Abstract
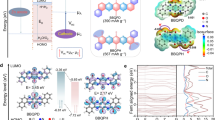
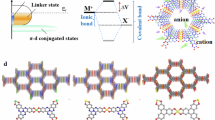
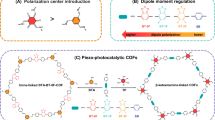
Covalent organic frameworks containing periodic redox-active motifs and conjugation structures are booming as competitive negative electrodes for ammonium-ion batteries. Introducing substantial single-electron active motifs linked by dynamic imine bonds can increase their capacity; however, this design is constrained by suboptimal single-electron redox efficiency and insufficient linkage stability. Here we unlock a multiple two-electron-transfer nitrobenzothiadiazole covalent organic framework via integrating alkynyl benzenes and nitro-functionalized four-electron benzothiadiazoles. The high degree of π-electron sp-conjugation along alkynyl linkages and strong electron-drawing effect of nitrobenzothiadiazole motifs in nitrobenzothiadiazole covalent organic framework promise high NH4+ accessibility of multi-two-electron nitro/thiazole sites (95.2% utilization) with a lower activation energy (25.93 vs. 35.99 kJ mol−1 of benzothiadiazole covalent organic framework).The fast octadeca-H-bonded NH4+ coordination in nitrobenzothiadiazole units liberates a high specific capacity of 317 mAh g−1 for nitrobenzothiadiazole covalent organic framework negative electrode. The alkynyl-bridged π-conjugation network establishes structural anti-dissolution to enable a cycling durability of 70,000 cycles. Paired with high-voltage Prussian blue analogue positive electrode, the ammonium-ion full battery delivers a specific energy of 86.1 Wh kg−1 (based on total active material mass) and a lifespan of 25,000 cycles. This work extends the design landscape of high-performance covalent organic frameworks for advanced ammonium-ion batteries.
Similar content being viewed by others
Data availability
All data that support the findings of this study are presented in the manuscript and Supplementary Information. Source data are provided with this paper.
References
Tian, Z. et al. High-capacity NH4+ charge storage in covalent organic frameworks. J. Am. Chem. Soc. 143, 19178–19186 (2021).
Peng, H. et al. Supramolecular engineering of cathode materials for aqueous zinc-ion energy storage devices: novel benzothiadiazole functionalized two-dimensional olefin-linked COFs. Angew. Chem. Int. Ed. 62, e202216136 (2023).
Liu, P. et al. Multi-H-bonded self-assembled superstructures for ultrahigh-capacity and ultralong-life all-organic ammonium-ion batteries. Energy Environ. Sci. 18, 5397–5406 (2025).
Tian, Z. et al. The role of hydrogen bonding in aqueous batteries: correlating molecular-scale interactions with battery performance. ACS Energy Lett. 9, 5179–5205 (2024).
Yao, N. et al. Identifying the lithium bond and lithium ionic bond in electrolytes. Chem 11, 102254 (2025).
Wu, H. et al. Alkaline-based aqueous sodium-ion batteries for large-scale energy storage. Nat. Commun. 15, 575 (2024).
Gao, H. et al. A pyrene-4,5,9,10-tetraone-based covalent organic framework delivers high specific capacity as a Li-ion positive electrode. J. Am. Chem. Soc. 144, 9434–9442 (2022).
Ye, L. et al. A rechargeable calcium–oxygen battery that operates at room temperature. Nature 626, 313–318 (2024).
Cui, H. et al. Regulating protons to tailor the enol conversion of quinone for high-performance aqueous zinc batteries. J. Am. Chem. Soc. 146, 15393–15402 (2024).
Karlsmo, M., Bouchal, R. & Johansson, P. High-performant all-organic aqueous sodium-ion batteries enabled by PTCDA electrodes and a hybrid Na/Mg electrolyte. Angew. Chem. Int. Ed. 60, 24709–24715 (2021).
Li, Z. et al. Electrolyte design enables rechargeable LiFePO4/graphite batteries from −80 °C to 80 °C. Angew. Chem. Int. Ed. 64, e202409409 (2025).
Li, H. et al. Developing high-power Li||S batteries via transition metal/carbon nanocomposite electrocatalyst engineering. Nat. Nanotechnol. 19, 792–799 (2024).
Lin, Y. et al. A covalent organic framework as a long-life and high-rate anode suitable for both aqueous acidic and alkaline batteries. Angew. Chem. Int. Ed. 62, e202218745 (2023).
Yu, X. et al. Activating organic electrode for zinc batteries via adjusting solvation structure of Zn ions. Angew. Chem. Int. Ed. 64, e202501359 (2025).
Rashad, M., Asif, M., Wang, Y., He, Z. & Ahmed, I. Recent advances in electrolytes and cathode materials for magnesium and hybrid-ion batteries. Energy Storage Mater. 25, 342–375 (2020).
Rodríguez-Pérez, I. A. et al. Mg-ion battery electrode: an organic solid’s herringbone structure squeezed upon Mg-ion insertion. J. Am. Chem. Soc. 139, 13031–13037 (2017).
Zhang, S.-J. et al. Coordination chemistry toward advanced Zn–I2 batteries with four-electron I–/I0/I+ conversion. J. Am. Chem. Soc. 147, 16350–16361 (2025).
Tian, Z. et al. A sustainable NH4+ ion battery by electrolyte engineering. Angew. Chem. Int. Ed. 61, e202213757 (2022).
Fei, H., Yang, F., Jusys, Z., Passerini, S. & Varzi, A. Ethylene glycol co-solvent enables stable aqueous ammonium-ion batteries with diluted electrolyte. Adv. Funct. Mater. 34, 2404560 (2024).
Zhang, S., Zhu, K., Gao, Y. & Cao, D. A long cycle stability and high rate performance organic anode for rechargeable aqueous ammonium-ion battery. ACS Energy Lett. 8, 889–897 (2023).
Bao, Z. et al. An acetate electrolyte for enhanced pseudocapacitve capacity in aqueous ammonium ion batteries. Nat. Commun. 15, 1934 (2024).
Liang, Y. et al. Interface storage mechanism in aqueous ammonium-ion supercapacitors with keggin-type polyoxometalates-modified Ag-BTC. Adv. Mater. 37, 2415545 (2025).
Li, M., Huang, X., Su, C. C. & Amine, K. Concerted formation of reversibly precipitated sulfur species and its importance for lean electrolyte lithium–sulfur batteries. J. Am. Chem. Soc. 146, 23182–23193 (2024).
Liang, G. et al. Initiating hexagonal MoO3 for superb-stable and fast NH4+ storage based on hydrogen bond chemistry. Adv. Mater. 32, 1907802 (2020).
Qi, X. et al. Phase engineering enables ultrahigh-capacity 1T/2H-MoS2 for advanced ammonium-ion storage. Energy Storage Mater. 75, 104063 (2025).
Ling, D. et al. Oxygen vacancy-enriched Bi2SeO5 nanosheets with dual mechanism for ammonium-ion batteries. ACS Nano 17, 25222–25233 (2023).
Zheng, S. et al. Orthoquinone–based covalent organic frameworks with ordered channel structures for ultrahigh performance aqueous zinc–organic batteries. Angew. Chem. Int. Ed. 61, e202117511 (2022).
Lin, L. et al. A semi-conductive organic cathode material enabled by extended conjugation for rechargeable aqueous zinc batteries. Energy Environ. Sci. 16, 89–96 (2023).
Huang, X. et al. Activating organic electrode via trace dissolved organic molecules. J. Am. Chem. Soc. 145, 25604–25613 (2023).
Hong, H. et al. Metal-free eutectic electrolyte with weak hydrogen bonds for high-rate and ultra-stable ammonium-ion batteries. Adv. Mater. 36, 2308210 (2024).
Yuan, Y. et al. High-capacity uranium extraction from seawater through constructing synergistic multiple dynamic bonds. Nat. Water 3, 89–98 (2025).
Wu, X. et al. Rocking-chair ammonium-ion battery: a highly reversible aqueous energy storage system. Angew. Chem. Int. Ed. 56, 13026–13030 (2017).
Wang, Q. et al. Unlocking the potential of ionic liquid-functionalized aqueous electrolytes for aqueous ammonium-bromine/ion batteries. Energy Storage Mater. 70, 103553 (2024).
Mittal, U., Colasuonno, F., Rawal, A., Lessio, M. & Kundu, D. A highly stable 1.3 V organic cathode for aqueous zinc batteries designed in-situ by solid-state electrooxidation. Energy Storage Mater. 46, 129–137 (2022).
Zou, G. et al. A symmetric aqueous magnesium ion supercapattery based on covalent organic frameworks. Adv. Energy Mater. 13, 2203193 (2023).
Han, J. et al. Concentrated electrolytes enabling stable aqueous ammonium-ion batteries. Adv. Mater. 34, 2201877 (2022).
Halder, A. et al. Interlayer hydrogen-bonded covalent organic frameworks as high-performance supercapacitors. J. Am. Chem. Soc. 140, 10941–10945 (2018).
Peng, C. et al. Reversible multi-electron redox chemistry of π-conjugated N-containing heteroaromatic molecule-based organic cathodes. Nat. Energy 2, 17074 (2017).
Liu, Y. et al. Redox-bipolar polyimide two-dimensional covalent organic framework cathodes for durable aluminium batteries. Angew. Chem. Int. Ed. 62, e202306091 (2023).
Emanuelsson, R., Sterby, M., Strømme, M. & Sjödin, M. An all-organic proton battery. J. Am. Chem. Soc. 139, 4828–4834 (2017).
Song, Z., Miao, L., Lv, Y., Gan, L. & Liu, M. NH4+ charge carrier coordinated H-bonded organic small molecule for fast and superstable rechargeable zinc batteries. Angew. Chem. Int. Ed. 62, e202309446 (2023).
Yu, Y.-X. Sodium/potassium intercalation on the Cu4S4 nanosheet accompanied by a surface phase transition and their competition with protons. ACS Appl. Energy Mater. 6, 10048–10060 (2023).
Tian, Z. et al. An ultrastable aqueous ammonium-ion battery using a covalent organic framework anode. Adv. Mater. 36, 2409354 (2024).
Chu, J. et al. Electronic band structure engineering of π-d conjugated metal-organic framework for sodium organic batteries. Nat. Commun. 16, 3549 (2025).
Acharjya, A., Longworth-Dunbar, L., Roeser, J., Pachfule, P. & Thomas, A. Synthesis of vinylene-linked covalent organic frameworks from acetonitrile: combining cyclotrimerization and aldol condensation in one pot. J. Am. Chem. Soc. 142, 14033–14038 (2020).
Zhang, B. et al. Crystalline dioxin-linked covalent organic frameworks from irreversible reactions. J. Am. Chem. Soc. 140, 12715–12719 (2018).
Yu, Y.-X. Theoretical insights into surface-pase transition and ion competition during alkali ion intercalation on the Cu4Se4 nanosheet. Phys. Chem. Chem. Phys. 26, 323–335 (2024).
Han, X. et al. Crystalline polyphenylene covalent organic frameworks. J. Am. Chem. Soc. 146, 89–94 (2024).
Zhou, Z. et al. Carbon dioxide capture from open air using covalent organic frameworks. Nature 635, 96–101 (2024).
Zheng, R. et al. Ammonium ion batteries: material, electrochemistry and strategy. Angew. Chem. Int. Ed. 62, e202301629 (2023).
Liu, J. et al. Superconjugated anthraquinone carbonyl-based covalent organic framework as anode material for high-performance aqueous ammonium-ion batteries. Angew. Chem. Int. Ed. 64, e202424494 (2025).
Yang, H., Tao, S., He, S. & Jiang, D. An ethynyl-linked sp-carbon-conjugated covalent organic framework through sonogashira cross-coupling reactions. J. Am. Chem. Soc. 147, 19667–19674 (2025).
Zhao, Q. et al. Improving active site local proton transfer in porous organic polymers for boosted oxygen electrocatalysis. Angew. Chem. Int. Ed. 63, e202414104 (2024).
Song, Z. et al. Anionic co-insertion charge storage in dinitrobenzene cathodes for high-performance aqueous zinc–organic batteries. Angew. Chem. Int. Ed. 61, e202208821 (2022).
Li, W. et al. Tuning electron delocalization of hydrogen-bonded organic framework cathode for high-performance zinc-organic batteries. Nat. Commun. 14, 5235 (2023).
Cao, Y. et al. Alkynyl boosted high-performance lithium storage and mechanism in covalent phenanthroline framework. Angew. Chem. Int. Ed. 62, e202302143 (2023).
Yang, H. et al. Tuning local charge distribution in multicomponent covalent organic frameworks for dramatically enhanced photocatalytic uranium extraction. Angew. Chem. Int. Ed. 62, e202303129 (2023).
Kurandina, D. et al. A porous crystalline nitrone-linked covalent organic framework. Angew. Chem. Int. Ed. 62, e202307674 (2023).
Kang, F. et al. Construction of crystalline nitrone-linked covalent organic frameworks via kröhnke oxidation. J. Am. Chem. Soc. 145, 15465–15472 (2023).
Lu, H. et al. Regulation of electron delocalization region in 2D heteroligand-based copper-organic framework to enhance NH4+ charge storage. Adv. Mater. 36, 2408396 (2024).
Du, L. et al. Coupling dual metal active sites and low-solvation architecture toward high-performance aqueous ammonium-ion batteries. Proc. Natl. Acad. Sci. USA 119, e2214545119 (2022).
Liu, J.-C. et al. P-n junction built-in electric field and electrochemical in-situ intercalation enabled ultra-stable and high-energy ammonium-ion storage. Nat. Energy 136, 110764 (2025).
Huang, Z.-H. et al. A high-capacity semiconductor organic polymer for stable aqueous ammonium-ion storage. Adv. Mater. 37, 2508001 (2025).
Gao, M. et al. 2D conjugated metal–organic frameworks embedded with iodine for high-performance ammonium-ion hybrid supercapacitors. Adv. Mater. 35, 2305575 (2023).
Wang, S. et al. Non-metal ion co-insertion chemistry in aqueous Zn/MnO2 batteries. Angew. Chem. Int. Ed. 60, 7056–7060 (2021).
Chen, Y., Song, Z., Lv, Y., Gan, L. & Liu, M. NH4+-modulated cathodic interfacial spatial charge redistribution for high-performance dual-ion capacitors. Nano-Micro Lett. 17, 117 (2025).
Wang, D. et al. Enhancing organic cathodes of aqueous zinc-ion batteries via nitro group modification. Chem. Sci. 16, 3630–3637 (2025).
Song, Z., Huang, Q., Lv, Y., Gan, L. & Liu, M. Multi-N-heterocycle donor-acceptor conjugated amphoteric organic superstructures for superior zinc batteries. Angew. Chem. Int. Ed. 64, e202418237 (2025).
Lam, V. N. et al. A decade of insights: delving into calendar aging trends and implications. Joule 9, 101796 (2025).
Yu, J. et al. Donor–acceptor porous aromatic framework cathode with fast redox kinetics for ultralow-temperature (−70 °c) potassium-organic batteries. Angew. Chem. Int. Ed. 64, e202507570 (2025).
Wang, F. et al. Constructing symmetric all-organic proton battery via scalable flux synthesis of vinylene-linked covalent organic frameworks. J. Am. Chem. Soc. 147, 36547–36556 (2025).
Cao, N. et al. The role of aromaticity in the cyclization and polymerization of alkyne-substituted porphyrins on Au(111). Nat. Chem. 15, 1765–1772 (2023).
Dai, J. et al. MoS2@polyaniline for aqueous ammonium-ion supercapacitors. Adv. Mater. 35, 2303732 (2023).
Ye, F. et al. Reversible ammonium ion intercalation/de-intercalation with crystal water promotion effect in layered VOPO4⋅2H2O. Angew. Chem. Int. Ed. 62, e202303480 (2023).
Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (No. 22272118, M.L.; NO. 22172111, L.G.; and NO. 22309134, Z.S.), the Shanghai Rising-Star Program (23YF1449200, Z.S.), the Zhejiang Provincial Science and Technology Project (NO. 2022C01182, Y.L.), and the Fundamental Research Funds for the Central Universities (Z.S.).
Author information
Authors and Affiliations
Contributions
Y.C. and Z.S. conceived the idea and designed the project. L.G. and M.L. supervised the experiments and edited the paper. Y.C., Y.Q., C. H., L.M., and Y. L. performed the data processing and analysis. Y.C. and D. Z. contributed to the theoretical simulations. Y.C., Z.S., L.G. and M.L. contributed to the manuscript review. All authors discussed the results and contributed to the completion of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Dinesh Shetty, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Y., Zhang, D., Qin, Y. et al. Multi-electron nitrobenzothiadiazole sp-conjugated-alkynyl covalent organic frameworks for ammonium-ion batteries. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70370-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70370-x