Abstract
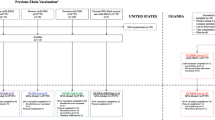
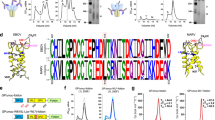
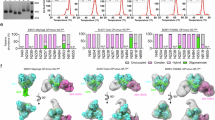
The highly lethal Ebola virus species—Zaire (EBOV), Sudan (SUDV), and Bundibugyo (BDBV)—pose persistent threats to global health. Current antibody therapies target EBOV but lack broad neutralization across ebolaviruses. Recent pan-ebolavirus strategies rely on antibody cocktails. Here, we identified two camelid-derived nanobodies (1A10 and BA2) that neutralize EBOV, SUDV, and BDBV in vitro and protect female rodents against these pathogens. High-resolution cryo-EM structures of their GP complexes showed that 1A10 and BA2 bind conserved but non-overlapping epitopes near the GP1 base and GP2’s internal fusion loop (IFL), and biochemical analyses revealed their distinct neutralization mechanisms. To further improve efficacy, we engineered a bispecific antibody (BA2-1A10) via GS linker-mediated IgG-Fc fusion, which provided highly potent protection against all three viruses in female rodents model and positions it as a strong broad-spectrum anti-ebolavirus candidate. Our work demonstrates a structure-guided bispecific nanobody strategy for pan-ebolavirus therapy and highlights compact antibodies for next-generation antivirals.
Similar content being viewed by others
Data availability
Source data are provided as a Source Data file. All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary information. Atomic coordinates and structure factors for the reported cryo-EM structures have been deposited with the Protein Data Bank and Electron Microscopy Data Bank under accession numbers 9VTS/EMD-65343 (https://www.rcsb.org/structure/unreleased/9VTS; https://www.ebi.ac.uk/emdb/EMD-65343), 9VT4/EMD-65314 (https://www.rcsb.org/structure/unreleased/9VT4; https://www.ebi.ac.uk/emdb/EMD-65314). Other structures for analysis, including 5JQ3, 7M8L, 6PCI, and 7SWD were obtained from the PDB. No new sequencing data has been generated as part of in this study. Source data are provided with this paper.
References
Carroll, S. A. et al. Molecular evolution of viruses of the family Filoviridae based on 97 whole-genome sequences. J. Virol. 87, 2608–2616 (2013).
Furuyama, W. & Marzi, A. Ebola virus: pathogenesis and countermeasure development. Ann. Rev. Virol. 6, 435–458 (2019).
Jain, S., Martynova, E., Rizvanov, A., Khaiboullina, S. & Baranwal, M. Structural and functional aspects of Ebola virus proteins. Pathogens 10, 1330 (2021).
O’Donnell, K. & Marzi, A. The Ebola virus glycoprotein and its immune responses across multiple vaccine platforms. Expert Rev. Vaccines 19, 267–277 (2020).
Cantoni, D. & Rossman, J. S. Ebolaviruses: New roles for old proteins. PLoS Negl. Trop. Dis. 12, e0006349 (2018).
Volchkov, V. E., Feldmann, H., Volchkova, V. A. & Klenk, H. D. Processing of the Ebola virus glycoprotein by the proprotein convertase furin. Proc. Natl. Acad. Sci. USA 95, 5762–5767 (1998).
Jeffers, S. A., Sanders, D. A. & Sanchez, A. Covalent modifications of the ebola virus glycoprotein. J. Virol. 76, 12463–12472 (2002).
Zhao, Y. et al. Toremifene interacts with and destabilizes the Ebola virus glycoprotein. Nature 535, 169–172 (2016).
Lennemann, N. J. et al. Comprehensive functional analysis of N-linked glycans on Ebola virus GP1. mBio 5, e00862–00813 (2014).
Iraqi, M. et al. N-Glycans mediate the Ebola virus-GP1 shielding of ligands to immune receptors and immune evasion. Front. Cell Infect. Microbiol. 10, 48 (2020).
Chandran, K., Sullivan, N. J., Felbor, U., Whelan, S. P. & Cunningham, J. M. Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. Science 308, 1643–1645 (2005).
Wang, H. et al. Ebola viral glycoprotein bound to its endosomal receptor Niemann-Pick C1. Cell 164, 258–268 (2016).
Gong, X. et al. Structural insights into the Niemann-Pick C1 (NPC1)-mediated cholesterol transfer and Ebola infection. Cell 165, 1467–1478 (2016).
Weissenhorn, W., Carfí, A., Lee, K. H., Skehel, J. J. & Wiley, D. C. Crystal structure of the Ebola virus membrane fusion subunit, GP2, from the envelope glycoprotein ectodomain. Mol. Cell 2, 605–616 (1998).
Lee, J. E. et al. Structure of the Ebola virus glycoprotein bound to an antibody from a human survivor. Nature 454, 177–182 (2008).
Gregory, S. M. et al. Structure and function of the complete internal fusion loop from Ebolavirus glycoprotein 2. Proc. Natl. Acad. Sci. USA 108, 11211–11216 (2011).
Lee, J. et al. Structure of the Ebola virus envelope protein MPER/TM domain and its interaction with the fusion loop explains their fusion activity. Proc. Natl. Acad. Sci. USA 114, E7987–e7996 (2017).
Coltart, C. E., Lindsey, B., Ghinai, I., Johnson, A. M. & Heymann, D. L. The Ebola outbreak, 2013-2016: old lessons for new epidemics. Philo. Trans. R. Soc. London. Ser. B Biol. Sci. 372, 20160297 (2017).
Qiu, X. et al. Reversion of advanced Ebola virus disease in nonhuman primates with ZMapp. Nature 514, 47–53 (2014).
Markham, A. REGN-EB3: first approval. Drugs 81, 175–178 (2021).
Rayaprolu, V. et al. Structure of the Inmazeb cocktail and resistance to Ebola virus escape. Cell Host Microbe 31, 260–272.e267 (2023).
Misasi, J. et al. Structural and molecular basis for Ebola virus neutralization by protective human antibodies. Science 351, 1343–1346 (2016).
Gaudinski, M. R. et al. Safety, tolerability, pharmacokinetics, and immunogenicity of the therapeutic monoclonal antibody mAb114 targeting Ebola virus glycoprotein (VRC 608): an open-label phase 1 study. Lancet 393, 889–898 (2019).
Ibrahim, S. K., Ndwandwe, D. E., Thomas, K., Sigfrid, L. & Norton, A. Sudan virus disease outbreak in Uganda: urgent research gaps. BMJ Global Health 7, e010982 (2022).
Towner, J. S. et al. Newly discovered ebola virus associated with hemorrhagic fever outbreak in Uganda. Plos Pathog. 4, e1000212 (2008).
El Ayoubi, L. W., Mahmoud, O., Zakhour, J. & Kanj, S. S. Recent advances in the treatment of Ebola disease: a brief overview. PLoS Pathog. 20, e1012038 (2024).
Taki, E. et al. Ebanga™: the most recent FDA-approved drug for treating Ebola. Front. Pharmacol. 14, 1083429 (2023).
Könning, D. et al. Camelid and shark single domain antibodies: structural features and therapeutic potential. Curr. Opin. Struct. Biol. 45, 10–16 (2017).
De Meyer, T., Muyldermans, S. & Depicker, A. Nanobody-based products as research and diagnostic tools. Trends Biotechnol. 32, 263–270 (2014).
Muyldermans, S. Nanobodies: natural single-domain antibodies. Annu Rev. Biochem. 82, 775–797 (2013).
Steeland, S., Vandenbroucke, R. E. & Libert, C. Nanobodies as therapeutics: big opportunities for small antibodies. Drug Discov. Today 21, 1076–1113 (2016).
Miller, E. H. & Chandran, K. Filovirus entry into cells - new insights. Curr. Opin. Virol. 2, 206–214 (2012).
Moller-Tank, S. & Maury, W. Ebola virus entry: a curious and complex series of events. Plos Pathog. 11, e1004731 (2015).
Schornberg, K. et al. Role of endosomal cathepsins in entry mediated by the Ebola virus glycoprotein. J. Virol. 80, 4174–4178 (2006).
Côté, M. et al. Small molecule inhibitors reveal Niemann-Pick C1 is essential for Ebola virus infection. Nature 477, 344–348 (2011).
Carette, J. E. et al. Ebola virus entry requires the cholesterol transporter Niemann-Pick C1. Nature 477, 340–343 (2011).
Simmons, J. A. et al. Ebolavirus glycoprotein directs fusion through NPC1+ endolysosomes. J. Virol. 90, 605–610 (2016).
Brecher, M. et al. Cathepsin cleavage potentiates the Ebola virus glycoprotein to undergo a subsequent fusion-relevant conformational change. J. Virol. 86, 364–372 (2012).
Spence, J. S., Krause, T. B., Mittler, E., Jangra, R. K. & Chandran, K. Direct visualization of Ebola virus fusion triggering in the endocytic pathway. mBio 7, e01857–01815 (2016).
Mittler, E., Alkutkar, T., Jangra, R. K. & Chandran, K. Direct Intracellular visualization of Ebola Virus-receptor interaction by in situ proximity ligation. mBio 12 10–1128 (2021).
Gilchuk, P. et al. Pan-ebolavirus protective therapy by two multifunctional human antibodies. Cell 184, 5593–5607.e5518 (2021).
Gilchuk, P. et al. Multifunctional pan-ebolavirus antibody recognizes a site of broad vulnerability on the ebolavirus glycoprotein. Immunity 49, 363–374.e310 (2018).
Milligan, J. C. et al. Asymmetric and non-stoichiometric glycoprotein recognition by two distinct antibodies results in broad protection against ebolaviruses. Cell 185, 995–1007.e1018 (2022).
Wec, A. Z. et al. Antibodies from a human survivor define sites of vulnerability for broad protection against Ebolaviruses. Cell 169, 878–890.e815 (2017).
Bornholdt, Z. A. et al. Isolation of potent neutralizing antibodies from a survivor of the 2014 Ebola virus outbreak. Science 351, 1078–1083 (2016).
Zhao, X. et al. Immunization-elicited broadly protective antibody reveals ebolavirus fusion loop as a site of vulnerability. Cell 169, 891–904.e815 (2017).
Dube, D. et al. The primed ebolavirus glycoprotein (19-kilodalton GP1,2): sequence and residues critical for host cell binding. J. Virol. 83, 2883–2891 (2009).
Oosterlaken, M. et al. Nanobody therapy rescues behavioural deficits of NMDA receptor hypofunction. Nature 645, 262–270 (2025).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Rubinstein, J. L. & Brubaker, M. A. Alignment of cryo-EM movies of individual particles by optimization of image translations. J. Struct. Biol. 192, 188–195 (2015).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Trabuco, L. G., Villa, E., Mitra, K., Frank, J. & Schulten, K. Flexible fitting of atomic structures into electron microscopy maps using molecular dynamics. Structure 16, 673–683 (2008).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. Sect. D. Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr D. Biol. Crystallogr. 66, 486–501 (2010).
Pettersen, E. F. et al. UCSF Chimera-a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Goddard, T. D. et al. UCSF ChimeraX: Meeting modern challenges in visualization and analysis. Protein Sci. 27, 14–25 (2018).
Copin, R. et al. The monoclonal antibody combination REGEN-COV protects against SARS-CoV-2 mutational escape in preclinical and human studies. Cell 184, 3949–3961.e3911 (2021).
Baum, A. et al. Antibody cocktail to SARS-CoV-2 spike protein prevents rapid mutational escape seen with individual antibodies. Science 369, 1014–1018 (2020).
Acknowledgements
This work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (Grant No. XDB0490000 to S.C., T.J., and P.G.), the National Key Research and Development Program (Grant No. 2022YFC2303300 to T.J. and P.G.), the National Natural Science Foundation of China (Grant No. 82302522 to M.W.), and the “USTC Research Funds of the Double First-Class Initiative (Grant No. YD9100002040). The funders had no role in the study design, data collection, and interpretation, or the decision to submit the work for publication. We thank Professor Huaju Chen for providing the original plasmids of EBOV-GPs. We thank Dr. Huan Ma, M.S Dongxiang Hong for helping with phage display library establish. We also thank Dr. Hengxia Jia for supporting the cryo-EM data collection and Dr. Bo Shu for helpful discussions in cryo-EM data proccessing and refinement. We thank the Institutional Center for Shared Technologies and Facilities of Wuhan Institute of Virology, Chinese Academy of Sciences, and the Cryo-EM Center at the University of Science and Technology of China for supporting cryo-EM data collection. We thank the National Biosafety Laboratory, Wuhan (CSTR: 31120.02.NBL), Chinese Academy of Sciences, for providing BSL-4 and ABSL-4 facility support for this project; the National Virus Resource Center; Experimental Animal Center, and Public Technology Service Center of Wuhan Institute of Virology, CAS, and the Animal Facility of USTC for their support.
Author information
Authors and Affiliations
Contributions
S.C. and T.C.J. conceived this study. S.C., T.C.J., P.G., M.H.W., and X.H.Z designed and directed the research; M.H.W, M.Y.L, J.H., and C.B.Y. immunized the camel, screened the Nbs, and analyzed sequences; M.H.W, M.Y.L., and Y.H.S. constructed the plasmids and produced antibodies; M.H.W performed the antibody characterization in vitro and engineering assay; L.X.Z and M.H.W performed the surface plasmon resonance (SPR) experiments; X.H.Z., E.T.L., M.H.W, C.P., B.Y.Z., and J.G.Z. performed the virus neutralization assay; M.H.W., Y.X.G., S.L.L., and Z.L.Z. performed the cryo-EM data collection, analysis, and deposition with the supervision of Tengchuan Jin (BA2 complex) and Peng Gong (1A10 complex). X.H.Z., Y.F.Y, W.J.L, F.X.L., M.Y.C., H.L., and F.H.Y. performed the rodents challenge studies; S.C., T.C.J, P.G., M.H.W., and X.H.Z. edited and revised the manuscript; all authors read and approved the paper.
Corresponding authors
Ethics declarations
Competing interests
T.C.T., M.H.W., S.C., and E.T.L. are named inventors on two patents covering the sequences of the 1A10 and BA2 nanobodies described in this study. These patents pertain to the use of said nanobodies for the prevention, treatment, or diagnosis of Ebola virus infection and impose no restrictions on data publication. All other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, M., Zhang, X., Li, W. et al. A highly potent nanobody-based bispecific therapeutic provides broad-spectrum protection against ebolavirus. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70464-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70464-6