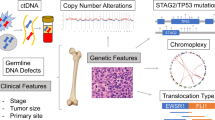
Abstract
Ewing sarcoma (EWS), a rare pediatric bone tumor, poses unique therapeutic challenges due to its distinct microenvironment and limited molecular understanding. To gain a comprehensive molecular and functional view of the tumors in their microenvironment, we perform a deep mass spectrometry-based proteomic analysis of 170 tumor samples from 74 patients from primary, relapsed, and metastatic tumors. Analysis of more than 10,000 proteins across patients reveals insights into cancer prognosis, chemo-resistance, and progression. Our analyses suggest that ferroptosis pathways may be associated with chemotherapy response in EWS, and we delineate molecular subclasses that correlate the tumor immune landscape with DNA damage repair, ubiquitin-related proteins, and patient outcomes. Multiplexed immunofluorescence imaging indicates possible associations between neutrophils and poorer prognosis, and between macrophages/T cells and a more favorable prognosis. Altogether, this investigation provides valuable insights into the intricate biology of EWS, paving the way for developing therapeutic strategies.
Similar content being viewed by others
Data availability
The raw mass spectrometry data generated in this study have been deposited in the PRIDE ProteomExchange database under accession code PXD050234. The processed proteomics data generated in this study are provided in the Supplementary data. The Image data used in this study are available in the BioImage Archive database under accession code S-BIAD1597. Source data are provided with this paper.
Code availability
The code for image processing and the Github MIT license document are available via the following GitHub79: [https://github.com/WIS-MICC-CellObservatory/Ewing-Sarcoma-Proteomics-and-Immune-Landscape].
References
Hawkins, C. et al. Advances and challenges in pediatric and childhood cancers. Cancer Cell 38, 429–432 (2020).
Sweet-Cordero, E. A. & Biegel, J. A. The genomic landscape of pediatric cancers: implications for diagnosis and treatment. Science 363, 1170–1175 (2019).
Grobner, S. N. et al. The landscape of genomic alterations across childhood cancers. Nature 555, 321–327 (2018).
Grunewald, T. G. P. et al. Ewing sarcoma. Nat. Rev. Dis. Primers 4, 5 (2018).
Riggi, N., Suva, M. L. & Stamenkovic, I. Ewing’s sarcoma. N. Engl. J. Med. 384, 154–164 (2021).
Tanner, J. M. et al. EWS/FLI is a master regulator of metabolic reprogramming in Ewing sarcoma. Mol. Cancer Res. 15, 1517–1530 (2017).
Elzi, D. J., Song, M., Houghton, P. J., Chen, Y. & Shiio, Y. The role of FLI-1-EWS, a fusion gene reciprocal to EWS-FLI-1, in Ewing sarcoma. Genes Cancer 6, 452–461 (2015).
Franzetti, G. A. et al. Cell-to-cell heterogeneity of EWSR1-FLI1 activity determines proliferation/migration choices in Ewing sarcoma cells. Oncogene 36, 3505–3514 (2017).
Gaspar, N. et al. Ewing sarcoma: current management and future approaches through collaboration. J. Clin. Oncol. 33, 3036–3046 (2015).
Flores, G. & Grohar, P. J. One oncogene, several vulnerabilities: EWS/FLI targeted therapies for Ewing sarcoma. J. Bone Oncol. 31, 100404 (2021).
Aynaud, M. M. et al. Transcriptional programs define intratumoral heterogeneity of Ewing sarcoma at single-cell resolution. Cell Rep. 30, 1767–1779.e1766 (2020).
Miller, H. E. et al. Reconstruction of Ewing sarcoma developmental context from mass-scale transcriptomics reveals characteristics of EWSR1-FLI1 permissibility. Cancers 12, 948 (2020).
Boone, M. A. et al. The FLI portion of EWS/FLI contributes a transcriptional regulatory function that is distinct and separable from its DNA-binding function in Ewing sarcoma. Oncogene 40, 4759–4769 (2021).
Scarborough, J. A. et al. Identifying states of collateral sensitivity during the evolution of therapeutic resistance in Ewing’s sarcoma. iScience 23, 101293 (2020).
Cillo, A. R. et al. Ewing sarcoma and osteosarcoma have distinct immune signatures and intercellular communication networks. Clin. Cancer Res. 28, 4968–4982 (2022).
Morales, E. et al. Role of immunotherapy in Ewing sarcoma. J. Immunother. Cancer 8, e000653 (2020).
Dyson, K. A. et al. Emerging trends in immunotherapy for pediatric sarcomas. J. Hematol. Oncol. 12, 78 (2019).
Stahl, D., Gentles, A. J., Thiele, R. & Gutgemann, I. Prognostic profiling of the immune cell microenvironment in Ewings sarcoma family of tumors. Oncoimmunology 8, e1674113 (2019).
Berger-Achituv, S. et al. A proposed role for neutrophil extracellular traps in cancer immunoediting. Front. Immunol. 4, 48 (2013).
Fujiwara, T. et al. Macrophage infiltration predicts a poor prognosis for human Ewing sarcoma. Am. J. Pathol. 179, 1157–1170 (2011).
Spurny, C. et al. Programmed cell death ligand 1 (PD-L1) expression is not a predominant feature in Ewing sarcomas. Pediatr. Blood Cancer 65, e26719 (2018).
Berghuis, D. et al. Pro-inflammatory chemokine-chemokine receptor interactions within the Ewing sarcoma microenvironment determine CD8(+) T-lymphocyte infiltration and affect tumour progression. J. Pathol. 223, 347–357 (2011).
Rokita, J. L. et al. Genomic profiling of childhood tumor patient-derived xenograft models to enable rational clinical trial design. Cell Rep. 29, 1675–1689 e1679 (2019).
Brohl, A. S. et al. The genomic landscape of the Ewing Sarcoma family of tumors reveals recurrent STAG2 mutation. PLoS Genet. 10, e1004475 (2014).
Tirode, F. et al. Genomic landscape of Ewing sarcoma defines an aggressive subtype with co-association of STAG2 and TP53 mutations. Cancer Discov. 4, 1342–1353 (2014).
Sheffield, N. C. et al. DNA methylation heterogeneity defines a disease spectrum in Ewing sarcoma. Nat. Med. 23, 386–395 (2017).
Yanovich, G. et al. Clinical proteomics of breast cancer reveals a novel layer of breast cancer classification. Cancer Res. 78, 6001–6010 (2018).
Arad, G. & Geiger, T. Functional impact of protein-RNA variation in clinical cancer analyses. Mol. Cell Proteom. 22, 100587 (2023).
Kitata, R. B., Yang, J. C. & Chen, Y. J. Advances in data-independent acquisition mass spectrometry towards comprehensive digital proteome landscape. Mass Spectrom. Rev. 42, 2324–2348 (2023).
Gotti, C. et al. Extensive and accurate benchmarking of DIA acquisition methods and software tools using a complex proteomic standard. J. Proteome Res. 20, 4801–4814 (2021).
Yoshihara, K. et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat. Commun. 4, 2612 (2013).
Machado, I. et al. Does PAX7 and NKX2.2 immunoreactivity in Ewing sarcoma have prognostic significance? Virchows Arch. 480, 909–917 (2022).
Jain, A., Zhang, J. & Rabbitts, T. LINGO-1 is a new therapy target and biomarker for Ewing sarcoma. Clin. Oncol. 2, 1183 (2017).
Louati, S., Senhaji, N., Chbani, L. & Bennis, S. EWSR1 rearrangement and CD99 expression as diagnostic biomarkers for Ewing/PNET sarcomas in a Moroccan population. Dis. Markers 2018, 7971019 (2018).
Bennani-Baiti, I. M. et al. Notch signalling is off and is uncoupled from HES1 expression in Ewing’s sarcoma. J. Pathol. 225, 353–363 (2011).
Schmidt, O. et al. Class I histone deacetylases (HDAC) critically contribute to Ewing sarcoma pathogenesis. J. Exp. Clin. Cancer Res. 40, 322 (2021).
El-Naggar, A. M. et al. Class I HDAC inhibitors enhance YB-1 acetylation and oxidative stress to block sarcoma metastasis. EMBO Rep. 20, e48375 (2019).
Zheng, X., Liang, Y. & Zhang, C. Ferroptosis regulated by hypoxia in cells. Cells 12, 1050 (2023).
Yang, Z. et al. HIF-1alpha drives resistance to ferroptosis in solid tumors by promoting lactate production and activating SLC1A1. Cell Rep. 42, 112945 (2023).
Morales, M. & Xue, X. Targeting iron metabolism in cancer therapy. Theranostics 11, 8412–8429 (2021).
DeRosa, A. & Leftin, A. The iron curtain: macrophages at the interface of systemic and microenvironmental iron metabolism and immune response in cancer. Front. Immunol. 12, 614294 (2021).
Pereira, M. et al. Acute iron deprivation reprograms human macrophage metabolism and reduces inflammation in vivo. Cell Rep. 28, 498–511 e495 (2019).
Manz, D. H., Blanchette, N. L., Paul, B. T., Torti, F. M. & Torti, S. V. Iron and cancer: recent insights. Ann. N. Y. Acad. Sci. 1368, 149–161 (2016).
Friedmann Angeli, J. P., Krysko, D. V. & Conrad, M. Ferroptosis at the crossroads of cancer-acquired drug resistance and immune evasion. Nat. Rev. Cancer 19, 405–414 (2019).
Zhou, N. & Bao, J. FerrDb: a manually curated resource for regulators and markers of ferroptosis and ferroptosis-disease associations. Database 2020, baaa021 (2020).
Volchenboum, S. L. et al. Gene expression profiling of Ewing sarcoma tumors reveals the prognostic importance of tumor-stromal interactions: a report from the Children’s Oncology Group. J. Pathol. Clin. Res. 1, 83–94 (2015).
Wilkerson, M. D. & Hayes, D. N. ConsensusClusterPlus: a class discovery tool with confidence assessments and item tracking. Bioinformatics 26, 1572–1573 (2010).
Sijts, E. J. & Kloetzel, P. M. The role of the proteasome in the generation of MHC class I ligands and immune responses. Cell Mol. Life Sci. 68, 1491–1502 (2011).
Kloetzel, P. M. The proteasome and MHC class I antigen processing. Biochim. Biophys. Acta 1695, 225–233 (2004).
Chen, Z. et al. Ferroptosis as a potential target for cancer therapy. Cell Death Dis. 14, 460 (2023).
Zhang, C., Liu, X., Jin, S., Chen, Y. & Guo, R. Ferroptosis in cancer therapy: a novel approach to reversing drug resistance. Mol. Cancer 21, 47 (2022).
Dos Santos, A. F., Fazeli, G., Xavier da Silva, T. N. & Friedmann Angeli, J. P. Ferroptosis: mechanisms and implications for cancer development and therapy response. Trends Cell Biol. 33, 1062–1076 (2023).
Zhao, Y., Huang, Z. & Peng, H. Molecular mechanisms of ferroptosis and its roles in hematologic malignancies. Front. Oncol. 11, 743006 (2021).
Hassannia, B., Vandenabeele, P. & Vanden Berghe, T. Targeting ferroptosis to iron out cancer. Cancer Cell 35, 830–849 (2019).
Dasgupta, S. & Gan, B. Ferroptosis vulnerability in MYCN-driven neuroblastomas. Clin. Transl. Med. 12, e963 (2022).
Badgley, M.A. et al. Cysteine depletion induces pancreatic tumor ferroptosis in mice. Science 368, 85–89 (2020).
Cheng, Q., Bao, L., Li, M., Chang, K. & Yi, X. Erastin synergizes with cisplatin via ferroptosis to inhibit ovarian cancer growth in vitro and in vivo. J. Obstet. Gynaecol. Res. 47, 2481–2491 (2021).
Paronetto, M. P. Ewing sarcoma protein: a key player in human cancer. Int. J. Cell Biol. 2013, 642853 (2013).
Mullard, M. et al. Ubiquitin-specific proteases as therapeutic targets in paediatric primary bone tumours? Biochem. Pharm. 194, 114797 (2021).
Shukla, N. et al. Proteasome addiction defined in Ewing sarcoma is effectively targeted by a novel class of 19S proteasome inhibitors. Cancer Res. 76, 4525–4534 (2016).
Vormoor, B. & Curtin, N. J. Poly(ADP-ribose) polymerase inhibitors in Ewing sarcoma. Curr. Opin. Oncol. 26, 428–433 (2014).
Liu, S. et al. The evolution and heterogeneity of neutrophils in cancers: origins, subsets, functions, orchestrations and clinical applications. Mol. Cancer 22, 148 (2023).
Shukrun, R. et al. Suggested role for neutrophil extracellular trap formation in Ewing sarcoma immune microenvironment. Cancer Sci. 115, 36–47 (2023).
Linde, I. L. et al. Neutrophil-activating therapy for the treatment of cancer. Cancer Cell 41, 356–372 e310 (2023).
Muller, T. et al. Automated sample preparation with SP3 for low-input clinical proteomics. Mol. Syst. Biol. 16, e9111 (2020).
Hughes, C. S. et al. Ultrasensitive proteome analysis using paramagnetic bead technology. Mol. Syst. Biol. 10, 757 (2014).
Demichev, V., Messner, C. B., Vernardis, S. I., Lilley, K. S. & Ralser, M. DIA-NN: neural networks and interference correction enable deep proteome coverage in high throughput. Nat. Methods 17, 41–44 (2020).
Rudolph, J. D. & Cox, J. A network module for the perseus software for computational proteomics facilitates proteome interaction graph analysis. J. Proteome Res. 18, 2052–2064 (2019).
Tyanova, S. et al. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 13, 731–740 (2016).
Szklarczyk, D. et al. STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 47, D607–D613 (2019).
Szklarczyk, D. et al. The STRING database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 45, D362–D368 (2017).
Shannon, P. et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504 (2003).
Monti, S., Tamayo, P., Mesirov, J. & Golub, T. Consensus clustering: a resampling-based method for class discovery and visualization of gene expression microarray data. Mach. Learn. 52, 91–118 (2003).
Ceranski, A. K. et al. Refined culture conditions with increased physiological relevance uncover oncogene-dependent metabolic signatures in Ewing sarcoma spheroids. Cell Rep. Methods 5, 100966 (2025).
Li, J. et al. Therapeutic targeting of the PLK1-PRC1-axis triggers cell death in genomically silent childhood cancer. Nat. Commun. 12, 5356 (2021).
Bankhead, P. et al. QuPath: open source software for digital pathology image analysis. Sci. Rep. 7, 16878 (2017).
Pachitariu, M. & Stringer, C. Cellpose 2.0: how to train your own model. Nat. Methods 19, 1634–1641 (2022).
Ram, S. et al. Pixelwise H-score: a novel digital image analysis-based metric to quantify membrane biomarker expression from immunohistochemistry images. PLoS ONE 16, e0245638 (2021).
Golani O. WIS-MICC-CellObservatory/Ewing-Sarcoma-Proteomics-and-Immune-Landscape. Zenodo, https://doi.org/10.5281/zenodo.18096815 (2025).
Acknowledgements
We thank the members of the Geiger lab for fruitful discussions and technical assistance. Opal slide imaging was made possible thanks to the support of the de Picciotto Cancer Cell Observatory in Memory of Wolfgang and Ruth Lesser, a research grant from the Quinquin Foundation, and a research grant from the Morris Kahn Institute for Human Immunology and the Fabrikant-Morse Families Research Fund for Humanity. This work was funded by the Israel Science Foundation grant #3106/21 and the European Council ERC-consolidator grant #101044574. This research was also generously supported by the Applebaum Foundation and the Vera and John Schwartz Family Center for Metabolic Biology.
Author information
Authors and Affiliations
Contributions
S.G. performed the experiments, analyzed the data and drafted the manuscript; R.S., S.G., R.E., and T.G. conceptualized, designed the research and interpreted the results; S.G., R.S., O.S., and M.M. assembled samples and clinical information; V.M., S.G., R.O., and L.F.A. performed the multiplexed imaging; V.M., O.G., and Y.A. acquired the multiplexed imaging data, developed the analytical pipelines and performed the image analyses; V.M., S.G., and S.M. performed the spheroid and ferroptosis analysis; L.B. established the proteomic sample preparation methods; V.M. contributed to figure preparation and the writing of the manuscript; B.D. initialized the research; T.G. supervised the research, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gordon, S., Mohan, V., Shukrun, R. et al. Proteomic landscape of Ewing sarcoma primary tumors and metastases. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70449-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70449-5