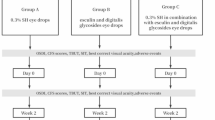
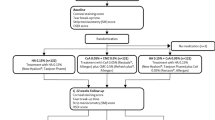
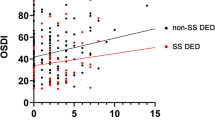
Abstract
The management of ocular surface inflammation, particularly in conditions like dry eye disease (DED), remains challenging due to multiple pathogenic causes and ocular barriers. Traditional eye drops usually provide transient and partial symptom relief, necessitating combined therapies to break the vicious cycle of oxidative stress and inflammation. To surmount these limitations, a polyphenol-based eye drop is developed via the self-polymerization of rosmarinic acid in the presence of hyaluronic acid, followed by co-assembly with cerium ions to afford monodispersed RHC NPs. To enhance the ocular retention efficiency, thiol groups are further decorated on NPs (termed as s-RHC NPs). In two mouse models of DED, topical administration of s-RHC NPs leads to comprehensive relief of symptoms, including inflammatory response suppression, corneal epithelial defect repair, and tear secretion recovery. We envision that this work might facilitate the treatment of DED and inspire the design of polyphenol-based nanoparticles via a self-polymerization strategy.
Similar content being viewed by others
Data availability
The RNA sequencing data generated in this study are deposited in the NCBI’s Sequence Read Archive (SRA) under accession code PRJNA1369795. The proteomics data generated in this study are deposited in the NCBI’s Sequence Read Archive (SRA) under accession code IPX0014408002 [https://www.iprox.cn/page/project.html?id=IPX0014408000]. Source data are available for Figs. 2d–i, 2k, 2m–p, 3b, d, 3f–h, 3o, 4c, 4f-j, 5c–f, 5i–l, 5n, 6c–f, 6i–l, 6n and Supplementary Figs. 1b,d, 2a, d, f, g, h, 4a, 5b, 7a–c, 8–10, 12b,c, 21a–c, 22a–c, 23a, b, 25a, b and 26 in the associated source data file. The authors declare that the remaining data are available within the Manuscript, Supplementary Information or Source Data file. Source data are provided in this paper.
References
Hakim, F. E. & Farooq, A. V. Dry eye disease: an update in 2022. J. Am. Med. Assoc. 327, 478–479 (2022).
Periman, L. M., Perez, V. L., Saban, D. R., Lin, M. C. & Neri, P. The immunological basis of dry eye disease and current topical treatment options. J. Ocul. Pharmacol. Ther. 36, 137–146 (2020).
Clayton, J. A. Dry eye. N. Engl. J. Med. 378, 2212–2223 (2018).
Han, H. et al. Polymer- and lipid-based nanocarriers for ocular drug delivery: Current status and future perspectives. Adv. Drug Deliv. Rev. 196, 114770 (2023).
Seah, I., Loh, X. J. & Su, X. A topical gel for extended ocular drug release. Nat. Biomed. Eng. 4, 1024–1025 (2020).
Bennett, N. H., Chinnery, H. R., Downie, L. E., Hill, L. J. & Grover, L. M. Material, immunological, and practical perspectives on eye drop formulation. Adv. Funct. Mater. 30, 1908476 (2020).
Cui, W. et al. Nanoceria-mediated cyclosporin A delivery for dry eye disease management through modulating immune-epithelial crosstalk. ACS Nano 18, 11084–11102 (2024).
Su, Y., Fan, X. & Pang, Y. Nano-based ocular drug delivery systems: an insight into the preclinical/clinical studies and their potential in the treatment of posterior ocular diseases. Biomater. Sci. 11, 4490–4507 (2023).
Shen, J. et al. Emerging co-assembled and sustained released natural medicinal nanoparticles for multitarget therapy of choroidal neovascularization. Adv. Mater. 36, 2314095 (2024).
Leal, J., Smyth, H. D. C. & Ghosh, D. Physicochemical properties of mucus and their impact on transmucosal drug delivery. Int. J. Pharm. 532, 555–572 (2017).
Carlson, T. L., Lock, J. Y. & Carrier, R. L. Engineering the mucus barrier. Annu. Rev. Biomed. Eng. 20, 197–220 (2018).
Lim, B., Kim, K. S., Ahn, J. Y. & Na, K. Overcoming antibiotic resistance caused by genetic mutations of Helicobacter pylori with mucin adhesive polymer-based therapeutics. Biomaterials 308, 122541 (2024).
TM, M. W., Lau, W. A.-O. & Khutoryanskiy, V. A.-O. Chitosan and its derivatives for application in mucoadhesive drug delivery systems. Polymers 10, 267 (2018).
Wang, Y. et al. Liposome-based permeable eyedrops for effective posterior segment drug delivery. Adv. Funct. Mater. 34, 2403142 (2024).
Shao, M. et al. Eye-drop nano-formulation of catalase self-assembled with thiolated chitosan for effective treatment of dry eye disease. Adv. Mater. 37, 2415353 (2025).
Yang, C.-J., Anand, A., Huang, C.-C. & Lai, J.-Y. Unveiling the power of gabapentin-loaded nanoceria with multiple therapeutic capabilities for the treatment of dry eye disease. ACS Nano 17, 25118–25135 (2023).
Kim, H., Rencoret, J., Elder, T. J., del Río, J. C. & Ralph, J. Biomimetic oxidative copolymerization of hydroxystilbenes and monolignols. Sci. Adv. 9, eade5519 (2023).
Zhou, T. et al. Rosmarinic acid-grafted gelatin nanogels for efficient diquafosol delivery in dry eye disease therapy. J. Control. Release 373, 306–318 (2024).
Guo, Y., Sun, Q., Wu, F.-G., Dai, Y. & Chen, X. Polyphenol-containing nanoparticles: synthesis, properties, and therapeutic delivery. Adv. Mater. 33, 2007356 (2021).
Lu, B. et al. Rosmarinic acid nanomedicine for rheumatoid arthritis therapy: targeted RONS scavenging and macrophage repolarization. J. Control. Release 362, 631–646 (2023).
Yang, L. et al. Biofilm microenvironment triggered self-enhancing photodynamic immunomodulatory microneedle for diabetic wound therapy. Nat. Commun. 14, 7658 (2023).
Zhao, C. et al. Synthetic lignin-derived therapeutic nano reagent as intestinal pH-sensitive drug carriers capable of bypassing the gastric acid environment for colitis treatment. ACS Nano 17, 811–824 (2023).
He, J., Ye, Y., Zhang, D., Yao, K. & Zhou, M. Visualized gallium/lyticase-integrated antifungal strategy for fungal keratitis treatment. Adv. Mater. 34, 2206437 (2022).
Deng, Z. et al. Metal polyphenol network/cerium oxide artificial enzymes therapeutic nanoplatform for MRI/CT-aided intestinal inflammation management. Nano Today 53, 102044 (2023).
Soh, M. et al. Ceria–zirconia nanoparticles as an enhanced multi-antioxidant for sepsis treatment. Angew. Chem. Int. Ed. 56, 11399–11403 (2017).
Zhou, F. et al. Redox homeostasis strategy for inflammatory macrophage reprogramming in rheumatoid arthritis based on ceria oxide nanozyme-complexed biopolymeric micelles. ACS Nano 17, 4358–4372 (2023).
Kim, C. K. et al. Ceria nanoparticles that can protect against ischemic stroke. Angew. Chem. Int. Ed. 51, 11039–11043 (2012).
Zhang, Y. et al. Nanozymes for nanohealthcare. Nat. Rev. Methods Prim. 4, 36 (2024).
Luo, R. et al. Oral microsphere formulation of M2 macrophage-mimetic Janus nanomotor for targeted therapy of ulcerative colitis. Sci. Adv. 10, eado6798 (2024).
Saifi, M. A., Seal, S. & Godugu, C. Nanoceria, the versatile nanoparticles: promising biomedical applications. J. Control. Release 338, 164–189 (2021).
Dogru, M., Kojima, T., Simsek, C. & Tsubota, K. Potential role of oxidative stress in ocular surface inflammation and dry eye disease. Invest. Ophthalmol. Vis. Sci. 59, DES163–DES168 (2018).
Sies, H. Oxidative stress: a concept in redox biology and medicine. Redox Biol. 4, 180–183 (2015).
Zhou, C. et al. JUN is a key transcriptional regulator of the unfolded protein response in acute myeloid leukemia. Leukemia 31, 1196–1205 (2017).
Palomer, X. et al. GADD45A: with or without you. Med. Res. Rev. 44, 1375–1403 (2024).
Liu, C. et al. Protein phosphatase 1 regulatory subunit 15 A promotes translation initiation and induces G2M phase arrest during cuproptosis in cancers. Cell Death Dis. 15, 149 (2024).
Hu, C. et al. Heat shock proteins: biological functions, pathological roles, and therapeutic opportunities. MedComm 3, e161 (2022).
Dubrez, L., Causse, S., Borges Bonan, N., Dumétier, B. & Garrido, C. Heat-shock proteins: chaperoning DNA repair. Oncogene 39, 516–529 (2020).
Huangfu, L., Li, R., Huang, Y. & Wang, S. The IL-17 family in diseases: from bench to bedside. Signal Transduct. Target. Ther. 8, 402 (2023).
Prabhu, K. S. et al. H2AX: a key player in DNA damage response and a promising target for cancer therapy. Biomed. Pharmacother. 175, 116663 (2024).
You, I.-C. et al. Macrophage phenotype in the ocular surface of experimental murine dry eye disease. Arch. Immunol. Ther. Exp. 63, 299–304 (2015).
Li, S. et al. Anti-oxidative and anti-inflammatory micelles: break the dry eye vicious cycle. Adv. Sci. 9, 2200435 (2022).
Andrabi, S. M. et al. Nitric Oxide: physiological functions, delivery, and biomedical applications. Adv. Sci. 10, 2303259 (2023).
Xiong, C. et al. A rabbit dry eye model induced by topical medication of a preservative benzalkonium chloride. Invest. Ophthalmol. Vis. Sci. 49, 1850–1856 (2008).
Agarwal, P. & Rupenthal, I. D. Modern approaches to the ocular delivery of cyclosporine A. Drug Discov. Today 21, 977–988 (2016).
Zhang, R. et al. Dose-dependent benzalkonium chloride toxicity imparts ocular surface epithelial changes with features of dry eye disease. Ocul. Surf. 18, 158–169 (2020).
Zhou, T. et al. miR-204-containing exosomes ameliorate GVHD-associated dry eye disease. Sci. Adv. 8, eabj9617 (2022).
Nagai, N. & Otake, H. Novel drug delivery systems for the management of dry eye. Adv. Drug Deliv. Rev. 191, 114582 (2022).
Yang, F. M. et al. The artemisinin analog SM934 alleviates dry eye disease in rodent models by regulating TLR4/NF-κB/NLRP3 signaling. Acta Pharmacol. Sin. 42, 593–603 (2021).
Chaudhari, P. et al. Rodent models for dry eye syndrome: standardization using benzalkonium chloride and scopolamine hydrobromide. Life Sci. 317, 121463 (2023).
Singh, S., Sharma, S. & Basu, S. Rabbit models of dry eye disease: current understanding and unmet needs for translational research. Exp. Eye Res. 206, 108538 (2021).
Hu, S. et al. A mussel-inspired film for adhesion to wet buccal tissue and efficient buccal drug delivery. Nat. Commun. 12, 1689 (2021).
Poinard, B., Kamaluddin, S., Tan, A. Q. Q., Neoh, K. G. & Kah, J. C. Y. Polydopamine coating enhances mucopenetration and cell uptake of nanoparticles. ACS Appl. Mater. Interfaces 11, 4777–4789 (2019).
Acknowledgements
Financial support from the Zhejiang Province Key Research and Development Program (2024C03073 received by H.H.), National Natural Science Foundation of China (22375128 received by Y.W., 82572387 received by H.H., 82271064 received by H.H., 22105126 received by Y.W.), and the Zhejiang Province Natural Science Foundation (LR23H120001 received by H.H.) is acknowledged.
Author information
Authors and Affiliations
Contributions
Z.W., Z.L., H.H., and Y.W. conceived the project and designed the experiments. Z.W. and R.X. synthesized materials. Z.W. and Y.G. performed in vitro cell experiments. Z.L. performed in vivo experiments. Z.W., Z.L., R.X., F.W., H.H., and Y.W. contributed to data collection and analysis. Z.W., Z.L., H.H., and Y.W. cowrote the manuscript. All authors discussed the results and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Chengchao Chu and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Z., Lv, Z., Ge, Y. et al. Self-polymerized polyphenol-based platform for the management of dry eye pathogenesis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70388-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70388-1