Abstract
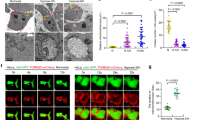
Mitochondria form contact sites with multiple organelles to coordinate diverse cellular processes. Melanosomes, lysosome-related organelles, undergo stepwise maturation to synthesize and store melanin, but how they interact with mitochondria remains unclear. Here we show that mitochondria–melanosome contacts dynamically increase during melanosome maturation and are mediated by STIM1–MFN2 interactions. Using a NanoBiT-based reporter system, MiMSBiT (Mitochondria–Melanosome contact reporter applying NanoBiT), to monitor reversible mitochondria–melanosome contacts in living cells, we demonstrate that STIM1 localizes to melanosomes and promotes their contact with mitochondrial MFN2. A transient decrease in melanosomal lumen calcium induces STIM1 clustering and enhances its association with MFN2. These contacts locally increase mitochondrial ATP availability, leading to melanosome lumen acidification via proton channel activation. This acidification facilitates PMEL fibrillation, a key step in melanosome maturation. Together, our findings reveal a mechanism by which mitochondria–melanosome contacts regulate melanosome maturation.
Similar content being viewed by others
Data availability
The mass spectrometry proteomics data generated in this study have been deposited in the ProteomeXchange Consortium via the jPOST partner repository under the dataset identifiers JPST003803 and PXD063939. Source data underlying all figures are provided with this paper. Other data supporting the findings of this study are available from the corresponding authors. Source data are provided with this paper.
References
Voeltz, G. K., Sawyer, E. M., Hajnóczky, G. & Prinz, W. A. Making the connection: how membrane contact sites have changed our view of organelle biology. Cell 187, 257–270 (2024).
Scorrano, L. et al. Coming together to define membrane contact sites. Nat. Commun. 10, 1287 (2019).
Tábara, L. C., Segawa, M. & Prudent, J. Molecular mechanisms of mitochondrial dynamics. Nat. Rev. Mol. Cell Biol. 26, 123–146 (2025).
Jenkins, B. C. et al. Mitochondria in disease: changes in shapes and dynamics. Trends Biochem. Sci. 49, 346–360 (2024).
Copeland, D. E. & Dalton, A. J. An association between mitochondria and the endoplasmic reticulum in cells of the pseudobranch gland of a teleost. J. Biophys. Biochem. Cytol. 5, 393–396 (1959).
de Brito, O. M. & Scorrano, L. Mitofusin 2 tethers endoplasmic reticulum to mitochondria. Nature 456, 605–610 (2008).
Rizzuto, R. et al. Ca(2+) transfer from the ER to mitochondria: when, how and why. Biochim. Biophys. Acta 1787, 1342–1351 (2009).
Giacomello, M. & Pellegrini, L. The coming of age of the mitochondria-ER contact: a matter of thickness. Cell Death Differ. 23, 1417–1427 (2016).
Rowland, A. A. & Voeltz, G. K. Endoplasmic reticulum-mitochondria contacts: function of the junction. Nat. Rev. Mol. Cell Biol. 13, 607–625 (2012).
Daniele, T. et al. Mitochondria and melanosomes establish physical contacts modulated by Mfn2 and involved in organelle biogenesis. Curr. Biol. 24, 393–403 (2014).
Raiborg, C., Malerød, L., Pedersen, N. M. & Stenmark, H. Differential functions of Hrs and ESCRT proteins in endocytic membrane trafficking. Exp. Cell Res. 314, 801–813 (2008).
Raiborg, C., Wesche, J., Malerød, L. & Stenmark, H. Flat clathrin coats on endosomes mediate degradative protein sorting by scaffolding Hrs in dynamic microdomains. J. Cell Sci. 119, 2414–2424 (2006).
Raposo, G. & Marks, M. S. Melanosomes-dark organelles enlighten endosomal membrane transport. Nat. Rev. Mol. Cell Biol. 8, 786–797 (2007).
Raiborg, C. & Stenmark, H. The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature 458, 445–452 (2009).
Wiriyasermkul, P., Moriyama, S. & Nagamori, S. Membrane transport proteins in melanosomes: regulation of ions for pigmentation. Biochim. Biophys. Acta Biomembr. 1862, 183318 (2020).
Le, L., Sirés-Campos, J., Raposo, G., Delevoye, C. & Marks, M. S. Melanosome biogenesis in the pigmentation of mammalian skin. Integr. Comp. Biol. 61, 1517–1545 (2021).
Cui, R. et al. Central role of p53 in the suntan response and pathologic hyperpigmentation. Cell 128, 853–864 (2007).
Buscà, R. & Ballotti, R. Cyclic AMP a key messenger in the regulation of skin pigmentation. Pigment Cell Res. 13, 60–69 (2000).
Cheli, Y. et al. {alpha}MSH and Cyclic AMP elevating agents control melanosome pH through a protein kinase A-independent mechanism. J. Biol. Chem. 284, 18699–18706 (2009).
Hirobe, T. Stimulation of dendritogenesis in the epidermal melanocytes of newborn mice by melanocyte-stimulating hormone. J. Cell Sci. 33, 371–383 (1978).
Zhou, D. et al. Mammalian pigmentation is regulated by a distinct cAMP-dependent mechanism that controls melanosome pH. Sci. Signal. 11, eaau7987 (2018).
Hearing, V. J. Biochemical control of melanogenesis and melanosomal organization. J. Investig. Dermatol. Symp. Proc. 4, 24–28 (1999).
Bhatnagar, V., Anjaiah, S., Puri, N., Darshanam, B. N. & Ramaiah, A. pH of melanosomes of B 16 murine melanoma is acidic: its physiological importance in the regulation of melanin biosynthesis. Arch. Biochem. Biophys. 307, 183–192 (1993).
Pfefferkorn, C. M., McGlinchey, R. P. & Lee, J. C. Effects of pH on aggregation kinetics of the repeat domain of a functional amyloid, Pmel17. Proc. Natl. Acad. Sci. USA 107, 21447–21452 (2010).
Watt, B., van Niel, G., Raposo, G. & Marks, M. S. PMEL: a pigment cell-specific model for functional amyloid formation. Pigment Cell Melanoma Res 26, 300–315 (2013).
McGlinchey, R. P. et al. The repeat domain of the melanosome fibril protein Pmel17 forms the amyloid core promoting melanin synthesis. Proc. Natl. Acad. Sci. USA 106, 13731–13736 (2009).
Shiiba, I. et al. ER-mitochondria contacts mediate lipid radical transfer via RMDN3/PTPIP51 phosphorylation to reduce mitochondrial oxidative stress. Nat. Commun. 16, 1508 (2025).
Ishida, M., Arai, S. P., Ohbayashi, N. & Fukuda, M. The GTPase-deficient Rab27A(Q78L) mutant inhibits melanosome transport in melanocytes through trapping of Rab27A effector protein Slac2-a/melanophilin in their cytosol: development of a novel melanosome-targetinG tag. J. Biol. Chem. 289, 11059–11067 (2014).
Tilokani, L., Nagashima, S., Paupe, V. & Prudent, J. Mitochondrial dynamics: overview of molecular mechanisms. Essays Biochem 62, 341–360 (2018).
Naon, D. et al. Splice variants of mitofusin 2 shape the endoplasmic reticulum and tether it to mitochondria. Science 380, eadh9351 (2023).
Ambrosio, A. L., Boyle, J. A., Aradi, A. E., Christian, K. A. & Di Pietro, S. M. TPC2 controls pigmentation by regulating melanosome pH and size. Proc. Natl. Acad. Sci. USA 113, 5622–5827 (2016).
Branon, T. C. et al. Efficient proximity labeling in living cells and organisms with TurboID. Nat. Biotechnol. 36, 880–887 (2018).
Dooley, C. M. et al. Slc45a2 and V-ATPase are regulators of melanosomal pH homeostasis in zebrafish, providing a mechanism for human pigment evolution and disease. Pigment Cell Melanoma Res. 26, 205–217 (2013).
Marvin, J. S. et al. iATPSnFR2: a high-dynamic-range fluorescent sensor for monitoring intracellular ATP. Proc. Natl. Acad. Sci. Usa. 121, e2314604121 (2024).
Zacharioudakis, E. et al. Modulating mitofusins to control mitochondrial function and signaling. Nat. Commun. 13, 3775 (2022).
Sheets, L., Ransom, D. G., Mellgren, E. M., Johnson, S. L. & Schnapp, B. J. Zebrafish melanophilin facilitates melanosome dispersion by regulating dynein. Curr. Biol. 17, 1721–1734 (2007).
Luik, R. M., Wang, B., Prakriya, M., Wu, M. M. & Lewis, R. S. Oligomerization of STIM1 couples ER calcium depletion to CRAC channel activation. Nature 454, 538–542 (2008).
Zhang, Z., Gong, J., Sviderskaya, E. V., Wei, A. & Li, W. Mitochondrial NCKX5 regulates melanosomal biogenesis and pigment production. J. Cell Sci. 132, jcs232009 (2019).
Le, L. et al. SLC45A2 protein stability and regulation of melanosome pH determine melanocyte pigmentation. Mol. Biol. Cell 31, 2687–2702 (2020).
Hellström, A. R. et al. Inactivation of Pmel alters melanosome shape but has only a subtle effect on visible pigmentation. PLoS Genet. 7, e1002285 (2011).
Rochin, L. et al. BACE2 processes PMEL to form the melanosome amyloid matrix in pigment cells. Proc. Natl. Acad. Sci. USA 110, 10658–10663 (2013).
Motiani, R. K. et al. STIM1 activation of adenylyl cyclase 6 connects Ca(2+) and cAMP signaling during melanogenesis. EMBO J. 37, e97597 (2018).
Sugiura, A. et al. MITOL regulates endoplasmic reticulum-mitochondria contacts via mitofusin2. Mol. Cell 51, 20–34 (2013).
Ban-Ishihara, R., Ishihara, T., Sasaki, N., Mihara, K. & Ishihara, N. Dynamics of nucleoid structure regulated by mitochondrial fission contributes to cristae reformation and release of cytochrome c. Proc. Natl. Acad. Sci. USA 110, 11863–11868 (2013).
Wang, Y. et al. Regulated complex assembly safeguards the fidelity of Sleeping Beauty transposition. Nucleic Acids Res. 45, 311–326 (2017).
Akagi, T., Sasai, K. & Hanafusa, H. Refractory nature of normal human diploid fibroblasts with respect to oncogene-mediated transformation. Proc. Natl. Acad. Sci. USA 100, 13567–13572 (2003).
Shinoda, H. et al. Acid-tolerant monomeric GFP from olindias formosa. Cell Chem. Biol. 25, 330–338 (2018).
Leonhardt, R. M., Vigneron, N., Rahner, C., Van den Eynde, B. J. & Cresswell, P. Endoplasmic reticulum export, subcellular distribution, and fibril formation by Pmel17 require an intact N-terminal domain junction. J. Biol. Chem. 285, 16166–16183 (2010).
Beyers, W. C., Detry, A. M. & Di Pietro, S. M. OCA7 is a melanosome membrane protein that defines pigmentation by regulating early stages of melanosome biogenesis. J. Biol. Chem. 298, 102669 (2022).
Strom, A. R. et al. HP1α is a chromatin crosslinker that controls nuclear and mitotic chromosome mechanics. eLife 10, e63972 (2021).
Ito, N. et al. MITOL regulates phosphatidic acid-binding activity of RMDN3/PTPIP51. J. Biochem. 171, 529–541 (2022).
Rappsilber, J., Mann, M. & Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2, 1896–1906 (2007).
Goedhart, J. & Luijsterburg, M. S. VolcaNoseR is a web app for creating, exploring, labeling and sharing volcano plots. Sci. Rep. 10, 20560 (2020).
Acknowledgements
We thank Michael S. Marks for the generous gift of PMEL fibril (I51) antibody. We thank Saki Asada for technical assistance. This study was partially supported by the MEXT/JSPS KAKENHI [Grant Nos. 22K15399, 22H05574, 24H01327, and 24K18382 (for IS), 23K14185 and 22K20637 (for NI), 21K06844 (for RI), 23K02691, 20H04911 and 20H03454 (for SY)], the Uehara Memorial Foundation (for IS and SY), the Kowa Life Science Foundation (to IS)], the Takeda Science Foundation (2025) (for SY), the Daiichi Sankyo Foundation of Life Science (for SY) and AMED [Grant No. JP20gm5010002 and JP25gm2110001 (to SY)].
Author information
Authors and Affiliations
Contributions
I.S. and S.Y. designed the experiments; I.S., N.I., K.U., Y.A., H.O., Y.I., and F.Y. performed the experiments; I.S. and S.Y. analyzed and interpreted the data; Y.M., H.A., S.N., Y.A., K.N., Y.H., and R.I. assisted in the analysis; I.S. and S.Y. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Qing Deng, David Peeney and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shiiba, I., Ishikawa, Y., Oshio, H. et al. STIM1-Mitofusin2 interactions tether mitochondria and melanosome contacts that promote melanosome maturation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70282-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70282-w