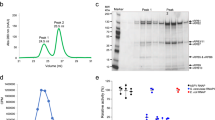
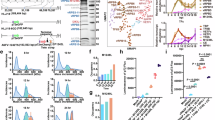
Abstract
African swine fever virus (ASFV) is a large DNA virus that poses a major threat to the global swine industry. Its virion is encapsulated by an icosahedral capsid predominantly composed of the structural protein p72, which constitutes approximately one-third of the total virion mass. Despite its abundance, the mechanisms regulating p72 stability remain poorly understood. Here, we demonstrate that host-mediated stabilization of p72 is essential for efficient ASFV replication. Mass spectrometry of p72 co-precipitates identified host cyclophilin A (CypA, also known as PPIA) as a key binding partner of p72. CypA interacts with p72 both in vitro and in vivo, specifically engaging exposed regions of p72 via its hydrophobic cavity. CypA interaction stabilizes p72 by reducing its ubiquitination and preventing proteasomal degradation, whereas cyclic CypA inhibitors destabilize p72 by disrupting this interaction and promoting its ubiquitination. Importantly, genetic or pharmacological inhibition of CypA markedly impairs ASFV replication. Mechanistically, CypA inhibition disrupts viral factory formation and virion assembly by decreasing p72 protein accumulation without affecting its transcription. Together, our findings uncover a host-dependent mechanism regulating capsid protein stability and highlight host CypA as a promising target for antiviral strategies against ASFV.
Similar content being viewed by others
Data availability
The mass spectrometry data generated in this study have been deposited in the ProteomeXchange Consortium via the iProX partner repository under accession code PXD073232. The RNA-seq data generated in this study have been deposited in the Gene Expression Omnibus (GEO) under accession code GSE300823. The previously published PDB structures referred to in this study include 3K0N, 1CWA, 1CWM, and 5HSV. All data are available within the Article, Supplementary Information, or Source Data file. Source data are provided with this paper.
References
You, S. et al. African swine fever outbreaks in China led to gross domestic product and economic losses. Nat. Food 2, 802–808 (2021).
Ata, E. B. et al. African swine fever virus: a raised global upsurge and a continuous threaten to pig husbandry. Microb. Pathogenesis 167, 105561 (2022).
Dixon, L., Sun, H. & Roberts, H. African swine fever. Antivir. Res. 165, 34–41 (2019).
Sánchez-Vizcaíno, J.M., Laddomada, A. & Arias, M.L. African swine fever virus. In Dis. Swine, 443–452 (Wiley, 2019).
Dixon, L. K., Chapman, D. A., Netherton, C. L. & Upton, C. African swine fever virus replication and genomics. Virus Res. 173, 3–14 (2013).
Salas, M. L. & Andrés, G. African swine fever virus morphogenesis. Virus Res. 173, 29–41 (2013).
Andrés, G., Simon-Mateo, C. & Vinuela, E. Assembly of African swine fever virus: role of polyprotein pp220. J. Virol. 71, 2331–2341 (1997).
Wang, N. et al. Architecture of African swine fever virus and implications for viral assembly. Science 366, 640–644 (2019).
Liu, S. et al. Cryo-EM structure of the African swine fever virus. Cell Host Microbe 26, 836–843. e833 (2019).
Andrés, G., Charro, D., Matamoros, T., Dillard, R. S. & Abrescia, N. G. The cryo-EM structure of African swine fever virus unravels a unique architecture comprising two icosahedral protein capsids and two lipoprotein membranes. J. Biol. Chem. 295, 1–12 (2020).
Liu, Q. et al. Structure of the African swine fever virus major capsid protein p72. Cell Res. 29, 953–955 (2019).
Cobbold, C., Windsor, M. & Wileman, T. A virally encoded chaperone specialized for folding of the major capsid protein of African swine fever virus. J. Virol. 75, 7221–7229 (2001).
Yu, Q. et al. p72 antigenic mapping reveals a potential supersite of vulnerability for African swine fever virus. Cell Discov. 10, 80 (2024).
Epifano, C., Krijnse-Locker, J., Salas, M. A. L., Rodríguez, J. M. & Salas, J. The African swine fever virus nonstructural protein pB602L is required for formation of the icosahedral capsid of the virus particle. J. Virol. 80, 12260–12270 (2006).
Song, J. et al. Autophagy promotes p72 degradation and capsid disassembly during the early phase of African swine fever virus infection. J. Virol. 99, e01701–e01724 (2024).
Hernáez, B., Guerra, M., Salas, M. L. & Andrés, G. African swine fever virus undergoes outer envelope disruption, capsid disassembly and inner envelope fusion before core release from multivesicular endosomes. PLoS Pathog. 12, e1005595 (2016).
Nigro, P., Pompilio, G. & Capogrossi, M. Cyclophilin A: a key player for human disease. Cell Death Dis. 4, e888–e888 (2013).
Liao, Y., Luo, D., Peng, K. & Zeng, Y. Cyclophilin A: a key player for etiological agent infection. Appl. Microbiol. Biotechnol. 105, 1365–1377 (2021).
Jin, S., Zhang, M. & Qiao, X. Cyclophilin A: promising target in cancer therapy. Cancer Biol. Ther. 25, 2425127 (2024).
Zheng, Y. et al. Transcriptome profiling in swine macrophages infected with African swine fever virus at single-cell resolution. Proc. Natl. Acad. Sci. 119, e2201288119 (2022).
Vogel, C. & Marcotte, E. M. Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat. Rev. Genet. 13, 227–232 (2012).
Davis, T. L. et al. Structural and biochemical characterization of the human cyclophilin family of peptidyl-prolyl isomerases. PLoS Biol. 8, e1000439 (2010).
Kim, K. et al. Cyclophilin A protects HIV-1 from restriction by human TRIM5α. Nat. Microbiol. 4, 2044–2051 (2019).
Zhao, Y. et al. TRIM5α restricts poxviruses and is antagonized by CypA and the viral protein C6. Nature 620, 873–880 (2023).
Sheng, X. et al. Host cyclophilin A facilitates SARS-CoV-2 infection by binding and stabilizing spike on virions. Signal Transduct. Target. Ther. 8, 459 (2023).
Fraser, J. S. et al. Hidden alternative structures of proline isomerase essential for catalysis. Nature 462, 669–673 (2009).
Gamble, T. R. et al. Crystal structure of human cyclophilin A bound to the amino-terminal domain of HIV-1 capsid. Cell 87, 1285–1294 (1996).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 1, 3 (2024).
Schrödinger, L. The PyMOL Molecular Graphics System, Version 2.6.0a0. (2025).
Sun, H. et al. OAS1 suppresses African swine fever virus replication by recruiting TRIM21 to degrade viral major capsid protein. J. Virol. 97, e01217–e01223 (2023).
Hyer, M. L. et al. A small-molecule inhibitor of the ubiquitin activating enzyme for cancer treatment. Nat. Med. 24, 186–193 (2018).
Liu, C. et al. Cyclophilin A stabilizes the HIV-1 capsid through a novel non-canonical binding site. Nat. Commun. 7, 10714 (2016).
Madiraju, C., Novack, J. P., Reed, J. C. & Matsuzawa, S. K63 ubiquitination in immune signaling. Trends Immunol. 43, 148–162 (2022).
Sheng, X., Xia, Z., Yang, H. & Hu, R. The ubiquitin codes in cellular stress responses. Protein Cell 15, 157–190 (2024).
Ohtake, F., Tsuchiya, H., Saeki, Y. & Tanaka, K. K63 ubiquitylation triggers proteasomal degradation by seeding branched ubiquitin chains. Proc. Natl. Acad. Sci. 115, E1401–E1408 (2018).
Vasco Ferreira, J., Rosa Soares, A., Silva Ramalho, J., Pereira, P. & Girao, H. K63 linked ubiquitin chain formation is a signal for HIF1A degradation by chaperone-mediated autophagy. Sci. Rep. 5, 10210 (2015).
Lu, W. et al. SKP2 loss destabilizes EZH2 by promoting TRAF6-mediated ubiquitination to suppress prostate cancer. Oncogene 36, 1364–1373 (2017).
Handschumacher, R. E., Harding, M. W., Rice, J., Drugge, R. J. & Speicher, D. W. Cyclophilin: a specific cytosolic binding protein for cyclosporin A. Science 226, 544–547 (1984).
Hopkins, S. & Gallay, P. Cyclophilin inhibitors: an emerging class of therapeutics for the treatment of chronic hepatitis C infection. Viruses 4, 2558–2577 (2012).
Dujardin, M., Bouckaert, J., Rucktooa, P. & Hanoulle, X. X-ray structure of alisporivir in complex with cyclophilin A at 1.5 Å resolution. Acta Crystallogr. Sect. F Struct. Biol. Commun. 74, 583–592 (2018).
Kallen, J., Mikol, V., Taylor, P. & Walkinshaw, M. D. X-ray structures and analysis of 11 cyclosporin derivatives complexed with cyclophilin A. J. Mol. Biol. 283, 435–449 (1998).
Pflügl, G. et al. X-ray structure of a decameric cyclophilin-cyclosporin crystal complex. Nature 361, 91–94 (1993).
Kallen, J. et al. Structure of human cyclophilin and its binding site for cyclosporin A determined by X-ray crystallography and NMR spectroscopy. Nature 353, 276–279 (1991).
Zydowsky, L. D. et al. Active site mutants of human cyclophilin A separate peptidyl-prolyl isomerase activity from cyclosporin A binding and calcineurin inhibition. Protein Sci. 1, 1092–1099 (1992).
Wang, W. et al. Establishment of an indirect immunofluorescence assay for the detection of African swine fever virus antibodies. J. Integr. Agric. 23, 228–238 (2024).
Chen, W. et al. A seven-gene-deleted African swine fever virus is safe and effective as a live attenuated vaccine in pigs. Sci. China Life Sci. 63, 623–634 (2020).
Cackett, G. et al. The African swine fever virus transcriptome. J. Virol. 94, 00119–00120 (2020).
Blome, S., Franzke, K. & Beer, M. African swine fever–a review of current knowledge. Virus Res. 287, 198099 (2020).
Gaudreault, N. N., Madden, D. W., Wilson, W. C., Trujillo, J. D. & Richt, J. A. African swine fever virus: an emerging DNA arbovirus. Front. Vet. Sci. 7, 215 (2020).
Wang, Y. et al. Structure of African swine fever virus and associated molecular mechanisms underlying infection and immunosuppression: a review. Front. Immunol. 12, 715582 (2021).
Rodríguez, J. M., García-Escudero, R., Salas, M. L. & Andrés, G. African swine fever virus structural protein p54 is essential for the recruitment of envelope precursors to assembly sites. J. Virol. 78, 4299–4313 (2004).
Suárez, C., Gutiérrez-Berzal, J., Andrés, G., Salas, M. L. & Rodríguez, J. M. African swine fever virus protein p17 is essential for the progression of viral membrane precursors toward icosahedral intermediates. J. Virol. 84, 7484–7499 (2010).
García-Escudero, R. N., Andrés, G. N., Almazán, F. & Viñuela, E. Inducible gene expression from African swine fever virus recombinants: analysis of the major capsid protein p72. J. Virol. 72, 3185–3195 (1998).
Franke, E. K., Yuan, H. E. H. & Luban, J. Specific incorporation of cyclophilin A into HIV-1 virions. Nature 372, 359–362 (1994).
Thali, M. et al. Functional association of cyclophilin A with HIV-1 virions. Nature 372, 363–365 (1994).
Towers, G. J. et al. Cyclophilin A modulates the sensitivity of HIV-1 to host restriction factors. Nat. Med. 9, 1138–1143 (2003).
Selyutina, A. et al. Cyclophilin A prevents HIV-1 restriction in lymphocytes by blocking human TRIM5α binding to the viral core. Cell Rep. 30, 3766–3777. e3766 (2020).
Li, H. et al. Cyclophilin A facilitates influenza B virus replication by stabilizing viral proteins. Iscience 26, 108515 (2023).
Yang, F. et al. Cyclophilin A is an essential cofactor for hepatitis C virus infection and the principal mediator of cyclosporine resistance in vitro. J. Virol. 82, 5269–5278 (2008).
Chatterji, U. et al. The isomerase active site of cyclophilin A is critical for hepatitis C virus replication. J. Biol. Chem. 284, 16998–17005 (2009).
Foster, T. L., Gallay, P., Stonehouse, N. J. & Harris, M. Cyclophilin A interacts with domain II of hepatitis C virus NS5A and stimulates RNA binding in an isomerase-dependent manner. J. Virol. 85, 7460–7464 (2011).
Yuan, W., Ge, H. & He, B. Pro-inflammatory activities induced by CyPA–EMMPRIN interaction in monocytes. Atherosclerosis 213, 415–421 (2010).
Li, Z., Min, W. & Gou, J. Knockdown of cyclophilin A reverses paclitaxel resistance in human endometrial cancer cells via suppression of MAPK kinase pathways. Cancer Chemother. Pharmacol. 72, 1001–1011 (2013).
DuShane, J. K. & Maginnis, M. S. Human DNA virus exploitation of the MAPK-ERK cascade. Int. J. Mol. Sci. 20, 3427 (2019).
Cheng, Y. et al. Virus-induced p38 MAPK activation facilitates viral infection. Theranostics 10, 12223 (2020).
Wöhnke, E. et al. Proteome analysis of swine macrophages after infection with two genotype II African swine fever isolates of different pathogenicity. Viruses 14, 2140 (2022).
Yurchenko, V. et al. Active site residues of cyclophilin A are crucial for its signaling activity via CD147. J. Biol. Chem. 277, 22959–22965 (2002).
Sun, S. et al. Cyclophilin A (CypA) interacts with NF-κB subunit, p65/RelA, and contributes to NF-κB activation signaling. PLoS One 9, e96211 (2014).
Dawar, F. U. et al. Potential role of cyclophilin A in regulating cytokine secretion. J. Leukoc. Biol. 102, 989–992 (2017).
Wang, S. et al. Cytokine storm in domestic pigs induced by infection of virulent African swine fever virus. Front. Vet. Sci. 7, 601641 (2021).
He, H. et al. Cyclophilin A inhibits rotavirus replication by facilitating host IFN-I production. Biochem. Biophys. Res. Commun. 422, 664–669 (2012).
Liu, W. et al. Cyclophilin A-regulated ubiquitination is critical for RIG-I-mediated antiviral immune responses. elife 6, e24425 (2017).
Rui, B. et al. LINC02888 promotes HGSOC progression and immune evasion via PPIB-mediated stabilization of LAPTM5 mRNA and inhibition of RIG-I-like receptor signaling. J. Transl. Med. 23, 640 (2025).
YOKOI, H. et al. Characterization of cyclophilin 40: highly conserved protein that directly associates with Hsp90. Biol. Pharm. Bull. 19, 506–511 (1996).
Wang, P. & Heitman, J. The cyclophilins. Genome Biol. 6, 1–6 (2005).
Hadpech, S. & Thongboonkerd, V. Current update on theranostic roles of cyclophilin A in kidney diseases. Theranostics 12, 4067 (2022).
Webb, A.E. Characterisation of Human Peptidyl Prolyl Isomerase-like Protein 1 (PPIL1) Mutations. PhD thesis, University of Leeds, 1–183 (2019).
Sánchez, E. G., Pérez-Núñez, D. & Revilla, Y. Mechanisms of entry and endosomal pathway of African swine fever virus. Vaccines 5, 42 (2017).
Rathakrishnan, A., Reis, A. L., Moffat, K. & Dixon, L. K. Isolation of porcine bone marrow cells and generation of recombinant African swine fever viruses. Afr. Swine Fever Virus. Methods Protoc. 2503, 73–94 (2022).
Zhao, D. et al. Replication and virulence in pigs of the first African swine fever virus isolated in China. Emerg. Microbes Infect. 8, 438–447 (2019).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Shevchenko, A., Tomas, H., Havli, J., Olsen, J. V. & Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 1, 2856–2860 (2006).
Gao, X. et al. Uncovering dark mass in population proteomics: Pan-analysis of single amino acid polymorphism relevant to cognition and aging. Cell Genom. 5, 100763 (2025).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized ppb-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Zhu, W. et al. Lateral flow assay for the detection of African swine fever virus antibodies using gold nanoparticle-labeled acid-treated p72. Front. Chem. 9, 804981 (2022).
Meng, K. et al. Structural design and assessing of Recombinantly expressed African swine fever virus p72 Trimer in Saccharomyces cerevisiae. Front. Microbiol. 13, 802098 (2022).
Zhu, F. et al. HAX1 mediates SARS-CoV-2 spike-triggered unfolded protein response in host cells. FEBS J. 292, 5948–5964 (2025).
Sheng, X. et al. Non-proteolytic ubiquitination of HBx controls HBV replication. Virol. Sin. 39, 338–342 (2024).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Wang, W. et al. Cell-adapted African swine fever virus Pig/HLJ/18 is highly attenuated but fails to induce immune protection against a challenge with its parental virus. J. Integr. Agric. https://doi.org/10.1016/j.jia.2025.03.017 (2025).
Shen, D. et al. A genome-wide CRISPR/Cas9 knockout screen identifies TMEM239 as an important host factor in facilitating African swine fever virus entry into early endosomes. Plos Pathog. 20, e1012256 (2024).
Matamoros, T. et al. African swine fever virus protein pE199L mediates virus entry by enabling membrane fusion and core penetration. MBio 11, 00789–00720 (2020).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, i884–i890 (2018).
Kim, D., Paggi, J. M., Park, C., Bennett, C. & Salzberg, S. L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 37, 907–915 (2019).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 1–21 (2014).
Yu, G., Wang, L.-G., Han, Y. & He, Q.-Y. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics J. Integr. Biol. 16, 284–287 (2012).
Ashburner, M. et al. Gene ontology: tool for the unification of biology. Nat. Genet. 25, 25–29 (2000).
Kanehisa, M. & Goto, S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Acknowledgements
We thank Dr. Geng Meng (China Agricultural University) for providing the properly folded recombinant p72 trimer protein. We are also grateful to Dr. Qingrun Li (Shanghai Advanced Research Institute, Chinese Academy of Sciences) for assistance with mass spectrometry analysis. This work was supported by the National Key Research and Development Program of China (2021YFD1800101, 2022YFD1800604 to D.Z.), the Central Public-interest Scientific Institution Basal Research Fund (CAAS-ZDRW202409 to D.Z., 1610302024005 and Y2026YC65 to X.S.), the Innovation Program of the Chinese Academy of Agricultural Sciences (CAAS-CSLPDCP-202301 to D.Z.), and the Heilongjiang Provincial Natural Science Foundation of China (JQ2023C005 to D.Z.).
Author information
Authors and Affiliations
Contributions
D.Z. and X.S. conceived and supervised the project. H.K. and L.Y. performed the majority of the experiments, with assistance from Z.Z., Y.Zhang, and L.L. L.Y. and L.S. provided support with bioinformatics analyses. W.W., Y.Zhu, E.S., F.L., and Z.B. contributed essential materials and methodology. D.Z., X.S., H.K., and L.Y. analyzed and organized the data. X.S. prepared the figures and wrote the manuscript. D.Z., X.S., and H.K. revised and edited the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Germán Andrés and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kong, H., Yang, L., Zhang, Z. et al. Cyclophilin A stabilizes the capsid protein p72 to facilitate African swine fever virus replication. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70430-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70430-2