features. Thus, our results indicate that dual-phagocytosis checkpoint blockade offers a promising therapeutic avenue to potentiate cancer immunotherapy.
Similar content being viewed by others
Data availability
Publicly available single-cell RNA sequencing data were obtained from GEO under accession code GSE182109. All data are included in the Supplementary Information or available from the authors, as are unique reagents used in this Article. The raw numbers for charts and graphs are available in the Source Data file whenever possible. Source data are provided with this paper.
References
Iwasaki, A. & Medzhitov, R. Regulation of adaptive immunity by the innate immune system. Science 327, 291–295 (2010).
Lee, D., Huntoon, K., Wang, Y., Jiang, W. & Kim, B. Y. Harnessing innate immunity using biomaterials for cancer immunotherapy. Adv. Mater. 33, 2007576 (2021).
Huntoon, K. et al. Targeting phagocytosis to enhance antitumor immunity. Trends Cancer 9, 650–665 (2023).
Lu, Y. et al. Immunological conversion of solid tumours using a bispecific nanobioconjugate for cancer immunotherapy. Nat. Nanotechnol. 17, 1332–1341 (2022).
von Roemeling, C. A. et al. Therapeutic modulation of phagocytosis in glioblastoma can activate both innate and adaptive antitumour immunity. Nat. Commun. 11, 1508 (2020).
Feng, M. et al. Phagocytosis checkpoints as new targets for cancer immunotherapy. Nat. Rev. Cancer 19, 568–586 (2019).
Chao, M. P. et al. Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell 142, 699–713 (2010).
Tseng, D. et al. Anti-CD47 antibody–mediated phagocytosis of cancer by macrophages primes an effective antitumor T-cell response. Proc. Natl. Acad. Sci. USA 110, 11103–11108 (2013).
Liu, X. et al. CD47 blockade triggers T cell–mediated destruction of immunogenic tumors. Nat. Med. 21, 1209–1215 (2015).
Majeti, R. et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell 138, 286–299 (2009).
Willingham, S. B. et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc. Natl. Acad. Sci. USA 109, 6662–6667 (2012).
Gholamin, S. et al. Disrupting the CD47-SIRPα anti-phagocytic axis by a humanized anti-CD47 antibody is an efficacious treatment for malignant pediatric brain tumors. Sci. Transl. Med. 9, eaaf2968 (2017).
Chao, M. P. et al. Therapeutic antibody targeting of CD47 eliminates human acute lymphoblastic leukemia. Cancer Res. 71, 1374–1384 (2011).
Sallman, D. A. et al. Magrolimab in combination with azacitidine in patients with higher-risk myelodysplastic syndromes: final results of a phase Ib study. J. Clin. Oncol. 41, 2815–2826 (2023).
Maute, R., Xu, J. & Weissman, I. CD47–SIRPα-targeted therapeutics: status and prospects. Immuno-Oncol. Technol. 13, 100070 (2022).
Barkal, A. A. et al. CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature 572, 392–396 (2019).
Abdelfattah, N. et al. Single-cell analysis of human glioma and immune cells identifies S100A4 as an immunotherapy target. Nat. Commun. 13, 767 (2022).
Gordon, S. R. et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 545, 495–499 (2017).
Barkal, A. A. et al. Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nat. Immunol. 19, 76–84 (2018).
Chen, J. et al. SLAMF7 is critical for phagocytosis of haematopoietic tumour cells via Mac-1 integrin. Nature 544, 493–497 (2017).
Martínez Murillo, R. & Martínez, A. Standardization of an orthotopic mouse brain tumor model following transplantation of CT-2A astrocytoma cells. Histol. Histopathol. 22, 1309–1326 (2007).
Macintyre, E. H., Pontén, J. & Vatter, A. The ultrastructure of human and murine astrocytes and of human fibroblasts in culture. Acta Pathol. Microbiol. Scand. Sect. A Pathol. 80, 267–283 (1972).
Letchuman, V. et al. Syngeneic murine glioblastoma models: reactionary immune changes and immunotherapy intervention outcomes. Neurosurg. Focus 52, E5 (2022).
Westermark, B. The deficient density-dependent growth control of human malignant glioma cells and virus-transformed glia-like cells in culture. Int. J. Cancer 12, 438–451 (1973).
Kosaka, A. et al. CD47 blockade enhances the efficacy of intratumoral STING-targeting therapy by activating phagocytes. J. Exp. Med. 218, e20200792 (2021).
Abdullah, A. et al. STING-mediated type-I interferons contribute to the neuroinflammatory process and detrimental effects following traumatic brain injury. J. Neuroinflammation 15, 1–17 (2018).
Hopfner, K.-P. & Hornung, V. Molecular mechanisms and cellular functions of cGAS–STING signalling. Nat. Rev. Mol. Cell Biol. 21, 501–521 (2020).
Martin, G. R., Blomquist, C. M., Henare, K. L. & Jirik, F. R. Stimulator of interferon genes (STING) activation exacerbates experimental colitis in mice. Sci. Rep. 9, 14281 (2019).
Mathur, V. et al. Activation of the STING-dependent type I interferon response reduces microglial reactivity and neuroinflammation. Neuron 96, 1290–1302. e1296 (2017).
Salvagno, C. et al. Therapeutic targeting of macrophages enhances chemotherapy efficacy by unleashing type I interferon response. Nat. Cell Biol. 21, 511–521 (2019).
Zitvogel, L., Galluzzi, L., Kepp, O., Smyth, M. J. & Kroemer, G. Type I interferons in anticancer immunity. Nat. Rev. Immunol. 15, 405–414 (2015).
Hayden, M. S. & Ghosh, S. Shared principles in NF-κB signaling. Cell 132, 344–362 (2008).
Garcia-Diaz, A. et al. Interferon receptor signaling pathways regulating PD-L1 and PD-L2 expression. Cell Rep. 19, 1189–1201 (2017).
Aldea, M. et al. Overcoming resistance to tumor-targeted and immune-targeted therapies. Cancer Discov. 11, 874–899 (2021).
Han, C. et al. Tumor cells suppress radiation-induced immunity by hijacking caspase 9 signaling. Nat. Immunol. 21, 546–554 (2020).
Tumeh, P. C. et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571 (2014).
Dong, S. et al. Adaptive design of mRNA-loaded extracellular vesicles for targeted immunotherapy of cancer. Nat. Commun. 14, 6610 (2023).
Shingu, T. et al. Qki deficiency maintains stemness of glioma stem cells in suboptimal environment by downregulating endolysosomal degradation. Nat. Genet. 49, 75–86 (2017).
Chen, C.-H. et al. Novel murine glioblastoma models that reflect the immunotherapy resistance profile of a human disease. Neuro-Oncol. 25, 1415–1427 (2023).
Najem, H. et al. STING agonist 8803 reprograms the immune microenvironment and increases survival in preclinical models of glioblastoma. J. Clin. Investig. 134, e175033 (2024).
Liu, Y. E. et al. Emerging phagocytosis checkpoints in cancer immunotherapy. Signal Transduct. Target. Ther. 8, 104 (2023).
Nishiga, Y. et al. Radiotherapy in combination with CD47 blockade elicits a macrophage-mediated abscopal effect. Nat. Cancer 3, 1351–1366 (2022).
Jeong, S. D. et al. Immunogenic cell death inducing fluorinated mitochondria-disrupting helical polypeptide synergizes with PD-L1 immune checkpoint blockade. Adv. Sci. 8, 2001308 (2021).
Yuan, H. et al. Multivalent bi-specific nanobioconjugate engager for targeted cancer immunotherapy. Nat. Nanotechnol. 12, 763–769 (2017).
Acknowledgements
This research was supported in part by Break Through Cancer Foundation, the National Institutes of Health/ National Institute of Neurological Disorders and Stroke Grant R01NS117828 (W.J.), the American Cancer Society Research Scholar Grant (RSG-22052-01-IBCD, W.J.), CPRIT Scholar in Cancer (RR250092, B.R.S.), The National Cancer Institute of the National Institutes of Health (1K08CA296776, B.R.S.), the James P. Allison Institute (P.S.), and the James P. Allison Institute’s Andrew Sabin Family Recruitment and Retention Fund (B.Y.S.K.).
Author information
Authors and Affiliations
Contributions
J.Ha., B.Y.S.K., and W.J. conceived the project and were responsible for all phases of the research. J.Ha. conducted the majority of the experiments and data analyses. Y.W., Y.M., A.W., S.D., S.D.J., J.L.E., M.C., Y-T.C., X.W., M.D., M.N.T., S.M., W.D., A.G., S.B., S.J., and B.S. assisted with data collection. J.S.W., S.M.R., F.D., I.E.M., S.P., S.D.F., C.I.E., K.A., J.Hu, S.G., V.K.P., F.F.L., K.H., and P.S. assisted with data analysis and interpretation. The manuscript was drafted by J.Ha., Y.W., B.Y.S.K., and W.J. and was revised and approved by all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Salvatore J. Coniglio and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ha, J., Wang, Y., Ma, Y. et al. Dual phagocytosis-checkpoint blockade revitalizes immune surveillance in mouse models of glioblastoma. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70221-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70221-9