Abstract
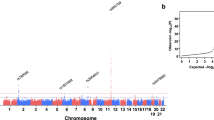
Polybrominated diphenyl ethers (PBDEs) are implicated in dyslipidemia, but the molecular basis of individual susceptibility remains elusive. Here we report an analysis based on the China National Human Biomonitoring cohort, where we integrate exposome, genomic, and metabolomic data to identify 3,571 genetic variants that interact with PBDE exposure to influence dyslipidemia risk. Metabolomic analysis highlights glycine and glycerophosphate as key mediators. A polygenic risk score derived from these PBDE-interactive variants significantly enhances dyslipidemia prediction in highly exposed individuals. Among these, rs9869609 emerges as a candidate causal variant, showing the strongest association with hypercholesterolemia risk (β = 1.18, FDR = 0.0078). Further functional validation using single-base CRISPR/Cas9 editing reveals that the rs9869609-G allele downregulates SLC6A20 expression by strengthening BHLHE40 binding, which further impairs glycine transport and promotes cholesterol accumulation, particularly under 2,2′,4,4′-Tetrabromodiphenyl ether exposure. Collectively, our study elucidates a gene-environment interaction mechanism through which genetic variants modulate lipid metabolism in response to PBDE exposure.
Similar content being viewed by others
Data availability
Source data are provided with this paper. The raw RNA-seq data are available in the GSA-Human repository under controlled access, accession code HRA014233 [https://ngdc.cncb.ac.cn/gsa-human/browse/HRA014233] (RNA-seq of HepG2 cells exposed to BDE47 and control). Access requests can be submitted via the GSA-Human System and are reviewed by the corresponding Data Access Committee (guidance: https://ngdc.cncb.ac.cn/gsa-human/document/GSA-Human_Request_Guide_for_Users_us.pdf). The timeframe for response to requests is expected within 15 working days. The genomic, metabolomic, and exposomic data of the study population involve sensitive biomonitoring data. Due to restrictions specified in agreements with collaborating institutions (cohort initiators) and the informed consent forms signed by study participants, these data cannot be made publicly available and are subject to controlled access. The request of these individual data is suggested by sending an email to the corresponding author Professor Xiaoming Shi (shixm@chinacdc.cn) and Professor Xiaoping Miao (xpmiao@whu.edu.cn). Requests should include name, affiliation, and contact details of the person requesting the data, which data are requested, and the purpose of requesting the data. Requests will be subject to consideration by the management committee of the corresponding institutes and the sample collection institutes. If approved, the corresponding author will send the request data by email. Time frame for a response will be within 3 months. Data requests under agreement will be considered for purposes of reproducing the data and subject to appropriate confidentiality obligations and restrictions. Applicants must promise that these individual data applied for will only be used for scientific research and cannot be publicly released. Source data are provided with this paper.
References
Chew, N. W. S. et al. The global burden of metabolic disease: data from 2000 to 2019. Cell Metab. 35, 414–428.e413 (2023).
GBD 2017 Risk Factor Collaborators. Global, regional and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 392, 1923–1994 (2018).
Lan, Y., Gao, X., Xu, H. & Li, M. 20 years of polybrominated diphenyl ethers on toxicity assessments. Water Res. 249, 121007 (2024).
Wei, J. et al. Metabolomics and lipidomics study unveils the impact of polybrominated diphenyl ether-47 on breast cancer mice. J. Hazard. Mater. 390, 121451 (2020).
Yin, W., Xu, R., Zou, J., Wang, Y. & Zhang, Y. Single and combined association between brominated flame retardants and cardiovascular disease: a large-scale cross-sectional study. Front. Public Health 12, 1357052 (2024).
Zhang, Q. et al. Associations between polybrominated diphenyl ethers (PBDEs) levels in adipose tissues and blood lipids in women of Shantou, China. Environ. Res. 214, 114096 (2022).
Boutot, M. E. et al. In utero exposure to persistent organic pollutants and childhood lipid levels. Metabolites 11, 657 (2021).
Zhao, X. et al. Polybrominated diphenyl ethers in serum from residents living in a brominated flame retardant production area: occurrence, influencing factors, and relationships with thyroid and liver function. Environ. Pollut. 270, 116046 (2021).
Klarin, D. et al. Genetics of blood lipids among ~300,000 multi-ethnic participants of the Million Veteran Program. Nat. Genet. 50, 1514–1523 (2018).
Noordam, R. et al. Multi-ancestry sleep-by-SNP interaction analysis in 126,926 individuals reveals lipid loci stratified by sleep duration. Nat. Commun. 10, 5121 (2019).
Wu, H., Eckhardt, C. M. & Baccarelli, A. A. Molecular mechanisms of environmental exposures and human disease. Nat. Rev. Genet. 24, 332–344 (2023).
Messerlian, C., Martinez, R. M., Hauser, R. & Baccarelli, A. A. Omics’ and endocrine-disrupting chemicals - new paths forward. Nat. Rev. Endocrinol. 13, 740–748 (2017).
Ortega-Azorín, C. et al. Associations of the FTO rs9939609 and the MC4R rs17782313 polymorphisms with type 2 diabetes are modulated by diet, being higher when adherence to the Mediterranean diet pattern is low. Cardiovasc. Diabetol. 11, 137 (2012).
Dietrich, S. et al. Gene-lifestyle interaction on risk of type 2 diabetes: a systematic review. Obes. Rev. 20, 1557–1571 (2019).
Brokken, L. J. & Giwercman, Y. L. Gene-environment interactions in male reproductive health: Special reference to the aryl hydrocarbon receptor signaling pathway. Asian J. Androl. 16, 89–96 (2014).
Zhou, Z. et al. Relationships among N,N-dimethylformamide exposure, CYP2E1 and TM6SF2 genes, and non-alcoholic fatty liver disease. Ecotoxicol. Environ. Saf. 228, 112986 (2021).
Migliore, L. & Coppedè, F. Gene-environment interactions in Alzheimer disease: the emerging role of epigenetics. Nat. Rev. Neurol. 18, 643–660 (2022).
Surendran, P. et al. Rare and common genetic determinants of metabolic individuality and their effects on human health. Nat. Med. 28, 2321–2332 (2022).
Luukkonen, P. K. et al. The PNPLA3 I148M variant increases ketogenesis and decreases hepatic de novo lipogenesis and mitochondrial function in humans. Cell Metab. 35, 1887–1896.e1885 (2023).
Luo, K. et al. Variant of the lactase LCT gene explains association between milk intake and incident type 2 diabetes. Nat. Metab. 6, 169–186 (2024).
Solon-Biet, S. M. et al. Branched chain amino acids impact health and lifespan indirectly via amino acid balance and appetite control. Nat. Metab. 1, 532–545 (2019).
Rom, O. et al. Glycine-based treatment ameliorates NAFLD by modulating fatty acid oxidation, glutathione synthesis, and the gut microbiome. Sci. Transl. Med. 12, eaaz2841 (2020).
Ghrayeb, A. et al. Serine synthesis via reversed SHMT2 activity drives glycine depletion and acetaminophen hepatotoxicity in MASLD. Cell Metab. 36, 116–129.e117 (2024).
Fu, A. et al. Glucose metabolism and pyruvate carboxylase enhance glutathione synthesis and restrict oxidative stress in pancreatic islets. Cell Rep. 37, 110037 (2021).
Cao, Z. et al. Cohort profile: China National Human Biomonitoring (CNHBM)-A nationally representative, prospective cohort in Chinese population. Environ. Int. 146, 106252 (2021).
Talmud, P. J. et al. Linkage of the cholesteryl ester transfer protein (CETP) gene to LDL particle size: use of a novel tetranucleotide repeat within the CETP promoter. Circulation 101, 2461–2466 (2000).
Reshetnikov, V. V. et al. Genes associated with cognitive performance in the Morris water maze: an RNA-seq study. Sci. Rep. 10, 22078 (2020).
Xue, A. et al. Genome-wide association analyses identify 143 risk variants and putative regulatory mechanisms for type 2 diabetes. Nat. Commun. 9, 2941 (2018).
Tokarz, J. et al. Endocrinology meets metabolomics: achievements, pitfalls, and challenges. Trends Endocrinol. Metab. 28, 705–721 (2017).
Shuai, Y. et al. TEAD4 modulated LncRNA MNX1-AS1 contributes to gastric cancer progression partly through suppressing BTG2 and activating BCL2. Mol. Cancer 19, 6 (2020).
Podleśny-Drabiniok, A. et al. BHLHE40/41 regulate microglia and peripheral macrophage responses associated with Alzheimer’s disease and other disorders of lipid-rich tissues. Nat. Commun. 15, 2058 (2024).
Bae, M. et al. SLC6A20 transporter: a novel regulator of brain glycine homeostasis and NMDAR function. EMBO Mol. Med. 13, e12632 (2021).
Chretien, Y. et al. Bile acid glycine and taurine conjugates in serum of patients with primary biliary cirrhosis: effect of ursodeoxycholic treatment. Gut 30, 1110–1115 (1989).
Mahajan, A. et al. Fine-mapping type 2 diabetes loci to single-variant resolution using high-density imputation and islet-specific epigenome maps. Nat. Genet. 50, 1505–1513 (2018).
Manolio, T. A. et al. Finding the missing heritability of complex diseases. Nature 461, 747–753 (2009).
Thomas, D. Gene-environment-wide association studies: emerging approaches. Nat. Rev. Genet. 11, 259–272 (2010).
Ritchie, M. D., Holzinger, E. R., Li, R., Pendergrass, S. A. & Kim, D. Methods of integrating data to uncover genotype-phenotype interactions. Nat. Rev. Genet. 16, 85–97 (2015).
Turner, A. W. et al. Single-nucleus chromatin accessibility profiling highlights regulatory mechanisms of coronary artery disease risk. Nat. Genet. 54, 804–816 (2022).
Todeschini, A. L., Georges, A. & Veitia, R. A. Transcription factors: specific DNA binding and specific gene regulation. Trends Genet. 30, 211–219 (2014).
Deplancke, B., Alpern, D. & Gardeux, V. The genetics of transcription factor DNA binding variation. Cell 166, 538–554 (2016).
Honma, S. et al. Dec1 and Dec2 are regulators of the mammalian molecular clock. Nature 419, 841–844 (2002).
Yao, W. et al. Transcriptome analysis reveals a protective role of liver X receptor alpha against silica particle-induced experimental silicosis. Sci. Total Environ. 747, 141531 (2020).
Andersen, M. E. et al. Assessing molecular initiating events (MIEs), key events (KEs) and modulating factors (MFs) for styrene responses in mouse lungs using whole genome gene expression profiling following 1-day and multi-week exposures. Toxicol. Appl. Pharmacol. 335, 28–40 (2017).
Andersen, M. E. et al. Combining transcriptomics and PBPK modeling indicates a primary role of hypoxia and altered circadian signaling in dichloromethane carcinogenicity in mouse lung and liver. Toxicol. Appl. Pharmacol. 332, 149–158 (2017).
Hua, W. et al. Transgenerational effects of arsenic exposure on learning and memory in rats: crosstalk between arsenic methylation, hippocampal metabolism, and histone modifications. Environ. Sci. Technol. 58, 6475–6486 (2024).
Pajoro, A., Severing, E., Angenent, G. C. & Immink, R. G. H. Histone H3 lysine 36 methylation affects temperature-induced alternative splicing and flowering in plants. Genome Biol. 18, 102 (2017).
Brown, M. S. & Goldstein, J. L. The SREBP pathway: regulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor. Cell 89, 331–340 (1997).
Martens, N. et al. Activation of liver X receptors and peroxisome proliferator-activated receptors by lipid extracts of brown seaweeds: a potential application in Alzheimer’s disease? Nutrients 15, 3004 (2023).
Darnerud, P. O., Eriksen, G. S., Jóhannesson, T., Larsen, P. B. & Viluksela, M. Polybrominated diphenyl ethers: occurrence, dietary exposure, and toxicology. Environ. Health Perspect. 109, 49–68 (2001).
Bröer, S. Amino acid transport across mammalian intestinal and renal epithelia. Physiol. Rev. 88, 249–286 (2008).
GTEx Consortium. The GTEx Consortium atlas of genetic regulatory effects across human tissues. Science. 369, 1318–1330 (2020).
Yue, J. T., Mighiu, P. I., Naples, M., Adeli, K. & Lam, T. K. Glycine normalizes hepatic triglyceride-rich VLDL secretion by triggering the CNS in high-fat fed rats. Circ. Res. 110, 1345–1354 (2012).
Guzior, D. V. & Quinn, R. A. Review: microbial transformations of human bile acids. Microbiome 9, 140 (2021).
Venkatesh, R., Srinivasan, K. & Singh, S. A. Effect of arginine:lysine and glycine:methionine intake ratios on dyslipidemia and selected biomarkers implicated in cardiovascular disease: a study with hypercholesterolemic rats. Biomed. Pharmacother. 91, 408–414 (2017).
Gao, H. et al. Perigestational low-dose BDE-47 exposure alters maternal serum metabolome and results in sex-specific weight gain in adult offspring. Chemosphere 233, 174–182 (2019).
Smith, G. D. & Ebrahim, S. Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease?. Int. J. Epidemiol. 32, 1–22 (2003).
Borén, J., Taskinen, M. R., Björnson, E. & Packard, C. J. Metabolism of triglyceride-rich lipoproteins in health and dyslipidaemia. Nat. Rev. Cardiol. 19, 577–592 (2022).
Jin, L. et al. Dynamic chromatin architecture of the porcine adipose tissues with weight gain and loss. Nat. Commun. 14, 3457 (2023).
Gianì, F. et al. Antioxidant defense capacity is reduced in thyroid stem/precursor cells compared to differentiated thyrocytes. Int. J. Mol. Sci. 24, 11509 (2023).
Karunakaran, D. et al. RIPK1 expression associates with inflammation in early atherosclerosis in humans and can be therapeutically silenced to reduce NF-κB activation and atherogenesis in mice. Circulation 143, 163–177 (2021).
Jiang, Y. et al. Hemocytes in blue mussel Mytilus edulis adopt different energy supply modes to cope with different BDE-47 exposures. Sci. Total Environ. 885, 163766 (2023).
Xia, B. et al. BDE-47 induces metabolic dysfunction-associated steatotic liver disease (MASLD) through CD36-mediated increased fatty acid uptake and PPARα-induced abnormal fatty acid oxidation in BALB/c mice. Toxicol. Lett. 391, 100–110 (2024).
Van Asselt, A. J. & Ehli, E. A. Whole-genome genotyping using DNA microarrays for population genetics. Methods Mol. Biol. 2418, 269–287 (2022).
An, S. et al. MetaPro: a web-based metabolomics application for LC-MS data batch inspection and library curation. Metabolomics 19, 57 (2023).
Sumner, L. W. et al. Proposed minimum reporting standards for chemical analysis Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics 3, 211–221 (2007).
Phapale, P., Rai, V., Mohanty, A. K. & Srivastava, S. Untargeted metabolomics workshop report: quality control considerations from sample preparation to data analysis. J. Am. Soc. Mass Spectrom. 31, 2006–2010 (2020).
Ma, Y., Zhao, Y., Zhang, J. F. & Bi, W. Efficient and accurate framework for genome-wide gene-environment interaction analysis in large-scale biobanks. Nat. Commun. 16, 3064 (2025).
Dong, Z. et al. Incorporating additive genetic effects and linkage disequilibrium information to discover gene-environment interactions using BV-LDER-GE. Genome Biol. 26, 332 (2025).
Cai, Y. et al. An atlas of genetic effects on cellular composition of the tumor microenvironment. Nat. Immunol. 25, 1959–1975 (2024).
Luo, Y. et al. New developments on the Encyclopedia of DNA Elements (ENCODE) data portal. Nucleic Acids Res. 48, D882–d889 (2020).
de Leeuw, C. A., Mooij, J. M., Heskes, T. & Posthuma, D. MAGMA: generalized gene-set analysis of GWAS data. PLoS Comput. Biol. 11, e1004219 (2015).
Gene Ontology, C. Gene ontology consortium: going forward. Nucleic Acids Res. 43, D1049–D1056 (2015).
Wang, H. et al. Identification of specific susceptibility loci for the early-onset colorectal cancer. Genome Med. 15, 13 (2023).
Rentzsch, P., Witten, D., Cooper, G. M., Shendure, J. & Kircher, M. CADD: predicting the deleteriousness of variants throughout the human genome. Nucleic Acids Res. 47, D886–d894 (2019).
Boyle, A. P. et al. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res. 22, 1790–1797 (2012).
Quan, C., Ping, J., Lu, H., Zhou, G. & Lu, Y. 3DSNP 2.0: update and expansion of the noncoding genomic variant annotation database. Nucleic Acids Res. 50, D950–d955 (2022).
Li, J. et al. Association of di(2-ethylhexyl) terephthalate and its metabolites with nonalcoholic fatty liver disease: an epidemiology and toxicology study. Environ. Sci. Technol. 58, 8182–8193 (2024).
Zhu, L., Ma, B. & Hites, R. A. Brominated flame retardants in serum from the general population in northern China. Environ. Sci. Technol. 43, 6963–6968 (2009).
Chong, J. et al. MetaboAnalyst 4.0: towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 46, W486–w494 (2018).
Acknowledgements
This research was supported by the National Key R&D Program of China (2022YFA0806600, 2024YFC3405804), the National Natural Science Foundation of China (82388102, 82230111, 82025030, 82222063, 82404355 U22A20404), the National Science Fund for Excellent Young Scholars (NSFC-82322058), the Young Elite Scientists Sponsorship Program by CAST (2022QNRC001), Open Fund of the Key Laboratory of Environmental and Population Health, Chinese Center for Disease Control and Prevention (2026-CKL-01), the National Science Fund for Distinguished Young Scholars of Hubei Province of China (2023AFA046), the Fundamental Research Funds for the Central Universities (2042025kf0027), the National Key R&D Program of China (2024YFC3405803) and National Natural Science Foundation of China (NSFC-82373663). We also thank the staff and participants of CNHBM for their important contributions in the cohort establishment and follow-up.
Author information
Authors and Affiliations
Contributions
N.H., B.L., and Y.L drafted the manuscript, with Q.J. revising and finalizing it. N.H., Q.J., and Y.Z.1 conducted the statistical analyses and interpreted the results. Y.L. and Z.L. coordinated cohort data collection, sample testing, and omics profiling. N.H., B.L., D.Z., Y.M., H.J., and Y.F.Z. conducted experimental work. D.Z., Z.W., Y.H.Z., and M.Z. processed data and prepared the visualizations. Y.Q. T.Q., Y.L., P.S., H.S., F.Z., and S.J. participated in population investigation and specimen collection. B.Y. provided technical expertise in pollutant and metabolite quantification. Y.Z.2, Y.L., X.M., J.T., and X.S. conceived the study design and supervised the project. All authors critically reviewed and approved the final manuscript for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Peer review
Peer review information
Nature Communications thanks Jean-Baptiste Fini and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hu, N., Li, B., Lu, Y. et al. Genetic variations interact with polybrominated diphenyl ether exposure to alter lipid homeostasis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70222-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70222-8