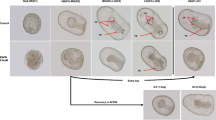
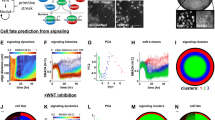
Abstract
The molecular control and ancestral state of dorsal–ventral patterning within spiralians remain unclear due to the remarkable diversity across species. Here we present a chromosome-level genome for the brachiopod Lingula anatina and apply functional transcriptomics to study dorsal–ventral patterning under BMP signalling control. We uncover asymmetrical activation of BMP signalling at the dorsal side of the gastrula, governed by ventral chordin expression and a ‘seesaw’ of BMP ligands. Using small-molecule drugs and recombinant proteins, we show that high BMP activity inhibits genes typically associated with neural patterning during gastrula and larval stages, similar to deuterostomes and non-spiralian protostomes. Our findings suggest deep conservation of this mechanism across all three major bilaterian clades, supported by striking similarities in BMP-regulated gene sets between brachiopods and Xenopus. We argue that the spiralian ancestor retained the ancestral bilaterian mechanism, although downstream network components have undergone developmental system drift.
Similar content being viewed by others
Data availability
Genome sequencing and RNA sequencing datasets are accessible through NCBI BioProject under accession number PRJNA1068743. The L. anatina genome is made available on NCBI with the accession GCA_051362555.1 [https://www.ncbi.nlm.nih.gov/datasets/genome/GCA_051362555.1/]. A L. anatina genome browser with a BLAST function is available at https://marinegenomics.oist.jp/lan_3_0/viewer/info?project_id=124. The genome sequence, gene models, and functional annotations from UniProt, Pfam, InterProScan, EggNOG and KOfam are also available at this location. Genomic, transcriptomic and imaging data from this study, including the genome assembly, gene annotations, repeat annotations, gene expression matrices and original Zeiss LSM imaging files, are available on Zenodo (https://doi.org/10.5281/zenodo.16916709). Source data are provided with this paper.
Code availability
Code for analyses in this study is available on GitHub (https://github.com/symgenoevolab/lingula_genome) and Zenodo (https://doi.org/10.5281/zenodo.18591213).
References
De Robertis, E. M. & Sasai, Y. A common plan for dorsoventral patterning in Bilateria. Nature 380, 37–40 (1996).
Ferguson, E. L. Conservation of dorsal-ventral patterning in arthropods and chordates. Curr. Opin. Genet. Dev. 6, 424–431 (1996).
De Robertis, E. M. Evo-devo: variations on ancestral themes. Cell 132, 185–195 (2008).
Sasai, Y., Lu, B., Steinbeisser, H. & De Robertis, E. M. Regulation of neural induction by the Chd and Bmp-4 antagonistic patterning signals in Xenopus. Nature 377, 757 (1995).
De Robertis, E. M. & Kuroda, H. Dorsal-ventral patterning and neural induction in Xenopus embryos. Annu. Rev. Cell Dev. Biol. 20, 285–308 (2004).
Sasai, Y. et al. Xenopus chordin: a novel dorsalizing factor activated by organizer-specific homeobox genes. Cell 79, 779–790 (1994).
Piccolo, S., Sasai, Y., Lu, B. & De Robertis, E. M. Dorsoventral patterning in Xenopus: inhibition of ventral signals by direct binding of chordin to BMP-4. Cell 86, 589–598 (1996).
Smith, W. C. & Harland, R. M. Expression cloning of noggin, a new dorsalizing factor localized to the Spemann organizer in Xenopus embryos. Cell 70, 829–840 (1992).
Zimmerman, L. B., Jesús-Escobar, J. M. & Harland, R. M. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell 86, 599–606 (1996).
Ray, R. P., Arora, K., Nüsslein-Volhard, C. & Gelbart, W. M. The control of cell fate along the dorsal–ventral axis of the Drosophila embryo. Development 113, 35–54 (1991).
Ferguson, E. L. & Anderson, K. V. Decapentaplegic acts as a morphogen to organize dorsal-ventral pattern in the Drosophila embryo. Cell 71, 451–461 (1992).
Srinivasan, S., Rashka, K. E. & Bier, E. Creation of a Sog morphogen gradient in the Drosophila embryo. Dev. Cell 2, 91–101 (2002).
Lynch, J. A. & Roth, S. The evolution of dorsal–ventral patterning mechanisms in insects. Genes Dev. 25, 107–118 (2011).
Lowe, C. J. et al. Dorsoventral patterning in hemichordates: insights into early chordate evolution. PLoS Biol. 4, e291 (2006).
Yu, J. K. et al. Axial patterning in cephalochordates and the evolution of the organizer. Nature 445, 613–617 (2007).
Lapraz, F., Besnardeau, L. & Lepage, T. Patterning of the dorsal–ventral axis in echinoderms: insights into the evolution of the BMP–chordin signaling network. PLoS Biol. 7, e1000248 (2009).
Arendt, D. & Nübler-Jung, K. Inversion of dorsoventral axis? Nature 371, 26 (1994).
Shimmi, O., Umulis, D., Othmer, H. & O’Connor, M. B. Facilitated transport of a Dpp/Scw heterodimer by Sog/Tsg leads to robust patterning of the Drosophila blastoderm embryo. Cell 120, 873–886 (2005).
Little, S. C. & Mullins, M. C. Bone morphogenetic protein heterodimers assemble heteromeric type I receptor complexes to pattern the dorsoventral axis. Nat. Cell Biol. 11, 637–643 (2009).
Bauer, M., Aguilar, G., Wharton, K. A., Matsuda, S. & Affolter, M. Heterodimerization-dependent secretion of bone morphogenetic proteins in Drosophila. Dev. Cell 58, 645–659 (2023).
Aono, A. et al. Potent ectopic bone-inducing activity of bone morphogenetic protein-4/7 heterodimer. Biochem. Biophys. Res. Commun. 210, 670–677 (1995).
Nishimatsu, S. & Thomsen, G. H. Ventral mesoderm induction and patterning by bone morphogenetic protein heterodimers in Xenopus embryos. Mech. Dev. 74, 75–88 (1998).
Massagué, J., Attisano, L. & Wrana, J. L. The TGF-beta family and its composite receptors. Trends Cell Biol. 4, 172–178 (1994).
Hoodless, P. A. MADR1, a MAD-related protein that functions in BMP2 signaling pathways. Cell 85, 489–500 (1996).
Nguyen, M., Park, S., Marqués, G. & Arora, K. Interpretation of a BMP activity gradient in Drosophila embryos depends on synergistic signaling by two type I receptors, SAX and TKV. Cell 95, 495–506 (1998).
Wrana, J. L., Attisano, L., Wieser, R., Ventura, F. & Massagué, J. Mechanism of activation of the TGF-beta receptor. Nature 370, 341–347 (1994).
Liu, F. et al. A human mad protein acting as a BMP-regulated transcriptional activator. Nature 381, 620–623 (1996).
Kretzschmar, M., Liu, F., Hata, A., Doody, J. & Massagué, J. The TGF-beta family mediator Smad1 is phosphorylated directly and activated functionally by the BMP receptor kinase. Genes Dev. 11, 984–995 (1997).
Genikhovich, G. et al. Axis patterning by BMPs: cnidarian network reveals evolutionary constraints. Cell Rep. 10, 1646–1654 (2015).
Eldar, A. et al. Robustness of the BMP morphogen gradient in Drosophila embryonic patterning. Nature 419, 304–308 (2002).
Ben-Zvi, D., Shilo, B.-Z., Fainsod, A. & Barkai, N. Scaling of the BMP activation gradient in Xenopus embryos. Nature 453, 1205–1211 (2008).
Mizutani, C. M. et al. Formation of the BMP activity gradient in the Drosophila embryo. Dev. Cell 8, 915–924 (2005).
Johnston, R. D. & Gelbart, W. M. Decapentaplegic transcripts are localized along the dorsal–ventral axis of the Drosophila embryo. EMBO J. 6, 2785–2791 (1987).
Arora, K., Levine, M. S. & O’Connor, M. B. The screw gene encodes a ubiquitously expressed member of the TGF-beta family required for specification of dorsal cell fates in the Drosophila embryo. Genes Dev. 8, 2588–2601 (1994).
Reversade, B. & De Robertis, E. M. Regulation of ADMP and BMP2/4/7 at opposite embryonic poles generates a self-regulating morphogenetic field. Cell 123, 1147–1160 (2005).
Inomata, H., Haraguchi, T. & Sasai, Y. Robust stability of the embryonic axial pattern requires a secreted scaffold for chordin degradation. Cell 134, 854–865 (2008).
Plouhinec, J.-L., Zakin, L. & De Robertis, E. M. Systems control of BMP morphogen flow in vertebrate embryos. Curr. Opin. Genet. Dev. 21, 696–703 (2011).
Lapraz, F., Haillot, E. & Lepage, T. A deuterostome origin of the Spemann organiser suggested by nodal and ADMPs functions in echinoderms. Nat. Commun. 6, 8927 (2015).
Finnerty, J. R., Pang, K., Burton, P., Paulson, D. & Martindale, M. Q. Origins of bilateral symmetry: Hox and dpp expression in a sea anemone. Science 304, 1335–1337 (2004).
Matus, D. Q. et al. Molecular evidence for deep evolutionary roots of bilaterality in animal development. Proc. Natl. Acad. Sci. USA 103, 11195–11200 (2006).
Saina, M., Genikhovich, G., Renfer, E. & Technau, U. BMPs and chordin regulate patterning of the directive axis in a sea anemone. Proc. Natl. Acad. Sci. USA 106, 18592–18597 (2009).
Mörsdorf, D., Knabl, P. & Genikhovich, G. Highly conserved and extremely evolvable: BMP signalling in secondary axis patterning of cnidaria and Bilateria. Dev. Genes Evol. 234, 1–19 (2024).
Genikhovich, G. & Technau, U. On the evolution of bilaterality. Development 144, 3392–3404 (2017).
Lowe, C. J., Clarke, D. N., Medeiros, D. M., Rokhsar, D. S. & Gerhart, J. The deuterostome context of chordate origins. Nature 520, 456–465 (2015).
Su, Y. H. et al. BMP controls dorsoventral and neural patterning in indirect-developing hemichordates providing insight into a possible origin of chordates. Proc. Natl. Acad. Sci. USA 116, 12925–12932 (2019).
Bier, E. & De Robertis, E. M. BMP gradients: a paradigm for morphogen-mediated developmental patterning. Science 348, aaa5838 (2015).
Lyons, D. C., Perry, K. J., Batzel, G. & Henry, J. Q. BMP signaling plays a role in anterior-neural/head development, but not organizer activity, in the gastropod Crepidula fornicata. Dev. Biol. 463, 135–157 (2020).
Webster, N. B., Corbet, M., Sur, A. & Meyer, N. P. Role of BMP signaling during early development of the annelid Capitella teleta. Dev. Biol. 478, 183–204 (2021).
Kuo, D.-H., Shankland, M. & Weisblat, D. A. Regional differences in BMP-dependence of dorsoventral patterning in the leech Helobdella. Dev. Biol. 368, 86–94 (2012).
Lanza, A. R. & Seaver, E. C. Activin/nodal signaling mediates dorsal-ventral axis formation before third quartet formation in embryos of the annelid Chaetopterus pergamentaceus. EvoDevo 11, 17 (2020).
Webster, N. B. & Meyer, N. P. Capitella teleta gets left out: possible evolutionary shift causes loss of left tissues rather than increased neural tissue from dominant-negative BMPR1. Neural Dev. 19, 4 (2024).
Lanza, A. R. & Seaver, E. C. Functional evidence that activin/nodal signaling is required for establishing the dorsal–ventral axis in the annelid Capitella teleta. Development 147, dev189373 (2020).
Martín-Zamora, F. M. et al. Annelid functional genomics reveal the origins of bilaterian life cycles. Nature 615, 105–110 (2023).
Kenny, N. J. et al. The Lophotrochozoan TGF-β signalling cassette-diversification and conservation in a key signalling pathway. Int. J. Dev. Biol. 58, 533–549 (2014).
Lambert, J. D., Johnson, A. B., Hudson, C. N. & Chan, A. Dpp/BMP2-4 mediates signaling from the D-quadrant organizer in a spiralian embryo. Curr. Biol. 26, 2003–2010 (2016).
Tan, S., Huan, P. & Liu, B. Molluskan dorsal–ventral patterning relying on BMP2/4 and chordin provides insights into spiralian development and evolution. Mol. Biol. Evol. 39, msab322 (2022).
Carrillo-Baltodano A. M. et al. Developmental system drift in dorsoventral patterning is linked to transitions to autonomous development in Annelida. Preprint at bioRxiv https://doi.org/10.1101/2025.05.29.656861 (2025).
Martín-Durán, J. M., Passamaneck, Y. J., Martindale, M. Q. & Hejnol, A. The developmental basis for the recurrent evolution of deuterostomy and protostomy. Nat. Ecol. Evol. 1, 5 (2016).
Luo, Y.-J. et al. The Lingula genome provides insights into brachiopod evolution and the origin of phosphate biomineralization. Nat. Commun. 6, 8301 (2015).
Simakov, O. et al. Insights into bilaterian evolution from three spiralian genomes. Nature 493, 526–531 (2013).
Kenny, N. J. et al. The gene-rich genome of the scallop Pecten maximus. Gigascience 9, giaa037 (2020).
Kwiatkowski, D. et al. The genome sequence of the bootlace worm, Lineus longissimus (Gunnerus, 1770). Wellcome Open Res. 6, 272 (2021).
Nishizawa, A., Sarashina, I., Tsujimoto, Y., IIjima, M. & Endo, K. Artificial fertilization, early development and chromosome numbers in the brachiopod Lingula anatina. Spec. Pap. Palaeontol. 84, 309–316 (2010).
Nesnidal, M. P., Helmkampf, M., Bruchhaus, I. & Hausdorf, B. Compositional heterogeneity and phylogenomic inference of metazoan relationships. Mol. Biol. Evol. 27, 2095–2104 (2010).
Nesnidal, M. P. et al. New phylogenomic data support the monophyly of Lophophorata and an ectoproct–phoronid clade and indicate that polyzoa and kryptrochozoa are caused by systematic bias. BMC Evol. Biol. 13, 253 (2013).
Laumer, C. E. et al. Spiralian phylogeny informs the evolution of microscopic lineages. Curr. Biol. 25, 2000–2006 (2015).
Luo, Y.-J. et al. Nemertean and phoronid genomes reveal lophotrochozoan evolution and the origin of bilaterian heads. Nat. Ecol. Evol. 2, 141–151 (2018).
Santagata, S. Genes with evidence of positive selection as potentially related to coloniality and the evolution of morphological features among the lophophorates and entoprocts. J. Exp. Zool. B Mol. Dev. Evol. 336, 267–280 (2021).
Marlétaz, F., Peijnenburg, K. T. C. A., Goto, T., Satoh, N. & Rokhsar, D. S. A new spiralian phylogeny places the enigmatic arrow worms among gnathiferans. Curr. Biol. 29, 312–318.e3 (2019).
Zverkov, O. A. et al. Dicyemida and orthonectida: two stories of body plan simplification. Front. Genet. 10, 443 (2019).
Lewin, T. D. et al. Phoronid genome supports a monophyletic Lophophorata. Curr. Biol. 35, 5633–5645 (2025).
Khalturin, K. et al. Polyzoa is back: the effect of complete gene sets on the placement of Ectoprocta and Entoprocta. Sci. Adv. 8, eabo4400 (2022).
Laumer, C. E. et al. Revisiting metazoan phylogeny with genomic sampling of all phyla. Proc. Biol. Sci. 286, 20190831 (2019).
Affolter, M., Marty, T., Vigano, M. A. & Jaźwińska, A. Nuclear interpretation of Dpp signaling in Drosophila. EMBO J. 20, 3298–3305 (2001).
Lapraz, F. et al. RTK and TGF-beta signaling pathways genes in the sea urchin genome. Dev. Biol. 300, 132–152 (2006).
Holtzhausen, A. et al. Novel bone morphogenetic protein signaling through Smad2 and Smad3 to regulate cancer progression and development. FASEB J. 28, 1248–1267 (2014).
Gaviño, M. A. & Reddien, P. W. A Bmp/Admp regulatory circuit controls maintenance and regeneration of dorsal–ventral polarity in planarians. Curr. Biol. 21, 294–299 (2011).
Ko, B. J. et al. Widespread false gene gains caused by duplication errors in genome assemblies. Genome Biol. 23, 205 (2022).
Rhie, A. et al. Towards complete and error-free genome assemblies of all vertebrate species. Nature 592, 737–746 (2021).
Simakov, O. et al. Deeply conserved synteny resolves early events in vertebrate evolution. Nat. Ecol. Evol. 4, 820–830 (2020).
Simakov, O. et al. Deeply conserved synteny and the evolution of metazoan chromosomes. Sci. Adv. 8, eabi5884 (2022).
Lewin, T. D., Liao, I. J.-Y. & Luo, Y.-J. Annelid comparative genomics and the evolution of massive lineage-specific genome rearrangement in bilaterians. Mol. Biol. Evol. 41, msae172 (2024).
Lewin, T. D., Liao, I. J.-Y. & Luo, Y.-J. Conservation of bilaterian genome structure is the exception, not the rule. Genome Biol. 26, 247 (2025).
Bishop, J. et al. The genome sequence of the sea mat, Membranipora membranacea (Linnaeus, 1767). Wellcome Open Res. 8, 38 (2023).
Lewin, T. D. et al. Fusion, fission, and scrambling of the bilaterian genome in bryozoa. Genome Res. 35, 78–92 (2025).
Cuny, G. D. et al. Structure-activity relationship study of bone morphogenetic protein (BMP) signaling inhibitors. Bioorg. Med. Chem. Lett. 18, 4388–4392 (2008).
Sanvitale, C. E. et al. A new class of small molecule inhibitor of BMP signaling. PLoS ONE 8, e62721 (2013).
Luo, Y.-J. & Su, Y.-H. Opposing nodal and BMP signals regulate left–right asymmetry in the sea urchin larva. PLoS Biol. 10, 1001402 (2012).
Panchision, D. M. et al. Sequential actions of BMP receptors control neural precursor cell production and fate. Genes Dev. 15, 2094–2110 (2001).
Lawson, K. A. et al. Bmp4 is required for the generation of primordial germ cells in the mouse embryo. Genes Dev. 13, 424–436 (1999).
Hogan, B. L. Bone morphogenetic proteins: multifunctional regulators of vertebrate development. Genes Dev. 10, 1580–1594 (1996).
Yatsu, N. On the development of Lingula anatina. J. Coll. Sci. Imp. Univ. Tokyo 17, 1 (1902).
Pascale, A., Amadio, M. & Quattrone, A. Defining a neuron: neuronal ELAV proteins. Cell. Mol. Life Sci. 65, 128–140 (2008).
Robinow, S. & White, K. The locus elav of Drosophila melanogaster is expressed in neurons at all developmental stages. Dev. Biol. 126, 294–303 (1988).
Acampora, D. et al. Forebrain and midbrain regions are deleted in Otx2-/- mutants due to a defective anterior neuroectoderm specification during gastrulation. Development 121, 3279–3290 (1995).
Pevny, L. & Placzek, M. SOX genes and neural progenitor identity. Curr. Opin. Neurobiol. 15, 7–13 (2005).
Bradford, D., Cole, S. J. & Cooper, H. M. Netrin-1: diversity in development. Int. J. Biochem. Cell Biol. 41, 487–493 (2009).
Zhang, X. et al. Pax6 is a human neuroectoderm cell fate determinant. Cell Stem Cell 7, 90–100 (2010).
Takebayashi-Suzuki, K., Kitayama, A., Terasaka-Iioka, C., Ueno, N. & Suzuki, A. The forkhead transcription factor FoxB1 regulates the dorsal–ventral and anterior–posterior patterning of the ectoderm during early Xenopus embryogenesis. Dev. Biol. 360, 11–29 (2011).
Yao, S. et al. Anaplastic lymphoma kinase is required for neurogenesis in the developing central nervous system of zebrafish. PLoS ONE 8, e63757 (2013).
Range, R. Specification and positioning of the anterior neuroectoderm in deuterostome embryos. Genesis 52, 222–234 (2014).
Biehs, B., François, V. & Bier, E. The Drosophila short gastrulation gene prevents Dpp from autoactivating and suppressing neurogenesis in the neuroectoderm. Genes Dev. 10, 2922–2934 (1996).
Kelman, Z. PCNA: structure, functions and interactions. Oncogene 14, 629–640 (1997).
Heichman, K. A. & Roberts, J. M. CDC16 controls initiation at chromosome replication origins. Mol. Cell 1, 457–463 (1998).
Paunesku, T. et al. Proliferating cell nuclear antigen (PCNA): ringmaster of the genome. Int. J. Radiat. Biol. 77, 1007–1021 (2001).
Cimprich, K. A. & Cortez, D. ATR: an essential regulator of genome integrity. Nat. Rev. Mol. Cell Biol. 9, 616–627 (2008).
Murakami, T. et al. Stable interaction between the human proliferating cell nuclear antigen loader complex Ctf18-replication factor C (RFC) and DNA polymerase epsilon is mediated by the cohesion-specific subunits, Ctf18, Dcc1, and Ctf8. J. Biol. Chem. 285, 34608–34615 (2010).
Hoitsma, N. M. et al. Structure and function relationships in mammalian DNA polymerases. Cell. Mol. Life Sci. 77, 35–59 (2020).
Bochman, M. L. & Schwacha, A. The Mcm complex: unwinding the mechanism of a replicative helicase. Microbiol. Mol. Biol. Rev. 73, 652–683 (2009).
Gross, R. E. et al. Bone morphogenetic proteins promote astroglial lineage commitment by mammalian subventricular zone progenitor cells. Neuron 17, 595–606 (1996).
Fainsod, A., Steinbeisser, H. & De Robertis, E. M. On the function of BMP-4 in patterning the marginal zone of the Xenopus embryo. EMBO J. 13, 5015–5025 (1994).
Moser, M. et al. BMPER, a novel endothelial cell precursor-derived protein, antagonizes bone morphogenetic protein signaling and endothelial cell differentiation. Mol. Cell. Biol. 23, 5664–5679 (2003).
Oelgeschläger, M., Larraín, J., Geissert, D. & De Robertis, E. M. The evolutionarily conserved BMP-binding protein twisted gastrulation promotes BMP signalling. Nature 405, 757–763 (2000).
Larraín, J. et al. Proteolytic cleavage of chordin as a switch for the dual activities of twisted gastrulation in BMP signaling. Development 128, 4439–4447 (2001).
Oelgeschläger, M. et al. The pro-BMP activity of twisted gastrulation is independent of BMP binding. Development 130, 4047–4056 (2003).
Onichtchouk, D. et al. Silencing of TGF-beta signalling by the pseudoreceptor BAMBI. Nature 401, 480–485 (1999).
Moos, M. Jr, Wang, S. & Krinks, M. Anti-dorsalizing morphogenetic protein is a novel TGF-beta homolog expressed in the Spemann organizer. Development 121, 4293–4301 (1995).
Hemmati-Brivanlou, A., Kelly, O. G. & Melton, D. A. Follistatin, an antagonist of activin, is expressed in the Spemann organizer and displays direct neuralizing activity. Cell 77, 283–295 (1994).
Dosch, R. & Niehrs, C. Requirement for anti-dorsalizing morphogenetic protein in organizer patterning. Mech. Dev. 90, 195–203 (2000).
Suzuki, A. et al. A truncated bone morphogenetic protein receptor affects dorsal–ventral patterning in the early Xenopus embryo. Proc. Natl. Acad. Sci. USA 91, 10255–10259 (1994).
Ambrosio, A. L. et al. Crossveinless-2 Is a BMP feedback inhibitor that binds chordin/BMP to regulate Xenopus embryonic patterning. Dev. Cell 15, 248–260 (2008).
Domazet-Lošo, T. & Tautz, D. A phylogenetically based transcriptome age index mirrors ontogenetic divergence patterns. Nature 468, 815–818 (2010).
Srivastava, M., Mazza-Curll, K. L., Wolfswinkel, J. C. & Reddien, P. W. Whole-body acoel regeneration is controlled by Wnt and Bmp-Admp signaling. Curr. Biol. 24, 1107–1113 (2014).
True, J. R. & Haag, E. S. Developmental system drift and flexibility in evolutionary trajectories. Evol. Dev. 3, 109–119 (2001).
Wang, Y.-C. & Ferguson, E. L. Spatial bistability of Dpp-receptor interactions during Drosophila dorsal–ventral patterning. Nature 434, 229–234 (2005).
Le Petillon, Y. et al. Nodal/activin pathway is a conserved neural induction signal in chordates. Nat. Ecol. Evol. 1, 1192–1200 (2017).
Garcia-Fernàndez, J., D’Aniello, S. & Escrivà, H. Organizing chordates with an organizer. BioEssays 29, 619–624 (2007).
Kraus, Y., Fritzenwanker, J. H., Genikhovich, G. & Technau, U. The blastoporal organiser of a sea anemone. Curr. Biol. 17, R874–R876 (2007).
Kraus, Y., Aman, A., Technau, U. & Genikhovich, G. Pre-bilaterian origin of the blastoporal axial organizer. Nat. Commun. 7, 11694 (2016).
Knabl, P., Ordoñez, J. F., Montenegro Cabrera, J. D., Wollesen, T. & Genikhovich, G. The anti-neural role of BMP signaling is a side effect of its global function in dorsoventral patterning. Preprint at bioRxiv https://doi.org/10.1101/2025.06.08.658475 (2025).
Martín-Durán, J. M. et al. Convergent evolution of bilaterian nerve cords. Nature 553, 45–50 (2018).
Martín-Durán, J. M. & Hejnol, A. A developmental perspective on the evolution of the nervous system. Dev. Biol. 475, 181–192 (2021).
Kuo, D.-H. & Weisblat, D. A. A new molecular logic for BMP-mediated dorsoventral patterning in the leech Helobdella. Curr. Biol. 21, 1282–1288 (2011).
Nielsen, C. Some aspects of spiralian development. Acta Zool. 91, 20–28 (2010).
Hejnol, A. A twist in time—the evolution of spiral cleavage in the light of animal phylogeny. Integr. Comp. Biol. 50, 695–706 (2010).
Henry, J. Q. Spiralian model systems. Int. J. Dev. Biol. 58, 389–401 (2014).
Amiel, A. R., Henry, J. Q. & Seaver, E. C. An organizing activity is required for head patterning and cell fate specification in the polychaete annelid Capitella teleta: new insights into cell-cell signaling in Lophotrochozoa. Dev. Biol. 379, 107–122 (2013).
van den Biggelaar, J. A. Development of dorsoventral polarity and mesentoblast determination in Patella vulgata. J. Morphol. 154, 157–186 (1977).
Lanza, A. R. & Seaver, E. C. An organizing role for the TGF-β signaling pathway in axes formation of the annelid Capitella teleta. Dev. Biol. 435, 26–40 (2018).
Lambert, J. D. Developmental patterns in spiralian embryos. Curr. Biol. 20, 72–77 (2010).
Martín-Durán, J. M. & Marlétaz, F. Unravelling spiral cleavage. Development 147, dev181081 (2020).
Henry, J. J. & Martindale, M. Q. Conservation and innovation in spiralian development. Hydrobiologia 402, 255–265 (1999).
Seaver, E. C. Variation in spiralian development: insights from polychaetes. Int. J. Dev. Biol. 58, 457–467 (2014).
Tagawa, K., Nishino, A., Humphreys, T. & Satoh, N. The spawning and early development of the Hawaiian acorn worm (hemichordate), Ptychodera flava. Zool. Sci. 15, 85–91 (1998).
Andrews, S. FastQC: a quality control tool for high throughput sequence data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (2010).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Cheng, H., Concepcion, G. T., Feng, X., Zhang, H. & Li, H. Haplotype-resolved de novo assembly using phased assembly graphs with hifiasm. Nat. Methods 18, 170–175 (2021).
Guan, D. et al. Identifying and removing haplotypic duplication in primary genome assemblies. Bioinformatics 36, 2896–2898 (2020).
Rao, S. S. et al. A 3D map of the human genome at kilobase resolution reveals principles of chromatin looping. Cell 159, 1665–1680 (2014).
Burton, J. N. et al. Chromosome-scale scaffolding of de novo genome assemblies based on chromatin interactions. Nat. Biotechnol. 31, 1119–1125 (2013).
Vasimuddin, M., Misra, S., Li, H. & Aluru, S. Efficient architecture-aware acceleration of BWA-MEM for multicore systems. In Proc. International Parallel and Distributed Processing Symposium 314–324 (IEEE, 2019).
Durand, N. C. et al. Juicer provides a one-click system for analyzing loop-resolution Hi-C experiments. Cell Syst. 3, 95–98 (2016).
Dudchenko, O. et al. De novo assembly of the Aedes aegypti genome using Hi-C yields chromosome-length scaffolds. Science 356, 92–95 (2017).
Durand, N. C. et al. Juicebox provides a visualization system for Hi-C contact maps with unlimited zoom. Cell Syst. 3, 99–101 (2016).
Buchfink, B., Reuter, K. & Drost, H.-G. Sensitive protein alignments at tree-of-life scale using DIAMOND. Nat. Methods 18, 366–368 (2021).
Kim, D., Paggi, J. M., Park, C., Bennett, C. & Salzberg, S. L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 37, 907–915 (2019).
Challis, R., Richards, E., Rajan, J., Cochrane, G. & Blaxter, M. B. Interactive quality assessment of genome assemblies. G3 10, 1361–1374 (2020).
Flynn, J. M. et al. RepeatModeler2 for automated genomic discovery of transposable element families. Proc. Natl. Acad. Sci. USA 117, 9451–9457 (2020).
Price, A. L., Jones, N. C. & Pevzner, P. A. De novo identification of repeat families in large genomes. Bioinformatics 21, 351–358 (2005).
Bao, Z. & Eddy, S. R. Automated de novo identification of repeat sequence families in sequenced genomes. Genome Res. 12, 1269–1276 (2002).
Ellinghaus, D., Kurtz, S. & Willhoeft, U. LTRharvest, an efficient and flexible software for de novo detection of LTR retrotransposons. BMC Bioinform. 9, 18 (2008).
Ou, S. & Jiang, N. LTR_retriever: a highly accurate and sensitive program for identification of long terminal repeat retrotransposons. Plant Physiol. 176, 1410–1422 (2018).
Hoff, K. J., Lomsadze, A., Borodovsky, M. & Stanke, M. Whole-genome annotation with BRAKER. Methods Mol. Biol. 1962, 65–95 (2019).
Brůna, T., Hoff, K. J., Lomsadze, A., Stanke, M. & Borodovsky, M. BRAKER2: automatic eukaryotic genome annotation with GeneMark-EP+ and AUGUSTUS supported by a protein database. NAR Genom. Bioinform. 3, lqaa108 (2021).
Hoff, K. J., Lange, S., Lomsadze, A., Borodovsky, M. & Stanke, M. B. Unsupervised RNA-seq-based genome annotation with GeneMark-ET and AUGUSTUS. Bioinformatics 32, 767–769 (2016).
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Barnett, D. W., Garrison, E. K., Quinlan, A. R., Strömberg, M. P. & Marth, G. T. BamTools: a C++ API and toolkit for analyzing and managing BAM files. Bioinformatics 27, 1691–1692 (2011).
Lomsadze, A., Burns, P. D. & Borodovsky, M. Integration of mapped RNA-Seq reads into automatic training of eukaryotic gene finding algorithm. Nucleic Acids Res. 42, e119 (2014).
Buchfink, B., Xie, C. & Huson, D. H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods 12, 59–60 (2015).
Stanke, M., Diekhans, M., Baertsch, R. & Haussler, D. Using native and syntenically mapped cDNA alignments to improve de novo gene finding. Bioinformatics 24, 637–644 (2008).
Stanke, M., Schöffmann, O., Morgenstern, B. & Waack, S. Gene prediction in eukaryotes with a generalized hidden Markov model that uses hints from external sources. BMC Bioinforma. 7, 62 (2006).
Smit, A. F. A., Hubley, R. & Green, P. RepeatMasker Open-4.0. http://www.repeatmasker.org (2015).
Simão, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Jones, P. et al. InterProScan 5: genome-scale protein function classification. Bioinformatics 30, 1236–1240 (2014).
Aramaki, T. et al. KofamKOALA: KEGG ortholog assignment based on profile HMM and adaptive score threshold. Bioinformatics 36, 2251–2252 (2020).
Huerta-Cepas, J. et al. eggNOG 5.0: a hierarchical, functionally and phylogenetically annotated orthology resource based on 5090 organisms and 2502 viruses. Nucleic Acids Res. 47, 309–314 (2018).
Cantalapiedra, C. P., Hernández-Plaza, A., Letunic, I., Bork, P. & Huerta-Cepas, J. eggNOG-mapper v2: functional annotation, orthology assignments, and domain prediction at the metagenomic scale. Mol. Biol. Evol. 38, 5825–5829 (2021).
Emms, D. M. & Kelly, S. OrthoFinder: phylogenetic orthology inference for comparative genomics. Genome Biol. 20, 238 (2019).
Hao, Z. et al. RIdeogram: drawing SVG graphics to visualize and map genome-wide data on the idiograms. PeerJ Comput. Sci. 6, e251 (2020).
Steenwyk, J. L. et al. OrthoSNAP: a tree splitting and pruning algorithm for retrieving single-copy orthologs from gene family trees. PLoS Biol. 20, e3001827 (2022).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Katoh, K., Misawa, K., Kuma, K.-I. & Miyata, T. MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30, 3059–3066 (2002).
Steenwyk, J. L. et al. ClipKIT: a multiple sequence alignment trimming software for accurate phylogenomic inference. PLoS Biol. 18, e3001007 (2020).
Minh, B. Q. et al. IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 37, 1530–1534 (2020).
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K. F., Haeseler, A. & Jermiin, L. S. ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Methods 14, 587–589 (2017).
Hoang, D. T., Chernomor, O., Haeseler, A., Minh, B. Q. & Vinh, L. S. UFBoot2: improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 35, 518–522 (2018).
Steenwyk, J. L. et al. PhyKIT: a broadly applicable UNIX shell toolkit for processing and analyzing phylogenomic data. Bioinformatics 37, 2325–2331 (2021).
Mendes, F. K., Vanderpool, D., Fulton, B. & Hahn, M. W. CAFE 5 models variation in evolutionary rates among gene families. Bioinformatics 36, 5516–5518 (2020).
Vences, M. et al. iTaxoTools 0.1: kickstarting a specimen-based software toolkit for taxonomists. Megataxa 6, 77–92 (2021).
Sanderson, M. J. r8s: inferring absolute rates of molecular evolution and divergence times in the absence of a molecular clock. Bioinformatics 19, 301–302 (2003).
Yu, P. B. et al. Dorsomorphin inhibits BMP signals required for embryogenesis and iron metabolism. Nat. Chem. Biol. 4, 33–41 (2008).
Yu, P. B. et al. BMP type I receptor inhibition reduces heterotopic ossification. Nat. Med. 14, 1363–1369 (2008).
Haas, B. J. et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 8, 1494–1512 (2013).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Law, C. W., Chen, Y., Shi, W. & Smyth, G. K. voom: precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol. 15, R29 (2014).
UniProt Consortium UniProt: the universal protein knowledgebase in 2023. Nucleic Acids Res. 51, 523–531 (2023).
Domazet-Loso, T., Brajković, J. & Tautz, D. A phylostratigraphy approach to uncover the genomic history of major adaptations in metazoan lineages. Trends Genet. 23, 533–539 (2007).
Barrera-Redondo, J., Lotharukpong, J. S., Drost, H.-G. & Coelho, S. M. Uncovering gene-family founder events during major evolutionary transitions in animals, plants and fungi using GenEra. Genome Biol. 24, 54 (2023).
Drost, H.-G., Gabel, A., Liu, J., Quint, M. & Grosse, I. myTAI: evolutionary transcriptomics with R. Bioinformatics 34, 1589–1590 (2017).
Acknowledgements
We acknowledge the permission from Amami City, Kagoshima, Japan, for collecting L. anatina specimens. We thank Ryo Koyanagi in the OIST DNA Sequencing Section for his support of RNA sequencing and Asuka Sentoku for support with the embryonic experiment at the University of Ryukyu. We also thank Keisuke Nakashima for a gift of chitin-binding probes and Jr-Kai Yu for support with microscopy. We are grateful to Konstantin Khalturin for facilitating the shipment of fixed embryos from Japan to the USA and to D. Marcela Bolaños for handling the shipment from the USA to Taiwan. This work was supported in part by multiple funding sources over the past decade, including a Japan Society for the Promotion of Science Grant-in-Aid for JSPS Research Fellows (15J01101), a Royal Society Newton International Fellowship (NIF\R1\201315), an Academia Sinica Career Development Award (AS-CDA-112-L06), an Academia Sinica Grand Challenge Program Seed Grant (AS-GCS-114-L08), and National Science and Technology Council Research Project Grants (113-2311-B-001-026 and 114-2311-B-001-024-MY3) to Y.-J.L., T.S. was supported by a JSPS Overseas Research Fellowship. Y.H.W. was supported by the Innovation Team Project of Universities in Guangdong Province (2023KCXTD028) and the National Natural Science Foundation of China General Program (42276104). We thank the Symbiosis Genomics and Evolution Lab members for their assistance and support, and three anonymous reviewers for their insightful comments.
Author information
Authors and Affiliations
Contributions
T.D.L., Y.-J.L. conceived the project. K.S., K.E., N.S. and Y.-J.L. collected specimens. Y.H.W. sequenced the chromosome-scale genome and tissue transcriptomes. T.D.L. and M.-E.C. annotated the genomes. Y.-J.L. conducted embryonic experiments and functional transcriptomics. T.S., K.S., L.-J.K., Y.-L.C. and Y.-J.L. performed whole mount in situ hybridisation. T.D.L., I.J.-Y.L. and Y.-J.L. prepared code for GitHub. K.H. prepared the genome browser. T.D.L. and Y.-J.L. analysed data and wrote the manuscript. K.E., P.W.H.H. and Y.H.W. discussed the results and edited the manuscript. All authors contributed to the revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Néva Meyer and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lewin, T.D., Sakagami, T., Shimizu, K. et al. Brachiopod genome unveils the evolution of BMP signalling in bilaterian body patterning. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70403-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70403-5