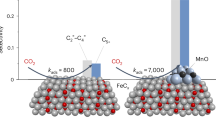
Abstract
Controlling the selectivity of chemical products on small Co nanoparticles is crucial in many catalytic applications. Reaction-driven structural changes offer an alternative methodology to regulate their properties. Herein, carbon-induced surface restructuring occurs on a 2Co/MnOₓ catalyst (cobalt nanoclusters with 2% mass loading on manganese oxide) during thermal CO2 hydrogenation, driven by the formation of bridging Co-C-O-Mn interfacial sites. This leads to a shift in selectivity from methane to CO, with a remarkable enhancement of the CO/CH4 product ratio from 0.89 to 13.4. Such a Co/MnOx system has unique interfacial properties, including strong carbonophilic and oxophilic characteristics. It chemisorbs reaction-derived CO and facilitates C-O bond breaking, promoting rapid CO dissociation and subsequent carbon coverage on Co nanoclusters. This restructuring of Co nanoclusters suppresses the hydrogenation of CO intermediates to methane. This effect is unique to 2Co/MnOₓ and absent at higher/lower Co loadings or other oxide supports. This insight shows how structural evolution during catalysis enables precise surface engineering, overcoming the structure-sensitivity limits of Co nanoclusters.
Similar content being viewed by others
Data availability
Data generated and analyzed in this study are included in the manuscript, Supplementary Information. All data are available from the corresponding author upon request. Source data are provided with this paper.
References
Parastaev, A. et al. Breaking structure sensitivity in CO2 hydrogenation by tuning metal-oxide interfaces in supported cobalt nanoparticles. Nat. Catal. 5, 1051–1060 (2022).
Rommens, K. T. & Saeys, M. Molecular views on Fischer–Tropsch synthesis. Chem. Rev. 123, 5798–5858 (2023).
Lee, S. et al. Subnanometer cobalt oxide clusters as selective low temperature oxidative dehydrogenation catalysts. Nat. Commun. 10, 954 (2019).
Wang, M. et al. Polynuclear cobalt cluster-based coordination polymers for efficient nitrate-to-ammonia electroreduction. J. Am. Chem. Soc. 146, 20439–20448 (2024).
Liu, Y. et al. Sampling the structure and chemical order in assemblies of ferromagnetic nanoparticles by nuclear magnetic resonance. Nat. Commun. 7, 11532 (2016).
den Breejen, J. P. et al. On the origin of the cobalt particle size effects in Fischer-Tropsch catalysis. J. Am. Chem. Soc. 131, 7197–7203 (2009).
Zhang, R., Wang, Y., Gaspard, P. & Kruse, N. The oscillating Fischer-Tropsch reaction. Science 382, 99–103 (2023).
Jiang, Q. et al. Surface oxygenate species on TiC reinforce cobalt-catalyzed Fischer-Tropsch synthesis. ACS Catal. 11, 8087–8096 (2021).
Have, I. C. T. et al. Uncovering the reaction mechanism behind CoO as active phase for CO2 hydrogenation. Nat. Commun. 13, 324 (2022).
Zhong, L. et al. Cobalt carbide nanoprisms for direct production of lower olefins from syngas. Nature 538, 84–87 (2016).
Xiang, Y., Kovarik, L. & Kruse, N. Rate and selectivity hysteresis during the carbon monoxide hydrogenation over promoted Co/MnOx catalysts. Nat. Commun. 10, 3953 (2019).
Gong, K. et al. Carbon-encapsulated metallic Co nanoparticles for Fischer-Tropsch to olefins with low CO2 selectivity. Appl. Catal. B: Environ. 316, 121700 (2022).
Zhang, S., Liu, X., Shao, Z., Wang, H. & Sun, Y. Direct CO2 hydrogenation to ethanol over supported Co2C catalysts: Studies on support effects and mechanism. J. Catal. 382, 86–96 (2020).
Wang, M. et al. Stabilizing Co2C with H2O and K promoter for CO2 hydrogenation to C2+ hydrocarbons. Sci. Adv. 9, eadg0167 (2023).
Ye, J. et al. Hydrogenation of CO2 for sustainable fuel and chemical production. Science 387, eadn9388 (2025).
Ye, R. et al. Design of catalysts for selective CO2 hydrogenation. Nat. Synth. 4, 288–302 (2025).
Feng, Y. et al. Carbon-supported molybdenum nitride with optimized triple-phase interfaces for unprecedented efficiency in reverse water-gas shift reactions. Joule 9, 102163 (2025).
Tao, F. & Salmeron, M. Surface restructuring and predictive design of heterogeneous catalysts. Science 386, eadq0102 (2024).
Wang, H. et al. Managing dynamic catalyst changes to upgrade reactors and reaction processes. Nat. Chem. Eng. 2, 169–180 (2025).
Santos, V. P. et al. Metal organic framework-mediated synthesis of highly active and stable Fischer-Tropsch catalysts. Nat. Commun. 6, 6451 (2015).
Haug, L. et al. Zirconium carbide mediates coke-resistant methane dry reforming on nickel-zirconium catalysts. Angew. Chem. Int. Ed. 61, e202213249 (2022).
Xin, H. et al. Reverse water gas-shift reaction product driven dynamic activation of molybdenum nitride catalyst surface. Nat. Commun. 15, 3100 (2024).
Zhao, Z. et al. Tuning the Fischer–Tropsch reaction over CoxMnyLa/AC catalysts toward alcohols: effects of La promotion. J. Catal. 361, 156–167 (2018).
Chen, H. et al. The role of manganese in CoMnOx catalysts for selective long-chain hydrocarbon production via Fischer-Tropsch synthesis. Nat. Commun. 15, 10294 (2024).
Sun, F. et al. Microstructure evolution of a Co/MnO catalyst for Fischer-Tropsch synthesis revealed by in situ XAFS studies. ChemCatChem 11, 2187–2194 (2019).
Aitbekova, A. et al. Low-temperature restructuring of CeO2-supported Ru nanoparticles determines selectivity in CO2 catalytic reduction. J. Am. Chem. Soc. 140, 13736–13745 (2018).
Koytsoumpa, E. I. & Karellas, S. Equilibrium and kinetic aspects for catalytic methanation focusing on CO2 derived substitute natural gas (SNG). Renew. Sustain. Energy Rev. 94, 536–550 (2018).
Xu, S. et al. CO poisoning of Ru catalysts in CO2 hydrogenation under thermal and plasma conditions: a combined kinetic and diffuse reflectance infrared fourier transform spectroscopy-mass spectrometry study. ACS Catal. 10, 12828–12840 (2020).
Kang, H. et al. Generation of oxide surface patches promoting H-spillover in Ru/(TiOx)MnO catalysts enables CO2 reduction to CO. Nat. Catal. 6, 1062–1072 (2023).
Kang, H. et al. Oxygen vacancy-dependent chemical intermediates on Ru/MnO catalysts dictate the selectivity of CO2 reduction. Appl. Catal. B: Environ. Energy 352, 124010 (2024).
Johnson, G. R., Werner, S. & Bell, A. T. An investigation into the effects of Mn promotion on the activity and selectivity of Co/SiO2 for Fischer-Tropsch synthesis: evidence for enhanced CO adsorption and dissociation. ACS Catal. 5, 5888–5903 (2015).
Bertolini, J., Dalmai-Imelik, G. & Rousseau, J. CO stretching vibration of carbon monoxide adsorbed on nickel (111) studied by high resolution electron loss spectroscopy. Surf. Sci. 68, 539–546 (1977).
Kalhara Gunasooriya, G. K. & Saeys, M. CO adsorption site preference on platinum: charge is the essence. ACS Catal. 8, 3770–3774 (2018).
Bala, K. et al. Copper and cobalt nanoparticles embedded in naturally derived graphite electrodes for the sensing of the neurotransmitter epinephrine. N. J. Chem. 42, 6604–6608 (2018).
Li, Y. et al. Single-atom Co-N-C catalysts for high-efficiency reverse water-gas shift reaction. Appl. Catal. B: Environ. 324, 122298 (2023).
Zhou, X., Price, G. A., Sunley, G. J. & Copéret, C. Small cobalt nanoparticles favor reverse water-gas shift reaction over methanation under CO2 hydrogenation conditions. Angew. Chem. Int. Ed. 62, e202314274 (2023).
Moya-Cancino, J. G. et al. In situ X-ray Raman scattering spectroscopy of the formation of cobalt carbides in a Co/TiO2 Fischer-Tropsch synthesis catalyst. ACS Catal. 11, 809–819 (2021).
Sun, Y.-N., Qin, Z.-H., Lewandowski, M., Shaikhutdinov, S. & Freund, H.-J. CO adsorption and dissociation on iron oxide supported Pt particles. Surf. Sci. 603, 3099–3103 (2009).
Kang, H. et al. Understanding the complexity in bridging thermal and electrocatalytic methanation of CO2. Chem. Soc. Rev. 52, 3627–3662 (2023).
Lee, D.-K., Lee, J.-H. & Ihm, S.-K. Effect of carbon deposits on carbon monoxide hydrogenation over alumina-supported cobalt catalyst. Appl. Catal. 36, 199–207 (1988).
McCarty, J. & Wise, H. Hydrogenation of surface carbon on alumina-supported nickel. J. Catal. 57, 406–416 (1979).
Yang, C. et al. The interplay between structure and product selectivity of CO2 hydrogenation. Angew. Chem. Int. Ed. 58, 11242–11247 (2019).
Xie, Z. et al. CO2 fixation into carbon nanofibres using electrochemical–thermochemical tandem catalysis. Nat. Catal. 7, 98–109 (2024).
van Ravenhorst, I. K. et al. On the cobalt carbide formation in a Co/TiO2 Fischer-Tropsch synthesis catalyst as studied by high-pressure, long-term operando X-ray absorption and diffraction. ACS Catal. 11, 2956–2967 (2021).
Weststrate, C. J. et al. Atomic and polymeric carbon on Co(0001): surface reconstruction, graphene formation, and catalyst poisoning. J. Phys. Chem. C. 116, 11575–11583 (2012).
He, L. et al. Robust and coke-free Ni catalyst stabilized by 1-2 nm-thick multielement oxide for methane dry reforming. ACS Catal. 11, 12409–12416 (2021).
Chen, Y. et al. Visualization of the active sites of zinc-chromium oxides and the CO/H2 activation mechanism in direct syngas conversion. J. Am. Chem. Soc. 146, 1887–1893 (2024).
van Ravenhorst, I. K. et al. Capturing the genesis of an active Fischer–Tropsch synthesis catalyst with operando X-ray nanospectroscopy. Angew. Chem. Int. Ed. 57, 11957–11962 (2018).
Frey, K. et al. Nanostructured MnOx as highly active catalyst for CO oxidation. J. Catal. 287, 30–36 (2012).
Wang, X., Shi, H. & Szanyi, J. Controlling selectivities in CO2 reduction through mechanistic understanding. Nat. Commun. 8, 513 (2017).
Banerjee, A., van Bavel, A. P., Kuipers, H. P. C. E. & Saeys, M. CO activation on realistic cobalt surfaces: kinetic role of hydrogen. ACS Catal. 7, 5289–5293 (2017).
Adams, R. D. & Cotton, F. A. Pathway of bridge-terminal ligand exchange in some binuclear metal carbonyls. Bis(pentahapto-cyclopentadienyldicarbonyliron) and its di(methyl isocyanide) derivative and bis(pentahapto-cyclopentadienylcarbonylnitrosylmanganese). J. Am. Chem. Soc. 95, 6589–6594 (1973).
Pan, S., Zhao, L., Dias, H. V. R. & Frenking, G. Bonding in binuclear carbonyl complexes M2(CO)9 (M = Fe, Ru, Os). Inorg. Chem. 57, 7780–7791 (2018).
Colton, R. & McCormick, M. J. μ2 Bridging carbonyl systems in transition metal complexes. Coord. Chem. Rev. 31, 1–52 (1980).
Hernández Mejía, C., van Deelen, T. W. & de Jong, K. P. Activity enhancement of cobalt catalysts by tuning metal-support interactions. Nat. Commun. 9, 4459 (2018).
Yang, F. et al. Carbon-involved near-surface evolution of cobalt nanocatalysts: an in situ study. CCS Chem. 3, 154–167 (2021).
Bremmer, G. M. et al. In situ TEM observation of the Boudouard reaction: multi-layered graphene formation from CO on cobalt nanoparticles at atmospheric pressure. Faraday Discuss. 197, 337–351 (2017).
Nakamura, J., Toyoshima, I. & Tanaka, K. -i Formation of carbidic and graphite carbon from CO on polycrystalline cobalt. Surf. Sci. 201, 185–194 (1988).
Piao, Y. et al. Identify Zr promotion effects in atomic scale for Co-based catalysts in Fischer-Tropsch synthesis. ACS Catal. 10, 7894–7906 (2020).
Ishihara, T. et al. Effect of alloying on CO hydrogenation activity over SiO2-supported CoNi alloy catalysts. J. Catal. 136, 232–241 (1992).
Zhang, J. et al. Co nanoparticles encapsulated in N-doped carbon nanotube materials derived from new metal-organic frameworks for oxygen electrocatalysis. J. Mater. Chem. A 13, 669–679 (2025).
Liu, Y. et al. Fischer-Tropsch synthesis to α-Olefins with low CO2 selectivity on a Co2C catalyst. Chem. Eng. J. 479, 147927 (2024).
Liu, Y. et al. In situ XAFS study on the formation process of cobalt carbide by Fischer-Tropsch reaction. Phys. Chem. Chem. Phys. 21, 10791–10797 (2019).
Li, W., Zierath, R. & Zou, S. Carbon-encapsulated Co nanoparticle and hollow carbon sphere composites as high-performance catalysts for oxygen reduction reaction. J. Phys. Chem. C. 127, 24489–24497 (2023).
Wang, L.-X., Wang, L. & Xiao, F.-S. Tuning product selectivity in CO2 hydrogenation over metal-based catalysts. Chem. Sci. 12, 14660–14673 (2021).
Li, Y. et al. Single-atom Co-NC catalysts for high-efficiency reverse water-gas shift reaction. Appl. Catal. B: Environ. 324, 122298 (2023).
Acknowledgements
This work was financially supported by the National Key Research and Development Program of China (2024YFB4006600 to Y.L.), the National Natural Science Foundation of China (22502018 to H.K., 22472168 to Y.L., and U23A20132 to R.Z.), Sichuan Science and Technology Program, China (2025ZNSFSC0901 to H.K.), Natural Science Foundation of Liaoning Province (2024-MSBA-57 to Y.L.), Dalian Science and Technology Innovation Fund (2024JJ12RC034 to Y.L.), Dalian Institute of Chemical Physics (DICP I202421 to Y.L.), the Energy Revolution S&T Program of Yulin Innovation Institute of Clean Energy (E411030705 to Y.L.) and the Postdoctoral Science Foundation of China (2024M762752 to L.L.). We also thank in-situ synchrotron XPS experiments supporting from BL02B beamline (31124.02.SSRF.BL02B01) at the Shanghai Synchrotron Radiation Facility (SSRF) in Shanghai, China.
Author information
Authors and Affiliations
Contributions
H.K. conceived and performed the experiments, analysed the data and wrote the paper. R.C. and R.Z. carried out the DFT calculations and wrote the related section. Y.Z., X.W., L.L., W.C., S.P., and G.C. analysed the data and provided helpful discussions. R.Z., S.P., and G.C. revised the paper. Y.L. conceived and directed the research, analysed the data, and wrote the paper. All authors contributed to the discussion and manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kang, H., Cao, R., Zhang, Y. et al. Reaction-induced modification of Co nanoclusters driven by Co-Mn interfacial sites to control selectivity in CO2 hydrogenation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70328-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70328-z