Abstract
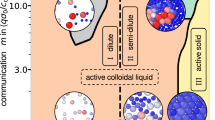
Elucidating how repulsive interactions evolve to generate ordered structures in nonequilibrium colloidal systems remains a central challenge, partly because few experimental platforms provide particle-resolved access to their structural evolution. Here we show that alginate hydrogel colloids confined within cyclohexyl bromide (CHB) emulsion droplets form a controllable model system in which electrostatic interactions evolve in time and drive ordering. Ba2+ ions diffusing from the surrounding aqueous phase progressively crosslink the alginate droplets, increasing their surface charge, while buoyancy compacts them into locally quasi-two-dimensional layers within the CHB phase. As electrostatic repulsion strengthens, the assembly evolves from a disordered state to a hexagonally ordered structure. By calibrating Brownian dynamics simulations to the experimentally measured lattice spring constant, we constrain the effective Debye screening length to ≈2.5–3 μm. Quantitative imaging further shows that ordering emerges once a dimensionless interaction parameter—defined as the ratio of electrostatic interaction energy to thermal energy—reaches values of ≈117–149. The ordered state exhibits reversible disordered–order behavior under mechanical and magnetic perturbations, demonstrating a robust nonequilibrium platform for probing charge-regulated colloidal ordering under confinement.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon request. Unprocessed raw data are provided as Supplementary Data 1. Source data are provided with this paper.
References
Lindsay, H. & Chaikin, P. Elastic properties of colloidal crystals and glasses. J. Chem. Phys. 76, 3774–3781 (1982).
Pusey, P. N. & Van Megen, W. Phase behaviour of concentrated suspensions of nearly hard colloidal spheres. Nature 320, 340–342 (1986).
Poon, W., Pusey, P. & Lekkerkerker, H. Colloids in suspense. Phys. World 9, 27 (1996).
Lee, H. M. et al. Direct measurements of the colloidal Debye force. Nat. Commun. 14, 3838 (2023).
Anjali, T. G. & Basavaraj, M. G. Shape-anisotropic colloids at interfaces. Langmuir 35, 3–20 (2018).
Velev, O. D. Self-assembly of unusual nanoparticle crystals. Science 312, 376–377 (2006).
Peeters, F. & Wu, X. Wigner crystal of a screened-Coulomb-interaction colloidal system in two dimensions. Phys. Rev. A 35, 3109 (1987).
Leunissen, M. E. et al. Ionic colloidal crystals of oppositely charged particles. Nature 437, 235–240 (2005).
Russell, E. R., Spaepen, F. & Weitz, D. A. Anisotropic elasticity of experimental colloidal Wigner crystals. Phys. Rev. E 91, 032310 (2015).
Leunissen, M. E. et al. Electrostatics at the oil–water interface, stability, and order in emulsions and colloids. Proc. Natl Acad. Sci. 104, 2585–2590 (2007).
Angelini, R. et al. Glass–glass transition during aging of a colloidal clay. Nat. Commun. 5, 4049 (2014).
Everts, J., Boon, N. & van Roij, R. Density-induced reentrant melting of colloidal Wigner crystals. Phys. Chem. Chem. Phys. 18, 5211–5218 (2016).
Ashcroft, N. & Mermin, N. Solid State Physics. Holt, Rinehart and Winston, New York (1976).
Gann, R., Chakravarty, S. & Chester, G. Monte Carlo simulation of the classical two-dimensional one-component plasma. Phys. Rev. B 20, 326 (1979).
Hockney, R. W. & Brown, T. A lambda transition in a classical electron film. J. Phys. C: Solid State Phys. 8, 1813 (1975).
Morf, R. Temperature dependence of the shear modulus and melting of the two-dimensional electron solid. Phys. Rev. Lett. 43, 931 (1979).
Thouless, D. Melting of the two-dimensional Wigner lattice. J. Phys. C: Solid State Phys. 11, L189 (1978).
Choi, K. H. et al. Direct measurement of electrostatic interactions between poly (methyl methacrylate) microspheres with optical laser tweezers. Soft Matter 15, 8051–8058 (2019).
Ahmadi, N. et al. A single-particle mechanofluorescent sensor. Nat. Commun. 15, 6094 (2024).
Jang, J. et al. Microfluidic antisolvent crystallization for chiral symmetry breaking. Lab Chip 24, 5055–5064 (2024).
Yoon, B., et al. Single particle semi‐reversible solvatochromic sensor. Small, 21, 2412189 (2025).
Jo, Y. K. & Lee, D. Biopolymer microparticles prepared by microfluidics for biomedical applications. Small 16, 1903736 (2020).
Han, S. W. et al. Colloidal pixel‐based micropatterning using uniform janus microparticles with tunable anisotropic particle geometry. Adv. Funct. Mater. 29, 1805392 (2019).
Ahmadi, N. et al. Gas‐shearing microfluidic fabrication of polydiacetylene–alginate colorimetric sensor beads. Small Struct. 6, 2400340 (2025).
Daradmare, S., Xia, M., Kim, J. & Park, B. J. Metal–organic frameworks/alginate composite beads as effective adsorbents for the removal of hexavalent chromium from aqueous solution. Chemosphere 270, 129487 (2021).
Jang, J. et al. Effects of alginate hydrogel cross-linking density on mechanical and biological behaviors for tissue engineering. J. Mech. Behav. Biomed. Mater. 37, 69–77 (2014).
Xia, M. et al. The recyclability of alginate hydrogel particles used as a palladium catalyst support. J. Ind. Eng. Chem. 73, 306–315 (2019).
Mørch, Ý. A., Donati, I., Strand, B. L. & Skjåk-Bræk, G. Effect of Ca2+, Ba2+, and Sr2+ on alginate microbeads. Biomacromolecules 7, 1471–1480 (2006).
Mazoyer, S., Ebert, F., Maret, G. & Keim, P. Dynamics of particles and cages in an experimental 2D glass former. Europhys. Lett. 88, 66004 (2010).
Zahn, K. et al. Elastic properties of 2D colloidal crystals from video microscopy. Phys. Rev. Lett. 90, 155506 (2003).
Barbhuiya, N. H. & Mishra, C. K. Anisotropic cage evolution in quasi-two-dimensional colloidal fluids. Phys. Rev. E. 110, L062602 (2024).
Park, B. J., Lee, B. & Yu, T. Pairwise interactions of colloids in two-dimensional geometric confinement. Soft Matter 10, 9675–9680 (2014).
Royall, C. P., et al. Re-entrant melting and freezing in a model system of charged colloids. J. Chem. Phys. 124, (2006).
Hsu, M. F., Dufresne, E. R. & Weitz, D. A. Charge stabilization in nonpolar solvents. Langmuir. 21, 4881–4887 (2005).
Sainis, S. K., Germain, V., Mejean, C. O. & Dufresne, E. R. Electrostatic interactions of colloidal particles in nonpolar solvents: Role of surface chemistry and charge control agents. Langmuir. 24, 1160–1164 (2008).
Espinosa, C. E., Guo, Q., Singh, V. & Behrens, S. H. Particle charging and charge screening in nonpolar dispersions with nonionic surfactants. Langmuir. 26, 16941–16948 (2010).
Choi, K. H. et al. Interpretation of electrostatic self-potential measurements using interface-trapped microspheres with surface heterogeneity. ACS Appl. Polym. Mater. 2, 1304–1311 (2020).
Park, B. J., Vermant, J. & Furst, E. M. Heterogeneity of the electrostatic repulsion between colloids at the oil–water interface. Soft Matter 6, 5327–5333 (2010).
Grimes, C. & Adams, G. Evidence for a liquid-to-crystal phase transition in a classical, two-dimensional sheet of electrons. Phys. Rev. Lett. 42, 795 (1979).
Tran, H. H., et al. Nanozyme‐shelled microcapsules for targeting biofilm infections in confined spaces. Adv. Healthc. Mater., 2402306
Kim, B. et al. Monodisperse microshell structured gelatin microparticles for temporary chemoembolization. Biomacromolecules 19, 386–391 (2018).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Arganda-Carreras, I. et al. Trainable Weka Segmentation: a machine learning tool for microscopy pixel classification. Bioinformatics 33, 2424–2426 (2017).
Ermak, D. L. & McCammon, J. A. Brownian dynamics with hydrodynamic interactions. J. Chem. Phys. 69, 1352–1360 (1978).
Higham, D. J. An algorithmic introduction to numerical simulation of stochastic differential equations. SIAM Rev. 43, 525–546 (2001).
Sainis, S. K., Merrill, J. W. & Dufresne, E. R. Electrostatic interactions of colloidal particles at vanishing ionic strength. Langmuir. 24, 13334–13337 (2008).
Kabsch, W. A solution for the best rotation to relate two sets of vectors. Found. Crystallogr. 32, 922–923 (1976).
Ma, X., Mishra, C. K., Habdas, P. & Yodh, A. Structural and short-time vibrational properties of colloidal glasses and supercooled liquids in the vicinity of the re-entrant glass transition. J. Chem. Phys. 155, 074902 (2021).
Acknowledgements
This study was supported by the National Research Foundation (NRF) of Korea (RS-2025-00516792 to B.J.P., RS-2024-00406741 to B.J.P., and RS-2025−16063688 to H.H). The research was conducted with the support of the equipment and facilities provided by the ACE Center at Kyung Hee University.
Author information
Authors and Affiliations
Contributions
B.J.P. conceived and supervised the project. I.H.J. C.C.R. and H.S.L. performed the microfluidic preparation and measured the behaviors of colloidal ordering. C.C.R. measured the zeta-potential of alginate hydrogel particles. I.H.J. and H.A. performed the image analysis. B.J.P. performed the BD simulations. H.H., H.A., and B.J.P. wrote the first draft. All authors contributed to the interpretation of experimental data and read, edited, commented on this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jung, I.H., Revadekar, C.C., Lee, H.S. et al. Nonequilibrium ordering dynamics of confined soft alginate hydrogel colloids driven by time-evolving electrostatic interactions. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70266-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70266-w