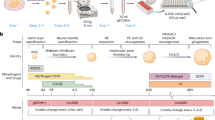
Abstract
The emergence of functional cerebellar circuits is heavily influenced by activity-dependent processes. However, the contribution of intrinsic Purkinje cell activity to cerebellar development remains less understood. Here, we demonstrate that before synaptic networks mature, Purkinje cell intrinsic activity is essential for regulating dendritic growth, establishing connections with cerebellar nuclei, and ensuring proper cerebellar function. Disrupting this activity during the postnatal period impairs motor function, with earlier perturbations causing more severe deficits. Importantly, only early developmental disruptions lead to pronounced defects in cellular morphology, highlighting key temporal windows for dendritic growth and maturation. Transcriptomic analyses reveal that early intrinsic activity drives the expression of activity-dependent genes, including Prkcg and Car8, which are essential for dendritic development. Our findings emphasize the importance of temporally regulated intrinsic activity in Purkinje cells in guiding cerebellar circuit development, providing a potential unifying mechanism underlying cerebellum-associated disorders.
Similar content being viewed by others
Data availability
Sequencing data generated in this study have been deposited in the National Center for Biotechnology Information (NCBI) Gene Expression Omnibus (GEO) under the accession number GSE294208 and are publicly available. Source data are provided with this paper.
Code availability
This study did not generate original codes. For LocoMouse, Compensatory Eye Movements, and EBC analyses, the codes we previously reported and deposited on GitHub: https://github.com/BaduraLab/DLC_analysis31, https://github.com/MSchonewille/iMove130, and https://github.com/francescafiocchi91/Eyeblink_Conditioning36, respectively.
References
Spitzer, N. C. Electrical activity in early neuronal development. Nature 444, 707–712 (2006).
Blankenship, A. G. & Feller, M. B. Mechanisms underlying spontaneous patterned activity in developing neural circuits. Nat. Rev. Neurosci. 11, 18–29 (2010).
Penn, A. A., Riquelme, P. A., Feller, M. B. & Shatz, C. J. Competition in retinogeniculate patterning driven by spontaneous activity. Science 279, 2108–2112 (1998).
McLaughlin, T., Torborg, C. L., Feller, M. B. & O’Leary, D. D. Retinotopic map refinement requires spontaneous retinal waves during a brief critical period of development. Neuron 40, 1147–1160 (2003).
Hanson, M. G. & Landmesser, L. T. Normal patterns of spontaneous activity are required for correct motor axon guidance and the expression of specific guidance molecules. Neuron 43, 687–701 (2004).
Hanson, M. G. & Landmesser, L. T. Increasing the frequency of spontaneous rhythmic activity disrupts pool-specific axon fasciculation and pathfinding of embryonic spinal motoneurons. J. Neurosci. 26, 12769–12780 (2006).
Tritsch, N. X., Yi, E., Gale, J. E., Glowatzki, E. & Bergles, D. E. The origin of spontaneous activity in the developing auditory system. Nature 450, 50–55 (2007).
Anton-Bolanos, N. et al. Prenatal activity from thalamic neurons governs the emergence of functional cortical maps in mice. Science 364, 987–990 (2019).
De Marco Garcia, N. V., Karayannis, T. & Fishell, G. Neuronal activity is required for the development of specific cortical interneuron subtypes. Nature 472, 351–355 (2011).
Herculano-Houzel, S. The human brain in numbers: a linearly scaled-up primate brain. Front. Hum. Neurosci. 3, 31 (2009).
Sathyanesan, A. et al. Emerging connections between cerebellar development, behaviour and complex brain disorders. Nat. Rev. Neurosci. 20, 298–313 (2019).
Cook, A. A., Fields, E. & Watt, A. J. Losing the beat: contribution of Purkinje cell firing dysfunction to disease, and its reversal. Neuroscience 462, 247–261 (2021).
Raman, I. M. & Bean, B. P. Resurgent sodium current and action potential formation in dissociated cerebellar Purkinje neurons. J. Neurosci. 17, 4517–4526 (1997).
Hausser, M. & Clark, B. A. Tonic synaptic inhibition modulates neuronal output pattern and spatiotemporal synaptic integration. Neuron 19, 665–678 (1997).
Leto, K. et al. Consensus paper: cerebellar development. Cerebellum 15, 789–828 (2016).
Kozareva, V. et al. A transcriptomic atlas of mouse cerebellar cortex comprehensively defines cell types. Nature 598, 214–219 (2021).
Sepp, M. et al. Cellular development and evolution of the mammalian cerebellum. Nature 625, 788–796 (2024).
White, J. J. et al. Cerebellar zonal patterning relies on Purkinje cell neurotransmission. J. Neurosci. 34, 8231–8245 (2014).
Hashimoto, K. et al. Postsynaptic P/Q-type Ca2+ channel in Purkinje cell mediates synaptic competition and elimination in developing cerebellum. Proc. Natl. Acad. Sci. USA 108, 9987–9992 (2011).
Mikuni, T. et al. Arc/Arg3.1 is a postsynaptic mediator of activity-dependent synapse elimination in the developing cerebellum. Neuron 78, 1024–1035 (2013).
Beekhof, G. C. et al. Differential spatiotemporal development of Purkinje cell populations and cerebellum-dependent sensorimotor behaviors. Elife 10, e63668 (2021).
Wu, B. et al. TRPC3 is a major contributor to functional heterogeneity of cerebellar Purkinje cells. Elife 8, e45590 (2019).
Moreno-Juan, V. et al. Prenatal thalamic waves regulate cortical area size prior to sensory processing. Nat. Commun. 8, 14172 (2017).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Zhou, H. et al. Cerebellar modules operate at different frequencies. Elife 3, e02536 (2014).
Gold, D. A., Gent, P. M. & Hamilton, B. A. ROR alpha in genetic control of cerebellum development: 50 staggering years. Brain Res. 1140, 19–25 (2007).
Lalonde, R. & Strazielle, C. Spontaneous and induced mouse mutations with cerebellar dysfunctions: behavior and neurochemistry. Brain Res. 1140, 51–74 (2007).
Reeber, S. L., Otis, T. S. & Sillitoe, R. V. New roles for the cerebellum in health and disease. Front. Syst. Neurosci. 7, 83 (2013).
Salman, M. S. & Tsai, P. The role of the pediatric cerebellum in motor functions, cognition, and behavior: a clinical perspective. Neuroimaging Clin. N. Am. 26, 317–329 (2016).
Machado, A. S., Darmohray, D. M., Fayad, J., Marques, H. G. & Carey, M. R. A quantitative framework for whole-body coordination reveals specific deficits in freely walking ataxic mice. Elife 4, e07892 (2015).
Kaiser, F. M. P. et al. Biallelic PAX5 mutations cause hypogammaglobulinemia, sensorimotor deficits, and autism spectrum disorder. J. Exp. Med. 219, e20220498 (2022).
Voges, K., Wu, B., Post, L., Schonewille, M. & De Zeeuw, C. I. Mechanisms underlying vestibulo-cerebellar motor learning in mice depend on movement direction. J. Physiol. 595, 5301–5326 (2017).
Bonnan, A., Rowan, M. M. J., Baker, C. A., Bolton, M. M. & Christie, J. M. Autonomous Purkinje cell activation instructs bidirectional motor learning through evoked dendritic calcium signaling. Nat. Commun. 12, 2153 (2021).
ten Brinke, M. M. et al. Evolving models of pavlovian conditioning: cerebellar cortical dynamics in awake behaving mice. Cell Rep. 13, 1977–1988 (2015).
Jirenhed, D. A., Bengtsson, F. & Hesslow, G. Acquisition, extinction, and reacquisition of a cerebellar cortical memory trace. J. Neurosci. 27, 2493–2502 (2007).
Osorio, C. et al. Pre-ataxic loss of intrinsic plasticity and motor learning in a mouse model of SCA1. Brain 146, 2332–2345 (2023).
Ebner, B. A. et al. Purkinje cell ataxin-1 modulates climbing fiber synaptic input in developing and adult mouse cerebellum. J. Neurosci. 33, 5806–5820 (2013).
Furrer, S. A. et al. Reduction of mutant ataxin-7 expression restores motor function and prevents cerebellar synaptic reorganization in a conditional mouse model of SCA7. Hum. Mol. Genet. 22, 890–903 (2013).
Lin, C. C. et al. Reduced cerebellar rhythm by climbing fiber denervation is linked to motor rhythm deficits in mice and ataxia severity in patients. Sci. Transl. Med. 17, eadk3922 (2025).
Tempia, F. et al. Parallel fiber to Purkinje cell synaptic impairment in a mouse model of spinocerebellar ataxia type 27. Front. Cell Neurosci. 9, 205 (2015).
Kurihara, H. et al. Impaired parallel fiber->Purkinje cell synapse stabilization during cerebellar development of mutant mice lacking the glutamate receptor delta2 subunit. J. Neurosci. 17, 9613–9623 (1997).
Sotelo, C. & Rossi, F. Purkinje Cell Migration and Differentiation (Springer, 2013).
Telley, L. et al. Dual function of NRP1 in axon guidance and subcellular target recognition in cerebellum. Neuron 91, 1276–1291 (2016).
Fleming, J. T. et al. The Purkinje neuron acts as a central regulator of spatially and functionally distinct cerebellar precursors. Dev. Cell 27, 278–292 (2013).
Wechsler-Reya, R. J. & Scott, M. P. Control of neuronal precursor proliferation in the cerebellum by Sonic Hedgehog. Neuron 22, 103–114 (1999).
Lewis, P. M., Gritli-Linde, A., Smeyne, R., Kottmann, A. & McMahon, A. P. Sonic hedgehog signaling is required for expansion of granule neuron precursors and patterning of the mouse cerebellum. Dev. Biol. 270, 393–410 (2004).
Stichel, C. C., Kagi, U. & Heizmann, C. W. Parvalbumin in cat brain: isolation, characterization, and localization. J. Neurochem 47, 46–53 (1986).
Schilling, K. & Oberdick, J. The treasury of the commons: making use of public gene expression resources to better characterize the molecular diversity of inhibitory interneurons in the cerebellar cortex. Cerebellum 8, 477–489 (2009).
Voerman, S., Broersen, R., Swagemakers, S. M. A., De Zeeuw, C. I. & van der Spek, P. J. Plasticity mechanisms of genetically distinct Purkinje cells. Bioessays 46, e2400008 (2024).
Saito, N., Kikkawa, U., Nishizuka, Y. & Tanaka, C. Distribution of protein kinase C-like immunoreactive neurons in rat brain. J. Neurosci. 8, 369–382 (1988).
Kato, K. Sequence of a novel carbonic anhydrase-related polypeptide and its exclusive presence in Purkinje cells. FEBS Lett. 271, 137–140 (1990).
Chen, D. H., Raskind, W. H. & Bird, T. D. Spinocerebellar ataxia type 14. Handb. Clin. Neurol. 103, 555–559 (2012).
Turkmen, S. et al. CA8 mutations cause a novel syndrome characterized by ataxia and mild mental retardation with predisposition to quadrupedal gait. PLoS Genet. 5, e1000487 (2009).
Kaya, N. et al. Phenotypical spectrum of cerebellar ataxia associated with a novel mutation in the CA8 gene, encoding carbonic anhydrase (CA) VIII. Am. J. Med. Genet B Neuropsychiatr. Genet. 156B, 826–834 (2011).
Yu, C. R. et al. Spontaneous neural activity is required for the establishment and maintenance of the olfactory sensory map. Neuron 42, 553–566 (2004).
Cajal, S.R. Histologie du Système Nerveux de l’Homme et des Vertébrés (Maloine, 1911).
Yamashiro, K. et al. AUTS2 governs cerebellar development, Purkinje cell maturation, motor function and social communication. iScience 23, 101820 (2020).
Hori, K., Shimaoka, K. & Hoshino, M. AUTS2 gene: keys to understanding the pathogenesis of neurodevelopmental disorders. Cells 11, 11 (2021).
Ibrahim, M. F., Power, E. M., Potapov, K. & Empson, R. M. Motor and cerebellar architectural abnormalities during the early progression of ataxia in a mouse model of SCA1 and how early prevention leads to a better outcome later in life. Front Cell Neurosci. 11, 292 (2017).
Serra, H. G. et al. RORalpha-mediated Purkinje cell development determines disease severity in adult SCA1 mice. Cell 127, 697–708 (2006).
Gibson, J. M. et al. A critical period for development of cerebellar-mediated autism-relevant social behavior. J. Neurosci. 42, 2804–2823 (2022).
Tsai, P. T. et al. Sensitive periods for cerebellar-mediated autistic-like behaviors. Cell Rep. 25, 357–367.e354 (2018).
Sudhof, T. C. Towards an understanding of synapse formation. Neuron 100, 276–293 (2018).
Kuwajima, T. et al. Optic chiasm presentation of Semaphorin6D in the context of Plexin-A1 and Nr-CAM promotes retinal axon midline crossing. Neuron 74, 676–690 (2012).
Nakanishi, Y. et al. Semaphorin 6D tunes amygdalar circuits for emotional, metabolic, and inflammatory outputs. Neuron 112, 2955–2972.e2959 (2024).
Guerrier, S. et al. The F-BAR domain of srGAP2 induces membrane protrusions required for neuronal migration and morphogenesis. Cell 138, 990–1004 (2009).
Schmidt, E. R. E., Kupferman, J. V., Stackmann, M. & Polleux, F. The human-specific paralogs SRGAP2B and SRGAP2C differentially modulate SRGAP2A-dependent synaptic development. Sci. Rep. 9, 18692 (2019).
Watt, A. J. et al. Traveling waves in developing cerebellar cortex mediated by asymmetrical Purkinje cell connectivity. Nat. Neurosci. 12, 463–473 (2009).
Eilers, J., Plant, T. D., Marandi, N. & Konnerth, A. GABA-mediated Ca2+ signalling in developing rat cerebellar Purkinje neurones. J. Physiol. 536, 429–437 (2001).
Rosenberg, S. S. & Spitzer, N. C. Calcium signaling in neuronal development. Cold Spring Harb. Perspect. Biol. 3, a004259 (2011).
Thompson, V. B., Jinnah, H. A. & Hess, E. J. Convergent mechanisms in etiologically-diverse dystonias. Expert Opin. Ther. Targets 15, 1387–1403 (2011).
Mbaya, E. et al. Calcium signalling-dependent mitochondrial dysfunction and bioenergetics regulation in respiratory chain Complex II deficiency. Cell Death Differ. 17, 1855–1866 (2010).
Ryu, C. et al. STIM1 regulates somatic Ca(2+) signals and intrinsic firing properties of cerebellar Purkinje neurons. J. Neurosci. 37, 8876–8894 (2017).
Nicholls, D. G. Mitochondria and calcium signaling. Cell Calcium 38, 311–317 (2005).
Zheng, H. et al. Mitochondrial oxidation of the carbohydrate fuel is required for neural precursor/stem cell function and postnatal cerebellar development. Sci. Adv. 4, eaat2681 (2018).
Nowicky, A. V. & Duchen, M. R. Changes in [Ca2+]i and membrane currents during impaired mitochondrial metabolism in dissociated rat hippocampal neurons. J. Physiol. 507, 131–145 (1998).
Datta, S. & Jaiswal, M. Mitochondrial calcium at the synapse. Mitochondrion 59, 135–153 (2021).
Inbar-Feigenberg, M. et al. Mitochondrial POLG related disorder presenting prenatally with fetal cerebellar growth arrest. Metab. Brain Dis. 33, 1369–1373 (2018).
Di Bella, D. et al. Mutations in the mitochondrial protease gene AFG3L2 cause dominant hereditary ataxia SCA28. Nat. Genet. 42, 313–321 (2010).
Leto, K., Carletti, B., Williams, I. M., Magrassi, L. & Rossi, F. Different types of cerebellar GABAergic interneurons originate from a common pool of multipotent progenitor cells. J. Neurosci. 26, 11682–11694 (2006).
Li, W. et al. Dendritic inhibition by Shh signaling-dependent stellate cell pool is critical for motor learning. J. Neurosci. 42, 5130–5143 (2022).
Solbach, S. & Celio, M. R. Ontogeny of the calcium binding protein parvalbumin in the rat nervous system. Anat. Embryol. 184, 103–124 (1991).
Collin, T. et al. Developmental changes in parvalbumin regulate presynaptic Ca2+ signaling. J. Neurosci. 25, 96–107 (2005).
Watanabe, M. & Kano, M. Climbing fiber synapse elimination in cerebellar Purkinje cells. Eur. J. Neurosci. 34, 1697–1710 (2011).
Consalez, G. G., Goldowitz, D., Casoni, F. & Hawkes, R. Origins, development, and compartmentation of the granule cells of the cerebellum. Front. Neural Circuits 14, 611841 (2020).
Sudarov, A. et al. Ascl1 genetics reveals insights into cerebellum local circuit assembly. J. Neurosci. 31, 11055–11069 (2011).
Hourez, R. et al. Aminopyridines correct early dysfunction and delay neurodegeneration in a mouse model of spinocerebellar ataxia type 1. J. Neurosci. 31, 11795–11807 (2011).
Dougherty, S. E. et al. Disruption of Purkinje cell function prior to huntingtin accumulation and cell loss in an animal model of Huntington disease. Exp. Neurol. 236, 171–178 (2012).
Tsai, P. T. et al. Autistic-like behaviour and cerebellar dysfunction in Purkinje cell Tsc1 mutant mice. Nature 488, 647–651 (2012).
Sathyanesan, A., Kundu, S., Abbah, J. & Gallo, V. Neonatal brain injury causes cerebellar learning deficits and Purkinje cell dysfunction. Nat. Commun. 9, 3235 (2018).
Leroux, S. et al. Intermittent hypoxia in a mouse model of apnea of prematurity leads to a retardation of cerebellar development and long-term functional deficits. Cell Biosci. 12, 148 (2022).
Kirischuk, S. et al. Modulation of neocortical development by early neuronal activity: physiology and pathophysiology. Front. Cell Neurosci. 11, 379 (2017).
Shakkottai, V. G. et al. Enhanced neuronal excitability in the absence of neurodegeneration induces cerebellar ataxia. J. Clin. Invest. 113, 582–590 (2004).
Liu, X. X. et al. BOD1 regulates the cerebellar IV/V lobe-fastigial nucleus circuit associated with motor coordination. Signal Transduct. Target Ther. 7, 170 (2022).
Mayoral-Palarz, K. et al. Cerebellar neuronal dysfunction accompanies early motor symptoms in spinocerebellar ataxia type 3. Dis. Model Mech. 15, dmm049514 (2022).
Zheng, N. & Raman, I. M. Synaptic inhibition, excitation, and plasticity in neurons of the cerebellar nuclei. Cerebellum 9, 56–66 (2010).
Titley, H. K. & Hansel, C. Asymmetries in cerebellar plasticity and motor learning. Cerebellum 15, 87–92 (2016).
Gao, Z., van Beugen, B. J. & De Zeeuw, C. I. Distributed synergistic plasticity and cerebellar learning. Nat. Rev. Neurosci. 13, 619–635 (2012).
Lorenzetto, E. et al. Genetic perturbation of postsynaptic activity regulates synapse elimination in developing cerebellum. Proc. Natl. Acad. Sci. USA 106, 16475–16480 (2009).
Kano, M. et al. Persistent multiple climbing fiber innervation of cerebellar Purkinje cells in mice lacking mGluR1. Neuron 18, 71–79 (1997).
Hills, L. B. et al. Deletions in GRID2 lead to a recessive syndrome of cerebellar ataxia and tonic upgaze in humans. Neurology 81, 1378–1386 (2013).
Watson, L. M. et al. Dominant mutations in GRM1 cause spinocerebellar ataxia type 44. Am. J. Hum. Genet. 101, 866 (2017).
Smeets, C. J. & Verbeek, D. S. Climbing fibers in spinocerebellar ataxia: a mechanism for the loss of motor control. Neurobiol. Dis. 88, 96–106 (2016).
Kose, A. et al. Electron microscopic localization of type I protein kinase C in rat Purkinje cells. J. Neurosci. 8, 4262–4268 (1988).
Chen, C. et al. Impaired motor coordination correlates with persistent multiple climbing fiber innervation in PKC gamma mutant mice. Cell 83, 1233–1242 (1995).
Daniel, H., Levenes, C. & Crepel, F. Cellular mechanisms of cerebellar LTD. Trends Neurosci. 21, 401–407 (1998).
De Michele, G. et al. Episodic ataxia and severe infantile phenotype in spinocerebellar ataxia type 14: expansion of the phenotype and novel mutations. J. Neurol. 269, 1476–1484 (2022).
Shimobayashi, E. & Kapfhammer, J. P. A new mouse model related to SCA14 carrying a pseudosubstrate domain mutation in PKCgamma shows perturbed Purkinje cell maturation and ataxic motor behavior. J. Neurosci. 41, 2053–2068 (2021).
Wong, M. M. K. et al. Neurodegeneration in SCA14 is associated with increased PKCgamma kinase activity, mislocalization and aggregation. Acta Neuropathol. Commun. 6, 99 (2018).
Verbeek, D. S., Goedhart, J., Bruinsma, L., Sinke, R. J. & Reits, E. A. PKC gamma mutations in spinocerebellar ataxia type 14 affect C1 domain accessibility and kinase activity leading to aberrant MAPK signaling. J. Cell Sci. 121, 2339–2349 (2008).
Shimobayashi, E. & Kapfhammer, J. P. Increased biological activity of protein Kinase C gamma is not required in Spinocerebellar ataxia 14. Mol. Brain 10, 34 (2017).
Metzger, F. & Kapfhammer, J. P. Protein kinase C activity modulates dendritic differentiation of rat Purkinje cells in cerebellar slice cultures. Eur. J. Neurosci. 12, 1993–2005 (2000).
Kaiyrzhanov, R. et al. Clinical and molecular spectrum of autosomal recessive CA8-related cerebellar ataxia. Mov. Disord. 39, 983–995 (2024).
Miterko, L. N. et al. Persistent motor dysfunction despite homeostatic rescue of cerebellar morphogenesis in the Car8 waddles mutant mouse. Neural Dev. 14, 6 (2019).
Jiao, Y. et al. Carbonic anhydrase-related protein VIII deficiency is associated with a distinctive lifelong gait disorder in waddles mice. Genetics 171, 1239–1246 (2005).
Shabani, K. & Hassan, B. A. The brain on time: links between development and neurodegeneration. Development 150, dev200397 (2023).
Millen, K. J. & Gleeson, J. G. Cerebellar development and disease. Curr. Opin. Neurobiol. 18, 12–19 (2008).
Zhang, X. M. et al. Highly restricted expression of Cre recombinase in cerebellar Purkinje cells. Genesis 40, 45–51 (2004).
Holt, G. R., Softky, W. R., Koch, C. & Douglas, R. J. Comparison of discharge variability in vitro and in vivo in cat visual cortex neurons. J. Neurophysiol. 75, 1806–1814 (1996).
Ferreira, T. A. et al. Neuronal morphometry directly from bitmap images. Nat. Methods 11, 982–984 (2014).
Willett, R. T. et al. Cerebellar nuclei excitatory neurons regulate developmental scaling of presynaptic Purkinje cell number and organ growth. Elife 8, e50617 (2019).
Kebschull, J. M. et al. Cerebellar nuclei evolved by repeatedly duplicating a conserved cell-type set. Science 370, eabd5059 (2020).
Arganda-Carreras, I. et al. Trainable Weka segmentation: a machine learning tool for microscopy pixel classification. Bioinformatics 33, 2424–2426 (2017).
Matsuda, T. & Cepko, C. L. Electroporation and RNA interference in the rodent retina in vivo and in vitro. Proc. Natl. Acad. Sci. USA 101, 16–22 (2004).
Xue, M., Atallah, B. V. & Scanziani, M. Equalizing excitation-inhibition ratios across visual cortical neurons. Nature 511, 596–600 (2014).
Luong, T. N., Carlisle, H. J., Southwell, A. & Patterson, P. H. Assessment of motor balance and coordination in mice using the balance beam. J. Vis. Exp. 10, 2376 (2011).
Mathis, A. et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat. Neurosci. 21, 1281–1289 (2018).
Ben-Shaul, Y. OptiMouse: a comprehensive open source program for reliable detection and analysis of mouse body and nose positions. BMC Biol. 15, 41 (2017).
Stahl, J. S. Using eye movements to assess brain function in mice. Vis. Res. 44, 3401–3410 (2004).
Schonewille, M. et al. Purkinje cell-specific knockout of the protein phosphatase PP2B impairs potentiation and cerebellar motor learning. Neuron 67, 618–628 (2010).
Heiney, S. A., Wohl, M. P., Chettih, S. N., Ruffolo, L. I. & Medina, J. F. Cerebellar-dependent expression of motor learning during eyeblink conditioning in head-fixed mice. J. Neurosci. 34, 14845–14853 (2014).
Boele, H. J. et al. Impact of parallel fiber to Purkinje cell long-term depression is unmasked in absence of inhibitory input. Sci. Adv. 4, eaas9426 (2018).
Picelli, S. et al. Full-length RNA-seq from single cells using Smart-seq2. Nat. Protoc. 9, 171–181 (2014).
Law, C. W. et al. RNA-seq analysis is easy as 1-2-3 with limma, Glimma and edgeR. F1000Res 5, ISCB Comm J-1408 (2016).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Csardi, G. & Nepusz, T. The igraph software package for complex network research. InterJournal Complex Systems 1695 http://igraph.org (2006).
Favuzzi, E. et al. Distinct molecular programs regulate synapse specificity in cortical inhibitory circuits. Science 363, 413–417 (2019).
Verhaagen, J. et al. Small scale production of recombinant adeno-associated viral vectors for gene delivery to the nervous system. Methods Mol. Biol. 1715, 3–17 (2018).
Kim, J. Y. et al. Viral transduction of the neonatal brain delivers controllable genetic mosaicism for visualising and manipulating neuronal circuits in vivo. Eur. J. Neurosci. 37, 1203–1220 (2013).
Acknowledgements
The authors kindly thank all personnel of the animal facility at Erasmus Medical Center Rotterdam, as well as Elize Haasdijk, Erika Sabel-Goedknegt, and Doriane Gisquet for excellent technical assistance. We also thank Guillermina López-Bendito for providing the Kir2.1 (Gt(ROSA)26Sortm2(CAG-KCNJ2/mCherry)Fmr) mouse model. This work was supported by H2020 European Research Council (ERC Starting Grant #680235), Erasmus MC Flagship Convergence Incentive Grant, and Dutch Research Council (NWO) through OCENW.M.22.046 to M.S.; Netherlands Organization for Health Research and Development (ZonMW Off-road grant 451001027), NWO through OCENW.XS5.121 and Incentive Grant for Women in STEM (19498) to C.O.; NWO VENI fellowship (016.Veni.192.270), and OCENW.XS21.1.087 to J.J.W.; China Scholarship Council (202407720046) to J.Y.; and Royal Netherlands Academy of Arts and Sciences (KNAW) Research Fund and the Leiden Regenerative Medicine Platform for AAV vector production to F.d.W.
Author information
Authors and Affiliations
Contributions
Conceptualization—C.O., L.L., G.M.v.W., and M.S. Methodology—C.O., J.J.W., and M.S. Investigation—C.O., J.J.W., P.T.-S., N.M., F.O., F.K., L.P., E.R., and M.S. Data curation—C.O., J.J.W., P.T.-S., J.Y., N.M., F.O., F.K., Z.H., I.S., S.T., and M.S. Formal analysis—C.O., J.J.W., P.T.-S., J.Y., F.R.F., I.S., and M.S. Resources—C.O., J.J.W., F.d.W., F.R.F., S.T., Z.O., M.C.G.N.H., W.F.J.I., G.L.-B., A.B., and M.S. Software—C.O., J.J.W., J.Y., F.R.F., S.T. and M.S. Visualization—C.O. Writing—original draft—C.O. Writing—review and editing—all authors. Funding acquisition—C.O., J.J.W., J.Y., F.d.W., and M.S. Supervision—C.O., J.J.W., and M.S.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Alanna Watt and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Osório, C., White, J.J., Torrents-Solé, P. et al. Purkinje cell intrinsic activity shapes cerebellar development and function. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70355-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70355-w