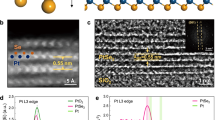
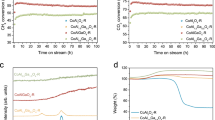
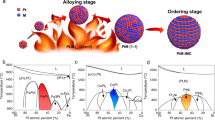
Abstract
Dry reforming of methane (DRM) is plagued by rapid catalyst deactivation, primarily due to carbon deposition exacerbated by exposed Al2O3 surfaces in conventional mixed-phase supports. Herein, we construct a well-defined Pt/TiO2-Al2O3 interface by depositing an ultra-thin anatase TiO2 overlayer onto Al2O3 via an in situ growth strategy to eliminate detrimental Al2O3 exposure. Characterization coupled with DFT calculations reveal that the Al2O3 support induces lattice contraction and electron enrichment of the ultra-thin TiO2 layer through interfacial stress and charge transfer. This concurrently activates lattice oxygen (Ti-O) and optimizes Pt charge density, endowing the catalyst with balanced CH4 activation and a heightened CH* → C* barrier. The resulting Pt/TiO2-Al2O3 catalyst achieves exceptional durability, maintaining 91% CH4 conversion at 800 °C for 100 h with negligible carbon deposition, outperforming Pt/Al2O3 and Pt/TiO2 benchmarks. This work demonstrates that engineering a continuous ultra-thin TiO2 overlayer on Al2O3 is a superior alternative to mixed-phase supports, providing a generalizable blueprint for coke-resistant catalyst design via precise interface control.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the Article and its Supplementary Information. The data generated in this study have been deposited in Figshare and are available via the https://doi.org/10.6084/m9.figshare.30731618. Source data are provided with this paper.
References
Pakhare, D. & Spivey, J. A review of dry (CO2) reforming of methane over noble metal catalysts. Chem. Soc. Rev. 43, 7813–7837 (2014).
Yentekakis, I. V., Panagiotopoulou, P. & Artemakis, G. A review of recent efforts to promote dry reforming of methane (DRM) to syngas production via bimetallic catalyst formulations. Appl. Catal. B Environ. 296, 120210 (2021).
Wang, Z. et al. Insight into the activity of Ni-based thermal catalysts for dry reforming of methane. J. Mater. Chem. A 12, 24802–24838 (2024).
Mondal, U. & Yadav, G. D. Methanol economy and net zero emissions: critical analysis of catalytic processes, reactors and technologies. Green. Chem. 23, 8361–8405 (2021).
Mosaad Awad, M. et al. Recent developments and current trends on catalytic dry reforming of methane: Hydrogen production, thermodynamics analysis, techno feasibility, and machine learning. Energy Convers. Manag. 304, 118252 (2024).
Alhassan, A. M. et al. Advances in catalytic dry reforming of methane (DRM): emerging trends, current challenges, and future perspectives. J. Clean. Prod. 423, 138638 (2023).
Chughtai, A. H., Ahmad, N., Younus, H. A., Laypkov, A. & Verpoort, F. Metal-organic frameworks: versatile heterogeneous catalysts for efficient catalytic organic transformations. Chem. Soc. Rev. 44, 6804–6849 (2015).
Usman, M., Wan Daud, W. M. A. & Abbas, H. F. Dry reforming of methane: Influence of process parameters—a review. Renew. Sustain. Energy Rev. 45, 710–744 (2015).
Cheng, F., Duan, X. & Xie, K. Dry reforming of CH4/CO2 by stable Ni nanocrystals on porous single-crystalline MgO monoliths at reduced temperature. Angew. Chem. Int. Ed. 60, 18792–18799 (2021).
Johnson, O. et al. Stabilizing catalytic performance in dry reforming of methane via surface-engineered intermetallic carbide nanoparticles. J. Am. Chem. Soc. 147, 16786–16791 (2025).
Wang, Q. et al. Interface engineering of oxygen-vacancy-rich MgO/Ni@NiAlO enables low-temperature coke-free methane dry reforming. Chin. J. Catal. 75, 9–20 (2025).
He, D. et al. Designing ultra-stable and surface-exposed Ni nanoparticles with dually confined microenvironment for high-temperature methane dry reforming. Adv. Funct. Mater. 35, 2412895 (2025).
Ding, X. et al. Engineering a nickel-oxygen vacancy interface for enhanced dry reforming of methane: a promoted effect of CeO2 introduction into Ni/MgO. ACS Catal. 13, 15535–15545 (2023).
Wang, Y. et al. Synergy of oxygen vacancies and Ni0 species to promote the stability of a Ni/ZrO2 catalyst for dry reforming of methane at low temperatures. ACS Catal. 13, 6486–6496 (2023).
Jin, B., Li, S., Liu, Y. & Liang, X. Engineering metal-oxide interface by depositing ZrO2 overcoating on Ni/Al2O3 for dry reforming of methane. Chem. Eng. J. 436, 135195 (2022).
Azancot, L., Bobadilla, L. F., Centeno, M. A. & Odriozola, J. A. IR spectroscopic insights into the coking-resistance effect of potassium on nickel-based catalyst during dry reforming of methane. Appl. Catal. B Environ. 285, 119822 (2021).
Azancot, L. et al. Evidence of new Ni-O-K catalytic sites with superior stability for methane dry reforming. Appl. Catal. B Environ. 307, 121148 (2022).
Zhao, Y. et al. Highly porous Ni/MgO-ZrO2 catalysts for dry reforming of methane and the effects of MgO addition on the mechanisms. Chem. Eng. J. 501, 157607 (2024).
Zhang, F. et al. Metal-support interactions and C1 chemistry: transforming Pt-CeO2 into a highly active and stable catalyst for the conversion of carbon dioxide and methane. ACS Catal. 11, 1613–1623 (2021).
Li, Y. et al. Catalytic performance of NixCoy catalysts supported on honeycomb-lantern-like CeO2 for dry reforming of methane: synergistic effect and kinetic study. Chem. Eng. Sci. 291, 119906 (2024).
Gangarajula, Y. et al. Operando induced strong metal-support interaction of Rh/CeO2 catalyst in dry reforming of methane. Appl. Catal. B Environ. 343, 123503 (2024).
Tauster, S. J., Fung, S. C. & Garten, R. L. Strong metal-support interactions. Group 8 noble metals supported on titanium dioxide. J. Am. Chem. Soc. 100, 170–175 (1978).
Chen, S. et al. Defective TiOx overlayers catalyze propane dehydrogenation promoted by base metals. Science 385, 295–300 (2024).
Lu, Z. et al. Modulation of metal-support interactions in Pt-TiOx synergistic catalysts for propane dehydrogenation. Chem. Eng. Sci. 297, 120304 (2024).
Jiang, F. et al. Propane dehydrogenation over Pt/TiO2-Al2O3 catalysts. ACS Catal. 5, 438–447 (2015).
Cui, A., Ren, P., Bai, Y., Yu, H. & Meng, H. Nanoparticle size effect of Pt and TiO2 anatase/rutile phases “volcano-type” curve for HOR electrocatalytic activity at Pt/TiO2-CNx nanocatalysts. Appl. Surf. Sci. 584, 152644 (2022).
Kim, M.-S. et al. Catalytic reduction of nitrate in water over Pd-Cu/TiO2 catalyst: effect of the strong metal-support interaction (SMSI) on the catalytic activity. Appl. Catal. B Environ. 142-143, 354–361 (2013).
Wu, Y. et al. Enhancing alkyne semi-hydrogenation through engineering metal-support interactions of Pd on oxides. Nano Res. 17, 3707–3713 (2024).
Panagiotopoulou, P., Christodoulakis, A., Kondarides, D. I. & Boghosian, S. Particle size effects on the reducibility of titanium dioxide and its relation to the water-gas shift activity of Pt/TiO2 catalysts. J. Catal. 240, 114–125 (2006).
Zhang, J., Zhang, M., Jin, Z., Wang, J. & Zhang, Z. Study of high-temperature hydrogen reduced Pt0/TiO2 by X-ray photoelectron spectroscopy combined with argon ion sputtering—Diffusion-encapsulation effect in relation to strong metal-support interaction. Appl. Surf. Sci. 258, 3991–3999 (2012).
Pongthawornsakun, B. et al. Deposition of Pt nanoparticles on TiO2 by pulsed direct current magnetron sputtering for selective hydrogenation of vanillin to vanillyl alcohol. Catal. Today 358, 51–59 (2020).
Fu, X.-P. et al. Boosting reactivity of water-gas shift reaction by synergistic function over CeO2-x/CoO1-x/Co dual interfacial structures. Nat. Commun. 14, 6851 (2023).
Wang, Q. et al. Operando investigation of toluene oxidation over 1D Pt@CeO2 derived from Pt cluster-containing MOF. J. Am. Chem. Soc. 143, 196–205 (2021).
Gong, C. et al. Construction of active Rh-TiOx interfacial sites on RhFeOx/P25 for highly efficient hydrogenation of CO2 to ethanol. ACS Catal. 14, 17582–17597 (2024).
Rocha, M. A. L. et al. Effect of the Pt oxidation state and Ce3+/Ce4+ ratio on the Pt/TiO2-CeO2 catalysts in the phenol degradation by catalytic wet air oxidation (CWAO). Catal. Today 250, 145–154 (2015).
Ferrari, P. et al. Controlling the adsorption of carbon monoxide on platinum clusters by dopant-induced electronic structure modification. Angew. Chem. Int. Ed. 55, 11059–11063 (2016).
Pakharukov, I. Y. et al. Concentration hysteresis in the oxidation of methane over Pt/γ-Al2O3: X-ray absorption spectroscopy and kinetic study. ACS Catal. 5, 2795–2804 (2015).
Cheng, Z. & Lo, C. S. Effect of support structure and composition on the catalytic activity of Pt nanoclusters for methane dehydrogenation. Ind. Eng. Chem. Res. 52, 15447–15454 (2013).
Wang, F. et al. Tuning the metal-support interaction in catalysts for highly efficient methane dry reforming reaction. Appl. Catal. B Environ. 180, 511–520 (2016).
Khatun, R. et al. Generation of active oxygen species by CO2 dissociation over defect-rich Ni-Pt/CeO2 catalyst for boosting methane activation in low-temperature dry reforming: Experimental and theoretical study. Appl. Catal. B Environ. 340, 123243 (2024).
Daniel, C. et al. Spatially resolved catalysis in microstructured reactors by IR spectroscopy: CO oxidation over mono- and bifunctional Pt catalysts. J. Catal. 272, 55–64 (2010).
Chein, R.-Y. & Fung, W.-Y. Syngas production via dry reforming of methane over CeO2 modified Ni/Al2O3 catalysts. Int. J. Hydrog. Energy 44, 14303–14315 (2019).
Marinho, A. L. A. et al. Highly active and stable Ni dispersed on mesoporous CeO2-Al2O3 catalysts for production of syngas by dry reforming of methane. Appl. Catal. B Environ. 281, 119459 (2021).
Farooqi, A. S. et al. Catalytic conversion of greenhouse gases (CO2 and CH4) to syngas over Ni-based catalyst: effects of Ce-La promoters. Arab. J. Chem. 13, 5740–5749 (2020).
da Fonseca, R. O. et al. Study of the effect of Gd-doping ceria on the performance of Pt/GdCeO2/Al2O3 catalysts for the dry reforming of methane. Catal. Today 355, 737–745 (2020).
da Fonseca, R. O., Rabelo-Neto, R. C., Simões, R. C. C., Mattos, L. V. & Noronha, F. B. Pt supported on doped CeO2/Al2O3 as catalyst for dry reforming of methane. Int. J. Hydrog. Energy 45, 5182–5191 (2020).
Liu, K. et al. Development of a highly stable ternary alloy catalyst for dry reforming of methane. ACS Catal. 13, 3541–3548 (2023).
Xu, M., Zhang, B., Zhang, R., Xia, L. & Liu, G. Zinc migration mediates isolated [PtFe3] in zeolite for propane dehydrogenation. ACS Catal. 15, 3215–3226 (2025).
Chen, H. et al. Dry reforming of methane on bimetallic Pt-Ni@CeO2 catalyst: a in situ DRIFTS-MS mechanistic study. Catal. Sci. Technol. 11, 5260–5272 (2021).
Buchalska, M. et al. On oxygen activation at rutile- and anatase-TiO2. ACS Catal. 5, 7424–7431 (2015).
Pechimuthu, N. A., Pant, K. K. & Dhingra, S. C. Deactivation studies over Ni-K/CeO2-Al2O3 catalyst for dry reforming of methane. Ind. Eng. Chem. Res. 46, 1731–1736 (2007).
Velisoju, V. K. et al. Overcoming the kinetic and deactivation limitations of Ni catalyst by alloying it with Zn for the dry reforming of methane. J. CO2 Util. 75, 102573 (2023).
Tan, X. et al. Titanium oxynitride interlayer to influence oxygen reduction reaction activity and corrosion stability of Pt and Pt-Ni alloy. ChemSusChem 8, 361–376 (2015).
Colombo, R. et al. Surface carbon formation and its impact on methane dry reforming kinetics on rhodium-based catalysts by operando Raman spectroscopy. Angew. Chem. Int. Ed. 63, e202408668 (2024).
Zhang, Y. et al. The tailored role of “defect” sites on γ-alumina: a key to yield an efficient methane dry reforming catalyst with superior nickel utilization. Appl. Catal. B Environ. 315, 121539 (2022).
Zhang, Z.-Y. et al. Promoted solar-driven dry reforming of methane with Pt/mesoporous-TiO2 photo-thermal synergistic catalyst: performance and mechanism study. Energy Convers. Manag. 258, 115496 (2022).
Bachiller-Baeza, B. et al. Transient studies of low-temperature dry reforming of methane over Ni-CaO/ZrO2-La2O3. Appl. Catal. B Environ. 129, 450–459 (2013).
Lu, Y. et al. Double-site doping of a V promoter on Nix-V-MgAl catalysts for the DRM reaction: simultaneous effect on CH4 and CO2 activation. ACS Catal. 11, 8749–8765 (2021).
Liu, Z. et al. Highly active ceria-supported Ru catalyst for the dry reforming of methane: in situ identification of Ruδ+-Ce3+ interactions for enhanced conversion. ACS Catal. 9, 3349–3359 (2019).
Kim, S., Lauterbach, J. & Sasmaz, E. Yolk-shell Pt-NiCe@SiO2 single-atom-alloy catalysts for low-temperature dry reforming of methane. ACS Catal. 11, 8247–8260 (2021).
Jiménez, J. D. et al. Identification of highly selective surface pathways for methane dry reforming using mechanochemical synthesis of Pd-CeO2. ACS Catal. 12, 12809–12822 (2022).
Li, K. et al. Ordered mesoporous Ni/La2O3 catalysts with interfacial synergism towards CO2 activation in dry reforming of methane. Appl. Catal. B Environ. 259, 118092 (2019).
Lv, J. et al. Dry-gel synthesis of hierarchical Ni-La@S-1 catalysts with stabilized Ni-La bimetals nanoparticles for dry reforming of methane. Nano Res. 17, 10216–10226 (2024).
Song, P. et al. Ni-Mg-Al catalysts for dry reforming of methane: effect of surface properties on coke formation and CO2 activation. Ind. Eng. Chem. Res. 63, 16077–16090 (2024).
Bobadilla, L. F., Garcilaso, V., Centeno, M. A. & Odriozola, J. A. Monitoring the reaction mechanism in model biogas reforming by in situ transient and steady-state DRIFTS measurements. ChemSusChem 10, 1193–1201 (2017).
Gong, Z.-J., Li, Y.-R., Wu, H.-L., Lin, S. D. & Yu, W.-Y. Direct copolymerization of carbon dioxide and 1,4-butanediol enhanced by ceria nanorod catalyst. Appl. Catal. B Environ. 265, 118524 (2020).
Chen, Y. et al. Continuous dimethyl carbonate synthesis from CO2 and methanol over BixCe1−xOδ monoliths: Effect of bismuth doping on population of oxygen vacancies, activity, and reaction pathway. Nano Res. 15, 1366–1374 (2022).
Song, X. & Boily, J.-F. Carbon dioxide binding at dry FeOOH mineral surfaces: evidence for structure-controlled speciation. Environ. Sci. Technol. 47, 9241–9248 (2013).
Zhang, Z.-Y. et al. Deep insight of the influence of Pt loading content with catalytic activity on light-assisted dry reforming of methane. Chem. Eng. Sci. 274, 118710 (2023).
Aso, R. et al. Direct identification of the charge state in a single platinum nanoparticle on titanium oxide. Science 378, 202–206 (2022).
Cai, Y. & Feng, Y. P. Review on charge transfer and chemical activity of TiO2: mechanism and applications. Prog. Surf. Sci. 91, 183–202 (2016).
Andersen, M. Revelations of the d band. Nat. Catal. 6, 460–461 (2023).
Hammer, B. & Norskov, J. K. Why gold is the noblest of all the metals. Nature 376, 238–240 (1995).
Tang, Y. et al. Synergy of single-atom Ni1 and Ru1 sites on CeO2 for dry reforming of CH4. J. Am. Chem. Soc. 141, 7283–7293 (2019).
Acknowledgements
The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China (22508298, R.R.Z., 22025802, G.Z.L., 22308257, G.Z.L.).
Author information
Authors and Affiliations
Contributions
G.Z.L., R.R.Z., S.S.Z., L.W., and X.W.Z. conceived and supervised the project. S.S.Z. synthesized the catalysts, performed catalytic experiments and density functional theory (DFT) calculations, and drafted the manuscript. S.Z.L. and R.C.L. contributed to the discussion of the DFT analysis. R.R.Z. and L.W. provided guidance on data interpretation and manuscript revision. All authors discussed the results and contributed to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Adel Boualouache, Changwei Hu, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, S., Wang, L., Lyu, S. et al. Modulation of Pt electron transfer via engineered ultra-thin TiO2-Al2O3 interfaces for coke-resistant methane dry reforming. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70338-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70338-x