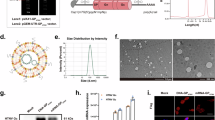
Abstract
Old World orthohantaviruses, including Hantaan virus (HTNV), cause hemorrhagic fever with renal syndrome (HFRS) in Eurasia. Available inactivated vaccines often induce low neutralizing antibodies and short-term protection. We evaluated nucleic acid vaccines expressing a prefusion-stabilized HTNV glycoprotein in female BALB/c mice. Both DNA and mRNA-LNP versions elicited robust neutralizing antibodies by strongly activating germinal centers, which protected mice against high-dose HTNV challenge. We further tested heterologous prime-boost regimens, where mice primed with inactivated vaccine received different boosters. All boosters increased neutralizing titers, but only the prefusion-stabilized glycoprotein mRNA-LNP vaccine raised titers to the level achieved by its own full primary vaccination course. This demonstrates the immunogen’s superiority in developing next-generation vaccines and its unique ability to potently recall memory B cells induced by suboptimal inactivated vaccines. Thus, prefusion-stabilized glycoprotein-based nucleic acid vaccines are promising candidates for advanced orthohantavirus vaccine development.
Similar content being viewed by others
Data availability
All data generated in this study are provided in the Source Data file associated with this paper. No sequencing or other omics data requiring deposition in public repositories were generated. Source data are provided with this paper. All reagents will be made available upon request after the completion of a material transfer agreement. Requests can be directed to the corresponding author. Source data are provided with this paper.
References
Meier, K., Thorkelsson, S. R., Quemin, E. R. J. & Rosenthal, M. Hantavirus replication cycle-an updated structural virology perspective. Viruses-Basel 13, 1561 (2021).
Vial, P. A. et al. Hantavirus in humans: a review of clinical aspects and management. Lancet Infect. Dis. 23, e371–e382 (2023).
Vaheri, A. et al. Uncovering the mysteries of hantavirus infections. Nat. Rev. Microbiol. 11, 539–550 (2013).
Jiang, H. et al. Hantavirus infection: a global zoonotic challenge. Virol. Sin. 32, 32–43 (2017).
Deng X., Xie Y., Du S. Distinct genotype of hantavirus infection in rodents in jiangxi province, China, in 2020–2021. Zoonoses 2 (2022),
Roda Gracia, J., Schumann, B. & Seidler, A. Climate variability and the occurrence of human puumala hantavirus infections in Europe: a systematic review. Zoonoses public health 62, 465–478 (2015).
Douglas K. O., Payne K., Sabino-Santos G. Jr., Agard J. Influence of climatic factors on human hantavirus infections in Latin America and the Caribbean: a Systematic Review. 11 (Pathogens Basel, Switzerland, 2021),
Cho, H. W., Howard, C. R. & Lee, H. W. Review of an inactivated vaccine against hantaviruses. Intervirology 45, 328–333 (2002).
Löber, C., Anheier, B., Lindow, S., Klenk, H. & Feldmann, H. The Hantaan virus glycoprotein precursor is cleaved at the conserved pentapeptide WAASA. Virology 289, 224–229 (2001).
Tischler, N. D., Gonzalez, A., Perez-Acle, T., Rosemblatt, M. & Valenzuela, P. D. Hantavirus Gc glycoprotein: evidence for a class II fusion protein. J. Gen. Virol. 86, 2937–2947 (2005).
Guardado-Calvo, P. et al. Mechanistic insight into bunyavirus-induced membrane fusion from structure-function analyses of the hantavirus envelope glycoprotein Gc. PLoS Pathog. 12, e1005813 (2016).
Serris, A. et al. The hantavirus surface glycoprotein lattice and its fusion control mechanism. Cell 183, 442–456.e416 (2020).
Bignon, E. A., Albornoz, A., Guardado-Calvo, P., Rey, F. A. & Tischler, N. D. Molecular organization and dynamics of the fusion protein Gc at the hantavirus surface. eLife 8, e46028 (2019).
Cifuentes-Munoz, N., Salazar-Quiroz, N. & Tischler, N. D. Hantavirus Gn and Gc envelope glycoproteins: key structural units for virus cell entry and virus assembly. Viruses 6, 1801–1822 (2014).
Paulsen, G. C. et al. Safety and immunogenicity of an Andes virus DNA vaccine by needle-free injection: a randomized, controlled phase 1 study. J. Infect. Dis. 229, 30–38 (2023).
Hooper, J. W., Josleyn, M., Ballantyne, J. & Brocato, R. A novel Sin Nombre virus DNA vaccine and its inclusion in a candidate pan-hantavirus vaccine against hantavirus pulmonary syndrome (HPS) and hemorrhagic fever with renal syndrome (HFRS). Vaccine 31, 4314–4321 (2013).
Hooper, J. W., Custer, D. M., Smith, J. & Wahl-Jensen, V. Hantaan/Andes virus DNA vaccine elicits a broadly cross-reactive neutralizing antibody response in nonhuman primates. Virology 347, 208–216 (2006).
Jiang, D. B. et al. Construction and evaluation of a DNA vaccine encoding Hantavirus glycoprotein N-terminal fused with lysosome-associated membrane protein. Vaccine 33, 3367–3376 (2015).
Jiang, D. B. et al. Hantavirus Gc induces long-term immune protection via LAMP-targeting DNA vaccine strategy. Antivir. Res. 150, 174–182 (2018).
Liu, R. et al. Investigation of a subunit protein vaccine for HFRS based on a consensus sequence between envelope glycoproteins of HTNV and SEOV. Virus Res. 334, 199149 (2023).
Willensky, S. et al. Crystal structure of glycoprotein C from a hantavirus in the post-fusion conformation. PLoS Pathog. 12, e1005948 (2016).
Rissanen, I. et al. Structural transitions of the conserved and metastable hantaviral glycoprotein envelope. J. Virol. 91, e00378–e00417 (2017).
Rissanen, I. et al. Molecular rationale for antibody-mediated targeting of the hantavirus fusion glycoprotein. eLife 9, e58242 (2020).
Rissanen, I. et al. Structural basis for a neutralizing antibody response elicited by a recombinant hantaan virus Gn immunogen. mBio 12, e0253120 (2021).
Duehr, J. et al. Neutralizing monoclonal antibodies against the Gn and the Gc of the andes virus glycoprotein spike complex protect from virus challenge in a preclinical hamster model. mBio 11, e00028–e00120 (2020).
Stass, R. et al. Mechanistic basis for potent neutralization of Sin Nombre hantavirus by a human monoclonal antibody. Nat. Microbiol. 8, 1293–1303 (2023).
Mittler, E. et al. Structural and mechanistic basis of neutralization by a pan-hantavirus protective antibody. Sci. Transl. Med. 15, eadg1855 (2023).
Engdahl, T. B. et al. Antigenic mapping and functional characterization of human New World hantavirus neutralizing antibodies. eLife 12, e81743 (2023).
McLellan, J. S. et al. Structure-based design of a fusion glycoprotein vaccine for respiratory syncytial virus. Science 342, 592–598 (2013).
Pallesen, J. et al. Immunogenicity and structures of a rationally designed prefusion MERS-CoV spike antigen. Proc. Natl. Acad. Sci. USA 114, E7348–E7357 (2017).
Hepojoki, J., Strandin, T., Vaheri, A. & Lankinen, H. Interactions and oligomerization of hantavirus glycoproteins. J. Virol. 84, 227–242 (2010).
Li, Z., Zhang, Y. & Sun, B. Current understanding of Th2 cell differentiation and function. Protein cell 2, 604–611 (2011).
Breitfeld, D. et al. Follicular B helper T cells express CXC chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J. Exp. Med. 192, 1545–1552 (2000).
Kim, C. H. et al. Subspecialization of CXCR5+ T cells: B helper activity is focused in a germinal center-localized subset of CXCR5+ T cells. J. Exp. Med. 193, 1373–1381 (2001).
Schaerli, P. et al. CXC chemokine receptor 5 expression defines follicular homing T cells with B cell helper function. J. Exp. Med. 192, 1553–1562 (2000).
Zheng, Y. et al. Long-term persistence of anti-hantavirus antibodies in sera of patients undergoing hemorrhagic fever with renal syndrome and subjects vaccinated against the disease. Infect. Dis. (Lond., Engl.) 48, 262–266 (2016).
Song, J. Y. et al. Immunogenicity and safety of a modified three-dose priming and booster schedule for the Hantaan virus vaccine (Hantavax): a multi-center phase III clinical trial in healthy adults. Vaccine 38, 8016–8023 (2020).
Cheng, L. F. et al. Incorporation of GM-CSF or CD40L enhances the immunogenicity of hantaan virus-like particles. Front Cell Infect. Microbiol 6, 185 (2016).
Arikawa, J., Yao, J. S., Yoshimatsu, K., Takashima, I. & Hashimoto, N. Protective role of antigenic sites on the envelope protein of Hantaan virus defined by monoclonal antibodies. Arch. Virol. 126, 271–281 (1992).
Xu, Z., Wei, L., Wang, L., Wang, H. & Jiang, S. The in vitro and in vivo protective activity of monoclonal antibodies directed against Hantaan virus: potential application for immunotherapy and passive immunization. Biochem.l Biophys. Res. Commun. 298, 552–558 (2002).
Hooper, J. W. et al. DNA vaccine-derived human IgG produced in transchromosomal bovines protects in lethal models of hantavirus pulmonary syndrome. Sci. Transl. Med. 6, 264ra162 (2014).
Vial, P. A. et al. A non-randomized multicentre trial of human immune plasma for treatment of hantavirus cardiopulmonary syndrome caused by Andes virus. Antivir. Ther. 20, 377–386 (2015).
Garrido, J. L. et al. Two recombinant human monoclonal antibodies that protect against lethal Andes hantavirus infection in vivo. Sci. Transl. Med. 10, eaat6420 (2018).
Mittler, E. et al. Human antibody recognizing a quaternary epitope in the Puumala virus glycoprotein provides broad protection against orthohantaviruses. Sci. Transl. Med. 14, eabl5399 (2022).
Milligan, J. C. et al. Asymmetric and non-stoichiometric glycoprotein recognition by two distinct antibodies results in broad protection against ebolaviruses. Cell 185, 995–1007.e1018 (2022).
Tian, H. et al. Anthropogenically driven environmental changes shift the ecological dynamics of hemorrhagic fever with renal syndrome. PLoS Pathog. 13, e1006198 (2017).
Kim, H. W. et al. Respiratory syncytial virus disease in infants despite prior administration of an antigenic inactivated vaccine. Am. J. Epidemiol. 89, 422–434 (1969).
Prince, G. A. et al. Enhancement of respiratory syncytial virus pulmonary pathology in cotton rats by prior intramuscular inoculation of formalin-inactivated virus. J. Virol. 57, 721–728 (1986).
Krarup, A. et al. A highly stable prefusion RSV F vaccine derived from structural analysis of the fusion mechanism. Nat. Commun. 6, 8143 (2015).
Stewart-Jones, G. B. E. et al. Structure-based design of a quadrivalent fusion glycoprotein vaccine for human parainfluenza virus types 1-4. Proc. Natl. Acad. Sci. USA 115, 12265–12270 (2018).
Hsieh, C. L. et al. Structure-based design of prefusion-stabilized SARS-CoV-2 spikes. Science 369, 1501–1505 (2020).
Wrapp, D. et al. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 367, 1260–1263 (2020).
Schaub, J. M. et al. Expression and characterization of SARS-CoV-2 spike proteins. Nat. Protoc. 16, 5339–5356 (2021).
Guardado-Calvo, P. & Rey, F. A. The Envelope proteins of the bunyavirales. Adv. virus Res. 98, 83–118 (2017).
Dey, A. et al. Immunogenic potential of DNA vaccine candidate, ZyCoV-D, against SARS-CoV-2 in animal models. Vaccine 39, 4108–4116 (2021).
Calarota, S. A., Kjerrström, A., Islam, K. B. & Wahren, B. Gene combination raises broad human immunodeficiency virus-specific cytotoxicity. Hum. Gene Ther. 12, 1623–1637 (2001).
Conry, R. M. et al. Safety and immunogenicity of a DNA vaccine encoding carcinoembryonic antigen and hepatitis B surface antigen in colorectal carcinoma patients. Clin. Cancer Res. 8, 2782–2787 (2002).
Tagawa, S. T. et al. Phase I study of intranodal delivery of a plasmid DNA vaccine for patients with Stage IV melanoma. Cancer 98, 144–154 (2003).
Hooper, J. et al. A phase 2a randomized, double-blind, dose-optimizing study to evaluate the immunogenicity and safety of a bivalent DNA vaccine for hemorrhagic fever with renal syndrome delivered by intramuscular electroporation. Vaccines 8, 377 (2020).
Jiang, D. B. et al. Recombinant DNA vaccine of Hantavirus Gn and LAMP1 induced long-term immune protection in mice. Antivir. Res. 138, 32–39 (2017).
Alameh, M. G. et al. Lipid nanoparticles enhance the efficacy of mRNA and protein subunit vaccines by inducing robust T follicular helper cell and humoral responses. Immunity 54, 2877–2892.e2877 (2021).
Han, X. et al. Adjuvant lipidoid-substituted lipid nanoparticles augment the immunogenicity of SARS-CoV-2 mRNA vaccines. Nat. Nanotechnol. 18, 1105–1114 (2023).
Gitlin, A. D. et al. HUMORAL IMMUNITY. T cells help controls the speed of the cell cycle in germinal center B cells. Sci. (N. Y., NY) 349, 643–646 (2015).
Brooks, J. F. et al. Molecular basis for potent B cell responses to antigen displayed on particles of viral size. Nat. Immunol. 24, 1762–1777 (2023).
Zhang, J. et al. A comprehensive investigation of Glycoprotein-based nucleic acid vaccines for Hantaan Virus. NPJ Vaccines 9, 196 (2024).
Wei, J. et al. Standardization, validation, and comparative evaluation of a convenient surrogate recombinant vesicular stomatitis virus plaque reduction test for quantification of Hantaan orthohantavirus (HTNV) neutralizing antibodies. Virol. J. 22, 31 (2025).
Yao, J. S. et al. Antibody-dependent enhancement of hantavirus infection in macrophage cell lines. Arch. Virol. 122, 107–118 (1992).
Yao, M. et al. N(6)-methyladenosine modifications enhance enterovirus 71 ORF translation through METTL3 cytoplasmic distribution. Biochem. Biophys. Res. Commun. 527, 297–304 (2020).
Ickenstein, L. M. & Garidel, P. Lipid-based nanoparticle formulations for small molecules and RNA drugs. Expert Opin. drug Deliv. 16, 1205–1226 (2019).
Zhang, H. et al. Single-dose recombinant VSV-based vaccine elicits robust and durable neutralizing antibody against Hantaan virus. NPJ Vaccines 9, 28 (2024).
Hu, Y. L. et al. Construction and evaluation of a DNA vaccine encoding the Crimean Congo hemorrhagic fever virus nucleocapsid protein, glycoprotein N-terminal and C-terminal fused with LAMP1. Front. Cell. Infect. Microbiol. 13, 1121163 (2023).
Ye, C. et al. An improved enzyme-linked focus formation assay revealed baloxavir acid as a potential antiviral therapeutic against hantavirus infection. Front. Pharmacol. 10, 1203 (2019).
Vaidya, S. R. et al. Development of a focus reduction neutralization test (FRNT) for the detection of mumps virus neutralizing antibodies. J. Virol. Methods 163, 153–156 (2010).
Yang, Q. et al. Establishment and optimization of a rapid and convenient viral RNA transcript copy reduction neutralization test (VcRNT) for quantification of Hantaan orthohantavirus (HTNV) neutralizing antibodies. Virology 608, 110542 (2025).
Acknowledgments
We thank the Center for Medical Innovation of Air Force Medical University for the technical support of the flow cytometry examination. F.L.Z. discloses support for the research of this work from the National Key Research and Development Program of China (No. 2022YFC2604200) and Key Research and Development Project of Shaanxi Province (No. 2019ZDLSF02-04). Z.K.X. discloses support for the research of this work from the National Natural Science Foundation of China (No. 82072268). W.Y. discloses support for the research of this work from the Air Force Medical University (No. 2021JSTS10). L.Z. discloses support for the research of this work from the Air Force Medical University (No. 2025KXKT115). Y.W. discloses support for the research of this work from the Air Force Medical University (No. 2022ZZXM044).
Author information
Authors and Affiliations
Contributions
Conceptualization: W.Y., Y.M.D., and F.L.Z. Methodology: W.Y., Y.M.D., Y.W., Q.Q.Y., H.Z., C.T.Y., J.W., L.Z., L.F.C., H.W.M., and H.L. Resources: C.T.Y., J.W.P., X.M.P., D.S.J., X.J.Y., X.L.J., and Y.C.D. Investigation: W.Y., Y.M.D., Q.Q.Y., H.Z., Y.W., J.W., and C.T.Y. Visualization: W.Y., Q.Q.Y., Z.K.X., Y.F.L., and F.L.Z. Funding acquisition: WY, YW, ZKX, and FLZ. Project administration: Z.K.X. and F.L.Z. Supervision: W.Y., Z.K.X., Y.F.L., and F.L.Z. Writing – original draft: W.Y., Y.M.D., and Q.Q.Y. Writing – review & editing: W.Y., Z.K.X., Y.F.L., and F.L.Z.
Corresponding authors
Ethics declarations
Competing interests
Authors W.Y., F.L.Z., Y.D., L.F.C., H.Z., L.Z., H.L., and Z.K.X. are inventors on a provisional patent application filed in China (Application No. CN202310156260.7) related to the prefusion-stabilized HTNV glycoprotein vaccine design described in this study. All other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Takaaki Koma, Faez Amokrane Nait Mohamed and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ye, W., Dang, Y., Wang, Y. et al. Prefusion-stabilized Hantaan virus glycoprotein nucleic acid vaccine elicits potent neutralizing antibody responses via germinal center activation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70285-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70285-7