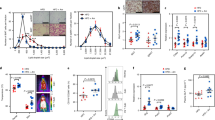
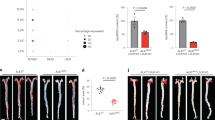
Abstract
Dietary fat absorption is among the most energy-demanding processes of nutrient uptake. Fatty acid activation, triglyceride synthesis, and the trafficking of chylomicrons through the secretory pathway - all require ATP. How enterocytes accommodate the surge in ATP consumption following fat uptake is unclear. We show that the purine biosynthesis/salvage pathway supplies necessary ATP and that Ankyrin Repeat Domain 9 (ANKRD9) couples ATP synthesis and lipoprotein trafficking. Ankrd9 regulates enzymes within the purine biosynthesis pathway to increase ATP synthesis and facilitate Golgi dynamics. Intracellular localization of ANKRD9 is lipid and ATP-dependent. Inactivation of Ankrd9 in mice reduces intestinal ATP despite intact mitochondrial and glycolytic function, alters Golgi morphology, delays ApoB/chylomicron trafficking, and causes lipid accumulation in enterocytes, along with a lean body phenotype. Taken together, the results reveal a previously unrecognized mechanism that regulates lipid absorption in enterocytes and identify ANKRD9 as a central component of this mechanism.
Similar content being viewed by others
Data availability
All the necessary data supporting the conclusions of this research are included within the manuscript, supplementary information, and Source Data files. The raw proteomic data have been deposited to the ProteomeXchange Consortium (https://www.ebi.ac.uk/pride) via the PRIDE partner repository with the dataset identifier PXD073421, with the project title “The role of ANKRD9 in mouse jejunal enteroids”. The raw data pertaining to lipidomics, metabolomics, and nucleotide measurements have been deposited in the MassIVE repository, with accession numbers MSV000100414 for lipidomics, MSV000100413 for metabolomics, and MSV000100415 for nucleotide measurements. The Source Data file accompanying this publication includes data from individual experiments utilized to derive the final figures. These data are provided as a Source Data file accompanying this publication. Source data are provided with this paper.
Code availability
We have developed custom code for the ANKRD9-correlation analysis derived from human single-nuclear sequencing data, which has been uploaded to GitHub: https://github.com/mingqizh/ANKRD9-Intestine-Integration/tree/main.
References
Bonora, M. et al. ATP synthesis and storage. Purinergic Signal 8, 343–357 (2012).
Gessner, P., Lum, J. & Frenguelli, B. G. The mammalian purine salvage pathway as an exploitable route for cerebral bioenergetic support after brain injury. Neuropharmacology 224, 109370 (2023).
Schultheisz, H. L., Szymczyna, B. R., Scott, L. G. & Williamson, J. R. Pathway engineered enzymatic de novo purine nucleotide synthesis. ACS Chem. Biol. 3, 499–511 (2008).
Murray, A. W. The biological significance of purine salvage. Annu Rev. Biochem 40, 811–826 (1971).
Camici, M. et al. Inborn errors of purine salvage and catabolism. Metabolites 13 (2023).
Parra, R. G., Espada, R., Verstraete, N. & Ferreiro, D. U. Structural and energetic characterization of the ankyrin repeat protein family. PLoS Comput. Biol. 11, e1004659 (2015).
Mosavi, L. K., Cammett, T. J., Desrosiers, D. C. & Peng, Z. Y. The ankyrin repeat as molecular architecture for protein recognition. Protein Sci. 13, 1435–1448 (2004).
Sedgwick, S. G. & Smerdon, S. J. The ankyrin repeat: a diversity of interactions on a common structural framework. Trends Biochem. Sci. 24, 311–316 (1999).
Wang, X. et al. Regulation of ANKRD9 expression by lipid metabolic perturbations. BMB Rep. 42, 568–573 (2009).
Lee, Y. et al. ANKRD9 is associated with tumor suppression as a substrate receptor subunit of ubiquitin ligase. Biochim. Biophys. Acta Mol. Basis Dis. 1864, 3145–3153 (2018).
Chen, F. Y. et al. BIK ubiquitination by the E3 ligase Cul5-ASB11 determines cell fate during cellular stress. J. Cell Biol. 218, 3002–3018 (2019).
Hayward, D. et al. ANKRD9 is a metabolically-controlled regulator of IMPDH2 abundance and macro-assembly. J. Biol. Chem. 294, 14454–14466 (2019).
Malinouski, M. et al. Genome-wide RNAi ionomics screen reveals new genes and regulation of human trace element metabolism. Nat. Commun. 5, 3301 (2014).
Ritterhoff, J. & Tian, R. Metabolism in cardiomyopathy: every substrate matters. Cardiovasc Res 113, 411–421 (2017).
Hargreaves, M. & Spriet, L. L. Skeletal muscle energy metabolism during exercise. Nat. Metab. 2, 817–828 (2020).
Spencer, R. P., Brody, K. R. & Bow, T. M. Intestinal levels of adenosine triphosphate, glutamate-pyruvatetransminase, and lactic dehydrogenase. Gastroenterology 44, 282–286 (1963).
Dunn, J. & Grider, M. H. Physiology, Adenosine Triphosphate, in StatPearls (Treasure Island, 2024).
Meddings, J. B. Lipid permeability of rat jejunum and ileum: correlation with physical properties of the microvillus membrane. Biochim. Biophys. Acta 943, 305–314 (1988).
Martinez-Guryn, K. et al. Small intestine microbiota regulate host digestive and absorptive adaptive responses to dietary lipids. Cell Host Microbe 23, 458–469 e455 (2018).
Yen, C. E., Nelson, D. W. & Yen, M. I. Intestinal triacylglycerol synthesis in fat absorption and systemic energy metabolism. J. Lipid Res. 56, 489–501 (2015).
Cooper, A. D. Hepatic uptake of chylomicron remnants. J. Lipid Res. 38, 2173–2192 (1997).
Christensen, N. J., Rubin, C. E., Cheung, M. C. & Albers, J. J. Ultrastructural immunolocalization of apolipoprotein B within human jejunal absorptive cells. J. Lipid Res. 24, 1229–1242 (1983).
Nakajima, K. et al. Apolipoprotein B-48: a unique marker of chylomicron metabolism. Adv. Clin. Chem. 64, 117–177 (2014).
Iqbal, J. & Hussain, M. M. Intestinal lipid absorption. Am. J. Physiol. Endocrinol. Metab. 296, E1183–E1194 (2009).
Abumrad, N. A. & Davidson, N. O. Role of the gut in lipid homeostasis. Physiol. Rev. 92, 1061–1085 (2012).
D’Aquila, T., Hung, Y. H., Carreiro, A. & Buhman, K. K. Recent discoveries on absorption of dietary fat: Presence, synthesis, and metabolism of cytoplasmic lipid droplets within enterocytes. Biochim. Biophys. Acta 1861, 730–747 (2016).
Pierson, H. et al. The function of ATPase copper transporter ATP7B in intestine. Gastroenterology 154, 168–180 e165 (2018).
Morel, E. et al. Lipid-dependent bidirectional traffic of apolipoprotein B in polarized enterocytes. Mol. Biol. Cell 15, 132–141 (2004).
Sarker, R. et al. Phosphorylation of NHE3-S(719) regulates NHE3 activity through the formation of multiple signaling complexes. Mol. Biol. Cell 28, 1754–1767 (2017).
Jackson, R. C., Weber, G. & Morris, H. P. IMP dehydrogenase, an enzyme linked with proliferation and malignancy. Nature 256, 331–333 (1975).
Crabtree, G. W. & Henderson, J. F. Rate-limiting steps in the interconversion of purine ribonucleotides in Ehrlich ascites tumor cells in vitro. Cancer Res. 31, 985–991 (1971).
Lanning, N. J. et al. A mitochondrial RNAi screen defines cellular bioenergetic determinants and identifies an adenylate kinase as a key regulator of ATP levels. Cell Rep. 7, 907–917 (2014).
Mansbach, C. M. & Siddiqi, S. A. The biogenesis of chylomicrons. Annu Rev. Physiol. 72, 315–333 (2010).
Hvorecny, K. L., Hargett, K., Quispe, J. D. & Kollman, J. M. Human PRPS1 filaments stabilize allosteric sites to regulate activity. Nat. Struct. Mol. Biol. 30, 391–402 (2023).
Hu, H. H. et al. Filamentation modulates allosteric regulation of PRPS. Elife 11 (2022).
Qian, X. et al. Conversion of PRPS hexamer to monomer by AMPK-mediated phosphorylation inhibits nucleotide synthesis in response to energy stress. Cancer Discov. 8, 94–107 (2018).
Lu, G. M. et al. Structural basis of human PRPS2 filaments. Cell Biosci. 13, 100 (2023).
Peng, M. et al. The IMPDH cytoophidium couples metabolism and fetal development in mice. Cell Mol. Life Sci. 81, 210 (2024).
Tabula Sapiens, C. et al. The Tabula Sapiens: a multiple-organ, single-cell transcriptomic atlas of humans. Science 376, eabl4896 (2022).
Consortium, G. T. The genotype-tissue expression (GTEx) project. Nat. Genet 45, 580–585 (2013).
Consortium, G. T. The GTEx Consortium atlas of genetic regulatory effects across human tissues. Science 369, 1318–1330 (2020).
Zhou, M. et al. Leveraging inter-individual transcriptional correlation structure to infer discrete signaling mechanisms across metabolic tissues. Elife 12 (2024).
Velez, L. M. et al. Genetic variation of putative myokine signaling is dominated by biological sex and sex hormones. Elife 11 (2022).
Cao, Y. et al. Liver-heart cross-talk mediated by coagulation factor XI protects against heart failure. Science 377, 1399–1406 (2022).
Seldin, M., Yang, X. & Lusis, A. J. Systems genetics applications in metabolism research. Nat. Metab. 1, 1038–1050 (2019).
Moschandrea, C. et al. Mitochondrial dysfunction abrogates dietary lipid processing in enterocytes. Nature 625, 385–392 (2024).
Thompson, A. et al. Tandem mass tags: a novel quantification strategy for comparative analysis of complex protein mixtures by MS/MS. Anal. Chem. 75, 1895–1904 (2003).
Li, J. et al. TMTpro reagents: a set of isobaric labeling mass tags enables simultaneous proteome-wide measurements across 16 samples. Nat. Methods 17, 399–404 (2020).
Chen, P. et al. A LC-MS/MS method for the analysis of intracellular nucleoside triphosphate levels. Pharm. Res. 26, 1504–1515 (2009).
Weinrich, T. W. et al. A day in the life of mitochondria reveals shifting workloads. Sci. Rep. 9, 13898 (2019).
Chen, L. et al. Mapping pesticide-induced metabolic alterations in human gut bacteria. Nat. Commun. 16, 4355 (2025).
Acknowledgements
This work was entirely funded by the National Institutes of Health (NIH) under grant R01DK071865 awarded to S.L., with partial support from NIH grant R35GM133510 to J.Z., and also supported by NIH grants DP1DK130640 and U54OD039864 to M.S. We thank Dr. Mark Donowitz of Johns Hopkins University School of Medicine for sharing the Caco2 cells, Dr. Jennifer Foulke-Abel of Johns Hopkins University School of Medicine for sharing the human enteroids, Dr. Nicholas Zachos of Johns Hopkins University School of Medicine for advice to enteroid culture, Dr. Edward C. Twomey of Johns Hopkins University School of Medicine for helping with the protein complex analysis, Mr. Michael Delannoy of Johns Hopkins Institute for Basic Biomedical Science Microscope Facility for EM sample preparation, Dr. Taekyung Ryu of Johns Hopkins University School of Medicine for uploading the proteomes data to the ProteomeXchange, and all members of Dr. Lutsenko laboratory for useful comments.
Author information
Authors and Affiliations
Contributions
Conceptualization, Y.W. and S.L.; Methodology, Y.W., L.C., Y.M., M.Z., A.G., M.S., J.Z., A.M.Z., X.D., R.N.C., and S.L.; Investigation, Y.W., L.C., Y.M., M.Z., M.S., J.Z., R.N.C., and S.L.; Visualization, Y.W., L.C., Y.M., M.Z., M.S., J.Z., and S.L.; Writing-Original Draft, Y.W. and S.L.; Funding Acquisition, S.L.; All authors contributed to editing and reviewing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Patrick Tso, Jie Luo, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Y., Chen, L., Ma, Y. et al. Enterocytes rely on purine biosynthesis/salvage pathway to facilitate dietary fat absorption. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70332-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70332-3