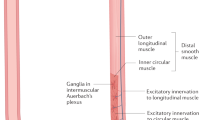
Abstract
Large animal models, while valuable, are expensive, time-consuming, and limited to discrete interventional or terminal timepoints, while existing benchtop models do not offer an accurate representation of the esophageal environment. Moreover, current pre-clinical models cannot effectively simulate swallowing dysfunction (dysphagia), restricting progress in understanding motility disorders like achalasia and hindering evidence-based dietary recommendations. In response, we present RoboGullet, a biomimetic soft-robotic model with independent localized longitudinal and circumferential muscle actuation, enabling, for the first time, simulation of both normal and diseased esophageal motility. We further enhance realism with a biohybrid variant, RoboGullet + , incorporating porcine esophageal mucosa/submucosa. We demonstrate this platform’s versatility through three key applications: assessing stent migration, simulating achalasia I-III within clinical diagnostic criteria, and analyzing bolus swallowing. Our findings reveal that: (1) stent migration increases over fivefold when incorporating longitudinal muscle movement versus isolated circumferential; (2) using a viscous non-Newtonian bolus improves high-resolution manometry diagnostic sensitivity of Achalasia III through increasing the Distal Latency diagnostic metric by 20.83%; and (3) stirring Greek-style yoghurt (common non-Newtonian dietary recommendation) significantly improves bolus transit versus unstirred for Achalasia Types I-II patients. This establishes RoboGullet+ as a powerful translational tool, advancing our understanding of esophageal motility and its therapeutic interventions.
Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information files. Any additional requests for information can be directed to the corresponding author. Source data is provided with this paper. Source data are provided with this paper.
References
Meyer, G. W. & Castell, D. O. 1—Physiology of the ōesophagus. Clin. Gastroenterol. 11, 439–451 (1982).
Patel, D. A., Yadlapati, R. & Vaezi, M. F. Esophageal motility disorders: current approach to diagnostics and therapeutics. Gastroenterology 162, 1617–1634 (2022).
Adkins, C. et al. Prevalence and characteristics of dysphagia based on a population-based survey. Clin. Gastroenterol. Hepatol. 18, 1970–1979.e2 (2020).
Pandolfino, J. E. & Gawron, A. J. Achalasia: a systematic review. J. Am. Med. Assoc. 313, 1841 (2015).
Mitra, T. et al. Clinical profile of patients presenting with dysphagia - an experience from a tertiary care center in North India. JGH Open 4, 472–476 (2019).
Sharma, P. & Kozarek, R. Role of esophageal stents in benign and malignant diseases. Am. J. Gastroenterol. 105, 258–273 (2010).
Vleggaar, F. P. & Siersema, P. D. Expandable stents for malignant esophageal disease. Gastrointest. Endosc. Clin. N. Am. 21, 377–388 (2011).
Thomas, S. et al. Fully-covered esophageal stent migration rates in benign and malignant disease: a multicenter retrospective study. Endosc. Int. Open 7, E751–E756 (2019).
Patel, D. A. & Vaezi, M. F. Achalasia and nutrition: is it simple physics or biology? Pract. Gastro. 40, 42–48 (2016).
Patel, D. A., Lappas, B. M. & Vaezi, M. F. An overview of achalasia and its subtypes. Gastroenterol. Hepatol. 13, 411–421 (2017).
Yadlapati, R. et al. Esophageal motility disorders on high-resolution manometry: Chicago classification version 4.0©. Neurogastroenterol. Motil. 33, e14058 (2021).
Edoardo, S. et al. Achalasia (Primer). Nat. Rev. Dis. Prim. 8, 28 (2022).
Basseri, B. et al. Apple sauce improves detection of esophageal motor dysfunction during high-resolution manometry evaluation of dysphagia. Dig. Dis. Sci. 56, 1723–1728 (2011).
Blonski, W. et al. Impedance manometry with viscous test solution increases detection of esophageal function defects compared to liquid swallows. Scand. J. Gastroenterol. 42, 917–922 (2007).
Hirano, I. Pathophysiology of achalasia and diffuse esophageal spasm. GI Motil. Online https://doi.org/10.1038/gimo22 (2006).
Park, C., Singh, M., Saeed, M. Y., Nguyen, C. T. & Roche, E. T. Biorobotic hybrid heart as a benchtop cardiac mitral valve simulator. Device 2, 100217 (2024).
Dynamic Digestion Models: General Introduction. in The Impact of Food Bioactives on Health (eds Verhoeckx, K. et al.) (Springer International Publishing, Cham, 2015). https://doi.org/10.1007/978-3-319-16104-4.
Alici, G. Softer is harder: what differentiates soft robotics from hard robotics?. MRS Adv. 3, 1557–1568 (2018).
Slawinski, P. & Terry, B. An automated intestinal biomechanics simulator for expediting robotic capsule endoscope development1. J. Med. Devices 8, 030901 (2014).
Condino, S. et al. Stomach simulator for analysis and validation of surgical endoluminal robots. Appl. Bionics Biomech. 8, 267–277 (2011).
Tharakan, A., Norton, I., Fryer, P. & Bakalis, S. Mass transfer and nutrient absorption in a simulated model of small intestine. J. Food Sci. 75, E339–E346 (2010).
Li, Y., Fortner, L. & Kong, F. Development of a gastric simulation model (GSM) incorporating gastric geometry and peristalsis for food digestion study. Food Res. Int. 125, 108598 (2019).
Bhattacharya, D., Ali, S. J. V., Cheng, L. K. & Xu, W. RoSE: a robotic soft esophagus for endoprosthetic stent testing. Soft Robot. 8, 397–415 (2021).
Dang, Y. et al. SoGut: a soft robotic gastric simulator. Soft Robot. 8, 273–283 (2021).
Nicosia, M. A., Brasseur, J. G., Liu, J.-B. & Miller, L. S. Local longitudinal muscle shortening of the human esophagus from high-frequency ultrasonography. Am. J. Physiol. 281, G1022–G1033 (2001).
Peerlinck, S., Willemyns, F., Reynaerts, D. & Gorissen, B. Biomimetic small intestinal peristalsis simulator using circumferential pneumatic artificial muscles (cirPAM). Adv. Mater. Technol. 9, 2301662 (2024).
Jamil, B., Oh, N., Lee, J.-G., Lee, H. & Rodrigue, H. A review and comparison of linear pneumatic artificial muscles. Int. J. Precis. Eng. Manuf. 11, 277–289 (2024).
Ge, J. Z., Calderon, A. A. & Perez-Arancibia, N. O. An earthworm-inspired soft crawling robot controlled by friction. in 2017 IEEE International Conference on Robotics and Biomimetics (ROBIO) 834–841 (IEEE, Macau, 2017). https://doi.org/10.1109/ROBIO.2017.8324521.
Park, C. et al. An organosynthetic dynamic heart model with enhanced biomimicry guided by cardiac diffusion tensor imaging. Sci. Robot. 5, eaay9106 (2020).
Wirekoh, J. & Park, Y.-L. Design of flat pneumatic artificial muscles. Smart Mater. Struct. 26, 035009 (2017).
Jung, H.-Y. et al. Asynchrony between the circular and the longitudinal muscle contraction in patients with nutcracker esophagus. Gastroenterology 128, 1179–1186 (2005).
Korsapati, H. et al. Dysfunction of the longitudinal muscles of the oesophagus in eosinophilic oesophagitis. Gut 58, 1056–1062 (2009).
Mittal, R. K., Hong, S. J. & Bhargava, V. Longitudinal muscle dysfunction in achalasia esophagus and its relevance. J. Neurogastroenterol. Motil. 19, 126–136 (2013).
Mittal, R. K. Regulation and dysregulation of esophageal peristalsis by the integrated function of circular and longitudinal muscle layers in health and disease. Am. J. Physiol. 311, G431–G443 (2016).
Joe, S., Totaro, M., Wang, H. & Beccai, L. Development of the ultralight hybrid pneumatic artificial muscle: modelling and optimization. PLoS ONE 16, e0250325 (2021).
Edwards, C. A. & Bohlen, P. J. Biology and Ecology of Earthworms. (Springer Science & Business Media, 1996).
Brasseur, J. G., Nicosia, M. A., Pal, A. & Miller, L. S. Function of longitudinal vs circular muscle fibers in esophageal peristalsis, deduced with mathematical modeling. World J. Gastroenterol. 13, 1335–1346 (2007).
Yang, W., Fung, T. C., Chian, K. S. & Chong, C. K. Instability of the two-layered thick-walled esophageal model under the external pressure and circular outer boundary condition. J. Biomech. 40, 481–490 (2007).
Liao, D. The oesophageal zero-stress state and mucosal folding from a GIOME perspective. World J. Gastroenterol. 13, 1347 (2007).
Carass, A. et al. Evaluating white matter lesion segmentations with refined Sørensen-Dice analysis. Sci. Rep. 10, 8242 (2020).
Arkin, E. M., Chew, L. P., Huttenlocher, D. P., Kedem, K. & Mitchell, J. S. B. An efficiently computable metric for comparing polygonal shapes. IEEE Trans. Pattern Anal. Mach. Intell. 13, 209–216 (1991).
Chen, F. J., Dirven, S., Xu, W. L. & Li, X. N. Soft actuator mimicking human esophageal peristalsis for a swallowing robot. IEEEASME Trans. Mechatron. 19, 1300–1308 (2014).
Miyashita, S. et al. Ingestible, controllable, and degradable origami robot for patching stomach wounds. in 2016 IEEE International Conference on Robotics and Automation (ICRA) pp 909–916. https://doi.org/10.1109/ICRA.2016.7487222 (2016).
Gyawali, C. P. & Kahrilas, P. J. A short history of high-resolution esophageal manometry. Dysphagia 38, 586–595 (2021).
Brasseur, J. G. & Dodds, W. J. Interpretation of intraluminal manometric measurements in terms of swallowing mechanics. Dysphagia 6, 100–119 (1991).
Passaretti, S. et al. Standards for oesophageal manometry A position statement from the Gruppo Italiano di Studio Motilità Apparato Digerente (GISMAD). Dig. Liver Dis. 32, 46–55 (2000).
Clouse, R. E. & Staiano, A. Topography of the esophageal peristaltic pressure wave. Am. J. Physiol. 261, G677–G684 (1991).
Peirlinck, M. Design of Biodegradable Esophageal Stents. (Universiteit Gent, Ghent, Belgium, 2013).
Silva, R. Esophageal stenting: how i do it. GE Port. J. Gastroenterol. 30, 35–44 (2023).
Ferhatoglu, M. F. & Kıvılcım, T. Anatomy of Esophagus. in Esophageal Abnormalities (ed Chai, J.) (IntechOpen, Rijeka, 2017). https://doi.org/10.5772/intechopen.69583.
Mittal, R. K. & Balaban, D. H. The esophagogastric junction. N. Engl. J. Med. 336, 924–932 (1997).
Mowlavi, S. et al. In vivo observations and in vitro experiments on the oral phase of swallowing of Newtonian and shear-thinning liquids. J. Biomech. 49, 3788–3795 (2016).
McCullough, G., Pelletier, C. & Steele, C. National dysphagia diet: what to swallow? ASHA Lead Arch. 8, 16–27 (2003).
Stone, C.-B. Achalasia: nutrition therapy. AGA GI Patient Center https://patient.gastro.org/achalasia-nutrition-therapy/ (2021).
Li, Y. & Kong, F. Simulating human gastrointestinal motility in dynamic in vitro models. Compr. Rev. Food Sci. Food Saf. 21, 3804–3833 (2022).
Mozafari, H. et al. Migration resistance of esophageal stents: the role of stent design. Comput. Biol. Med. 100, 43–49 (2018).
Christie, K. N., Thomson, C. & Hopwood, D. A comparison of membrane enzymes of human and pig oesophagus; the pig oesophagus is a good model for studies of the gullet in man. Histochem. J. 27, 231–239 (1995).
Durcan, C. et al. Experimental investigations of the human oesophagus: anisotropic properties of the embalmed muscular layer under large deformation. Biomech. Model. Mechanobiol. 21, 1169–1186 (2022).
Yang, W., Fung, T. C., Chian, K. S. & Chong, C. K. Finite element simulation of food transport through the esophageal body. World J. Gastroenterol. 13, 1352–1359 (2007).
Yang, W., Fung, T. C., Chian, K. S. & Chong, C. K. Directional, regional, and layer variations of mechanical properties of esophageal tissue and its interpretation using a structure-based constitutive model. J. Biomech. Eng. 128, 409–418 (2005).
Lin, C. X. et al. Friction behavior between endoscopy and esophageal internal surface. Wear 376–377, 272–280 (2017).
O’Leary, S. A., Doyle, B. J. & McGloughlin, T. M. The impact of long term freezing on the mechanical properties of porcine aortic tissue. J. Mech. Behav. Biomed. Mater. 37, 165–173 (2014).
Caon, T. & Simões, C. M. O. Effect of freezing and type of mucosa on ex vivo drug permeability parameters. AAPS PharmSciTech 12, 587–592 (2011).
Das, K. K. et al. Performance and predictors of migration of partially and fully covered esophageal self-expanding metal stents for malignant dysphagia. Clin. Gastroenterol. Hepatol. 19, 2656–2663.e2 (2021).
Jaber, F. et al. A comprehensive analysis of reported adverse events and device failures associated with esophageal self-expandable metal stents: an FDA MAUDE Database Study. Dig. Dis. Sci. 69, 2765–2774 (2024).
Seven, G. et al. Partially versus fully covered self-expanding metal stents for benign and malignant esophageal conditions: a single center experience. Surg. Endosc. 27, 2185–2192 (2013).
Hong, S. J., Bhargava, V., Jiang, Y., Denboer, D. & Mittal, R. K. A unique esophageal motor pattern that involves longitudinal muscles is responsible for emptying in achalasia esophagus. Gastroenterology 139, 102–111 (2010).
Salvador, R. et al. Manometric pattern progression in esophageal achalasia in the era of high-resolution manometry. Ann. Transl. Med. 9, 906–906 (2021).
Salvador, R. et al. The natural history of achalasia: Evidence of a continuum—“the evolutive pattern theory”. Dig. Liver Dis. 50, 342–347 (2018).
Blonski, W., Vela, M., Hila, A. & Castell, D. O. Normal values for manometry performed with swallows of viscous test material. Scand. J. Gastroenterol. 43, 155–160 (2008).
Ang, D. et al. Diagnostic yield of high-resolution manometry with a solid test meal for clinically relevant, symptomatic oesophageal motility disorders: serial diagnostic study. Lancet Gastroenterol. Hepatol. 2, 654–661 (2017).
Qazi, W. M., Ekberg, O., Wiklund, J., Kotze, R. & Stading, M. Assessment of the food-swallowing process using bolus visualisation and manometry simultaneously in a device that models human swallowing. Dysphagia 34, 821–833 (2019).
Dellon, E. S. et al. Viscous topical is more effective than nebulized steroid therapy for patients with eosinophilic esophagitis. Gastroenterology 143, 321–324.e1 (2012).
Steiger, C. et al. Ingestible electronics for diagnostics and therapy. Nat. Rev. Mater. 4, 83–98 (2019).
Rafeedi, T. et al. Wearable, epidermal devices for assessment of swallowing function. Npj Flex. Electron. 7, 1–19 (2023).
Kim, Y. et al. Simulator-based training method in gastrointestinal endoscopy training and currently available simulators. Clin. Endosc. 56, 1–13 (2023).
Elisha, G. et al. Modeling based insights into mechanical dysfunction in esophageal motility disorders. PLOS Computational Biology 21, e1013778 (2025).
Peirlinck, M. et al. An in silico biomechanical analysis of the stent–esophagus interaction. Biomech. Model. Mechanobiol. 17, 111–131 (2018).
Sommer, G. et al. Multiaxial mechanical response and constitutive modeling of esophageal tissues: Impact on esophageal tissue engineering. Acta Biomater. 9, 9379–9391 (2013).
Din, S., Xu, W., Cheng, L. K. & Dirven, S. A Stretchable Array of Electronic Receptors for Esophageal Swallowing Robot for Biomimetic Simulations of Bolus Transport. IEEE Sens. J. 18, 5497–5506 (2018).
Dirven, S. et al. Design and Characterization of a Peristaltic Actuator Inspired by Esophageal Swallowing. IEEEASME Trans. Mechatron. 19, 1234–1242 (2014).
Xavier, M. S., Fleming, A. J. & Yong, Y. K. Finite Element Modeling of Soft Fluidic Actuators: Overview and Recent Developments. Adv. Intell. Syst. 3, 2000187 (2021).
Chen, J. et al. Determination of oral mucosal Poisson’s ratio and coefficient of friction from in-vivo contact pressure measurements. Comput. Methods Biomech. Biomed. Engin. 19, 357–365 (2016).
May, A., Nachbar, L., Schneider, M., Neumann, M. & Ell, C. Push-and-pull enteroscopy using the double-balloon technique: method of assessing depth of insertion and training of the enteroscopy technique using the Erlangen Endo-Trainer. Endoscopy 37, 66–70 (2005).
Neumann, M. et al. The Erlangen Endo-Trainer: life-like simulation for diagnostic and interventional endoscopic retrograde cholangiography. Endoscopy 32, 906–910 (2000).
Vick, L. R., Vick, K. D., Borman, K. R. & Salameh, J. R. Face, content, and construct validities of inanimate intestinal anastomoses simulation. J. Surg. Educ. 64, 365–368 (2007).
Lu, X. & Gregersen, H. Regional distribution of axial strain and circumferential residual strain in the layered rabbit oesophagus. J. Biomech. 34, 225–233 (2001).
Karcher, A., Schäfer, J., Cattaneo, G. & Sanchez, D. Development of a measurement setup to determine the frictional properties of tissuemimicking materials for vascular models. Curr. Dir. Biomed. Eng. 10, 356–359 (2024).
Kim, J.-S. et al. Experimental investigation of frictional and viscoelastic properties of intestine for microendoscope application. Tribol. Lett. 22, 143–149 (2006).
Peng, L., Roch, T., Bonn, D. & Weber, B. The decrease of static friction coefficient with interface growth from single to multiasperity contact. Phys. Rev. Lett. 134, 176202 (2025).
Dionisio, P. et al. High resolution esophageal manometry (HRM): topographical mapping of esophageal motor function in scleroderma: 45. J. Am. Coll. Gastroenterol. 103, S18 (2008).
Acknowledgements
The authors thank Dr. Gang Shen for support with frictional testing. N.A.P. gratefully acknowledges the Visiting Professor position in the MIT Department of Mechanical Engineering during his academic leave from Northwestern University in Fall 2023 and Winter 2024. S.K. acknowledges Mark Fitzmaurice and Sword Medical Ltd. for the loan of equipment. Funding Taighde Éireann— Research Ireland grant GOIPG/2023/3917 (SK) Taighde Éireann—Research Ireland grant 13/RC/2073_P2 (E.D.O’C.) Karl van Tassel (1925) Career Development Professorship (GT) Department of Mechanical Engineering, MIT (GT) Division of Gastroenterology, Brigham and Women’s Hospital (GT).
Author information
Authors and Affiliations
Contributions
Conceptualization: S.K., W.W.C., G.T., and E.D.O.’C. Methodology: S.K., G.T., N.A.P., and E.D.O.’C. Investigation: S.K. Funding acquisition: S.K., G.T., and E.D.O.’C. Project administration: G.T. and E.D.O.’C. Supervision: G.T. and E.D.O.’C. Writing—original draft: S.K. Writing—review & editing: S.K., N.A.P., W.W.C., G.T., and E.D.O.’C.
Corresponding authors
Ethics declarations
Competing interests
Complete details of all relationships for profit and not for profit for G.T. can be found at the following link: https://www.dropbox.com/sh/szi7vnr4a2ajb56/AABs5N5i0q9AfT1IqIJAE-T5a?dl=0. The other authors declare that they have no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kilroy, S., Patankar, N.A., Chan, W.W. et al. A Soft-Robotic Biomimetic Benchtop Model for Esophageal Motility Simulation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70260-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70260-2