Abstract
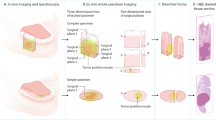
Tumour-free margins ( >5 mm) in head and neck malignancies improve prognosis, possibly impacting the need for adjuvant treatment. In this phase II prospective, non-randomised trial (NCT05499065), we investigate the use of fluorescence imaging using the tumour targeting fluorescent tracer cetuximab-IRDye800CW combined with fresh frozen sectioning to identify inadequate margins intraoperatively in patients with oral squamous cell carcinoma. Primary objectives of our study are intraoperative detection of inadequate margins and additional resections, and feasibility of implementing fluorescence-guided fresh frozen sectioning (FG-FFS) into the clinical workflow. In this study, 10 out of 20 patients show inadequate margins after initial surgery; FG-FFS correctly classifies margin in 19 patients. Additional resections increase tumour-free margin rates from 50% to 85%, where anatomical boundaries limit further improvement in one patient. Intraoperative margin adjustment reduces the need for adjuvant treatment in seven patients. These findings demonstrate that FG-FFS is a reliable and feasible method for intraoperative margin assessment without delaying surgery.
Similar content being viewed by others
Data availability
Due to patient privacy regulations, raw patient data are not available; only de-identified imaging data, safety data, clinical details, and laboratory data are available upon request. Proposals should be directed to the corresponding author (f.j.voskuil@umcg.nl); to gain access, data requestors will need to sign a data access agreement. Data will be saved for a minimum of 25 years, in accordance with Dutch legislation. Upon request, data can be made available to third parties within 6 weeks and can be accessed for 6 weeks thereafter. The source data underlying Figs. 2 and 3 and Tables 1 and 2 are provided as a Source data file; the remaining data are available in the Article. The study protocol is available as Supplementary Note in the Supplementary Information file. Source data are provided with this paper.
References
Slootweg, P. J., Hordijk, G. J., Schade, Y., Van Es, R. J. J. & Koole, R. Treatment failure and margin status in head and neck cancer. A critical view on the potential value of molecular pathology. Oral Oncol. 38, 500–503 (2002).
Hafström, A., Wahlberg, P., Klasson, S., Greiff, L. & Sjövall, J. Predictors of survival in advanced oral cancers after salvage surgery with free tissue flap reconstruction. Eur. Arch. Oto-Rhino-Laryngol. 280, 2953–2964 (2023).
Stepan, K. O., Li, M. M., Kang, S. Y. & Puram, S. V. Molecular margins in head and neck cancer: current techniques and future directions. Oral Oncol. 110, 104893 (2020).
Woolgar, J. A. & Triantafyllou, A. A histopathological appraisal of surgical margins in oral and oropharyngeal cancer resection specimens. Oral Oncol. 41, 1034–1043 (2005).
Hoffmannová, J. et al. Hemimandibulectomy and therapeutic neck dissection with radiotherapy in the treatment of oral squamous cell carcinoma involving mandible: a critical review of treatment protocol in the years 1994-2004. Int. J. Oral Maxillofac. Surg. 39, 561–567 (2010).
Smits, R. W. H. et al. Intraoperative assessment of the resection specimen facilitates achievement of adequate margins in oral carcinoma. Front. Oncol. 10, 614593 (2020).
Lee, D. H. et al. Close surgical margins in oral and oropharyngeal cancer: do they impact prognosis?. Cancers 14, 2990 (2022).
Wu, C., Gleysteen, J., Teraphongphom, N. T., Li, Y. & Rosenthal, E. In-vivo optical imaging in head and neck oncology: basic principles, clinical applications and future directions review-Article. Int. J. Oral Sci. 10, 10 (2018).
Kovács, A. F. Relevance of positive margins in case of adjuvant therapy of oral cancer. Int. J. Oral Maxillofac. Surg. 33, 447–453 (2004).
Zhang, L. et al. Immediate resection of positive margins improves local control in oral tongue cancer. Oral Oncol. 141, 106402 (2023).
Aaboubout, Y. et al. Intraoperative assessment of resection margins by Raman spectroscopy to guide oral cancer surgery. Analyst 148, 4116–4126 (2023).
Li, M. M. et al. Margin analysis in head and neck cancer: state of the art and future directions. Ann. Surg. Oncol. 26, 4070–4080 (2019).
DiNardo, L. J., Lin, J., Karageorge, L. S. & Powers, C. N. Accuracy, utility, and cost of frozen section margins in head and neck cancer surgery. Laryngoscope 110, 1773–1776 (2000).
Ribeiro, N. F. F., Godden, D. R. P., Wilson, G. E., Butterworth, D. M. & Woodwards, R. T. M. De frozen sections help achieve adequate surgical margins in the resection of oral carcinoma?. Int. J. Oral Maxillofac. Surg. 32, 152–158 (2003).
Baddour, H. M., Magliocca, K. R. & Chen, A. Y. The importance of margins in head and neck cancer. J. Surg. Oncol. 113, 248–255 (2016).
Aaboubout, Y. et al. Intraoperative assessment of resection margins in oral cavity cancer: this is the way. J. Vis. Exp. https://doi.org/10.3791/62446 (2021).
Buchakjian, M. R., Tasche, K. K., Robinson, R. A., Pagedar, N. A. & Sperry, S. M. Association of main specimen and tumor bed margin status with local recurrence and survival in oral cancer surgery. JAMA Otolaryngol. Head Neck Surg. 142, 1191–1198 (2016).
Barroso, E. M. et al. Performance of intraoperative assessment of resection margins in oral cancer surgery: a review of literature. Front. Oncol. 11, 628297 (2021).
Singh, A., Qayyumi, B. & Chaturvedi, P. An update on surgical margins in the head neck squamous cell carcinoma: assessment, clinical outcome, and future directions. Curr. Oncol. Rep. 22, 82 (2020).
Noorlag, R., De Bree, R. & Witjes, M. J. H. Image-guided surgery in oral cancer: toward improved margin control. Curr. Opin. Oncol. 34, 170–176 (2022).
Rosenthal, E. L. et al. Safety and tumor specificity of cetuximab-IRDye800 for surgical navigation in head and neck cancer. Clin. Cancer Res. 21, 3658–3666 (2015).
Voskuil, F. J. et al. Fluorescence-guided imaging for resection margin evaluation in head and neck cancer patients using cetuximab-800CW: a quantitative dose-escalation study. Theranostics 10, 3994–4005 (2020).
Gowrishankar, S. et al. Fluorescence-guided surgery for assessing margins in head and neck cancer: a review. JAMA Surg. 109, 1018–1023 (2025).
Krishnan, G. et al. Fluorescent molecular imaging can improve intraoperative sentinel margin detection in oral squamous cell carcinoma. J. Nucl. Med. 63, 1162–1168 (2022).
de Wit, J. G. et al. EGFR-targeted fluorescence molecular imaging for intraoperative margin assessment in oral cancer patients: a phase II trial. Nat. Commun. 14, 4952 (2023).
Higginson, J. A. et al. Diagnostic accuracy of intraoperative margin assessment techniques in surgery for head and neck squamous cell carcinoma: a meta-analysis. Oral. Oncol. 142, 106419 (2023).
Gao, R. W. et al. Determination of tumor margins with surgical specimen mapping using near-infrared fluorescence. Cancer Res. 78, 5144–5154 (2018).
Warram, J. M. et al. Fluorescence imaging to localize head and neck squamous cell carcinoma for enhanced pathological assessment. J. Pathol. Clin. Res. 2, 104–112 (2016).
van Keulen, S. et al. Rapid, non-invasive fluorescence margin assessment: optical specimen mapping in oral squamous cell carcinoma. Oral Oncol. 88, 58–65 (2019).
Van Keulen, S. et al. The sentinel margin: intraoperative ex vivo specimen mapping using relative fluorescence intensity. Clin. Cancer Res. 25, 4656–4662 (2019).
Balasamy, S. & Sundramoorthy, A. K. EGFR-targeted fluorescence imaging for precision margin assessment in oral cancer surgery. Oral Oncol. 150, 106712 (2024).
Heeman, W. et al. A guideline for clinicians performing clinical studies with fluorescence imaging. J. Nucl. Med. 63, 640–645 (2022).
Sharma, A. K. & Sharma, A. (eds) Processing. In Chromosome Techniques: Theory and Practice. 71–90 (Butterworths, 1980).
Gao, R. W. et al. Safety of panitumumab-IRDye800CW and cetuximab-IRDye800CW for fluorescence-guided surgical navigation in head and neck cancers. Theranostics 8, 2488–2495 (2018).
Andersen, A. O. et al. Optical molecular imaging in oral-and oropharyngeal squamous cell carcinoma using a novel uPAR-targeting near-infrared imaging agent FG001 (ICG-Glu-Glu-AE105): an explorative phase II clinical trial. Theranostics 15, 52–67 (2025).
Voskuil, F. J. et al. Intraoperative imaging in pathology-assisted surgery. Nat. Biomed. Eng. 6, 503–514 (2022).
Rounds, C. C. et al. Improved intraoperative identification of close margins in oral squamous cell carcinoma resections using a dual aperture fluorescence ratio approach: first in-human results. J. Biomed. Opt. 29, 016003 (2024).
Fassler, C. et al. Digital mapping of resected cancer specimens: the visual pathology report. J. Pathol. Inf. 15, 100399 (2024).
Linssen, M. D. et al. Roadmap for the development and clinical translation of optical tracers cetuximab-800CW and trastuzumab-800CW. J. Nucl. Med. 60, 418–423 (2019).
Acknowledgements
We would like to thank Karien Kreeft-Polman and Ellen van den Ende-Schaap for their help in patient recruitment. Also, we would like to thank Maaike Barentsen, Tim Ramcharan, Lilo Janssens, Marco Abma and Erik Bleuel for their assistance in specimen processing. Finally, we would like to thank all patients who participated in this study.
Author information
Authors and Affiliations
Contributions
The study was conceptualised by T.S.N., B.K., B.v.d.V., M.J.H.W. and F.J.V. Fluorescent tracer was produced and made available by H.K.H. and H.H.B. Data acquisition was performed by T.S.N., B.K., D.R., M.J.H.W. and F.J.V. Intraoperative clinical decision-making was performed by T.S.N., B.K., J.B., S.A.H.J.d.V., G.B.H., B.E.C.P., J.J.D., K.P.S., B.v.d.V., M.J.H.W. and F.J.V. Data analysis was performed by T.S.N., B.K., D.R., M.J.H.W., F.J.V. T.S.N. and B.K. wrote and revised the manuscript with input, feedback, and acceptance from all authors. The study was supervised by M.J.H.W. and F.J.V.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks A. Dimitrios Colevas, Jason Warram, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nijboer, T.S., Keizers, B., Boeve, K. et al. Intraoperative fluorescence-guided fresh frozen sectioning for margin control in head and neck cancer: phase 2 clinical trial. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70264-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70264-y