Abstract
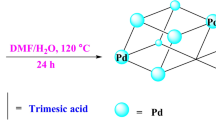
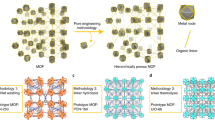
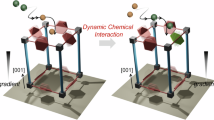
Immobilizing enzymes in a metal–organic framework (MOF) is an effective approach to improve their stability and reusability, but the small pore sizes of most MOFs exclude many industrially relevant enzymes. Here, we present a dynamic bond-mediated approach that enables enzyme encapsulation beyond pore size constraints. We constructed a series of mesoporous MOFs by integrating robust trivalent metal–carboxylate clusters with dynamic divalent metal–pyridyl units. Systematic variation of metal combinations and linker lengths precisely tunes the framework stability and dynamics, enabling reversible dissociation and reformation of metal–pyridyl bonds. These dynamic bonds function as molecular gates, permitting enzymes larger than the intrinsic pores to infiltrate while preserving framework integrity. The strategy was applied to encapsulate diverse enzymes, preserving high enzymatic activity and enhancing operational stability. Furthermore, it supports the co-immobilization of multi-enzyme systems, such as NahK and GlmU, for efficient cascade synthesis of high-value glycosylated donors.
Similar content being viewed by others
Data availability
Previously published protein structures used in this study were obtained from the Protein Data Bank (PDB) under the following accession codes: 3CP5 (Cyt C) 1W4Y (HRP) 7×32 [https://doi.org/10.2210/pdb7X32/pdb] (NTR) 1TRH (Lipase) 3VIK (BGL) 4OCO (NahK) 1FWY (GlmU) Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Center, under deposition numbers CCDC 2489486 (Cr-L1-Cu), 2489487 (Cr-L1-Pd),2489488 (Fe-L1-Cu), and 2489489 (Cr-L3-Pd). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within the article and its supplementary information. Source data are provided with this paper.
References
Buller, R. et al. From nature to industry: harnessing enzymes for biocatalysis. Science 382, eadh8615 (2023).
Vanella, R. et al. Understanding activity-stability tradeoffs in biocatalysts by enzyme proximity sequencing. Nat. Commun. 15, 2024 (1807).
Wang, K.-Y. et al. Bioinspired framework catalysts: from enzyme immobilization to biomimetic catalysis. Chem. Rev. 123, 5347–5420 (2023).
Chen, Y. et al. How can proteins enter theinterior of a MOF? investigation of cytochrome c translocation into a MOF consisting of mesoporous cages with microporous windows. J. Am. Chem. Soc. 134, 13188–13191 (2012).
Cheng, C. et al. Construction of degradable liposome-templated microporous metal-organic frameworks with commodious space for enzymes. Chin. Chem. Lett. 35, 109812 (2024).
Deng, H. et al. Large-pore apertures in a series of metal-organic frameworks. Science 336, 1018–1023 (2012).
Feng, D. et al. Stable metal-organic frameworks containing single-molecule traps for enzyme encapsulation. Nat. Commun. 6, 5979 (2015).
Liu, Y., Zhou, B., Zhang, S., Cao, C. & Qi, L. Fabrication of hierarchically mesoporous multishelled Metal–Organic Frameworks via template-induced assembly/grinding strategy for immobilization of enzyme and enhancing catalytic performance. ACS Appl. Mater. Interfaces 17, 58905–58913 (2025).
Wang, S. & Urban, M. W. Self-healing polymers. Nat. Rev. Mater. 5, 562–583 (2020).
Yuan, Y. et al. Hyaluronic acid-modified MOF nanoparticles for encapsulating asparaginase in T-cell acute lymphoblastic leukemia treatment. Chin. Chem. Lett. 37, 111222 (2026).
Chowdhury, R., Stromer, B., Pokharel, B. & Kumar, C. V. Control of enzyme–solid interactions via chemical modification. Langmuir 28, 11881–11889 (2012).
Chen, S.-Y. et al. Probing interactions between Metal–Organic Frameworks and freestanding enzymes in a hollow structure. Nano Lett 20, 6630–6635 (2020).
Liang, K. et al. Biomimetic mineralization of metal-organic frameworks as protective coatings for biomacromolecules. Nat. Commun. 6, 7240 (2015).
Wang, X. et al. A solid-state crystallization strategy for direct enzyme encapsulation in Zr-MOFs: eliminating harsh pH and thermal requirements of liquid-phase synthesis. Angew. Chem. Int. Ed. 64, e202509275 (2025).
Lin, S.-W. et al. Decoding the biomimetic mineralization of Metal–Organic Frameworks in water. ACS Nano 18, 25170–25182 (2024).
Wei, T.-H. et al. Rapid mechanochemical encapsulation of biocatalysts into robust metal–organic frameworks. Nat. Commun. 10, 5002 (2019).
Lyu, F., Zhang, Y., Zare, R. N., Ge, J. & Liu, Z. One-pot synthesis of protein-embedded Metal–Organic Frameworks with enhanced biological activities. Nano Letters 14, 5761–5765 (2014).
Feng, Y. et al. A dynamic defect generation strategy for efficient enzyme immobilization in robust metal–organic frameworks for catalytic hydrolysis and chiral resolution. Angew. Chem. Int. Ed. 62, e202302436 (2023).
Pisklak, T. J., Macías, M., Coutinho, D. H., Huang, R. S. & Balkus, K. J. Hybrid materials for immobilization of MP-11 catalyst. Top. Catal. 38, 269–278 (2006).
Li, Z., Rayder, T. M., Luo, L., Byers, J. A. & Tsung, C.-K. Aperture-opening encapsulation of a transition metal catalyst in a metal–organic framework for CO2 hydrogenation. J. Am. Chem. Soc. 140, 8082–8085 (2018).
Morabito, J. V. et al. Molecular encapsulation beyond the aperture size limit through dissociative linker exchange in Metal–Organic Framework crystals. J. Am. Chem. Soc. 136, 12540–12543 (2014).
Liu, Q., Cong, H. & Deng, H. Deciphering the spatial arrangement of metals and correlation to reactivity in multivariate Metal–Organic Frameworks. J. Am. Chem. Soc. 138, 13822–13825 (2016).
Wang, K. et al. A series of highly stable mesoporous metalloporphyrin Fe-MOFs. J. Am. Chem. Soc. 136, 13983–13986 (2014).
Shahnawaz Khan, M., Khalid, M. & Shahid, M. What triggers dye adsorption by metal organic frameworks? The current perspectives. Mater. Adv. 1, 1575–1601 (2020).
Liu, Q. et al. Mesoporous cages in chemically robust MOFs are created by a large number of vertices with reduced connectivity. J. Am. Chem. Soc. 141, 488–496 (2019).
Schoedel, A. et al. Network diversity through decoration of trigonal-prismatic nodes: two-step crystal engineering of cationic metal–organic materials. Angew. Chem. Int. Ed. 123, 11623–11626 (2011).
Zhou, J. et al. Linking oxidative and reductive clusters to prepare crystalline porous catalysts for photocatalytic CO2 reduction with H2O. Nat. Commun. 13, 4681 (2022).
Schoedel, A. et al. The asc trinodal platform: two-step assembly of triangular, tetrahedral, and trigonal-prismatic molecular building blocks. Angew. Chem. Int. Ed. 52, 2902–2905 (2013).
Rieth, A. J., Wright, A. M. & Dincă, M. Kinetic stability of metal–organic frameworks for corrosive and coordinating gas capture. Nat. Rev. Mater. 4, 708–725 (2019).
Deria, P. et al. Perfluoroalkane functionalization of NU-1000 via solvent-assisted ligand incorporation: synthesis and CO2 adsorption studies. J. Am. Chem. Soc. 135, 16801–16804 (2013).
Giannozzi, P. et al. Advanced capabilities for materials modelling with Quantum ESPRESSO. J. Phys.: Condens. Matter 29, 465901 (2017).
Giannozzi, P. et al. Quantum ESPRESSO: a modular and open-source software project for quantum simulations of materials. J. Phys.: Condens. Matter 21, 395502 (2009).
Giannozzi, P. et al. Quantum ESPRESSO toward the exascale. J. Chem. Phys. 152, 154105 (2020).
Li, P. et al. Toward design rules for enzyme immobilization in hierarchical mesoporous Metal-Organic Frameworks. Chem 1, 154–169 (2016).
Qin, W. et al. Recent progress in small molecule fluorescent probes for nitroreductase. Chin. Chem. Lett. 29, 1451–1455 (2018).
Kienle, D. F., Falatach, R. M., Kaar, J. L. & Schwartz, D. K. Correlating structural and functional heterogeneity of immobilized enzymes. ACS Nano 12, 8091–8103 (2018).
Zou, X. et al. Investigating the effect of two-point surface attachment on enzyme stability and activity. J. Am. Chem. Soc. 140, 16560–16569 (2018).
Li, W. et al. Protein β-O-glucosylation by Legionella LtpM through short consensus sequons G-T/S and S-G. Nat. Chem. Biol. 21, 1086–18177 (2025).
Balana, A. T. et al. Post-translational glycosylation diminishes α-synuclein pathology formation. Nat. Chem. Biol. 20, 553–554 (2024).
Chen, X. et al. Glycosaminoglycans modulate long-range mechanical communication between cells in collagen networks. Proc. Natl. Acad. Sci. 119, e2116718119 (2022).
Masuko, S. et al. Chemoenzymatic synthesis of uridine diphosphate-GlcNAc and uridine diphosphate-GalNAc analogs for the preparation of unnatural glycosaminoglycans. J. Org. Chem. 77, 1449–1456 (2012).
Schultz, V. L. et al. Chemoenzymatic synthesis of 4-fluoro-N-acetylhexosamine uridine diphosphate donors: chain terminators in glycosaminoglycan synthesis. J. Org. Chem. 82, 2243–2248 (2017).
Gorbitz, C. What is the best crystal size for the collection of X-ray data? Refinement of the structure of glycyl-l-serine based on data from a very large crystal. Acta Crystallogr. Sect. B 55, 1090–1098 (1999).
Sheldrick, G. A short history of SHELX. Acta Crystallogr. A 64, 112–122 (2008).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Schlipf, M. & Gygi, F. Optimization algorithm for the generation of ONCV pseudopotentials. Comput. Phys. Commun. 196, 36–44 (2015).
van Setten, M. J. et al. The PseudoDojo: Training and grading an 85-element optimized norm-conserving pseudopotential table. Comput. Phys. Commun. 226, 39–54 (2018).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Acknowledgments
This work was supported by the National Key Research and Development Program of China (2025YFA1511500; S.Y., Y.C.L.), the National Natural Science Foundation of China (223B2107, 22271141, T2541071; S.Y., Y.C.L., X.Z., M.Q.), and the Natural Science Foundation of Jiangsu Province (BK20250064, BK20240032; S.Y., Y.C.L.). We thank the staff of the BL17B beamline (https://cstr.cn/31129.02.NFPS.BL17B) at the National Facility for Protein Science in Shanghai (NFPS, https://cstr.cn/31129.02.NFPS), Shanghai Advanced Research Institute, Chinese Academy of Sciences, for their technical support in Single-crystal XRD/PXRD. The theoretical calculations were conducted using the computing facilities of the High-Performance Computing Center (HPCC) at Nanjing University.
Author information
Authors and Affiliations
Contributions
Y.C.L. and M.Q. contributed equally to this work. S.Y., J.-L.Z., and X.Z. conceived the original idea. Y.C.L. and M.Q. performed the synthesis. Y.C.L. and M.Q. performed the comprehensive structural characterization, property measurements, and data analysis. L.G. conducted the DFT calculations. Y.X.Z. performed the ICP-OES measurements. Y.Y. and J.Z. performed the CLSM measurements. Y.C.L., M.Q., S.Y., J.-L.Z., and X.Z. drafted the manuscript. All authors contributed to the revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Lien-Yang Chou, Fa-Kuen Shieh and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Qiao, M., Gao, L. et al. Dynamic bond-driven encapsulation of enzymes in metal–organic frameworks beyond pore size constraints. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70249-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70249-x